Abstract
Early-onset parkinsonism (EO parkinsonism), defined as subjects with disease onset before the age of 40 or 50 years, can be the main clinical presentation of a variety of conditions that are important to differentiate. Although rarer than classical late-onset Parkinson’s disease (PD) and not infrequently overlapping with forms of juvenile onset PD, a correct diagnosis of the specific cause of EO parkinsonism is critical for offering appropriate counseling to patients, for family and work planning, and to select the most appropriate symptomatic or etiopathogenic treatments. Clinical features, radiological and laboratory findings are crucial for guiding the differential diagnosis. Here we summarize the most important conditions associated with primary and secondary EO parkinsonism. We also proposed a practical approach based on the current literature and expert opinion to help movement disorders specialists and neurologists navigate this complex and challenging landscape.
Keywords
INTRODUCTION
The term “early-onset Parkinson’s disease” (EOPD, or young-onset PD - YOPD) refers to cases of PD with onset between the age of 21 and 40 years, as reported by Quinn et al. in their seminal paper from 1987, or between 21 and 50 years, according to other authors [1–4]. Compared with idiopathic cases of PD (iPD), patients with EOPD usually present a slower progression of the motor symptoms, a prevalence of bradykinesia over tremor, focal dystonia at onset or during off-state, satisfactory response even to low doses of levodopa, earlier motor complications (such as motor fluctuations and dyskinesias), a lower incidence of cognitive impairment and non-motor symptoms, while anxiety and depression are frequent [1, 5–16]. A positive family history can be often identified in these patients, suggesting an important role of genetics in the pathogenesis of several of these cases [15, 16].
Based on retrospective observational studies, classical EOPD accounts for about 3–7% of all cases of PD in the Western world and up to 10–14% in Japan [1, 17–19], with an incidence between 0.29 and 3.3 per 100,000 persons-years in the current literature [8, 21].
In patients with EO parkinsonism, parkinsonian features can present as part of more complicated syndromes, with associated additional neurological manifestations (dystonia, pyramidal signs, cognitive impairment), psychiatric manifestations, systemic symptoms, alterations on brain imaging, and age of onset between 21 and 40 or 50 years.
Although rare, cases of EOPD or EO parkinsonism demand prompt diagnosis, as some forms can respond to treatments (i.e., PRKN-associated PD) and early interventions can prevent severe complications (i.e., Wilson’s disease or ataxia telangiectasia). Primary forms of EO parkinsonism are more frequent than secondary cases due, among the others, to drug exposures or vascular accidents [22]. A significant proportion of primary forms of EO parkinsonism are due to genetic causes. Given the impressive advancements in gene-based therapeutic approaches, some patients may qualify for these treatments in the near future [23].
Because of the broad range of the differential diagnosis, significant diagnostic delays are frequent with important implications for genetic counseling for these patients and their families.
In this review, we summarized the most important genetic and systemic causes of EOPD and EO parkinsonism (Table 1, Fig. 1). We also provide a practical approach for the clinical evaluation, examination, and genetic counseling of these patients to help guide the diagnostic process and the referral to appropriate available or future treatments.
Demographic and radiological features of the spectrum of causes of EO parkinsonism
The table summarizes the different etiologies that can manifest with EO parkinsonism. Typical age of onset, mode of heritability, response to levodopa, characteristic imaging findings and distinctive clinical features are reported. AD, autosomal dominant; ALS, amyotrophic lateral sclerosis; AR, autosomal recessive; CC, corpus callosum; GRE, gradient echo; HSP, hereditary spastic paraplegia; MCP, medium cerebellar peduncle; NBIA, neurodegeneration with brain iron accumulation; OA, optic atrophy; PEO, Progressive external ophthalmoplegia; SANDO, sensory ataxic neuropathy, dysarthria and ophthalmoparesis; SNGP, supranuclear gaze palsy; SWI, susceptibility weighted imaging; XD, X-linked dominant; XR, X-linked recessive; WM, white matter; +, positive response; ++, robust response; +/–, inconsistent or limited response.
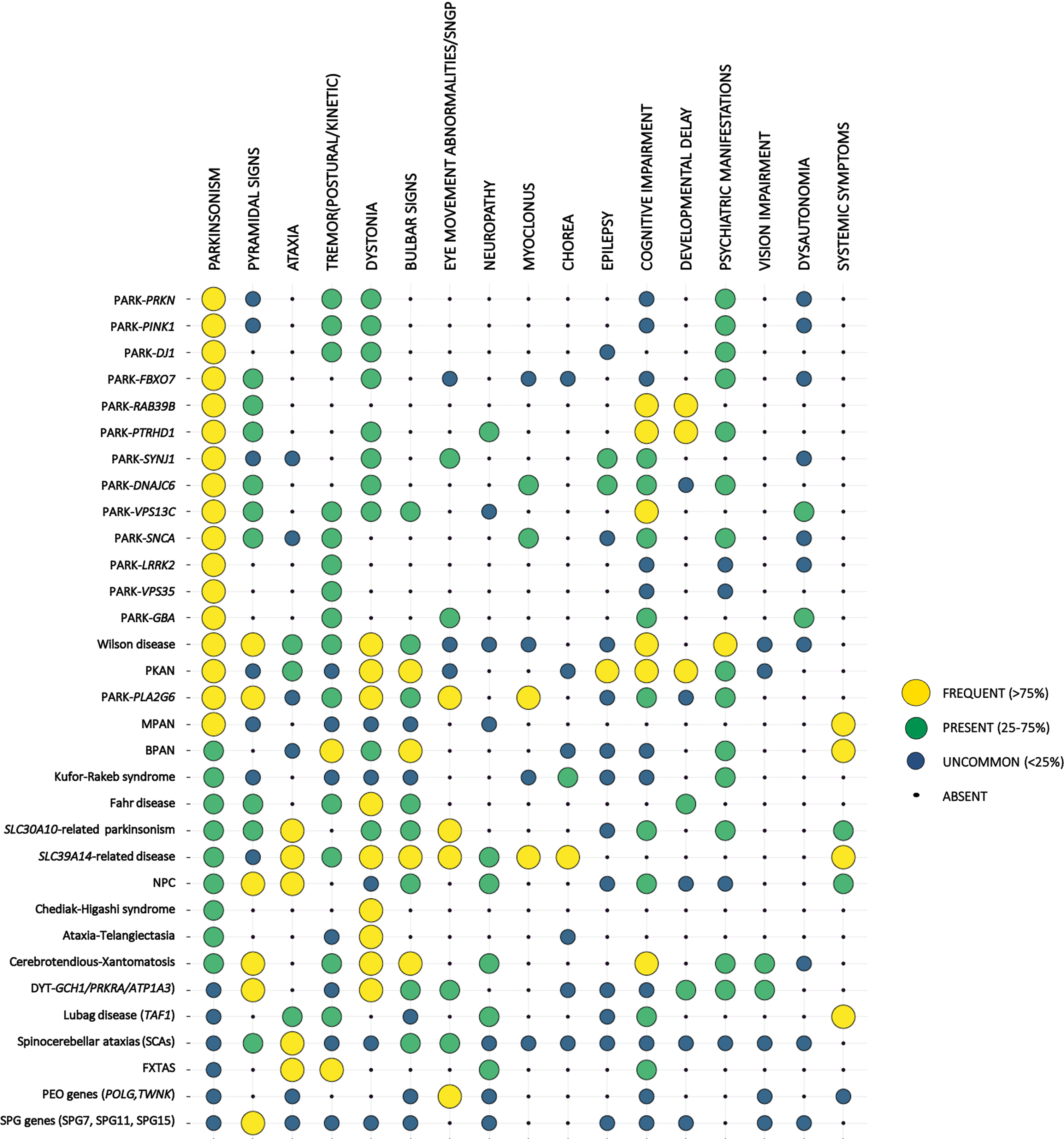
Clinical presentation of the different forms of EO parkinsonism. Monogenic forms of EOPD and other forms of EO parkinsonism are listed on the left. For each condition the frequency of the listed clinical features (top row) is represented by the size and the color of the corresponding dot. Symptoms are described as frequent (if expected to be found in more than 75% of cases of a specific form of EO parkinsonism), present (expected to be found in 25–75% of cases), uncommon (expected to be found in < 25 of cases), or absent (never reported in the specific phenotype), based on data reported in the literature and authors’ experience. Conditions were ordered according to the frequency of parkinsonian features.
Although some of these forms can sometimes manifest before the age of 20 year, thus falling into the juvenile-onset PD (JOPD) category, here we will focus on the presentation of these various forms in the age range of 21 to 50 years.
The various etiologies of EO parkinsonism underlie distinct pathogenic mechanisms, particularly related to the genetic mutations. In general, earlier onset parkinsonism, whether in genetic or idiopathic cases, has been found to be associated with a more prominent degeneration of the basal ganglia, in certain cases lacking the classical Lewy bodies (such as in cases of
METHODS
This work represents an expert opinion on this topic supported by the current literature. A narrative review of the literature was performed through a PubMed research using to the following key words: “early onset Parkinson’s disease”, “early onset parkinsonism”, “young onset Parkinson’s disease”, “young onset Parkinsonism”, “neurodegeneration with brain iron accumulation”, “secondary parkinsonism”, “parkinsonism” plus “lysosomal storage disease”, “mitochondrial disease”, “metabolic diseases”, “dystonia”, “ataxia”, “spasticity”, “spastic paraplegia”, “mental retardation”, “basal ganglia calcification”, “manganese”, “infectious disease”, “vascular diseases”, “Wilson’s disease”.
We focused only on the works referring to cases of EO parkinsonism. We included the more relevant and comprehensive cohort studies and reviews on the topics, as well as data from single case reports or small cohort studies for the rarer conditions.
Parkinson-predominant EO parkinsonism
Genetic forms
Patients with mutations in
Very rare cases of rapidly progressive parkinsonism with median age at onset of 29 have been associated with mutations in
Rarer autosomal recessive forms of PD present an early onset and are associated with atypical parkinsonism and complex phenotypes, often with a more severe course and are therefore discussed in the corresponding paragraphs below.
Mutations in
Mutations in
GBA mutations are the most frequent genetic risk factor of PD, found in 5–20% of patients with PD across different populations [98–102]. A recent study screening a large cohort of PD patients identified an enrichment in GBA variants in EOPD [102]. Different variants of this gene have variable frequency across populations [103]. For example, the N370S, 84GG, E326K, and R496H variants are more frequent among Ashkenazi Jews, while the other common variant, L444P, is mostly found in non-Ashkenazi Jewish European subjects [99]. Compared with non-GBA carriers, PD patients with GBA mutations have an earlier onset of PD (median age 58) [98, 104], are more likely to develop PD before 50 years of age [98, 105], and have a higher risk and earlier presentation of non-motor features (i.e., dementia, dysautonomia, psychiatric manifestations) compared with idiopathic PD [99, 107].
Secondary EO parkinsonism
Secondary causes of parkinsonism should be considered as well when assessing patients with EOPD, as they may represent treatable conditions, different from the majority of the degenerative processes. Drug-induced parkinsonism (secondary to the use of dopamine blocking agents, dopamine depletors, or, more rarely, associated with other drugs, such as calcium antagonists), and vascular parkinsonism are the most common forms of secondary parkinsonism [108]. Symmetric presentations and lack of the classical resting tremor are more frequent in these patients [109]. Although these conditions are more common among older subjects, they can be seen also in younger patients. When first evaluating a patient with EOPD, imaging studies of the brain should be considered to rule out rarer causes of secondary parkinsonism, such as vascular malformations (fistula, cavernoma, or arteriovenous malformations), single strategic lesions in the basal ganglia circuitry, supratentorial tumors or metastatic neoplastic lesions, as well as obstructive hydrocephalus [110]. Additional neurological symptoms can be frequently appreciated in secondary forms of EOPD and can help guide the diagnosis.
Systemic diseases that can present with parkinsonism in young age are represented by hepatic insufficiency (associated with bilateral hyperintensities of the basal ganglia on T1-weighted sequences or hyperintensities in the cerebellum and middle cerebellar peduncles on T2-weighted images), systemic lupus erythematosus (where characteristic serological biomarkers and systemic symptoms are crucial for the diagnosis), as well as infectious diseases (such as HIV and possible opportunistic infections) [109]. A high number of cases of secondary parkinsonism were observed during the pandemic of Spanish flu [111].
The use of street drugs (such as amphetamines derivatives or MPTP), as well as exposure to carbon monoxide, manganese - as described in the paragraph “Metals in the brain” -, cyanide and methanol can represent additional causes of secondary parkinsonism in young patients [112].
A recent work also showed a correlation with sport-related head trauma and EO parkinsonism, in subjects where the most common genetic causes of EOPD were excluded [113]. Although more studies will be necessary to further clarify this association, traumatic brain injuries have been already correlated with parkinsonism, thus this trigger may be taken into consideration among the other factors as a causative or precipitating event for EO parkinsonism in predisposed subjects [114].
EO parkinsonism associated with additional characteristic features
EO parkinsonism and metals in the brain
Brain metal accumulation can manifest clinically as early-onset parkinsonism in several neurological disorders. Various metals can be accumulated in the brain and other body tissues in these conditions (e.g., copper in Wilson’s disease, iron in neurodegeneration with brain iron accumulation (NBIA), calcium in Fahr’s disease, and manganese in hypermanganesemia). Some of these disorders are genetically inherited, while others are acquired [115–118].
Typically, the clinical suspect for these conditions emerges after brain imaging studies (i.e., brain MRI for Wilson’s disease, NBIA, and hypermanganesemia, and head CT for brain calcifications) [119]. However, associated systemic findings, such as hepatic involvement in Wilson’s disease and hypermanganesemia, can be the first clue to suspect metal accumulation in the brain [115, 120].
Wilson’s disease
Wilson’s disease is distributed worldwide, with an estimated prevalence of 1 case per 30,000 live births in most populations [146]. However, in some isolated populations, such as a small mountain village on the island of Crete, the prevalence is much higher (1 in 15 births), due to high rates of consanguinity [121].
Wilson’s disease is an autosomal recessive disorder caused by loss-of-function mutations of
Copper accumulation is associated with a progressive neurodegeneration of basal ganglia leading to a complex movement disorder often associated with bulbar symptoms. The movement disorder is usually characterized by tremor, dystonia, and/or parkinsonism. Bulbar symptoms consist of dysarthria, drooling and/or dysphagia. In addition, a range of additional neurologic features may be present, including cerebellar ataxia, chorea, hyperreflexia, seizures, cognitive impairment, and psychiatric features [115].
While parkinsonism is rarely found in isolation or as the predominant feature in Wilson’s disease, this is commonly seen in combination with dystonia and/or tremor at presentation. Indeed, isolated parkinsonism occurs in less than 2% of neurologic presentations but parkinsonism in combination with other movement disorders is seen in the majority of the cases [115].
On brain MRI, T2 hyperintensity of the putamina and other deep brain nuclei is the most common abnormality. Involvement of the midbrain tegmentum can appear as a face of the “giant panda” on axial images. Axial T2 at the pons level may also show the face of a “miniature panda”. On T1-weighted images, these same areas show hypointensity. However, patients with severe hepatic dysfunction show areas of T1 hyperintensity, especially in the globus pallidus, similar to that seen in acquired hepatocerebral degeneration attributed to manganese deposition [126].
Neurodegeneration with brain iron accumulation
NBIA is a heterogeneous group of inherited movement disorders characterized by abnormal iron accumulation in basal ganglia and extrapyramidal movement disorders. The clinical features of NBIA range from global neurodevelopmental disorder in infancy to mild parkinsonism associated with minimal cognitive impairment in adulthood [116]. To date, ten NBIA genes have been identified, each associated with a different clinical syndrome and specific brain MRI features [118]. All forms of NBIA combined are considered to be ultra-rare with less than 1/1,000,000 affected world-wide, with the most common form being pantothenate kinase-associated neurodegeneration (PKAN), which accounts for approximately 50% of cases [127].
NBIA disorders presenting with parkinsonism as part of their clinical presentation include PKAN,
Mutations in
Mutations in
The clinical differential diagnosis among NBIA disorders is primarily based on the age at onset and on the different brain MRI features. PKAN, BPAN and MPAN typically have an early onset in childhood, PLAN and KRS begin in early adulthood, while neuroferritinopathy and aceruloplasminemia are adult-onset disorders [116]. Notably, BPAN-associated EOPD, due to mutations in
Fahr’s disease
The presence of neurological and psychiatric symptoms associated with bilateral basal ganglia calcifications identifies a clinical syndrome defined as Fahr’s disease [117]. This denomination mainly refers to idiopathic forms where no metabolic or other underlying causes are identified. Fahr’s disease commonly affects people after their 40s; therefore, this disorder is not a frequent cause of EOPD. Patients are usually in good health in their youth and tend to develop symptoms later in adulthood [145]. Calcifications are typically symmetrical and bilateral and involve the caudate, putamen, globus pallidus, thalamus, deep cortex, dentate nucleus, and, less frequently, the white matter [146]. CT scan is considered the gold standard for diagnosis identifying calcifications as hyperdense lesions [146]. Recently, mutations in five different genes (
Brain manganese accumulation
Manganese brain accumulation can be due to genetic or acquired causes. Genetically-determined brain manganese accumulations are ultra-rare conditions, with less than 100 cases reported world-wide so far [149]. There are two known genetic forms of brain manganese accumulation, associated with recessive mutations of
Considering non-genetic forms, environmental exposure to high quantities of manganese is known to be neurotoxic and causes manganism, a clinical syndrome characterized by an extrapyramidal movement disorder (e.g., dystonia and/or parkinsonism) associated with T1 hyperintensity of the basal ganglia on brain MRI resulting from manganese accumulation in the basal ganglia. Manganism has been described in workers inhaling manganese-loaded dust or fumes (e.g., mining and welding industries), in individuals drinking manganese-contaminated water, and in drug addicts using intravenous drugs contaminated with potassium permanganate [150]. Brain accumulation of manganese is also observed in subjects with advanced hepatic cirrhosis or portosystemic shunts (acquired hepatocerebral degeneration) in which impaired biliary excretion of manganese results in manganese accumulation in the basal ganglia possibly causing debilitating movement disorders [151].
EO parkinsonism associated with metabolic conditions
A number of metabolic disorders can present with EO parkinsonism. Diseases caused by mutations in genes encoding for mitochondrial proteins are usually characterized by short stature, diabetes, ophthalmoplegia, deafness, cardiomyopathy, as well as neurological disorders such as myoclonus, seizures, and stroke like episodes [152]. The majority of the reports of parkinsonism and mitochondrial diseases consist of single case descriptions, as summarized in previous works [153, 154]. Although in most of these cases the neurological symptoms, including parkinsonism, manifest during childhood, onset after the age of 21 year is possible. Thus they should be considered in the differential diagnosis of EOPD. Some of these conditions are described below (paragraph “EO parkinsonism associated with other neurological symptoms”). Matrilineal heritability, due to mutations of genes in the mitochondrial DNA, can be a key factor for suspecting these diseases.
Parkinsonism has also been reported in a growing number of conditions caused by impairment of the lysosomal/lipid metabolisms, such as Gaucher disease (GD) (as described in paragraph “Monogenic forms of EOPD”). Parkinsonism has been described in single cases of Niemann Pick type C (NPC), although screenings of large cohorts of patients with PD failed to confirm the association between mutations of the NPC-causative genes, NPC1 and NPC2, and PD. On the contrary, variants of the SMPD1 gene, that is responsible for Niemann Pick types A and B, have been associated with PD across different populations [155–162]. NPC and GD are far more common among Ashkenazi Jews.
Chediak-Higashi syndrome (CHS) is a rare lysosomal disorder caused by mutations of the
Although the onset is more common during childhood, EOPD has also been reported in cases of gangliosidosis GM1, as well as late onset GM2 gangliosidosis (also called late onset Tay-Sachs disease, LOTS), and adult onset neuronal ceroid lipofuscinosis, usually in the context of additional neurological manifestations [15, 169–171]. Some of the conditions, such as LOTS, are more prevalent among Ashkenazi Jewish, although other clusters have been reported also within an Amish community in Pennsylvania and non-Jewish French Canadians in the area of St. Lawrence [172].
Parkinsonism can present in almost half of the cases of ataxia telangiectasia (A-T) (41% cases), usually in the second or third decade, when the other symptoms associated with this systemic disorder are already present (summarized in [173]). In a few cases levodopa has been reported to be beneficial in A-T.
Finally, young-onset parkinsonism can be present in the context of hemochromatosis, a hereditary condition caused by aberrant iron accumulation in the peripheral tissues causing, among the others, hepatic toxicity, anemia, diabetes, cardiac manifestations and hypogonadism [174–176]. In these cases, parkinsonism develops between the fourth and seventh decade and brain MRI usually shows hypointensity in the basal ganglia and substantia nigra [175, 176]. This condition is more frequent across people of Northern European descent, originally arising within the Celtic population [177].
EO parkinsonism associated with other neurological symptoms
Dystonia-parkinsonism syndromes
Parkinsonism may be part of the clinical spectrum of syndromes defined as “dystonia-parkinsonisms”. This term applies to rare conditions where the severity of dystonia usually equals that of parkinsonism. Unlike the parkinsonian syndromes with additional dystonia (i.e.,
DYT-GCH1 typically presents with lower-limb dystonia, causing gait disturbance and a tendency to fall. Clinical features like diurnal fluctuation and sleep benefit are typically associated with this form and can be helpful in differentiating it from other forms of EOPD. The response to levodopa of both dystonic and parkinsonian signs is remarkable with low-doses (i.e., < 300–400 mg/daily) [178] with no development of the levodopa-induced dyskinesias.
Mutations in protein kinase, interferon-inducible double-stranded RNA-dependent activator (DYT-PRKRA) cause an autosomal recessive form of young onset, progressive generalized dystonia and mild parkinsonism [179]. A remarkable involvement of oromandibular and bulbar regions with a sardonic smile, tongue protrusion, significant speech and swallowing disturbances is typical of this form. The axial involvement of the neck and trunk can lead to opisthotonic postures. Parkinsonism is usually not a prominent feature, appears later than dystonia and is characterized by bradykinesia and rigidity, rather than resting tremor. The disorder was initially described in three Brazilian families [179]. In a few cases, bilateral striatal degeneration has been observed on brain MRI [180, 181]. However, the three described cases presented with a very young age of onset, before the age of 20 [180, 181].
Rapid-onset dystonia parkinsonism (RODP) due to mutations in the
DYT-TAF1 is an adult-onset X-linked recessive neurodegenerative disorder, affecting predominantly men (male to female ratio of 93:1), described in patients from the island of Panay, Philippines, and very rarely in the general population [186, 187]. Dystonia is usually focal at onset, involving the cranio-cervical region with retrocollis, oromandibular dystonia, blepharospasm, and tongue protrusion, and it has a tendency to generalize as the disease progresses [188]. Deep brain stimulation of the internal pallidum (GPi-DBS) has proven effective with long-lasting benefit, especially when performed in the early stage of the disease [189, 190]. Ten years after the onset there is usually a decrease in the severity of dystonia and parkinsonism becomes gradually more prominent [188, 191]. This temporal pattern is quite unique and helps differentiate this condition from other disorders where there is a progression of both dystonic and parkinsonian features.
Parkinsonism-ataxia
Parkinsonism may be part of the clinical presentation of a range of spino-cerebellar ataxias (SCAs) associated with polyglutamine expansion (SCA1,2,3,6,7,8,10,12,17), other SCAs (19,21,22,28), and Fragile-X tremor ataxia syndrome (FXTAS) [192–194]. Characteristic features of the underlying disease (i.e. mental retardation, physiognomic features (long face, low set ears), action tremor in FXTAS; slow saccades in SCA2; pyramidal signs in SCA3; macular degeneration in SCA7; dementia, psychiatric features, chorea in SCA17; seizures in SCA10; cognitive impairment in SCA21; ophthalmoparesis, ptosis, spasticity in SCA28) can sometimes be overlooked by physicians but they are important to guide the diagnosis [195, 196]. SCA1,2,3 and 17 should be suspected in patients presenting with a phenotype suggestive of multiple system atrophy (MSA), as they can present with prominent parkinsonism and dysautonomia, although the classical “hot cross bun” sign on brain MRI, that is characteristic of MSA, is only rarely present in these forms [197].
Parkinsonism, PEO, or optic atrophy
Parkinsonism preceded by development of progressive external complete ophthalmoplegia (e.g., on both horizontal and vertical plane) has been reported in a few cases of subjects carrying mutations in the mitochondrial-related genes
Parkinsonism and spastic paraplegia
Mutations in the
Other SPG syndromes may display parkinsonism as part of the clinical picture, in particular in SPG7 [204–206] and SPG15 [207, 208].
Parkinsonism and intellectual disability
Monogenic forms of early intellectual disability and early or juvenile onset parkinsonism have been described. The combination of early-onset parkinsonism and intellectual disability is a distinctive feature of patients with mutations in
Phenotypes associated with mutations in
A dopamine-responsive parkinsonian syndrome has been reported in patients with mutations in the
Interestingly, a recent review found that impaired cognition or neuropsychiatric disorders are also an early presentation in 92% of patients with EOPD associated with mutations in
Parkinsonism and seizures
Seizures with onset in infancy or childhood are seen in ∼50% [224] of patients with parkinsonism associated with mutations in
HOW TO APPROACH A PATIENT WITH EO PARKINSONISM
Clinical history
A detailed clinical history can provide important information to reach a diagnosis in patients presenting with EO parkinsonism. An initial presentation of pure parkinsonism within the 3rd decade, with no prominent additional clinical manifestations, is more characteristic of monogenic forms of PD (such as PARK-
Certain clinical features are very suggestive of specific forms of EO parkinsonism (Fig. 1). For example, epilepsy can be associated with
Because of the possibility of preventing severe complications when this condition is recognized and promptly treated, genetic analysis of
EOPD is usually slowly progressive. On the contrary, abrupt onset can be suggestive of RODP, viral, immune-mediated, or vascular (stroke, cavernomas or arteriovenous malformations) conditions. While acute forms usually have a more static presentation, the majority of the other forms of EO parkinsonism are progressive, especially the more complicated ones (such as NBIA, Fahr’s disease, and metabolic conditions) compared with pure monogenic forms of parkinsonism (such as PARK-
Non-motor symptoms can also help discriminating among different causes of EO parkinsonism. Symptoms such as dysautonomia, RBDs, hyposmia, which are typically found in patients with iPD, are much rarer, if not absent, in the majority of patients with EOPD. Their prominent presentation can be associated with mutations of the
As part of the assessment of the clinical history, a detailed investigation of patients’ family history is mandatory, especially in the context of genetic forms. If family history suggests an autosomal dominant pattern of inheritance, genetic screening should include
Finally, patients’ ancestry should always be inquired, since some genetic mutations associated with EO parkinsonism present high prevalence in selected ethnic groups. For example, the
Clinical exam
When approaching a patient with EOPD, a comprehensive clinical examination is essential to collect all elements that will aid in the diagnostic process. While the classic onset of iPD is in one body part or hemi-body, the initial presentation of EO parkinsonism observed in forms of NBIA and in Fahr’s disease is often symmetric and involves both halves of the body equally since the first stages. Similarly, acquired parkinsonisms due to exposure to dopamine-blocking agents and to vascular lesions in the basal ganglia typically have a symmetric presentation. Some forms of EO parkinsonism exhibit a characteristic distribution and preferential involvement of the limbs or the cranial district. DYT-
Additional movement disorders and neurological signs can accompany parkinsonism in several cases. Dystonia is characteristic of forms of monogenic EOPD (
Imaging studies
Specific neuroradiological signs may strongly suggest the genetic diagnosis. For example, the T2*/SWI hypointensity of basal ganglia is a key feature of NBIA. The presence of basal ganglia calcifications on CT scan should address the genetic investigation to the FBGC forms. T2-hyperintensity of the putamina and other deep brain nuclei are the most common abnormality seen in Wilson’s disease. T2-hyperintensity of the white matter in the dentate nuclei, cerebellar peduncles, and internal capsule, substantia nigra, globus pallidus, striatum is found in CTX. Conversely, T1-hyperintensity of the basal ganglia can indicate manganese accumulation. Metabolic forms, such as NPC, CHS, and A-T can instead present cortical and cerebellar atrophy, particularly in advanced stages of the disease. A detailed description of specific MRI neuroradiological features associated with each form is reported in Table 1. All forms of Parkinson’s disease and the vast majority of degenerative parkinsonisms are associated with a reduction of the dopaminergic transporter (DaT) at the presynaptic dopaminergic terminal, as shown by dopaminergic functional imaging (e.g., Ioflupane I 123 SPECT) [233]. Fahr’s disease, hereditary spastic paraplegias, and spinocerebellar ataxias can be associated with abnormalities of the presynaptic dopaminergic terminal, more frequently if clinical parkinsonism is present. Differently, parkinsonian disorders associated with brain manganese accumulation (acquired or inherited) and non-degenerative dystonias display a normal Ioflupane I 123 SPECT imaging [234, 235]. A summary of functional imaging features of early-onset parkinsonisms is reported in Supplementary Table 1.
GENETIC COUNSELING
Next generation sequencing (NGS) panels or diagnostic exome sequencing represent the best diagnostic approach in the absence of strong candidate genes. With these techniques, all genes related to EOPD and EO parkinsonism are sequenced simultaneously, which is a cost and time effective method to identify possible mutations. Shortcomings related to this approach include a low sequence efficiency for difficult-to-sequence DNA regions and the risk of missing gross structural alterations. Each variant identified by gene panels should be confirmed by the Sanger approach. Moreover, dosage assay techniques (i.e., Multiplex Ligation-dependent Probe Amplification (MLPA) and qPCR) should always be implemented when analyzing
As an alternative approach, each single gene can also be analyzed by classic Sanger sequencing, a highly reliable but expensive technique, that can be used as a first approach only when few candidate genes are suspected as causative. Sanger sequencing should always be complemented with dosage assay techniques. Pathogenic mutations of
Genetic counseling of patients with EOPD involves the entire family. Whether genetic testing should be offered to asymptomatic family members is still very controversial. Except for genetic forms in which a disease-modifying therapeutic approach exists (e.g., Wilson’s disease) and an early genetic diagnosis can be useful to the patient, no defined guidelines are available for genetic counseling of family members of patients with EO parkinsonism.
The identification of causative genes of EOPD and EO parkinsonism contributed to increase our understanding of the genetic architecture and pathogenic mechanisms of these conditions. These discoveries led to the selection of targeted drugs for specific genetic forms (e.g.,
CONCLUSIONS
EO parkinsonism can be the manifestation of a broad range of conditions. A prompt and accurate diagnosis is crucial for counseling of these patients, prognostic implications, and possibly early treatments to prevent further complications, such as in the case of Wilson’s disease. Specific genetic mutations can be more difficult to distinguish only based on phenotype in case of monogenic forms of EOPD, because of the extensive overlap of their clinical presentation. On the contrary, complicated, and rarer forms related to genetic and metabolic conditions, or metals deposition in the brain can be more easily differentiated, because of the characteristic clinical features, radiological and laboratory findings. Although rare, awareness of the different forms of EO parkinsonism by movement disorder specialists will certainly improve the management and care of these patients.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
