Abstract
Background:
Gait impairments are common in Parkinson’s disease (PD). The pathological mechanisms are complex and not thoroughly elucidated, thus quantitative and objective parameters that closely relate to gait characteristics are critically needed to improve the diagnostic assessments and monitor disease progression. The substantia nigra is a relay structure within basal ganglia brainstem loops that is centrally involved in gait modulation.
Objective:
We tested the hypothesis that quantitative gait biomechanics are related to the microstructural integrity of the substantia nigra and PD-relevant gait abnormalities are independent from bradykinesia-linked speed reductions.
Methods:
Thirty-eight PD patients and 33 age-matched control participants walked on a treadmill at fixed speeds. Gait parameters were fed into a principal component analysis to delineate relevant features. We applied the neurite orientation dispersion and density imaging (NODDI) model on diffusion-weighted MR-images to calculate the free-water content as an advanced marker of microstructural integrity of the substantia nigra and tested its associations with gait parameters.
Results:
Patients showed increased duration of stance phase, load response, pre-swing, and double support time, as well as reduced duration of single support and swing time. Gait rhythmic alterations associated positively with the free-water content in the right substantia nigra in PD, indicating that patients with more severe neurodegeneration extend the duration of stance phase, load response, and pre-swing.
Conclusion:
The results provide evidence that gait alterations are not merely a byproduct of bradykinesia-related reduced walking speed. The data-supported association between free-water and the rhythmic component highlights the potential of substantia nigra microstructure imaging as a measure of gait-dysfunction and disease-progression.
INTRODUCTION
Gait and balance deficits are common phenomena associated with Parkinson’s disease, limit patients’ mobility, can cause falls, and pose a substantial burden on the quality of life of the affected [1–3]. The most noticeable manifestations comprise postural instabilities and freezing of gait, as being captured in the motor part of the Unified Parkinson’s disease Rating Scale (UPDRSIII) as well as the Hoehn and Yahr staging scale (H&Y) [4, 5].
Complementarily to clinical examination, biomechanical analyses can identify gait alterations early in the disease, even in the prodromal stage, and in patients that are not evidently suffering from postural instabilities or freezing [6–8]. Beyond their descriptive value, biomechanical analyses may establish clear links between gait impairments and their underlying pathophysiological mechanisms, provide relevant biomarkers for the diagnosis of Parkinson’s disease, and aid in monitoring therapeutic effects [1, 9]. It is well established that the self-selected and maximal walking speed is reduced, and that biomechanical parameters like step length, step duration, or double support time are altered in people with Parkinson’s disease [10–13]. However, it was shown that walking speed directly affects several gait parameters like step length, swing time, and swing time variability [14]. Thus, in light of the generally reduced walking speed in people with Parkinson’s disease, for a deeper understanding it is crucial to investigate whether differences in gait biomechanics merely relate to speed reductions or constitute genuine pathological mechanisms. Furthermore, most gait studies have investigated gait speeds above 1.0 km/h [10, 14–16]. Since the reduction of inertial forces during very slow walking (0.5 km/h or 1.0 km/h) could increase balancing requirements, investigating the biomechanics at these velocities could provide additional insight into gait impairments. To address these issues, the first aim of our study was to investigate gait biomechanics at fixed and very slow gait velocities.
The pathological mechanisms underlying gait impairments are far from understood. Although bradykinesia and rigidity affect gait [1, 17] it is not clear whether gait impairments derive secondarily or instead constitute a distinct and multidimensional symptom. For example, Levodopa substitution effectively manages bradykinesia, rigidity, and tremor, but exerts mixed results on gait dysfunction: although medication improves some gait parameters like stride velocity and length, other characteristics may deteriorate or remain unaltered [18–20]. Similarly, high-frequency deep brain stimulation of the subthalamic nucleus, a well-established treatment for alleviating motor symptoms, effectively improves bradykinesia and tremor while showing inconsistent outcomes on gait [21, 22]. Interestingly, the substantia nigra pars reticulata has been proposed as an alternative stimulation target to specifically address gait impairments [23, 24]. As one of the output centers of the basal ganglia, the substantia nigra is integrated within the locomotor control systems [25, 26]. The progressive degeneration of dopaminergic neurons in the substantia nigra in people with Parkinson’s disease is reflected in microstructural alterations that can be examined in vivo with diffusion-weighted imaging by measuring the displacement patterns of water molecules [27]. In the substantia nigra of people with Parkinson’s disease, reduced fractional anisotropy and increased mean diffusivity may indicate tissue degeneration [27–29]. However, these parameters are biased in areas of crossing fibers or gray matter because they cannot account for the increased orientation dispersion in such regions [30, 31]. Furthermore, atrophy based partial volume with free-water can have an impact on the diffusion model [32]. More advanced models disentangle free-water from diffusion-weighted-imaging-derived parameters and have shown an increased amount of free water within the substantia nigra in people with Parkinson’s disease [33, 34]. Consequently, free water imaging of the substantia nigra has been suggested as a promising tool for the differential diagnosis and monitoring of over-all disease progression in Parkinson’s disease. However, these studies could not demonstrate a relationship between elevated free-water and clinical motor scores like the UPDRSIII. Biomechanical analyses of specific motor tasks like walking can complement and standardize the clinical assessment and reveal additional important information about motor impairments that are not captured by clinical scores. Considering the substantia nigra being integrated within the gait network, being a potential target for deep brain stimulation in patients exhibiting gait impairments, and demonstrating increased free-water, a pivotal question that has not been investigated is whether the microstructural integrity of the substantia nigra relates to gait impairments. Therefore, our second aim was to investigate the statistical relationship between microstructural characteristics of the substantia nigra and altered gait performance in people with Parkinson’s disease. To this end, we applied the neurite orientation dispersion and density imaging (NODDI) model that is based on advanced imaging sequences to disentangle between orientation dispersion, intracellular volume, and free-water, and thus provides more specific markers of brain tissue [30]. We hypothesized that biomechanical gait parameters are affected by gait speed but nonetheless show marked differences between people with Parkinson’s disease and age-matched healthy control subjects. We further hypothesized that the gait parameters correlate with microstructural properties of the substantia nigra.
MATERIALS AND METHODS
Participants and study protocol
Participants were recruited at the Department of Neurology, University Medical Centre of the Johannes Gutenberg University Mainz. In total, 38 people with Parkinson’s disease and 33 age-matched healthy control subjects participated in our study. The diagnosis of Parkinson’s disease was based on the Movement Disorder Society Clinical Diagnostic Criteria for Parkinson’s disease [35]. Demographics and disease-related information are presented in Table 1. Participants walked on a treadmill, and anatomical and diffusion-weighted MR images were recorded. A movement disorder specialist (S.G.) evaluated the severity of Parkinson’s disease-related symptoms by the Hoehn and Yahr scale [5] and the motor disability by the Movement Disorder Society Unified Parkinson’s Disease Rating Scale part III (UPDRSIII) [4]. The Parkinson’s disease group was tested using their medication according to their usual schedule. All but two newly diagnosed patients received dopamine replacement therapy at the time of study participation. The local ethics committee of the State Medical Association of Rhineland-Palatinate approved this study, which was conducted in accordance to the principles of good clinical practice of the Declaration of Helsinki. All participants gave written informed consent.
Participant demographics and clinical information
Participant demographics and clinical information
PD, people with Parkinson’s disease; HC, healthy controls; N, number of participants; UPDRSIII, motor section of the revised Unified Parkinson’s disease Rating Scale; LEDD, Levodopa equivalent daily dosage (all patients received dopamine replacement therapy at the time of participation) *Mean±standard deviation (range); 1,2Data only available for 34 and 36 PD patients, respectively.
Gait examination
To determine kinematic variables of very slow gait speeds, participants walked on an instrumented treadmill (Zebris, Zebris Medical GmbH, 88316 Isny, Germany). This allowed us to fix the gait speed at very slow velocities (0.5 km/h and 1.0 km/h), effectively controlling the gait speed for both groups. All subjects were given enough time to accustom themselves to the experimental procedure to avoid a protective gait pattern. For safety reasons, one experimenter stood next to the treadmill to prevent potential falls. Each walking trial was between 30 to 60 seconds long. Ground reaction force data were recorded with the software WinFDM-T. This provided seven biomechanical single leg variables reflecting either absolute length or time measures (step length, and step time) or characteristics about the rhythmic organization of the gait cycle (relative duration of stance phase, load response, single support, pre-swing, swing phase), and three bipedal variables (absolute length and time: stride length, stride time, cadence; rhythmic organization: relative duration of double support time).
Imaging data acquisition
Brain imaging was performed in a 3T Siemens TrioTim MRI scanner with a 32-channel head coil (Siemens Healthcare, Erlangen, Germany). Earplugs were used to attenuate scanner noise. Head motion was restricted with a foam pillow and foam padding. We first acquired a 3D T1-weighted magnetization prepared rapid gradient echo (MP-RAGE) sequence (echo time [TE] = 2.52 ms, repetition time [TR] = 1900 ms, inversion time [TI] = 900 ms, flip angle = 9°, matrix size = 256×256, field of view [FOV] = 256×256 mm2, slice thickness = 1 mm, voxel size = 1×1×1 mm3). Following this, we obtained multi-shell DWI data with the following parameters: TE = 111 ms, TR = 10800 ms, flip angle = 90°, matrix size = 128×128, FOV = 2304×2304 mm2, slice thickness = 2 mm, voxel size = 2×2×2 mm3. Diffusion-weighting of b = 900 s/mm2, b = 1800 s/mm2, and b = 2700 s/mm2 was applied in 30 unique directions, each. For each b-value we additionally acquired six non diffusion-weighted volumes (b0-volumes). Finally, one additional b0-volume was acquired in the anterior-posterior, and one in the posterior-anterior direction.
Image analysis
Pre-processing of diffusion-weighted scans was performed with the MRTrix3 toolbox (https://www.mrtrix.org/) [36]. In brief, we applied the functions dwidenoise to correct spatially varying thermal noise [37] and mrdegibbs to remove Gibbs ringing artefacts [38]. We corrected susceptibility induced (EPI) distortions, motion artefacts, eddy currents, and spatial distortions using dwifslpreproc.
The NODDI model was fitted to the preprocessed diffusion-weighted images by accelerated microstructure imaging via convex optimization (AMICO, Version 1.2.7, https://github.com/daducci/AMICO) [43]. This provided orientation dispersion (OD), intracellular volume fraction (vic) and isotropic volume fraction (viso) as markers of microstructural integrity. While OD describes the angular variation of neurite orientation, vic characterizes the neurite density, and viso reflects isotropic, unrestricted water movement corresponding to the free-water compartment. We linearly aligned the first b0-image to the T1-weighted anatomical image in SPM12 (https://www.fil.ion.ucl.ac.uk/spm/software/spm12/) [44]. The co-registration matrix was then applied to OD, vic, and viso maps. T1-weighted preprocessing involved segmentation of gray matter, white matter, and cerebrospinal fluid, and calculation of the transformation from T1-weighted into MNI152-standard-space. NODDI images were then normalized to standard-space in SPM12 using the T1-weighted transform. All images were spatially resampled to 1 mm isotropic resolution. Substantia nigra volume was calculated based on the normalized T1-weighted images.
Statistical analysis
If not stated otherwise, all statistical analyses were performed using IBM SPSS Statistics, Version 22.0 (SPSS, Chicago, Illinois, USA). The significance threshold was set to p < 0.05 (two-tailed). Effect sizes were calculated and interpreted as Cohen’s d (small: d
s
= 0.2, medium: d
s
= 0.5, and large: d
s
= 0.8) or partial eta squared (small:
Demographic characteristics of people with Parkinson’s disease and healthy subjects were compared by Fisher’s exact test for categorical data or independent samples t-tests for non-categorical data. We performed two-factorial mixed model ANOVAs with “speed” as within-subject-factor (0.5 km/h vs. 1.0 km/h) and “group” as between-subject factor (people with Parkinson’s disease vs. healthy controls) to investigate speed effects and group differences for all gait parameters. We applied FDR-correction (at p < 0.05) to accommodate for the number of tests. We then conducted a principal component analysis (PCA) to reduce the dimensionality of the data [46]. Because stance phase and swing phase together make up 100% of the normalized gait cycle, meaning that stance phase is defined as 100% minus swing phase, these two parameters provide redundant information. Therefore, we excluded the stance phase from the PCA. Most meaningful components were selected based on a cumulative percentage threshold of 80% [47]. The first two principal components contributed to a cumulative percentage of total variation of 80%. To identify group differences of the resulting principal components, we performed independent samples t-tests. To evaluate the discriminative value of the two principal components, we calculated a logistic regression (stepwise inclusion, “group” as the dependent variable, and age and sex as covariates) and a receiver-operator-curve analysis for binary classification.
We then tested whether linear relationships between microstructural properties of the substantia nigra and each of the two principal components differed between people with Parkinson’s disease and healthy controls. To this end, we performed separate general linear models with the NODDI parameters as dependent variables and the gait components and groups as independent variables. Age and gender were added as covariables. For statistical inference we performed permutation tests using the randomise tool in FSL6 (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/FSL) [48]. This consisted of 5000 permutations with the threshold free cluster enhancement option to control for multiple comparisons. Voxels within the left and right substantia nigra were segmented according to a mask provided by Ilinsky et al. [49]. We then extracted the individual mean values of voxels within the identified cluster to perform partial correlations with the principal components, corrected for age and gender.
We further investigated whether the gait components and NODDI parameters showing significant interactions statistically related to the patients’ clinical status. To this end, partial correlation analyses between these variables (viso and principal component two) and the H&Y- and UPDRSIII-scores, adjusted for age, sex, and substantia nigra-volume were performed. We further subdivided the Parkinson’s disease group by H&Y stages and performed ANCOVAs with viso and principal component two as dependent variables, H&Y as fixed factor (four levels), and age and sex as covariates. Substantia nigra volume was included as a covariate to control for potential volume-related confounders.
Data availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
RESULTS
In total, we included data of 38 people with Parkinson’s disease and 33 age-matched healthy control subjects. Demographics and disease related information are summarized in Table 1. There were no statistically significant differences in age (t = 0.677, df = 69, p = 0.5) or sex (Fisher’s exact test p = 0.239).
Gait examination
We first examined the speed effects and group differences between people with Parkinson’s disease and HC for all specific gait parameters (Table 2). All parameters were significantly affected by gait speed (all p < 0.0001, corrected, all
Repeated-measures ANOVAs of the gait variables
*Values are presented as mean (± standard deviation); PD, people with Parkinson’s disease; HC, healthy controls; F, F-statistic; p, p-value; p (FDR), false discovery rate corrected p-value;
PCA resulted in two principal components (Fig. 1, Supplementary Table 1). These components cumulatively explained 82.7% of the variance. The first component accounted for 41.9% of the variance and could be assigned mainly to variables describing length and time characteristics (step length, stride length, step time, stride time, and cadence; absolute weights between 0.891 and 0.982). For simplicity, we term the first principal component “length and time component”. The second principal component accounted for 40.8% of the variance and mainly resembled rhythmic characteristics of the gait cycle (load response, single support, pre-swing, swing-phase; absolute weights between 0.697 and 0.995). For this component, increased gait speed (i.e., 1.0 km/h) provided higher weights. We term this principal component “rhythmic component”. In people with Parkinson’s disease, the length and time component was significantly reduced (t = –2.298, df = 69, p = 0.025; 95% CI = [–0.99, –0.07], d s = 0.55), while the rhythmic component was significantly increased (t = 4.775, df = 69, p < 0.0001, 95% CI = [0.58, 1.41], d s = 1.13) (Fig. 2A, B). The stepwise logistic regression with age and gender as covariates included the rhythmic component in the first step, and had a classification accuracy of 73.2%. Adding the distance and time component in a second step increased the accuracy to 78.9%. Besides, the area under the curve for classification was 0.79 and 0.66 for the rhythmic component and the distance and time component, respectively (Fig. 2D).
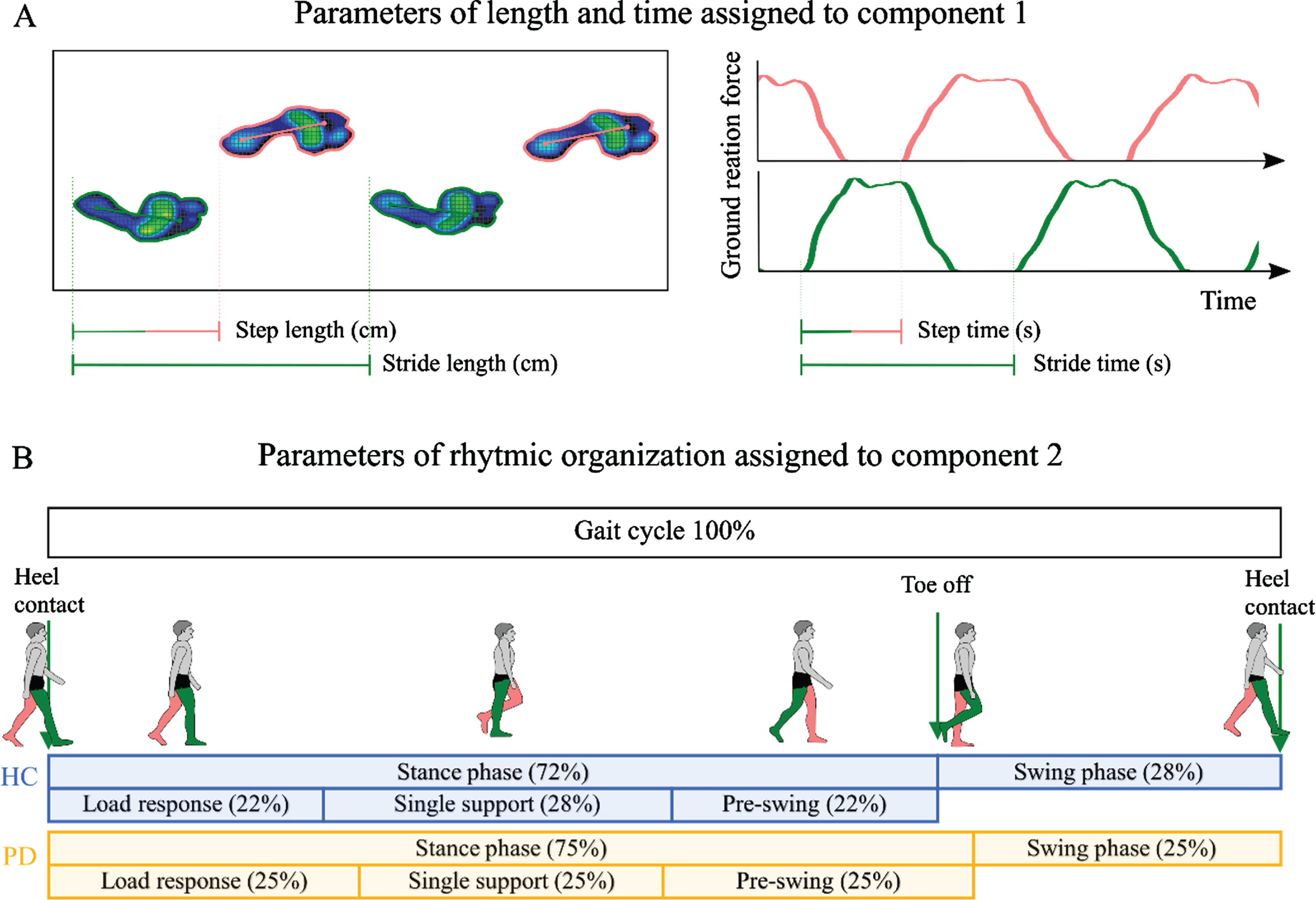
Gait parameters and their assignment to principal components. A) The left box shows four steps on the treadmill (left foot in pink; right foot in green). Step length is calculated as the distance between the initial contact of one foot and the initial contact of the opposite foot. Stride length is the distance between successive points of initial contact of the same foot. The graphs on the right show the ground reaction forces plotted against time. Step time is the time between the initial contact of one foot and the initial contact of the opposite foot. Stride time is the time between successive initial contacts of the same foot. Step length, stride length, step time, stride time, and cadence (steps per minute, not shown) were assigned to principal component 1 according to their weights (all > 0.89). B) Parameters of the rhythmic organization of gait were assigned to principal component 2. The gait cycle, here presented for the right leg (green) consists of the stance and swing phase. The stance phase begins with the initial heel contact and ends when the toe leaves the ground. It can be subdivided into load response, single support, and pre-swing phase. Swing phase is the time during which the leg swings forward (i.e. the time between “toe off” and “heel contact”). Data shown for walking with 1.0 km/h. Duration of stance phase, load response, and pre-swing are significantly increased in people with Parkinson’s disease. Duration of single support and swing phase are significantly reduced (all p < 0.005, corrected). HC, healthy controls (blue); PD, people with Parkinson’s disease (orange).
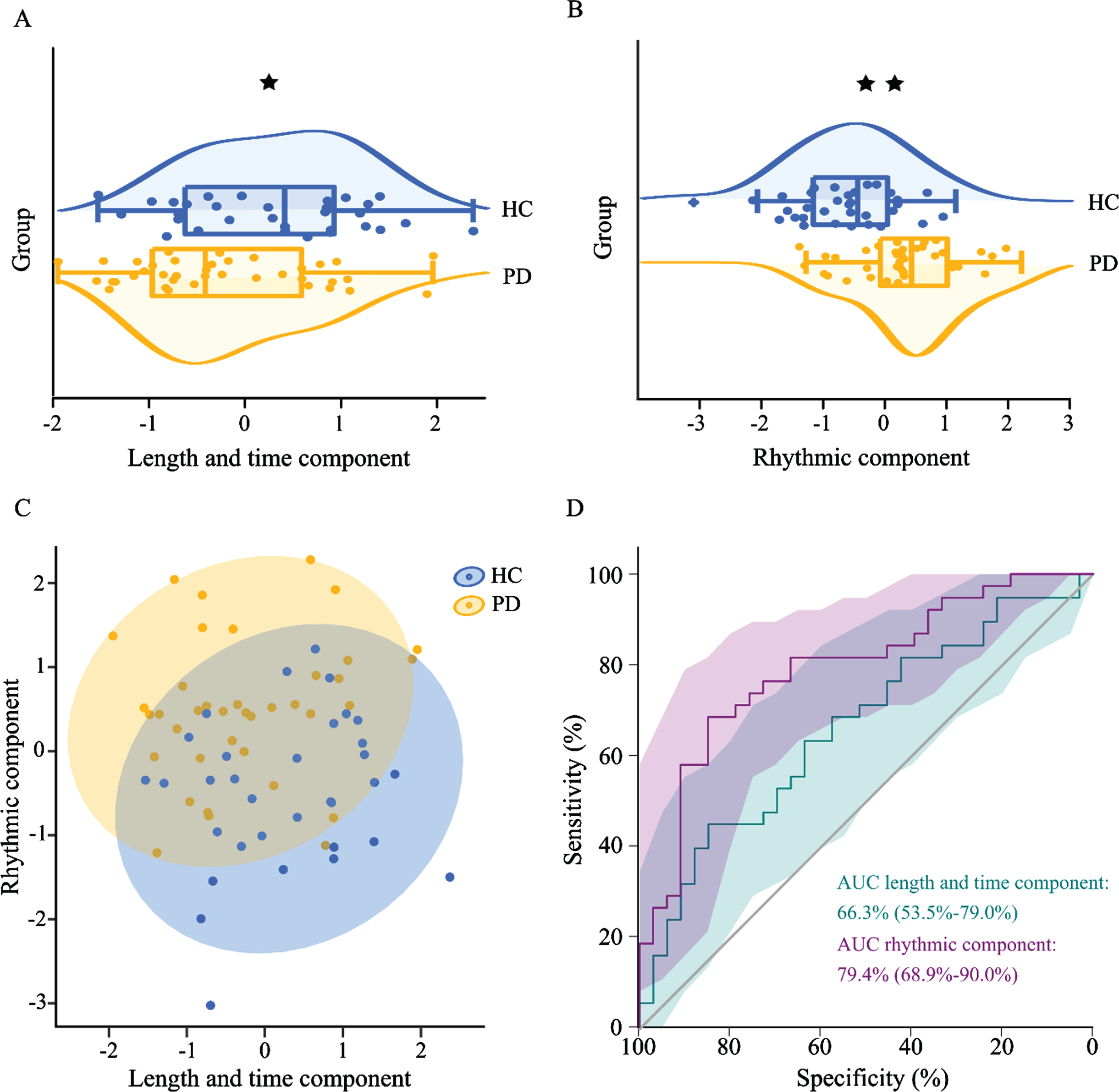
Altered gait characteristics in people with Parkinson’s disease. A, B) Significant differences between the two groups for the length and time component (p = 0.025) and the rhythmic component (p < 0.001). C) Scatterplot of the length and time component and the rhythmic component. D) Classification accuracy for the two components (AUC length and time component: 0.663, p = 0.019; AUC rhythmic component: 0.794, p < 0.001). Logistic regression including both components had an accuracy of 78.9%. HC: healthy controls (blue); PD: people with Parkinson’s disease (orange); *p < 0.05; **p < 0.001.
Correlations between free-water content and the rhythmic component
There were significantly different associations between viso and the rhythmic component among the two participant groups in one cluster containing 19 voxels (T-scores between 2.82 and 3.58; p < 0.05). This cluster was located at the lateral boundary of the substantia nigra (Center of gravity: x = 12; y = –22; z = –10; Fig. 3; Supplementary Table 2). To verify that the results were not driven by extreme values, we excluded two people with Parkinson’s disease and one healthy subject due to being outliers and performed partial correlation analyses. These showed that in people with Parkinson’s disease, high values of viso related to increased values of the rhythmic component (r = 0.48, p = 0.005), whereas the relationship was negative in healthy controls (r = –0.6, p < 0.001). There were no significantly different associations between any of the NODDI parameters with the distance and time component.
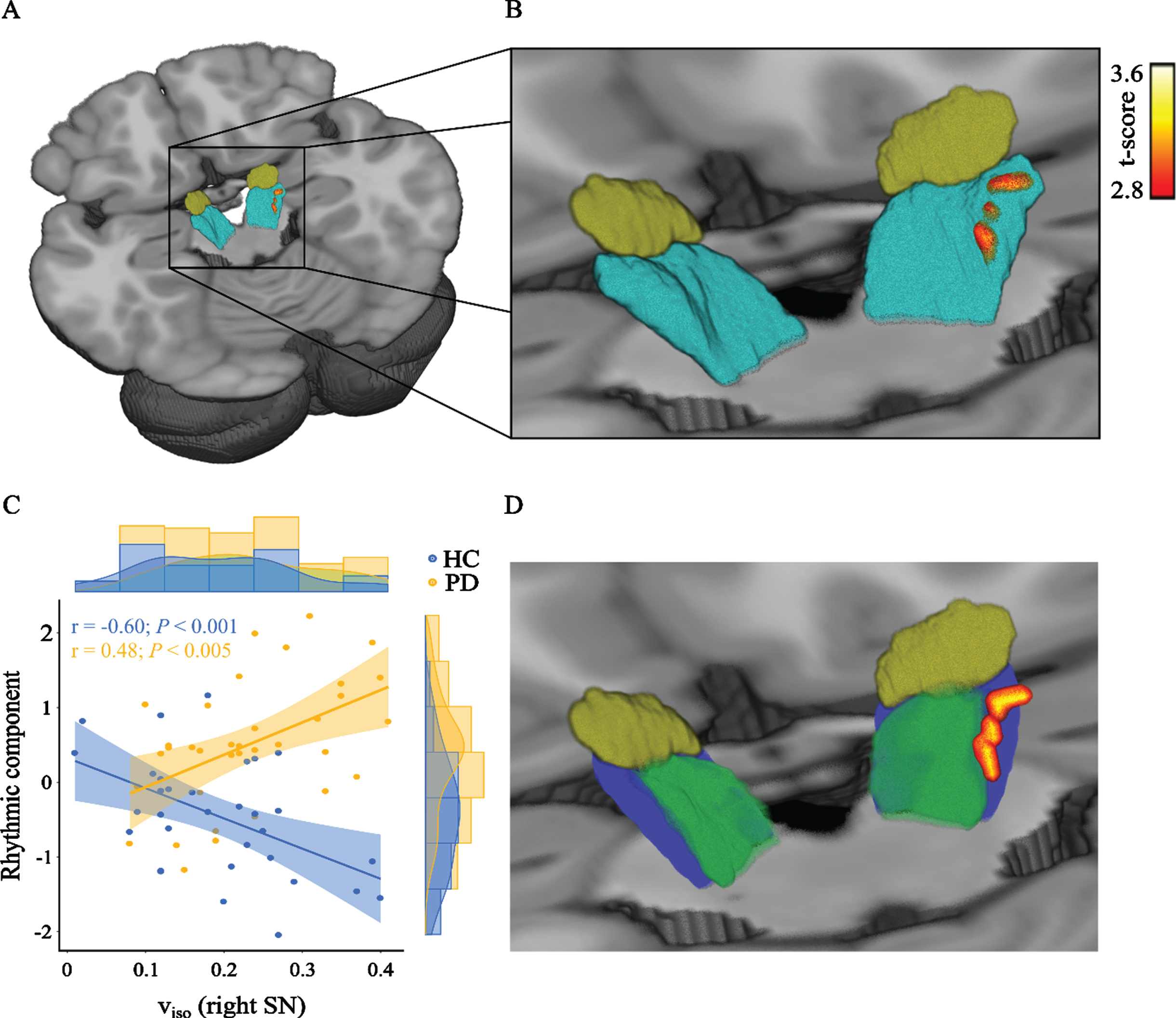
Associations between free-water and the rhythmic component are different between people with Parkinson’s disease and healthy controls. A) The figure shows the cluster containing significant voxels within the right substantia nigra (teal). For illustration the cluster was smoothed. For orientation, the subthalamic nucleus (brown) is also shown. B) Same as (A), but with a magnified field of view. C) Scatterplot including the regression line and its 95% confidence interval between the gait rhythm component and the mean viso within that cluster. The partial correlations were corrected for age and gender. D) The significant voxels shown in relation to the substantia nigra pars compacta (green) and the substantia nigra pars reticulata (blue), as masked by the CIT168 Reinforcement Learning Atlas [81]. The subthalamic nucleus (brown) is shown for orientation. HC, healthy controls (blue); PD, people with Parkinson’s disease (orange); viso, isotropic volume fraction; SN, substantia nigra.
To visualize the white matter pathways that are traversing the identified cluster within the substantia nigra, we additionally overlaid a mask composed of the significant voxels on a population-average diffusion MRI Template from the human connectome project [50]. This evidenced that tracts passing the cluster are highly connected to the primary motor cortex, primary sensory cortex, supplementary motor area, and somatosensory cortex (Fig. 4).
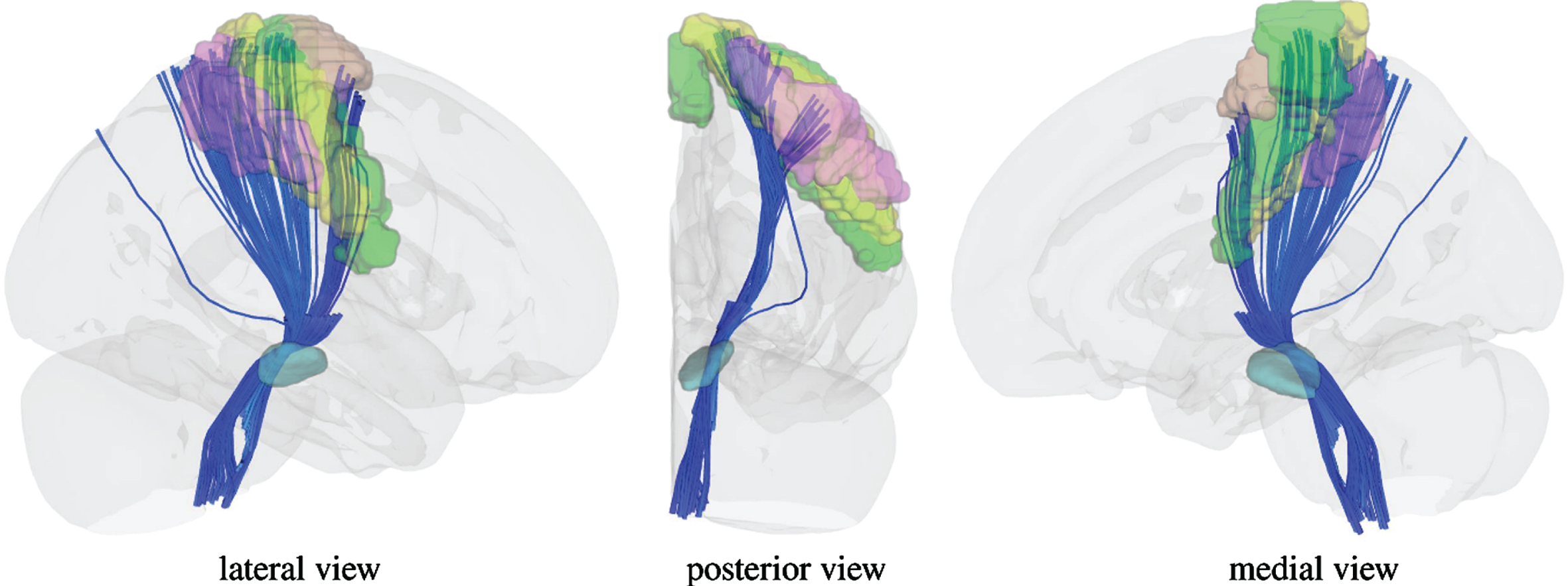
White matter pathways traversing the identified cluster within the substantia nigra. White matter pathways are connecting the most medial part of the primary motor cortex (green), primary sensory cortex (yellow), supplementary motor area (brown), and somatosensory cortex (purple). To demonstrate the connections we used the Human Connectome Project Multi-Modal Parcellation 1.0 atlas by Glasser & Coalson et al. [82].
Partial correlations showed that there were significant linear correlations between viso and H&Y (r = 0.43; p = 0.016; Supplementary Figure 1A) and the rhythmic component and H&Y (r = 0.46, p = 0.008; Supplementary Figure 1B). No statistically significant correlations were attested with the UPDRSIII (all p > 0.05).
To test whether gait parameters and viso are influenced by the disease stage we further subdivided the Parkinson’s disease group by H&Y stages and performed ANCOVAs (Fig. 5). We found significant differences between the four subgroups for both viso (F = 2.991, p = 0.048,
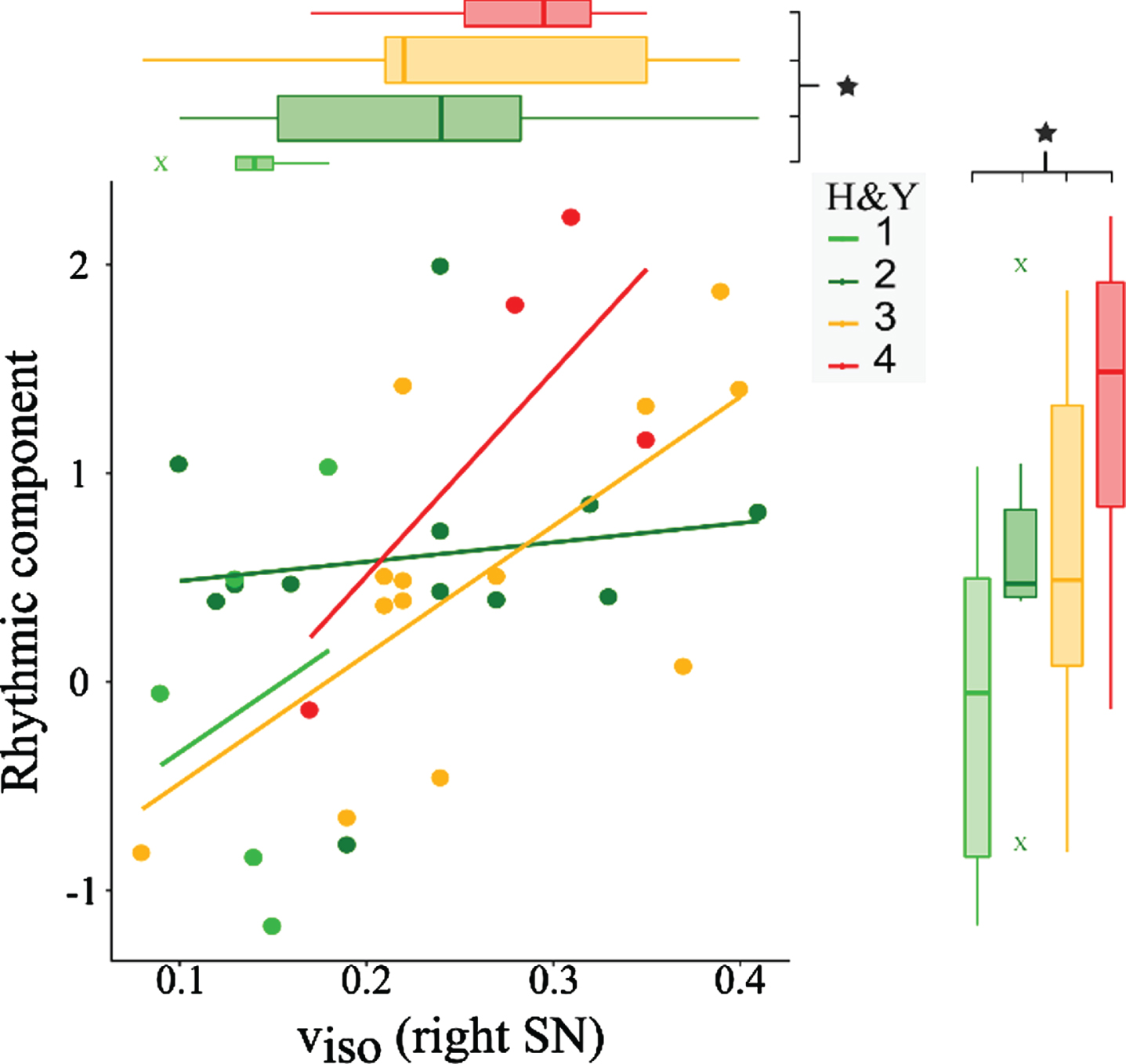
Effects of disease stage on gait and substantia nigra free-water. ANCOVAs with H&Y stage (light green = 1; dark green = 2; orange = 3; red = 4) as independent factor and age, gender, and volume as covariates show that the gait rhythm component (p = 0.009) and the free-water within the substantia nigra (p = 0.048) are significantly increased for patients with later disease stage. The scatter plot shows individual values of the rhythmic component and viso, including the regression lines for each subgroup. viso, isotropic volume fraction; SN, substantia nigra.
DISCUSSION
The present work links altered gait biomechanics in Parkinson’s disease to microstructural properties within the substantia nigra. We demonstrated that modifying gait speed affected all biomechanical gait parameters. Nonetheless, fixing the gait speed across the two groups enabled us to provide strengthened evidence in support of the hypothesis that Parkinson’s disease affects biomechanical gait characteristics. Among all gait parameters we identified two principal components that reflected either absolute distance and time characteristics or the rhythmic dimension of gait. Furthermore, we found statistically significantly different relationships between the rhythmic component and the free-water content across the two groups: while people with Parkinson’s disease displayed a positive correlation, healthy control participants presented a negative correlation. Moreover, we found evidence that both the rhythmic component and the free-water content within the substantia nigra can be associated with the disease state. Taken together, these findings suggest that the interactions between substantia nigra microstructure and gait function are affected in people with Parkinson’s disease, highlighting the potential of the substantia nigra as a target for future therapeutical interventions.
Altered gait biomechanics of very slow walking in Parkinson’s disease
Our first aim was to investigate gait characteristics during treadmill walking when gait speed is very slow and fixed between people with Parkinson’s disease and healthy controls. It was recently shown that gait speed affects the arm swing in people with Parkinson’s disease [51]. Gait speed also affects swing time and swing time variability [14]. However, in these studies gait speed was chosen based on a percentage of self-selected walking speed and therefore heterogeneous, was not fixed for both groups, and substantially faster than in our study. While other treadmill studies prevalently investigated self-selected or maximal gait speed [10–13], we fixed the gait speed to avoid any confounding effects that may originate from differences in maximal or preferred speed. We first found that gait speed indeed affected all parameters of the lower extremities. Nonetheless, we further observed increased duration of double support, load response, and pre-swing time in people with Parkinson’s disease. In addition, we found reduced duration of single support and swing phase. Step length, step time, stride length, stride time, and cadence, all reflecting length and time characteristics, were not significantly different between the two groups when analyzed separately. This indicates to reconsider the strategies for correcting or controlling gait speed in future studies that investigate gait function to prevent a potential overestimation of disease effects. On the other hand, our findings corroborate that especially gait alterations in the rhythm domain are not merely a byproduct of reduced walking speed but likely resemble independent, disease-related disturbances that are observable even when gait speed is fixed.
The self-selected speed during treadmill walking is most often chosen above 1.0 km/h in people with Parkinson’s disease and further increased in healthy subjects [10, 14–16]. Slower self-selected walking speed might pose a compensational mechanism to prevent falls in Parkinson’s disease [16]. Compared to natural walking, gait speeds of 0.5 km/h or 1.0 km/h are very slow and have so far been widely neglected when investigating people with Parkinson’s disease. To modulate walking speed, one can manipulate either step length or cadence, or a combination of both. Both step length and cadence are governed by a power law to find the most economical combination, even at very slow speeds [52]. Accordingly, healthy subjects should reduce both step length and cadence in compliance with the power law when walking at a very slow gait speed. In addition, the displacement of the lower extremities caused by the moving belt of the treadmill provides proprioceptive feedback which might ameliorate biomechanical gait alterations in Parkinson’s disease. Taken together, this might cause length and time parameters to converge in both groups, potentially accounting for the lack of observable differences. Interestingly, all these variables were assigned to the first component of our PCA with very high weights. In contrast to individual length and time variables, this component was significantly reduced in people with Parkinson’s disease. This indicates that combining the common gait characteristics within multiple length and time parameters may eliminate variance that does not contribute to explaining group differences and suggests that PCA can considerably increase the statistical power of the gait analysis. Further, while step length, step time, stride length, and stride time had positive weights, cadence was represented by negative weights. Thus, this component may additionally reveal differences in the relation between stride length and cadence, which could be altered in Parkinson’s disease [6].
Regarding the rhythmic organization of gait cycles, we found an increased duration of the stance phase, load response, pre-swing, and double support time, while the duration of swing phase and single support time were reduced in people with Parkinson’s disease. Very slow gait speed may reduce the stability provided by inertial forces, provoking enhanced balancing demands on the musculoskeletal system. Furthermore, reduced mediolateral forces indicate the need for increased lateral balance control [53]. Augmenting this, trunk rigidity and reduced arm swing potentially limit the ability to transfer weight or shift the center of mass for people with Parkinson’s disease [54, 55]. Increasing the time where both feet are in contact with the ground (i.e. the relative duration of load response, pre-swing, and double support) may facilitate utilization of proprioceptive feedback or active control mechanisms to meet the balancing demands [52]. Furthermore, rigidity could play an additional role for rhythmic alterations [17], and an attenuated force production rate [56] could induce diminished propelling of the legs during stepping, which may shorten the time of the swing phase. All the variables related to rhythm were represented by high loadings in the rhythmic component of the PCA. Due to the positive weights, high values of this component represented prolonged load response, pre-swing, and double support. Contrarily, high values reflected reductions in single support and swing phase due to the negative weights. Importantly, the values of this component were enhanced in people with Parkinson’s disease, reflecting increases in load response, pre-swing, and double support and reductions in single support and swing phase. Thus, we were able to condense most of rhythmic parameters’ individual information in one relevant component.
The rhythmic organization of gait is associated with free-water content in the substantia nigra
Because the substantia nigra is involved in the gait network [25] and constitutes an alternative target for deep brain stimulation [24], we investigated the associations between its microstructural characteristics and gait impairments. We found that extracellular water content in a lateral cluster of the right substantia nigra correlated positively with the rhythm component in people with Parkinson’s disease, while the relationship was negative in healthy controls. Our results thus indicate that patients with elevated free-water increase the duration of load response, pre-swing, and double support time at the expense of reduced swing phase and single support time.
In people with Parkinson’s disease, the posterior part of the substantia nigra is characterized by elevated free-water, which represents pathological alterations occurring in the process of neurodegeneration [32–34]. Corroborating the relevance of the substantia nigra microstructural integrity for gait functionality, free-water imaging in the posterior part of the substantia nigra contributes to predicting posture and gait scores of the UPDRSIII [57]. The cluster identified by our analysis is located at the lateral part of the substantia nigra, potentially overlapping with the pars reticulata. The pars reticulata is an integral part of the gait network as it receives input from the striatum, nucleus accumbens, and subthalamic nucleus, and provides tonically active GABAergic projections towards lower brainstem regions involving the pedunculopontine nucleus and mesencephalic locomotor region [25, 58]. Within the pars reticulata, enhanced degeneration might cause perturbations in the balance of inhibitory and excitatory input, potentially altering its activity. Indeed, the substantia nigra pars reticulata is overactive in Parkinson’s disease and exerts exaggerated inhibition on the pedunculopontine nucleus and the mesencephalic locomotor region [25, 26]. Our results indicate that neurodegeneration affects especially the rhythmic organization of the gait cycle. This is particularly intriguing because it has been shown that repetitive stimulation of the pars reticulata modulates the walking patterns in rats [59]. Together, this suggests a continual modulatory role of the substantia nigra pars reticulata during walking. In Parkinson’s disease, progressive neurodegeneration may impair gait function, which eventually causes alterations in walking patterns.
However, due to possible partial volume effects and the relatively small size of the two compartments of the substantia nigra, we cannot completely rule out that the identified cluster overlaps, to some extent, with the pars compacta. Dopaminergic neurons within this region predominantly project along the nigrostriatal pathway to the dorsal striatum [60], and the loss of such dopaminergic neurons is the pathological hallmark in PD [61]. Given their role within the basal ganglia, concomitant extensive functional alterations in wide-spread networks have been shown previously [62–65]. Therefore, it is possible that degeneration and the associated reduction of dopaminergic signaling affects communication in gait-related brain circuits, which could ultimately manifest as biomechanical gait alterations.
Since our results emerged from correlative testing it is important to also consider an alternative interpretation. It might as well be possible that impairments of the rhythmic organization of gait are not directly originating from neurodegeneration in the substantia nigra, but instead affect the microstructural integrity of this region. In that case, specifically targeting the rhythmic component with training and exercise interventions might initiate protective mechanisms that slow down degeneration [66]. Because our results indicate that the rhythmic organization of gait is modulated by walking speed, interventions that systematically vary gait speed may have a positive impact. Indeed, treadmill training at various velocities (between 80% and 130% of over-ground walking speed) has been shown to promote alterations of functional connectivity that potentially reflect neural plasticity within cortical and subcortical regions [67]. Treadmill training also affects the basal ganglia dopaminergic systems both in humans and rats [68, 69]. Whether exercise interventions promote protective mechanisms that affect the microstructural integrity of the substantia nigra poses a highly interesting question worth pursuing. Therefore, future studies should utilize diffusion weighted imaging and the NODDI model to investigate if training and exercise targeting the rhythmic organization of gait also protect the microstructural integrity of the substantia nigra.
In addition to the pathophysiological implications, the identified association may help unraveling the mixed results regarding the substantia nigra as a target for deep brain stimulation. So far, studies have shown efficacy for the combined stimulation of the subthalamic nucleus and substantia nigra in freezing of gait, but no improvement has been reported for exclusive substantia nigra stimulation [23, 71]. Concerning kinematic gait parameters, stimulation of the substantia nigra (alone) improved anticipatory postural adjustments and swing time asymmetry [24, 70]. In this regard, deep brain stimulation might interfere with pathological activity in distinct regions within the substantia nigra to target information processing of interconnected areas like the mesencephalic locomotor region or pedunculopontine nucleus. This could specifically affect the rhythmic organization of gait, as demonstrated for swing time symmetry [24]. Another important aspect pertains to the question of whether microstructural integrity could be used as a predictor for the efficacy of substantia nigra deep brain stimulation. Intriguingly, in our sample, the average free-water within the identified voxels was not significantly different between the two groups. These disparities may be explained by the lateral location of the cluster, which does not necessarily overlap with the regions of interest investigated in previous studies [32–34]. Nonetheless, our results demonstrate increased extracellular free-water in some patients, and these patients concomitantly displayed increased gait impairments in the rhythmic organization of the gait cycle. It could be possible that deep brain stimulation might not be an optimal treatment for these patients. Thus, it seems crucial to investigate in future studies if microstructural integrity affects the efficacy of substantia nigra deep brain stimulation and could provide a reliable biomarker for the identification of responders or non-responders.
Receiving GABAergic signaling from the substantia nigra pars reticulata, the pedunculopontine nucleus is another relevant region for gait functionality. As such, this area has been considered an additional target for deep brain stimulation to alleviate axial motor deficits in Parkinson’s disease [72, 73]. Interestingly, microstructural characteristics in this region provide an independent marker for predicting future postural instability and gait difficulties [74]. A potential relationship between diffusion-weighted-imaging-derived parameters of the pedunculopontine nucleus and biomechanical parameters of gait has, however, not been investigated so far and should thus be a target for future studies, as well.
Clinical relevance
We investigated the discriminative value of treadmill walking at very slow speeds. While logistic regression analysis including the rhythmic component provided an accuracy of 73.2%, the inclusion of the length and time component improved the accuracy to 78.9%. This demonstrates that condensing the information of a high number of gait variables to only two components can be a valuable approach to facilitate gait analysis. Especially the rhythmic component may represent a valuable marker to aid the clinical diagnosis, even when gait velocity is fixed. The accuracy of the clinical diagnosis differentiating Parkinson’s disease from other neurological diseases is only 74% when performed by non-experts and 80% by movement disorder specialists [75]. As an objective and easy-to-perform analysis, the application of treadmill walking in the clinical routine could facilitate the diagnosis, especially if biomechanical alterations are detected before gait impairments are clinically manifest. In this regard, we found elevations of the rhythmic component with increasing disease stage, as demonstrated in the subgroup as well as correlation analyses. Importantly, our results show that gait alterations are not only emerging at or after stage 3, but are already present earlier. Gait symptoms progress in the course of Parkinson’s disease and are thought to evolve more rapidly than other features [76]. Thus, biomechanical analyses may be more sensitive to progressive and subclinical alterations than the H&Y, further corroborating their diagnostic value.
We further demonstrate a relationship between the free-water within the substantia nigra and H&Y stages, indicative of the relationship between neurodegeneration and disease progression. Similar to the associations between gait and disease stage, these results highlight the potential of free-water imaging as a useful tool to monitor disease progression or the effects of future disease-modifying trials, as suggested in recent studies [32, 33].
As a limitation, we did not test maximal speed, general mobility, or balance [77, 78]. In contrast to people with Parkinson’s disease, healthy subjects with increased free-water had reduced values of the rhythmic component, representing increased duration of swing phase and single support time, and decreased duration of load response, pre-swing, and double support time. In healthy subjects, reduced integrity of the substantia nigra thus lead to different behavior than in people with Parkinson’s disease. Missing external criteria like mobility or balance scores prevents us from drawing specific conclusions about the functional implication of reduced values of the rhythmic component in healthy subjects. Future studies should investigate the meaning of non-disease-related rhythmic alterations. For example, healthy subjects with increased atrophy within the substantia nigra might adapt the rhythmic characteristics of gait by utilizing pathways that are impaired in people with Parkinson’s disease.
Interestingly, the associations between the free-water content and the rhythmic gait characteristics were lateralized in the right hemisphere. These findings could potentially relate to handedness or asymmetries in the clinical features. Out of our 38 patients, three were left-handed, while none of the 33 healthy subjects were left-handed. Thus, with the available data we could not test any potential effects of handedness and it is necessary to perform similar analyses with a higher number of left-handed subjects to evaluate if microstructural alterations in the substantia nigra of the non-dominant hemisphere (i.e. the left hemisphere for left-handers) relate to rhythmic gait alterations. Regarding asymmetries in the expression of clinical features, additional sub-group analyses dividing patients with right-sided symptom dominance (N = 23) and patients with left-sided symptom dominance (N = 12) did not show any significant differences in the gait or free-water parameters, thus we could not provide any evidence for the potential relevance of asymmetries for our findings.
Another limitation is that we performed the clinical examination and investigated gait function exclusively in the dopaminergic “ON” state. Since both gait biomechanics as well as the clinical evaluation are affected by the presence of medication effects [18–20, 79], the results provided here might be over- or underestimating pathological effects in the absence of dopaminergic replacement therapy. However, we intended to capture the gait biomechanics in a state that patients most likely experience throughout the day and thus performed the study without interfering with their usual medication schedule. Nonetheless, it would be interesting to repeat the gait analysis in the dopaminergic “OFF” state to investigate the effects of medication in addition to drawing a more disease specific picture.
As a final limitation, PCA does not provide the ability to perform a cross validation test like the leave one out cross validation or k-fold cross validation used in other machine learning techniques like support vector machines (see for example Di Lazzaro et al., 2020 [80]). Therefore, additional studies with a similar treadmill paradigm like the one used here are needed to replicate the results of our PCA-analysis.
CONCLUSION
The current study provides evidence that alterations in gait biomechanics involving the rhythmic organization and length and time characteristics of gait are not merely a consequence of reduced gait speed in people with Parkinson’s disease but reflect genuine pathological characteristics. Nonetheless, it is crucial to correct for gait speed variations in order to prevent a potential overestimation in future studies. When doing so, especially the rhythmic organization of the gait cycle seems to be highly relevant and may represent a valuable marker to aid the clinical diagnosis. The association between free-water in the substantia nigra and the rhythmic gait component corroborates the role of the substantia nigra for gait impairments in Parkinson’s disease.
Footnotes
ACKNOWLEDGMENTS
We thank the participants for their time and effort.
The authors would like to acknowledge the support of FTN/TransMed, the German Research Foundation (DFG; CRC-TR-128), and the Boehringer Ingelheim Fonds (BIF-03).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
