Abstract
Background
Freezing of gait (FoG) is a debilitating symptom of Parkinson's disease (PD) with limited response to dopaminergic medication and subthalamic deep brain stimulation (STN-DBS). Substantia nigra pars reticulata (SNr) stimulation could improve FoG.
Objective
To analyze the effect of combined STN-SNr stimulation at different frequencies on FoG.
Methods
We performed a double-blind, cross-over, randomized pilot trial involving STN-DBS treated PD patients with FoG. Participants received: high-frequency (HF) STN-DBS (S), combined HF-STN and SNr stimulation (C1), and combined HF-STN and low-frequency (LF) SNr stimulation (C2), for one month each. The primary endpoint was the score change in the New-Freezing-of-Gait-Questionnaire (NFOG-Q). Secondary analyses were performed on motor complications, axial symptoms, daily living activities, psychiatric symptoms, sleep, and patient preference.
Results
Fifteen patients received at least one combined stimulation. No significant difference in NFOG-Q scores was found between S, C1, and C2; one-third of patients showed a clinically significant improvement (≥8 points) with combined stimulations. Motor complications improved significantly with C1 and C2 (C1-S: 3.6 ± 3.8 vs. 4.9 ± 3.8, p = 0.046; C2-S: 2.7 ± 3.1 vs. 4.9 ± 3.8, p = 0.005). 80% of patients preferred the combined STN-SNr stimulation while blinded. All adverse events were manageable.
Conclusions
Our study did not prove a statistically significant improvement in NFOG-Q with STN-SNr stimulation; however, one-third of patients experienced a clinically meaningful FoG improvement, and the majority preferred to maintain STN-SNr stimulation. STN-SNr stimulation was both safe and effective in addressing motor complications and improving sleep quality, highlighting the importance of further exploration into the effects of combined STN-SNr stimulation.
Plain Language Summary
We aimed to identify novel therapeutic approaches for freezing of gait (FoG), a disabling symptom of Parkinson's disease (PD) associated with risk of falls and poor life quality. FoG affects 31 up to 87% of PD patients and can be difficult to manage with standard PD medications. FoG may remain unchanged or even worsen in patients treated with Deep Brain Stimulation (DBS). We focused on DBS-treated PD patients and stimulated simultaneously two areas of the brain: the subthalamic nucleus (STN), the typical target for treating PD motor symptoms, and the substantia nigra pars reticulata (SNr). 15 PD patients with FoG received different types of brain stimulation for one month each: STN stimulation alone and combined STN + SNr stimulation at two different stimulation frequencies. We measured improvement of FoG (using a validated questionnaire) and other PD symptoms (including motor fluctuations, anxiety and depression, and sleep quality), as well as safety and tolerability of the combined STN + SNr stimulation. Finally, we observed the patient preference for the type of stimulation provided. We found no statistically significant change of FoG scores with STN + SNr stimulation, but one-third of patients experienced a clinically meaningful reduction in FoG. STN + SNr stimulations helped improve motor fluctuations and dyskinesia. Moreover, 80% of the patients preferred the combined stimulation over the standard STN stimulation. We believe this data suggest that combined STN + SNr stimulation could be a valuable area for further research aiming to improve FoG and can be safely attempted in clinical practice in DBS-treated patients with disabling FoG.
Keywords
Introduction
Freezing of gait (FoG) is a frequent symptom in Parkinson's disease (PD), with prevalence ranging from 31 to 87%, and incidence increasing by 10% each year. 1 FoG is associated with poor quality of life, increased morbidity, and higher risk of falls.2–6 The therapeutic management of FoG is challenging: it can initially respond to dopaminergic medications, being present only in the Off therapeutic condition (‘Off-FoG’), but as disease progresses it tends to become unresponsive to dopaminergic therapies, and sometimes may be worsened or even induced by an increase in dopamine levels.7,8 Deep brain stimulation (DBS), either targeting the subthalamic nucleus (STN) or the globus pallidus internus (GPi), reliably improves parkinsonian motor symptoms; however, predicting FoG response to DBS can be challenging.9–11 An improvement in patients with a pre-surgical Off-FoG is expected, although it may remain unchanged or even worsen in some patients as an effect of surgery, chronic stimulation, or disease progression.9,12–14 In fact, individual patients’ characteristics, electrode position, stimulation parameters and disease progression may all have a role in determining the efficacy of DBS on FoG. 9
One proposed treatment for FoG unresponsive to standard high frequency (HF) STN-DBS (i.e., 130 Hz) or dopaminergic medication, is low frequency (LF) stimulation at 60 or 80 Hz, adapting the total electrical energy delivered (TEED).12,15 While this strategy has shown positive results regarding axial symptoms, including FoG, its use may be limited by the worse control of cardinal motor symptoms.4,6,15 Since a defective motor processing of descending nigropontine projections to spinal motor neurons appears to be associated to the emergence of gait disorders, a new stimulation paradigm based on the co-stimulation of the substantia nigra pars reticulata (SNr) and STN has been proposed with promising results.16–18 Another study with a small sample size analyzed the effects of LF-SNr or HF-STN stimulation alone or in combination: four out of six patients obtained a marked improvement of FoG when LF-SNr and HF-STN were combined. 4
To date, the evidence on SNr stimulation effect on FoG remains scarce. More importantly, no study has ever compared data between LF- and HF-SNr stimulation versus standard STN stimulation, and data concerning control of cardinal motor symptoms and best stimulation frequency remain unknown.
In this context, we conducted a two-center, randomized, cross-over, double-blind pilot trial to analyze the effect on FoG of STN-DBS stimulation combined to a stimulation of the SNr at HF (C1) or LF (C2) vs. the standard HF STN stimulation (S) at 130 Hz in a cohort of PD patients with a therapy-resistant FoG.
Secondary objectives were: to evaluate the safety and tolerability of each combined stimulation, to analyze its effect on motor complications, axial motor symptoms, activity of daily living (ADLs), psychiatric symptoms and sleep, and to assess patient's preference on the type of stimulation, according to a patient-centered outcome evaluation.
Methods
Study design and participants
Two centers (Centre Expert Parkinson's Disease, Toulouse, France and Movement Disorders Center of the University of Turin, Turin, Italy) participated in the present study (ClinicalTrials.gov number: NCT05415774). Study design and protocol were discussed, defined, and harmonized between the two centers before IRB submission. The French center received funds for a monocentric study, which was the sole reason for the separate study coordination. In both centers, the study was conducted between July 2022 and May 2023.
Patients were consecutively included in the study according to the following criteria: a) diagnosis of PD defined by the Movement Disorder Society (MDS) Criteria 19 ; b) bilateral STN-DBS implantation (either with Medtronic Activa™ PC or Boston Vercise™ devices) at least 6 months prior to inclusion; c) oral medication and stimulation parameters stable in the last month before study inclusion; d) FoG not optimally managed by dopaminergic therapy and standard DBS-STN settings, defined by a New Freezing of Gait Questionnaire (NFOG-Q), Part II Item II ≥ 2, or Part III Item VII ≥ 2 20 ; d) Mini-Mental State Examination (MMSE) score ≥ 25 21 ; e) Hoehn and Yahr (H&Y) stage ≤ 3 in the Med ON/Stim On condition 22 ; f) ventral contact of electrodes implanted in the SNr bilaterally, according to an imaging-based analysis of volume of tissue activated (VTA) models; g) ability to provide informed consent.
Patients with a diagnosis of atypical parkinsonism, severe FoG before DBS (i.e., MDS-sponsored revision of the Unified Parkinson's Disease Rating Scale (UPDRS) item 2.13 in Med On ≥2 or MDS-UPDRS item 3.11 ≥ 2 in Med On), or patients under legal protection were excluded from the study.
Local institutional review boards provided ethics approval (IRB approval numbers: RC31/21/0609 for Toulouse center and 0119241 - 352/2022 for Turin center). Trials were conducted in accordance with the Declaration of Helsinki and International Conference on Harmonization Good clinical Practice Guidelines. All patients provided written informed consent. The clinical trial report has been written in accordance with the Consolidated Standards of Reporting Trials (CONSORT) statement (see Supplemental Table 1 and Supplemental Figure 1).
Randomization and blinding
As this is a cross-over study, the sequence of different stimulation parameters has been randomized. Each patient received three types of stimulation (S, C1, and C2; Supplemental Figure 2), making 6 possibilities for the administration order of the treatments in agreement with a William's design with 3 periods and 6 sequences. 23 A computer program generated the randomization list, with randomization independently performed in each center (see Supplemental Table 2).
One investigator per center (MF and CAA), in charge of changing DBS parameter settings, was unblinded to stimulation parameters and aware of the randomization sequence. Another investigator per center (CL, SG) oversaw clinical assessments, being blinded to stimulation parameters and randomization sequence, as well as the patient.
Procedures
All patients received in a different order for a total study period of 3 months: 1) one month of HF STN-DBS (“S”); 2) one month of combined HF stimulation of the STN and SNr at 130 Hz (“C1”); 3) one month of combined HF stimulation of the STN and LF stimulation of the SNr at 60 Hz (“C2”). A 4-week stimulation period was considered an appropriate timeframe to overcome any carryover effects and to properly assess the clinical and subjective effects of the new stimulation parameters.12,24
The TEED, calculated according to the formula proposed by Koss and colleagues, 25 remained stable in all the three stimulation settings. Intensity of SNr stimulation was initially set at the same intensity of STN stimulation and then adapted according to the occurrence of adverse events (AEs). Specifically, intensity was set 0.1 V (or 0.1 mA) below the minimum value determining pyramidal tract AEs or any other stimulation-induced AEs reported by patients and persisting > 2 min.
The detailed methods to obtain the 3 stimulations were reported in Supplemental Figure 3.
If, over the course of the month, the patient did not tolerate the stimulation setting, he/she was switched to the next stimulation setting in agreement with the randomization order, without being excluded from the trial. The decision for a premature stimulation setting switch was made in agreement with the blinded investigator and the patient during an additional visit. In case of aggravation of parkinsonian motor symptoms or fluctuations, the blinded clinician was allowed to increase the treatment of maximum 100 mg of daily levodopa. In case of dyskinesia, a decrement of maximum 100 mg of daily levodopa was allowed. Likewise, intensity adjustment of -/+ 0.5 V (or mA) were allowed.
Upon experiencing all three stimulation settings, while still blinded, patients selected the one they had most appreciated. Following this choice, the blinding was removed.
Endpoints
Primary endpoint was the change in mean scores of the NFOG-Q at the conclusion of each period of combined C1 and C2 stimulation, individually examined, in comparison to the S stimulation.
Secondary endpoints were: the number and type of AEs collected at each visit; the differences between S-stimulation and C1- and C2-stimulation regarding the following outcomes: a) The Timed up and go test (TUG): b) MDS-UPDRS part III, considering both total score and axial score (i.e., the sum of the items 3.1, 3.9, 3.10, 3.11 and 3.12, corresponding to speech, arising from the chair, gait, FoG, and postural stability) 26 ; c) MDS-UPDRS part IV for motor complications (total score and sum of the items 4.1 and 4.2 for dyskinesia and 4.3 and 4.4 for motor fluctuations) 26 ; d) MDS-UPDRS part I and II for non-motor and motor symptoms impact on ADLs; e) Hospital Anxiety and Depression Scale-D (HAD-S) 27 ; f) the PD Sleep scale-2 (PDSS-2). 28 Finally, we considered the number of patients who chose to keep one of the combined stimulations (C1 or C2) at the end of the study for chronic therapeutic stimulation.
AEs were classified as follows: i) AEs inducing voltage minimal adjustment over the months, without requiring a stimulation parameter switching; ii) AEs related to global stimulation parameter switching; iii) AEs not related to stimulation adaptation.
Patients’ assessment at baseline and after one month of each stimulation setting (S, C1 and C2) was detailed in figure S2. Of note, after study inclusion and before randomization (baseline visit), MDS-UPDRS part III and TUG were assessed in four different conditions: MedOff/StimOn condition, MedOff/StimOff, MedON/StimOff and MedON/StimOn. All other assessments were performed in the MedON/StimOn condition.
The analysis of the VTA to check for the coverage of SNr with the stimulation settings C1 and C2 is reported in the Supplemental Material.
Statistical analysis
We performed a pilot study to compare the effect of combined STN + HF or LF SNr stimulation vs. standard STN stimulation on FoG, with the aim of acquiring high-quality information, laying the groundwork for the initiation of a larger study based on the outcomes. Considering the sample size of previous similar studies,16,29 the number of patients treated with STN-DBS in the two centers each year (20/center/year), and the low number of patients developing non-responsive FoG after surgery (about 10% at 1 year),13,30 we opted as feasible and meaningful the inclusion of a minimum of 6 patients per center for a pilot trial.
All statistical analyses have been performed on the modified intention-to-treat (mITT) population (all randomized patients who have had at least one stimulation setting, meeting the inclusion criteria, with at least one post-period evaluation after one month).
Descriptive statistics was used for the entire recruited population with percentage for qualitative variables and mean with standard deviation (SD), range (minimum and maximum) or median with interquartile range (IQR) for quantitative variables. To compare the score change between the three stimulation settings (S, C1 and C2) a linear mixed effect regression model was performed with change score as the dependent variable, period (visit), stimulation (S, C1, C2) and sequence (interaction between period and stimulation) as fixed effects and a random “subject” effect. The model was adjusted on center (France, Italy).
To compare the percentage of SNr coverage by the VTA in C1 and C2 stimulation settings, as well as baseline characteristics of patients who experienced a NFOG-Q score improvement ≥ 8 points (responders) compared to the rest of the sample (non-responders), 31 we used a Fischer and Mann-Whitney test, respectively. Likely, baseline variables of responders vs. non-responders were compared by means of qui-square test or Wilcoxon Mann-Whitney test, as appropriate.
All the analyses were performed with SAS® Version 9.4., using two-tailed p-values with a level of significance of 0.05. Trial results are presented considering pooled data from the two centers.
Results
A total of 16 PD patients have been recruited and randomized in the two centers (6 at the Turin center and 10 at the Toulouse center). One patient did not tolerate either C1 or C2 stimulation parameters due to a severe resting tremor suddenly appearing few seconds after parameter changes and was excluded from efficacy data analysis (mITT population).
Thus, data are presented for the 15 patients (13 males and 2 females) who received at least one combined stimulation setting. Out of the 15 included patients, 13 (86.6%) underwent all three stimulation settings (S, C1, C2), one patient underwent a single stimulation setting (C2), and another underwent two stimulation settings (S and C2).
At baseline, the mean age of the cohort was 66 ± 5.8 years, with a PD duration of 16.8 ± 6.3 years. The average FoG duration was 4.3 ± 3.8 years. All demographic and clinical baseline characteristics are summarized in Table 1.
Demographic and clinical data of patients at baseline.

Results are reported as mean ± SD. Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS); Medication (Med); Stimulation (Stim).
Axial symptoms were calculated by the sum of the following MDS-UPDRS items: 3.1, 3.9, 3.10, 3.11, 3.12.
* The presence and type FoG here is based on the levodopa challenge test evaluation, considering item 3.11 of the MDS-UPDRS. The only patient with Off- and On-FoG received an item 3.11 score of “2” in Off and “1”’ in On.
** For sub-items 3.10 and 3.11 pre-DBS in Med ON and Med OFF we have data from 12 out of 15 patients.
Freezing of gait
The change of the NFOG-Q score showed no significant difference between S and C1 (16.8 ± 7.9 vs. 16.6 ± 9.0, respectively; p = 0.64) or between S and C2 (16.8 ± 7.9 vs. 15.7 ± 8.5, respectively; p = 0.19) (Table 2, Figure 1).
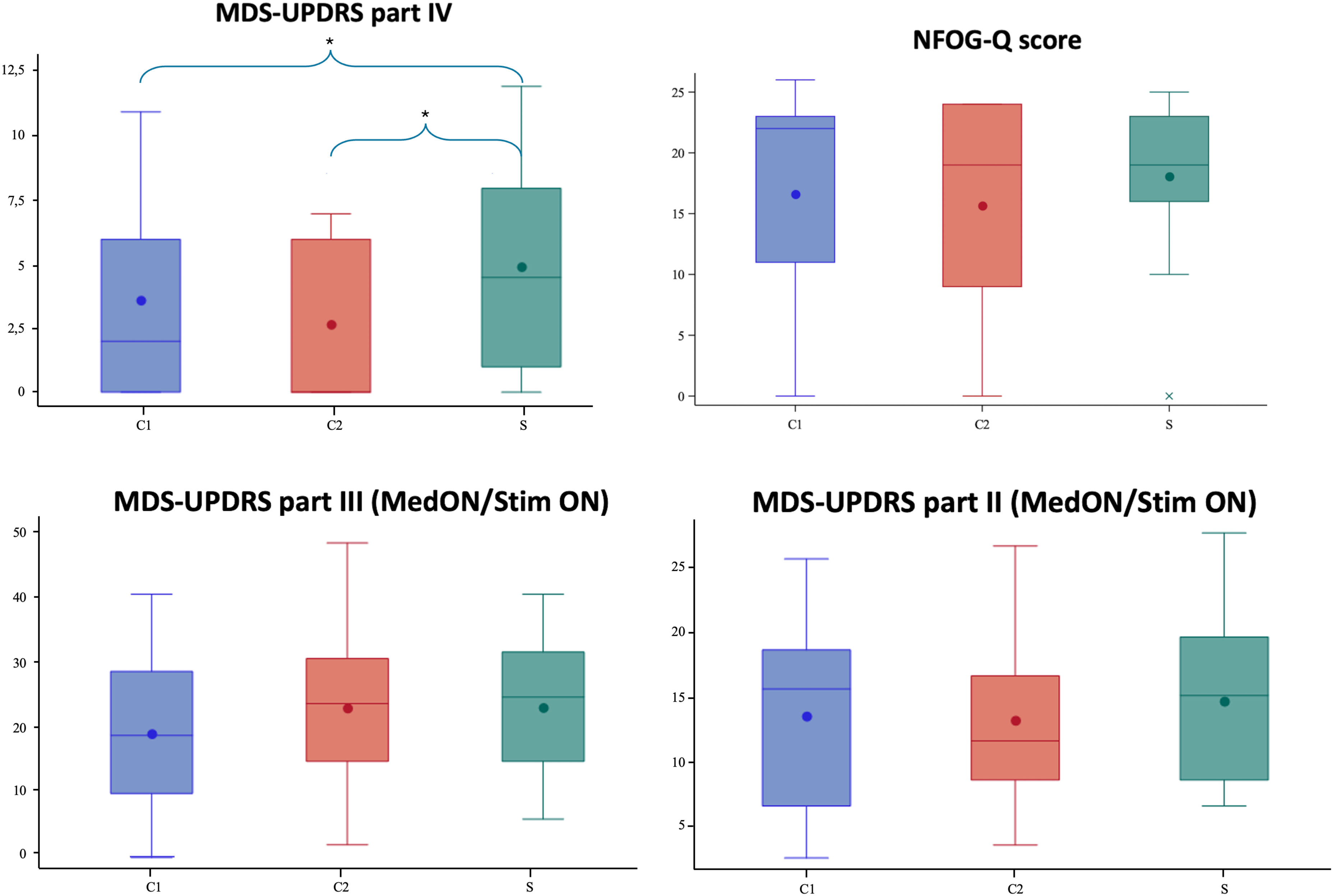
Clinical outcomes after one moth of the three stimulation settings: C1, C2, and S. Results are presented as box plots; New Freezing of Gait Questionnaire (NFOG-Q); Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS); Stimulation of the subthalamic nucleus (S); stimulation of the subthalamic nucleus and of the substantia nigra at high frequency (C1); stimulation of the subthalamic nucleus and of the substantia nigra at low frequency (C2)
Comparison of score changes in primary and secondary outcomes between the combined STN-SNr stimulation and standard STN stimulation.
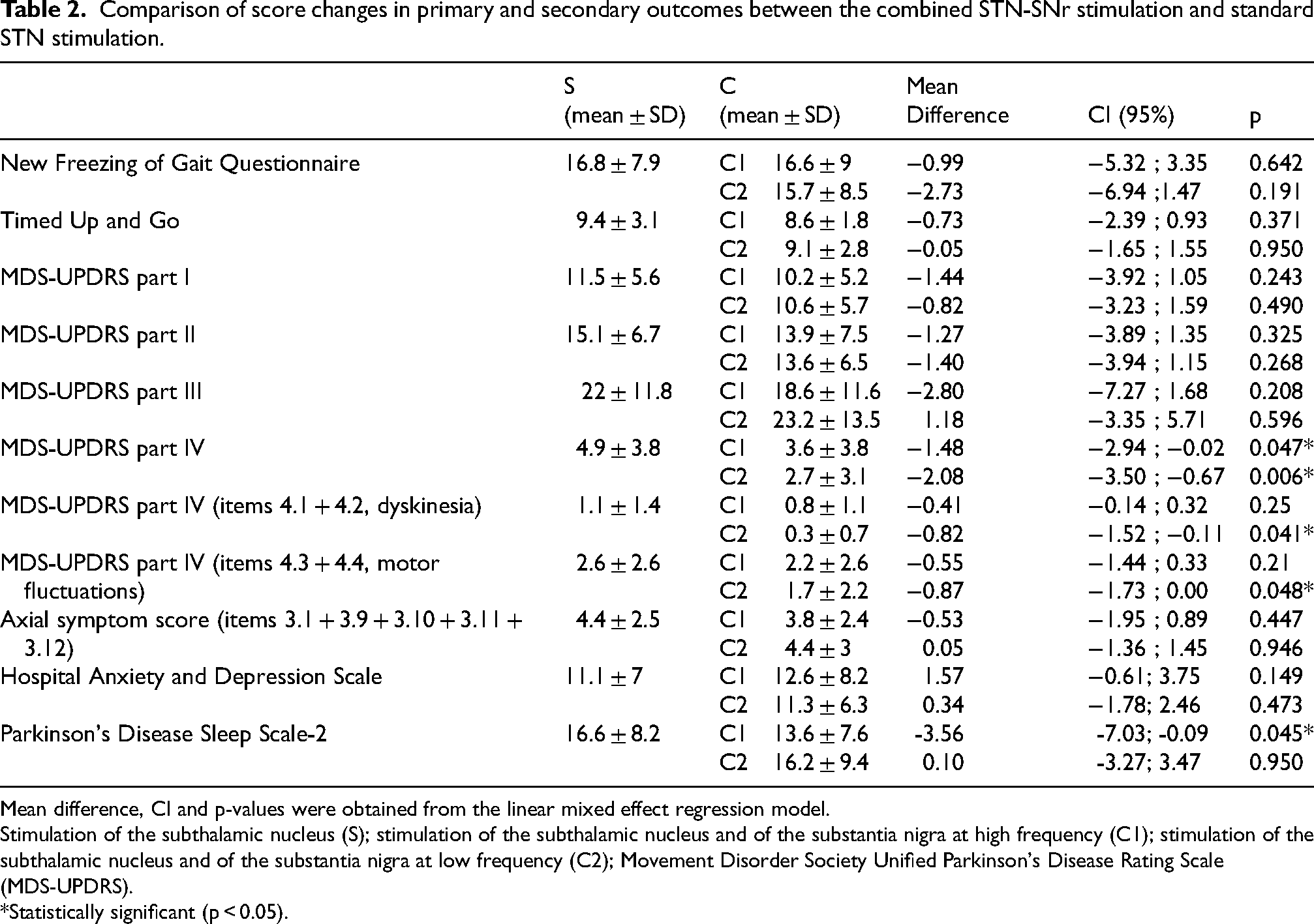
Mean difference, CI and p-values were obtained from the linear mixed effect regression model.
Stimulation of the subthalamic nucleus (S); stimulation of the subthalamic nucleus and of the substantia nigra at high frequency (C1); stimulation of the subthalamic nucleus and of the substantia nigra at low frequency (C2); Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS).
*Statistically significant (p < 0.05).
One-third of patients (n = 5, “responders”) showed a NFOG-Q score improvement ≥ 8 points at C1 (n = 2) or at C2 (n = 3), which is considered a clinically significant change for this scale. 31
Age, sex, disease duration, DBS duration, FoG duration, LEDD, and MDS-UPDRS III and IV were not significantly different between responders vs. not-responders.
Motor complications, motor and axial symptoms
We found a significant improvement of motor complications (MDS-UPDRS part IV) with the combined STN-SNr stimulation, both comparing C1 vs. S (3.6 ± 3.8 vs 4.9 ± 3.8; p = 0.047) and C2 vs. S (2.7 ± 3.1 vs 4.9 ± 3.8; p = 0.006) (Table 2, Figure 2). Only dyskinesia (sum of items 4.1 and 4.2) presented a significant improvement on the C2 vs. S (0.3 ± 0.7 vs. 1.1 ± 1.4; p = 0.041), while motor fluctuations (sum of items 4.3 and 4.4) was not significatively different across conditions (Table 2).
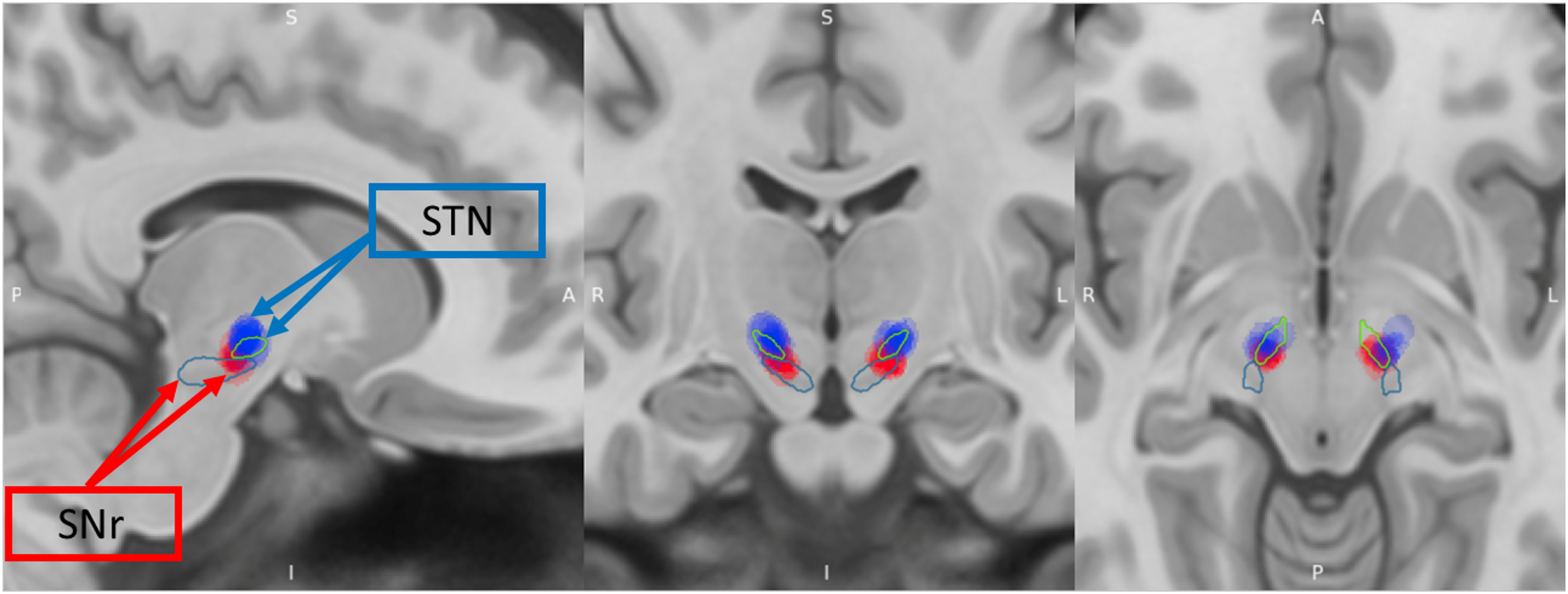
Volume of tissue activated of the subthalamic nucleus and substantia nigra pars reticulata for all patient included in the study in the stimulation setting C1/C2.
No significative differences at the MDS-UPDRS part III total/axial score and TUG time were found between the three conditions (Table 2, Figure 2).
Activities of daily living and non-motor symptoms
ADLs (MDS-UPDRS part II) did not show a statistical difference between C1 vs. S (13.9 ± 7.5 vs 15.1 ± 6.7; p = 0.32) and C2 vs. S (13.6 ± 6.5 vs 15.1 ± 6.7; p = 0.26).
Sleep quality (PDSS-2 scale) showed an improvement in C1 vs. S (13.6 ± 7.6 and 16.6 ± 8.2; p = 0.04) but not in C2 vs. S (16.2 ± 9.4 and 16.6 ± 8.2; p = 0.95) (Table 2). No significant differences across conditions were observed for MDS-UPDRS part I score and HADS score (Table 2).
Safety
We observed a total of 66 AEs. None of them was severe and all were reversible. Twenty-five (37.88%) AEs led to stimulation configuration switches (3 patients during S, 4 patients during C1, 3 patients during C2 stimulation), sixteen (24.24%) required a change of the stimulation intensity (5 patients during C2 stimulation), and twenty-five (37.88%) did not require any intervention and were recorded only after the post-period evaluation (Table 3, Supplemental Table 3). The most frequently reported AEs leading to a switch in stimulation settings included increased falls and Off-time for the S stimulation (n = 4), increased dyskinesia, falls, dysarthria for the C1 stimulation (n = 7) and increased Off-time for C2 (n = 2). AEs necessitating an intensity change (either reduction or increase) were exclusively reported during C2 stimulation, with the most common being increased FoG, dyskinesia, and psychiatric symptoms such as impulsivity, agitation, depression or apathy, and anxiety (Table 3, Supplemental Table 3). Only one patient required a revision of dopaminergic therapy with a reduction of 50 mg of daily levodopa intake due to dyskinesia increase during C1 stimulation.
Adverse events possibly related to stimulation changes.
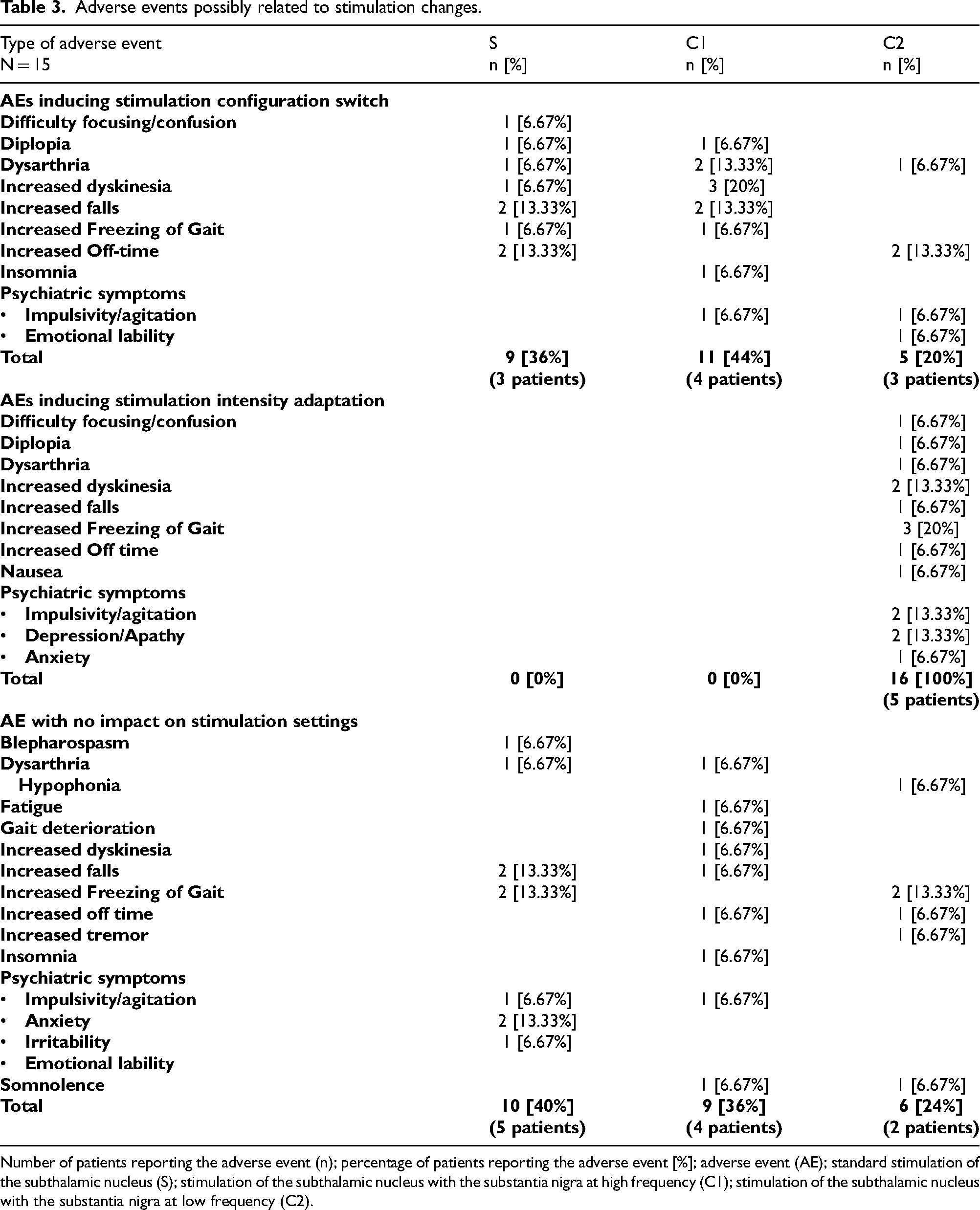
Number of patients reporting the adverse event (n); percentage of patients reporting the adverse event [%]; adverse event (AE); standard stimulation of the subthalamic nucleus (S); stimulation of the subthalamic nucleus with the substantia nigra at high frequency (C1); stimulation of the subthalamic nucleus with the substantia nigra at low frequency (C2).
VTA analysis and open-label observation
SNr VTA coverage data are summarized in Supplemental Table 4.
Upon the conclusion of the study, 80% of patients (n = 12) opted to maintain the combined stimulation of the STN-SNr, with six patients at LF (C2) and six patients at HF (C1). The average improvement in NFOG-Q score for patients maintaining the combined STN-SNr stimulation was 27% compared with the score reported in the S stimulation. All 5 “responders” (i.e., patients experiencing an improvement of ≥ 8 points in the NFOG-Q) maintained the combined stimulation.
Three months after the completion of the double-blind study phase (open-label assessment), all patients were still treated with the stimulation parameters chosen at the end of the study.
Patients maintaining the combined STN-SNr stimulation showed significantly higher percentage of SNr VTA coverage than those maintaining standard STN stimulation after study completion (5.95 ± 3.06 and 2.44 ± 1.04, respectively; p = 0.004) (Supplemental Table 4); conversely SNr coverage did not significantly differ among patients who had a ≥ 8 points score improvement at the NFOG-Q vs. the ones who did not (p = 0.34).
Discussion
In this pilot, randomized, double-blind trial aiming to assess the effect of combined HF-STN with HF- or LF-SNr stimulation to improve disabling FoG in PD patients, we found no statistically significant difference in NFOG-Q scores. Nonetheless, using the combined STN-SNr stimulation, one third of patients reached an improvement ≥8 points at the NFOG-Q, which is considered a clinically significant improvement. 31 Motor complications and sleep quality showed benefit from the combined stimulation when compared to the standard stimulation, with both C1 (i.e., HF-STN and HF-SNr) and C2 (i.e., HF-STN and LF-SNr) improving MDS-UPDRS-IV and only C1 improving PDSS2. Finally, 80% of patients included in the trial chose a combined STN-SNr stimulation after study completion for chronic therapeutic stimulation in clinical practice.
As previously suggested,16,31–34 concomitant stimulation of the SNr was globally safe and well tolerated, with all AEs manageable and reversible, often requiring only minor adjustments in stimulation intensity. Interestingly, the evaluation of AEs seems to suggest that concomitant HF-SNr stimulation is better tolerated than LF-SNr stimulation, considering that 8 patients in LF-SNr required stimulation adjustments or early interruption versus 4 patients in HF-SNr (see Supplemental Table 3).
The rationale behind SNr targeting relies on SNr sending non-dopaminergic projection to brainstem structures involved in locomotion; consequently, the SNr stimulation holds the potential to modulate locomotor integration.35–37 Animal research demonstrated GABAergic inhibitory output from the SNr to the pedunculopontine nucleus (PPN).38–40 Given the efferent monosynaptic GABAergic transmission from SNr to the PPN, 38 HF stimulation at the level of SNr might attenuate an over-inhibitory drive and modulate mesencephalic pathways and locomotor integration.4,16,22,41–43 In particular, the dorsolateral part of the SNr has mainly GABAergic and cholinergic projection neurons, and HF stimulation of the SNr demonstrated to suppress the pathological overactivity of the PPN. 44 Consistently, HF-SNr stimulation improved forelimb akinesia in a rat model of PD and recording of basal ganglia activity showed task-related STN and SNr single-unit activity changes during gait-like movements.45,46 This evidence indicates a nucleus-specific motor- and attentional-related change of basal ganglia firing rates, pointing toward an integrative role of attentional and motor aspects of gait, and further supporting the potential clinical benefit of a combined STN-SNr stimulation. 46
A few clinical studies on SNr stimulation on PD patients have been performed so far. A cross-over, double-blind, randomized controlled clinical trial on 12 PD patients showed FoG improvement with the combined stimulation of STN and SNr at HF, using a scoring based on a single course assessment; similar to our study, a substantial proportion of patients (10 out of 12) expressed a preference for continuing the combined stimulation over standard STN stimulation. 16
Another study analyzed the combination of LF stimulation of the ventral area of the STN (to target SNr) and HF stimulation of the STN in 6 patients which were chosen to undergo STN-DBS for the presence of dopaminergic unresponsive gait disorders, obtaining an improvement in gait disorders in four patients. 4 The use of LF stimulation of the SNr is further supported by intra-operative findings from another study, which demonstrates that even LF stimulation of the SNr significantly decreases cell firing and modulates synaptic plasticity. 47
Additionally, the role of SNr stimulation in improving axial symptoms has been recently proposed in a cross-over study on 25 PD patients. The Authors used a complex “dual-frequency” programming (interleave-interlink, IL-IL), which consisted in the application of two asynchronous LF stimulations in two adjacent STN areas: the peripheral areas received LF stimulation, optimal to reduce gait and balance issues, while the central area received HF stimulation resulting from the overlap of the two LF stimulation, in order to maintain the control of appendicular symptoms. 12 The analysis of the VTA in the IL-IL programming showed a ventral extension into the SNr, proposed as one of the mechanisms leading to the improvement in axial symptoms. 12
We conducted a pilot study, which was not designed to demonstrate a statistically significant benefit on our primary outcome, but rather to observe trends in FoG improvement provided by LF or HF stimulation of the SNr. One third of patients exhibited a clinically significant improvement in the scale (i.e., a reduction of ≥ 8 points), 31 and 80% chose to maintain either C1 or C2 stimulations after study completion; both outcomes were equally distributed among the C1 and C2 stimulation groups. The NFOG-Q appears to lack sufficient reliability to detect small effect sizes, as changes must exceed 35% to overcome measurement error. 31 In fact, our results revealed a slight improvement of FoG with both C1 and C2 stimulations, although it did not reach the statistically significant threshold. Considering the clinometric properties of the NFOG-Q, our findings suggest that future studies should either examine HF and LF-SNr stimulation in a larger sample size, adequately powered to demonstrate a benefit using the NFOG-Q as the primary outcome or contemplate adopting an alternative outcome measure allowing a smaller sample size. It is important to note that there is currently no validated instrumental objective assessment method for reliably evaluating FoG over an extended period in a real-world setting. Interestingly, 80% of patients (n = 12) chose to maintain the combined stimulation of the STN-SNr while still blind for the stimulation settings (6 patients the HF-STN/LF-SNr and 6 patients HF-STN/HF-SNr stimulation).
It is possible that different factors have influenced the choice in different patients, including the good tolerability and lack of severe AEs with both combined stimulations. However, the blinded choice for combined STN-SNr stimulation over STN alone must have been based also on some benefit perceived by patients. It remains to be defined if this choice was also dependent on the significant improvement of motor complications with the combined STN-SNr stimulations, with most patients showing a mild but statistically significant improvement of dyskinesia. Moreover, the improvement in sleep quality obtained by C1 may have led some patients to opt for HF STN-HF SNr stimulation. Regarding this latter point, a pilot randomized, cross-over, double-blind study has recently suggested a possible role of STN-SNr DBS in improving sleep, in particular restless legs and immobility at night items of the PDSS, 48 likely due to its interconnection with mesencephalic region, PPN, and consecutively the “downstream” locus coeruleus. Our study endorses the possible role of combined HF stimulation of STN and SNr on improving sleep quality.
Finally, VTA coverage analysis confirmed that SNr was properly stimulated by both C1 and C2 settings. Due to the limited sample size, attributing significance to the difference found in SNr coverage is challenging. Further studies could help to define the role of coverage and the
Our study has some limitations worthy of consideration for the results interpretation. First of all, this is a pilot trial, due to the sample size unpowered for the primary outcome measure. Furthermore, the study includes a small proportion of women. Part of the gender imbalance can be explained by the lower prevalence of women among PD patients and the lower percentage of women undergoing DBS (about 30% of patients treated with DBS). 49
In addition, although NFOG-Q is a widely used outcome measure for studies on FoG (useful to assess FoG impact over a prolonged time period), it showed to be barely adequate for the use in clinical trials, due to possible recall bias, patients’ difficulties with evaluating FoG based on self-perception, the inability to assess other provoking circumstances of FoG and daytime fluctuations,31,50 highlighting the need for reliable and objective measures of FoG. At the same time, we coupled its use with the TUG, a recommended measure for gait assessment. We should also consider that there is no consensus on digital technology that can detect FoG, particularly regarding not only its presence but also its severity. A final consideration on study limitations could be made regarding a possible comparison of LF STN-DBS, which has demonstrated at least short-term effectiveness on FoG, versus combined HF STN + SNr DBS at LF and HF. We chose not to include this further comparison as a compromise for study feasibility, but such an assessment is worth testing.
These shortcomings notwithstanding, we believe that this study expands our knowledge on the safety and the possible usefulness of combined STN and SNr stimulation in PD, not only to improve FoG (which may have a variable and subjective response) but also to improve motor complications and some nonmotor features like sleep quality. Our study was not powered to demonstrate superiority of one type of combined stimulation (i.e., high- vs. low-frequency) vs. standard stimulation, but our results suggest that both can be safely attempted in PD patients treated with STN-DBS when FoG issues or motor complications persist despite various changes in standard stimulation settings. Attention should be paid in wisely adjusting the intensity of nigral stimulation (especially when using the low frequencies) due to the possible interference with mesolimbic circuitry and, thus, the possible onset of neurobehavioral side effects. Based on this trial, we suggest that further studies, possibly including a larger sample size and quantitative kinematic assessments, should be performed to better understand the role of combined STN-SNr stimulation for clinical practice.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241292315 - Supplemental material for Subthalamic and nigral stimulation for freezing of gait in Parkinson's disease: Randomized pilot trial
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241292315 for Subthalamic and nigral stimulation for freezing of gait in Parkinson's disease: Randomized pilot trial by Carlo Alberto Artusi, Claudia Ledda, Silvia Gallo, Domiziana Rinaldi, Corrado Campisi, Vanessa Rousseau, Claire Thalamas, Raquel Barbosa, Fabienne Ory-Magne, Christine Brefel-Courbon, Olivier Rascol, Amaury de Barros, Estelle Harroch, Maurizio Zibetti and Mario Giorgio Rizzone, Alberto Romagnolo, Gabriele Imbalzano, Leonardo Lopiano, Jean Luc Houeto, Margherita Fabbri in Journal of Parkinson's Disease
Supplemental Material
sj-docx-2-pkn-10.1177_1877718X241292315 - Supplemental material for Subthalamic and nigral stimulation for freezing of gait in Parkinson's disease: Randomized pilot trial
Supplemental material, sj-docx-2-pkn-10.1177_1877718X241292315 for Subthalamic and nigral stimulation for freezing of gait in Parkinson's disease: Randomized pilot trial by Carlo Alberto Artusi, Claudia Ledda, Silvia Gallo, Domiziana Rinaldi, Corrado Campisi, Vanessa Rousseau, Claire Thalamas, Raquel Barbosa, Fabienne Ory-Magne, Christine Brefel-Courbon, Olivier Rascol, Amaury de Barros, Estelle Harroch, Maurizio Zibetti and Mario Giorgio Rizzone, Alberto Romagnolo, Gabriele Imbalzano, Leonardo Lopiano, Jean Luc Houeto, Margherita Fabbri in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We acknowledge the collaboration of the patients who participated in this study and the collaboration of Marie Helène Fabre and Dr Clémence Leung for patients’ selection.
ORCID iDs
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by the ARI-CHU Project (Toulouse University Hospital).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Carlo Alberto Artusi received speaker honoraria from Zambon, Bial, Lusofarmaco. Domiziana Rinaldi received travel grants from AbbVie. Mario Giorgio Rizzone received grant support and speaker honoraria from UCB. Maurizio Zibetti received speaker honoraria from Medtronic, Zambon Pharma and AbbVie. Leonardo Lopiano received honoraria for lecturing and travel grants from Medtronic, UCB Pharma, and AbbVie. Alberto Romagnolo has received speaker honoraria from Zambon. Claudia Ledda, Corrado Campisi, Raquel Barbosa, Amaury De Barros, Silvia Gallo, Estelle Harroch, Vanessa Rousseau, Clarie Thalamas, Gabriele Imbalzano, Jean Luc Houeto report no disclosures. Christine Brefel-Courbon has received research grant from Association France Parkinson, and fees for lectures and consultancies from Aguettant, Orkyn, NHC, Zambon and AbbVie. Olivier Rascol has acted as a scientific advisor for drug companies developing antiparkinsonian medications (Abbott, Abbvie, Acorda, Adamas, BIAL, Biogen, Boehringer-Ingelheim, Cynapsus, GSK, Impax, Merck, Osmotica, Oxford-Biomedica, Lundbeck, Novartis, Prexton, Servier, Sunovion, TEVA, UCB, Zambon). Fabienne Ory-Magne has received honoraria for serving as an advisory board member from Abbvie, Medtronic, Orphalan, Aguettant and Orkyn, and for consultancy activities from Aguettant, Abbvie, Orphalan, Ellivie, Homeperf and Orkyn. Margherita Fabbri received Honoraria to speak from AbbVie, ORKYN, and BIAL, consultancies from BIAL and LVL Medical; Grant from France Parkinson, HORIZON 2022 and MSA Coalition.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
