Abstract
Background:
L-DOPA-induced dyskinesia (LID), occurring with aberrant processing of exogenous L-DOPA in the dopamine-denervated striatum, is a main complication of levodopa treatment in Parkinson’s disease.
Objective:
To characterize the effects of the vesicular antagonist tetrabenazine (TBZ) on L-DOPA-induced behavior, neurochemical signals, and underlying protein expressions in an animal model of Parkinson’s disease.
Methods:
20-week-old MitoPark mice were co-treated or separately administered TBZ and L-DOPA for 14 days. Abnormal involuntary movements (AIMs) and locomotor activity were analyzed. To explore dopamine (DA) transmission, fast scan cyclic voltammetry was used to assess presynaptic DA dynamics in striatal slices following treatments. PET imaging with 4-[18F]-PE2I, ADAM and immunoblotting assays were used to detect receptor protein changes in the DA-denervated striatum. Finally, nigrostriatal tissues were collected for HPLC measures of DA, serotonin and their metabolites.
Results:
A single injection of TBZ given in the interval between the two L-DOPA/Carbidopa treatments significantly attenuated L-DOPA-induced AIMs expression and locomotor hyperactivity. TBZ was shown to reduce tonic and phasic release of DA following L-DOPA treatment in DA-denervated striatal tissue. In the DA-depleted striatum, TBZ decreased the expression of L-DOPA-enhanced D1 receptors and the serotonin reuptake transporter. Neurochemical analysis indicated that TBZ attenuated L-DOPA-induced surges of DA levels by promoting DA turnover in the nigrostriatal system.
Conclusions:
Our findings demonstrate that TBZ diminishes abnormal striatal DA transmission, which involves the ability of TBZ to modulate the presymptomatic dynamics of DA, and then mitigate aberrant release of exogenous L-DOPA from nerve terminals. The results support the potential of repositioning TBZ to counteract LID development.
INTRODUCTION
Dopamine (DA) replacement therapy with L-DOPA is the standard treatment for Parkinson’s disease (PD). However, after years of daily exposure, most PD patients begin suffering from fluctuating motor responses and debilitating involuntary choreic and dystonic movements (abnormal involuntary movements, AIMs) also known as L-DOPA-induced dyskinesia (LID) [1, 2]. Although the mechanisms that underlie LID are complex, the condition mainly results from altered L-DOPA-DA metabolism, accompanied by hyperactivation of striatal DA receptors and related signaling pathways [3–7]. Importantly, as degeneration progresses, synthesis of DA from exogenously administered L-DOPA is gradually shifted to serotonergic neurons and non-neuronal cells. However, these cells lack precise control of DA release and reuptake mechanisms. Therefore, they cannot buffer extracellular DA levels, which subsequently result in narrowing of the therapeutic window in patients with PD [8]. Current therapeutic strategies for LID treatment include titrating L-DOPA intake, addition of amantadine as a blocker at NMDA receptors, and utilizing DA receptor agonists [7, 9]. Although these approaches sometimes manifest anti-dyskinetic effects, the long-term results have been disappointing, especially in very advanced PD patients afflicted with co-morbidities [10]. A large number of emerging investigations have focused on presynaptic release of various neurotransmitters, such as adenosine, acetylcholine (ACh), GABA, or Serotonin (5-HT), that interact with dopaminergic pathways and have been shown to be possible pharmacological targets to suppress L-DOPA-induced dyskinesia [11, 12]. However, although these alternative approaches produced positive results in animals, the majority of recent clinical trials in patients showed no or minimal LID improvement [13].
In the MitoPark mouse, a homozygous disruption of mitochondrial transcription factor A (TFAM), specifically in midbrain DA neurons, results in mitochondrial dysfunction and a slow. progressive loss of DA. The MitoPark phenotype results in steadily progressive loss of DA afferents in areas of the basal ganglia, starting in early adulthood, which manifests the associated motor symptoms typical of PD in human [14, 15]. Therefore, MitoPark mouse has also been studied as a preclinical animal model to recapitulate LID in response to DA replacement therapy [14, 16].
TBZ is a benzoquinolizine pharmacophore that selectively interferes with the vesicular monoamine reservoir and was proven to be beneficial in hyperkinetic movement disorders [17]. In striatal areas, the vast majority of vesicular monoamine transporters (VMAT-2) are present on DA terminals; therefore, analogs of TBZ are utilized as imaging markers for striatal DA terminals [18–22]. In human postmortem brain tissue, TBZ was shown to deplete DA without affecting other neurotransmitter levels in the striatum [23]. Although administration of higher doses of TBZ in rats has significant effects on all striatal monoamines, doses at or below 2.0 mg/kg of TBZ were shown to preferentially deplete striatal DA [24]. Fast scan cyclic voltammetry in freely moving rats shows that TBZ almost abolishes phasic DA release in the accumbens, but increases extracellular DA by inducing reverse transport by the DA transporter [25]. At a clinical level, the therapeutic rationale of using TBZ for targeting hyperkinetic movement disorders is supported by the observation of symptomatic improvement [26]. Although clinical data have shown that TBZ may also improve LID in patients with PD, it only focused on clinical viability, not elucidated the underlying mechanism of antidyskinetic properties [27].
Hence, in this study we used L-DOPA-responsive, DA-deficient MitoPark mice as a model for LID to determine if TBZ can mitigate aberrant DA release in the nigrostriatal system and reduce dyskinesia. We designed a therapeutic regimen, in which TBZ is given in the interval between the administration of L-DOPA/Carbidopa, over 2 weeks and found that this dosing schedule could suppress LID development. In addition, neurochemical and cellular analysis revealed target-specific modulation of monoamine neurotransmission and dyskinesia-related biochemical markers, suggesting that administration of TBZ can optimize L-DOPA therapy. Importantly, this pharmacological approach to reduce LID development in MitoPark mice sheds light on neurophysiological mechanisms of TBZ action on nigrostriatal monoamines from L-DOPA-primed animals and repositions TBZ against LID development and expression in PD.
MATERIALS AND METHODS
Animals
20-week-old male wild type and MitoPark mice were housed in the animal facility at National Defense Medical Center (NDMC), which has full AAALAC accreditation. The mice were housed at 25°C with a 12/12 h light/dark cycle and continuous water and food supply. All efforts were made to reduce animal suffering and to minimize the number of animals used. All animal protocols were approved by the NDMC Animal Care and Use Committee (IACUC 20-129 and IACUC 20-267) in accordance with NIH guidelines. See Fig. 2A, C, E; Fig. 3A; and Supplementary Figures 1 and 2 for experimental protocols and times of drug administration. The breeding scheme for generating MitoPark mice has been previously described [15, 28]. MP mice used in these experiments were heterozygous for DAT-cre expression (DAT/DATcre), and homozygous for the loxP-flanked Tfam gene (Tfaml ∘xP/Tfaml ∘xP). Breeding pairs to generate MP mice were sent to the NDMC in Taiwan from a colony maintained at the NIDA (NIH) Intramural Program, and genotyping was performed at the NDMC.
Treatments
L-DOPA and Carbidopa (Sigma-Aldrich, St. Louis, MO) were dissolved in 0.9% saline. L-DOPA (10 mg/kg) was injected i.p. together with carbidopa (5 mg/kg) to inhibit peripheral dopa decarboxylase. L-DOPA/Carbidopa was injected twice a day (morning and late afternoon) and initiated in the MitoPark mice beginning at 20 weeks of age and continued for 14 days. Behavioral analyses were performed one day before administration and weekly after initiation of L-DOPA/Carbidopa treatment. TBZ, dissolved in saline, or saline alone (vehicle) in all experiments was given intraperitoneally. In the first protocol, Mitopark mice were coadministered TBZ (1 mg/kg) and L-DOPA/Carbidopa twice per day for 2 weeks to model the previously observational study of TBZ in treatment of LID patients. In the second protocol, the MitoPark mice received a single injection of TBZ (1 mg/kg) in the interval between twice-daily administrations of L-DOPA/Carbidopa for 2 weeks. L-DOPA (10 mg/kg, i.p.) combined with carbidopa (5 mg/kg) was administered twice per day (10 and 18) for two weeks to induced dyskinesia. This group is represented by the symbol “MP LID”. Co-administration of TBZ (1 mg/kg) and L-DOPA (10 mg/kg) combined with carbidopa (5 mg/kg) was administered at two times (10:00 and 18:00) per day for two weeks. “MP + L-DOPA + TBZ Co-administration” is the symbol for this group. In contrast, L-DOPA (10 mg/kg) combined with carbidopa (5 mg/kg) was administered at 10:00 and 18:00 while the TBZ (1 mg/kg) was administered at 14:00. This group is termed as “MP + L-DOPA + TBZ (administered separately)”.
Behavioral tests
AIM test
Animals were observed 120 min after the administration of L-DOPA (10 mg/kg & Carbidopa 5 mg/kg) on 0, 1, 4, 7, and 14 days. A webcam was used to simultaneously record the upper and the side of each mouse (Fig. 1) for 1 min every 20 min for a total of 6 recordings.

AIM scoring, including axial and body posture deformity, limb movement, paw movement and rearing time. Amounts of abnormal movement are defined in the included table.
The AIM scoring used was a modified version of the AIM behavior grading of mice proposed by Ding [29] and Sebastianutto [30]. The modified AIM scoring standards are shown in the table in Fig. 1 (and Supplementary Video 1). The severity of dyskinesia was recorded every 20 min with the AIM score of the most severe symptom according to the above-mentioned scoring standard. The higher the AIM score, the more marked the abnormal involuntary movement behaviors. The total AIM score of normal mice should be below 4 [31].
Locomotor activity
Horizontal and vertical movements were evaluated using activity boxes (45×45 cm) after habituation in a low-noise environment for 1 hour. The protocol uses a grid of infrared beams to monitor the movement of mice over a 1 h period, including the distance of horizontal movement and the number of rearings.
[18F]FE-PE2I positron emission tomography (PET) imaging of DA transporter function
Our previous papers have described the preparation of [18F]FE-PE2I [32, 33]. Mice were injected with 0.3 mCi [18F]FE-PE2I into the tail vein during anesthesia (5% isoflurane/oxygen mixture for induction, 2% for maintenance) and transferred to a radiation barrier box. After 20 min, the mouse was moved to the PET scanner for a static image scan for about 20 min, with the energy window set to 250 to 700 KeV. 3D images of the brain were then captured. Images were smoothed using a Gaussian algorithm. The specific uptake rate (SUR) was calculated as SUR = (VOI of striatum)- (VOI of cerebellum)/VOI of cerebellum [34, 35].
Fast scan cyclic voltammetry (FSCV) for DA dynamics measurements in brain slices
Striatal brain slice preparation
A brief description of the brain slice preparation process is presented here, details are described in our previous studies [36, 37]. After sacrifice by decapitation, the brain was removed and transferred to an oxygenated (95% O2/5% CO2), cold, cutting solution (in mM: sucrose 194, NaCl 30, KCl 4.5, MgCl2 1, NaH2PO4 1.2, glucose 10, and NaHCO3 26). After 30 s, the brain tissue was cut into coronal 280μm slices containing striatum with a vibrating blade microtome (VT 100, Leica) in a chamber filled with cutting solution; these brain slices were then transferred to a chamber with a solution containing oxygenated artificial cerebrospinal fluid (aCSF; in mM: NaCl 126, KCl 3, MgCl2 1.5, CaCl2 2.4, NaH2PO4 1.2, glucose 11, NaHCO3 26) at 30°C for 30 min and were used in the FSCV experiments below.
Fast scan cyclic voltammetry (FSCV) for DA measurements in brain slices
The detailed protocols of the FSCV recording used in this study are described in our earlier papers [38, 39]. The coronal brain slices (280μm) from the 22-week-old mice were moved into the chamber (0.5 ml, 31–33°C) filled with aCSF and with a perfusion rate of 2 ml/min for recording. A carbon fiber (7μm diameter; Goodfellow Corp., Oakdale, PA, USA) was positioned between the separated tips of the bipolar stimulation electrodes (FHC Inc., Bowdoin, ME, USA) in the dorsal striatum, with a depth of 100μm. The carbon fiber electrode consisted of a pipette filled with 4M potassium acetate/150 mM KCl solution with the fiber inside the pipette. The FSCV used a triangular waveform (400 V/s scan rate, 7 ms duration) applied every 100 ms to drive the potential of the carbon fiber from –0.4 to 1.0 V and back to –0.4 V, using a 10V voltage stimulus intensity at 25 Hz to induce DA release. Data collection and post-mortem analysis were carried out using an A/D board (PCI 6052E and PCI-6711E, National Instruments, Austin, TX, USA) and customized software based on LabView (TarHeel CV, courtesy of Drs. Joseph Cheer and Michael Heien, University of North Carolina).
DA release in striatal slices detected by fast scan cyclic voltammetry (FSCV) in WT, MP, and MP LID
To examine the effect of L-DOPA or TBZ on the ability to release DA in striatum, we administered the DA precursor L-DOPA (0.1μM) or TBZ (0.1μM) for 30 min into striatal slices. Finally, to examine the effect of co-administration of L-DOPA and TBZ on DA release, we administered 0.1μM L-DOPA for 140 min into the striatal slices. After for 30 min, we co-administered 0.1μM L-DOPA and 0.1μM TBZ for 30 min.
The FSCV data was fit to a linear regression model (Prism 6.02; GraphPad, San Diego, CA, USA) to calculate DA Release probability [40] as follows. The DA concentration differences induced by different pulses (2, 5, 10) and a single pulse were used to calculate the slope of the linear regression [36, 38]. Each electrode was calibrated with 0.1μM DA standard solution before and after the experiment, to allow the current signals obtained to be converted into DA concentrations. All signals used in the analysis matched the expected voltage-current curve for DA [41].
High performance liquid chromatography (HPLC) analysis of DA and serotonin and their metabolites
Details of our HPLC system is described in our previous paper [42]. Briefly, male mice (22 weeks old) were sacrificed by decapitation 10 min after a challenge of L-DOPA injection (10 mg/kg, i.p. and CB 5 mg/kg). The dorsal striatum and substantia nigra were then dissected according to a mouse brain atlas. The tissue was immediately frozen on dry ice and stored at –80°C until use. Tissues were homogenized in 1 mM of oxalic acid and then centrifuged at 15,000 g for 40 min at 4°C. The resulting supernatants were filtered through a 0.22-μm syringe filter (Millipore, Bedford, MA, USA) and then injected into the HPLC system for measurement of DA, 5-HT, and their metabolites [DA metabolites: 3,4-dihydroxyphenylacetic acid (DOPAC) and homovanillic acid (HVA); serotonin metabolite: 5-hydroxyindole acetic acid (5-HIAA)]. Neuronal activity was estimated by calculating the DA and 5-HT turnover rates. The DA turnover rate was calculated as [DOPAC] + [HVA])/[DA], and the 5-HT turnover rate as [5-HIAA]/[5-HT]. Linear regression was used to construct a standard curve in the concentration range 10–1,000 nM to compare the sample with the standard curve and determine the content of DOPAC, HVA, DA, 5-HIAA, and 5-HT.
The HPLC system consisted of a TSKgel, ODS-80TM C18 column (Tosoh, Japan), a pump (LC-10AD, Shimadzu, Japan), and an electrochemical detector (Coulochem II, ESA, Chelmsford, MA, U.S.A.) containing 5010 analytical cells and 5020 guard cells. The voltages of the analytical cell were set at 40 and 250 mV (detecting potential) and the guard cell (mobile phase cell) at 350 mV. The mobile phase (MDTM mobile phase, ESA), which consisted of 75 mM sodium dehydrogenate phosphate (monohydrate), 1.7 mM 1-octanesulfonic acid (sodium salt), 100μl/L triethylamine, 25μM EDTA, and 10% acetonitrile, at a pH 3.00, was delivered at a flow rate of 1.0 ml/min.
Western blot analysis
Mice (22 weeks old) were sacrificed by decapitation 10 min after a challenge of L-DOPA injection (10 mg/kg, CB i.p.). The dorsal striatum was dissected according to a mouse brain atlas. The tissues were homogenized in RIPA buffer (TAAR-ZBZ5, Biotools Co., Ltd., Taiwan) with a protease inhibitor cocktail (ab20111, Abcam, Cambridgeshire, United Kingdom). The BCA Protein Assay Kit II (K813, Biovision, Lausen, Switzerland) was used to measure protein concentration. Proteins were electrophoresed onto a 12% SDS-polyacrylamide gel and electrotransferred onto a polyvinyl difluoride membrane. After incubating with blocking buffer (2% bovine serum albumin and Tris-buffered saline with Tween 20 (TBST)) for 45 min, the membrane was incubated overnight with primary antibodies against VMAT2 (1:1000, rabbit, GTX78428, GeneTex, Irvine, TX), D1R (1:1000, rabbit, GTX100354, GeneTex, Irvine, TX), D2R (1:1000, rabbit, GTX17570, GeneTex, Irvine, TX), 5HT1A (1:1000, rabbit, GTX02545, GeneTex, Irvine, TX), 5HT1B (1:1000, rabbit, NB100-56350, Novus, Centennial, CO), pDARPP32 (1:1000, rabbit, #5393, Cell Signaling Technology, Danvers, MA), DARPP32 (1:1000, rabbit, #2306, Cell Signaling Technology, Danvers, MA), and β-actin (1:5000, rabbit, ab8227, Abcam, Cambridge, United Kingdom). After washing 3 times, the membranes were incubated 1 h with goat HRP-linked anti-rabbit IgG antibodies (1:20,000, ab6721, Abcam, Cambridge, United Kingdom), developed with an enhanced chemiluminescence (ECL) detection kit (Amersham Life Sciences, Piscataway, NJ, USA) and imaged with UVP ChemStudio Plus (Analytik Jena AG, Germany). The results were normalized to the level of β-actin used as a loading control, and the intensities of immunoreactivity were quantified relative to the expression of the corresponding control using NIH Image J.
Statistical analysis
All data were shown as mean±SEM and were analyzed using an unpaired t-test or a one or two-way analysis of variance (ANOVA) followed by a Bonferroni post hoc test for multiple comparisons using appropriate software (Prism 6.02; GraphPad, San Diego, CA, USA). A p-value <0.05 was considered statistically significant.
RESULTS
TBZ alters LID development in MitoPark mice
Dyskinesia was monitored to evaluate whether L-DOPA could induce involuntary movements in MitoPark mice. We adapted an AIM score system used previously, in which general dyskinesia includes degrees of the twitching of trunk and latency of body elevation, while limb dyskinesia is measured as severity of abnormal displacement of the limbs involved (Fig. 1). We thus calculated overall AIM scores on days 1, 7, 14 after initiation of L-DOPA/Carbidopa treatment to evaluate the progression of LIDs (Fig. 2A; Supplementary Video 2). As shown in Fig. 2B, 20-week-old MitoPark mice developed obvious LIDs after 1 week of L-DOPA/Carbidopa treatment (p < 0.001). Furthermore, MitoPark mice still exhibited LIDs after 2-week period of treatment with L-DOPA/Carbidopa. Since severe dyskinetic movements persisted in MitoPark mice after 4-5 weeks of chronic L-DOPA treatment [43], in this study, we wanted to investigate if TBZ could prevent dyskinetic development in MitoPark mice under the 2-weeks treatment of L-DOPA/Carbidopa. Based on the previously established protocol in a human clinical study [27], TBZ was co-administered with L-DOPA/Carbidopa twice a day for 2 weeks (Fig. 2C). Unexpectedly, co-administration of TBZ and L-DOPA in MitoPark mice did not attenuate LID development over the 2- week study period (Fig. 2D). In contrast, a single injection of TBZ given in the interval between the two L-DOPA/Carbidopa treatments for 2 weeks (separately administered TBZ) significantly attenuated LID development (Fig. 2E, F; Supplementary Figure 3; Supplementary Video 3). Importantly, by day 14, separately administered TBZ could reduce the AIM scores in the period of LID induction by L-DOPA injection (Fig. 2G, p < 0.001). Compared to the control group (MitoPark mice without L-DOPA/Carbidopa treatment), 2-week L-DOPA-primed MitoPark mice (MP LID) showed higher average AIM scores in the dyskinetic period of 20 to 1200 min after L-DOPA injection (Fig. 2H, MP vs. MP LID p < 0.001) (Supplementary Video 4), while separately administered TBZ could reverse the L-DOPA-increased average AIM expression (Fig. 2H, MP LID vs. MP + L-DOPA + TBZ (administered separately) p < 0.001) (Supplementary Video 5). Taken together, this regimen of TBZ administration in the interval between the L-DOPA/Carbidopa treatments could mitigate dyskinesia development in L-DOPA-primed MitoPark mice.
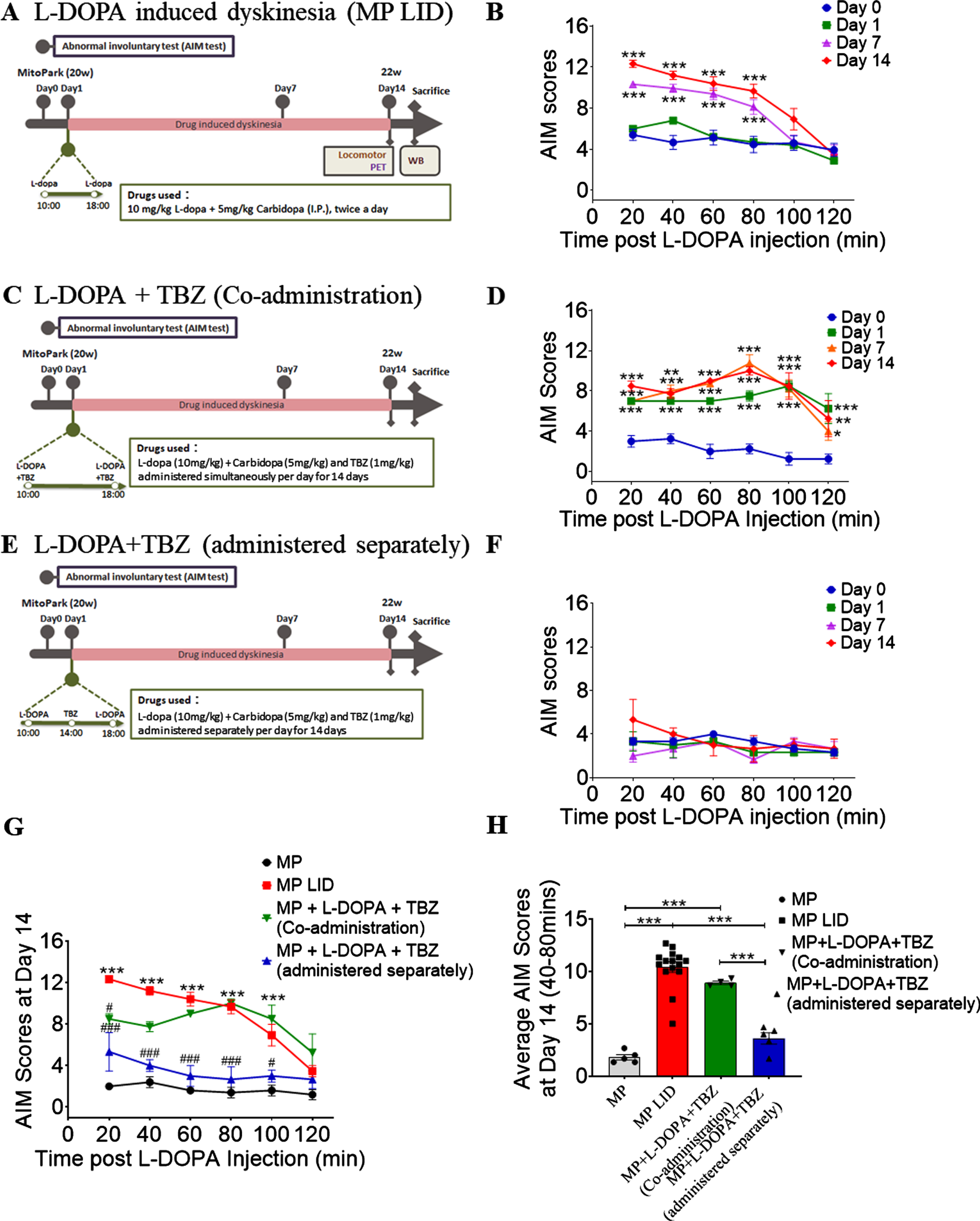
Protocol for L-DOPA induced dyskinesia and TBZ administration affecting L-DOPA induced dyskinesia in MP LID mice. (A) L-DOPA, combined with carbidopa, was administered daily at two times (10 and 18) for two weeks to induce dyskinesia. (B) Dyskinesia could be observed after several days of L-DOPA and Carbidopa injection with increments in AIM scores. Two-way analysis of variance (ANOVA) followed by a Bonferroni post hoc test for multiple comparisons. ***p < 0.001 indicates comparison with Day 0. (C) Co-administration of TBZ and L-DOPA protocol where TBZ was administered simultaneously with L-DOPA and Carbidopa in MP mice. (D) Dyskinesia could be observed with significant changes in AIM scores during co-administration of TBZ and L-DOPA to MP LID mice. ANOVA followed by a Bonferroni post hoc test for multiple comparisons. *p < 0.05, **p < 0.01, ***p < 0.001 indicates comparison with Day 0. (E) Administration protocol for TBZ and L-DOPA injection, in which L-DOPA and TBZ were given separately. (F) Dyskinesia was suppressed in MP mice by the separate administration protocol without any significant variation in the AIM score. (G) AIM scores at Day 14 of the MP LID induction period revealed significant differences in LID and the groups with TBZ administered separately. ANOVA followed by a Bonferroni post hoc test for multiple comparisons. MP LID compared to untreated MitoPark, ***p < 0.001; MP + L-DOPA + TBZ compared to untreated MitoPark, #p < 0.05, # # #p < 0.001. (H) AIM score over 20 to 120 minutes in 14-day dyskinesia period after L-DOPA injection. Unpaired t-test; ***p < 0.001. The data represent mean±SEM.
TBZ administered in the interval of L-DOPA treatment improves locomotor hyperactivity in MitoPark mice
Next, we determined if TBZ could also modify locomotor activity in L-DOPA-primed MitoPark mice. Locomotor activities, including horizontal ambulation and rearing duration, were analyzed for 60 min following L-DOPA injection (Fig. 3A). After one injection of L-DOPA, the distance traveled by MitoPark mice was shorter than that by WT mice (Fig. 3B, C, p < 0.05). However, there was no difference in rearing duration between 20-week-old MitoPark mice and age-matched WT littermates. Following 14-days administrations of L-DOPA and Carbidopa, the horizontal travel distance and rearing duration of LID MitoPark mice (MP-LID) were longer than those of WT or MitoPark mice (Fig. 3C, p < 0.001), suggesting that MP-LID develop abnormal locomotor behavior when L-DOPA is given on day 14. Notably, administration of TBZ in the L-DOPA interval reduced the L-DOPA-induced locomotor hyperactivities (Fig. 3C, p < 0.05), but did not exacerbate horizontal locomotor reduction resulting from the continuing degeneration of DA terminals in MitoPark mice (Fig. 3C, p < 0.05). These data thus demonstrate that MP-LID mice manifest sensitivity to DA replacement and our designed regimen of TBZ treatment, and that modulating abnormal DA transmission could improve the pattern of locomotor behavior in L-DOPA-primed MitoPark mice.
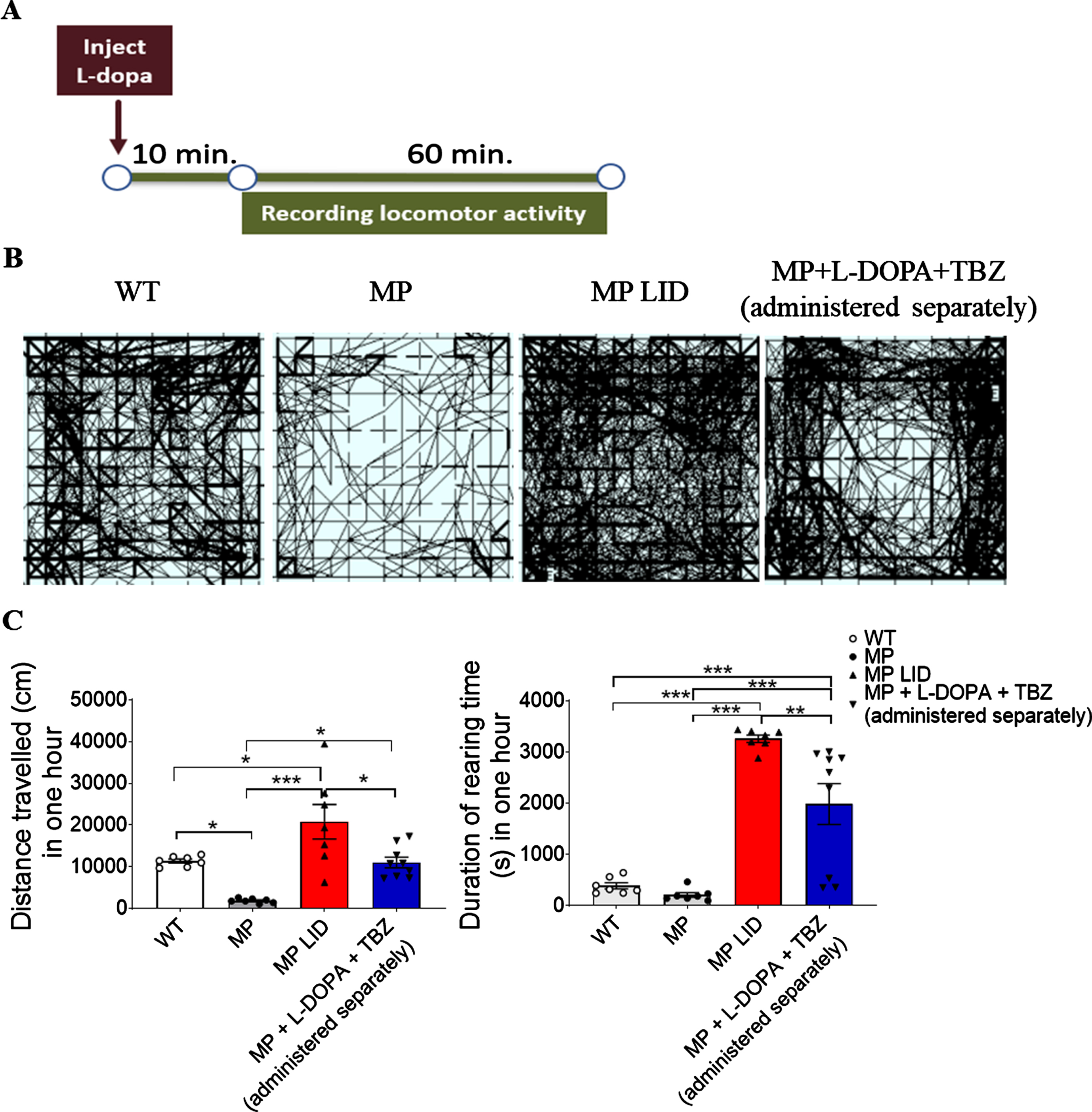
Open field horizontal distance recordings. (A) Experimental protocol. The recording started at 10 minutes after L-DOPA injection and the locomotor activity was recorded for the next 60 minutes. (B) Representative recording maps which reveal LID increasing locomotion significantly compared with untreated MP mice. TBZ administration partially ameliorates the hyperactivity in MP LID mice. (C) Left panel shows significant increases in moving distance in the MP LID mice and that TBZ administration suppresses this increment. Right panel shows that sustained rearing in MP LID mice could be ameliorated by TBZ administration. Unpaired t-test; *p < 0.05, **p < 0.01, ***p < 0.001.
TBZ reduces tonic and phasic DA release in the MP-LID striatum after L-DOPA administration
In order to gain some insight into how DA release relates to the behavioral changes noted above, we employed FSCV. After administration of the DA precursor L-DOPA in striatal slices with an intact DA system (WT group) for 15 min, DA release in the dorsal striatum of the WT group was significantly increased (Fig. 4A, B). Interestingly, the administration of L-DOPA could also significantly increase the DA release in the dorsal striatum from MP and LID groups (Fig. 4A, B); the DA percentage change from baseline after administration of L-DOPA was higher than in the WT group. After 14 days of L-DOPA treatment in MP mice (MP-LID group), the percentage of DA concentration increased by L-DOPA was even more significant (Fig. 4G). Importantly, the increment in DA concentration induced by L-DOPA in MP LID could be suppressed close to the DA levels of WT animals after TBZ infusion (Fig. 4C-F).
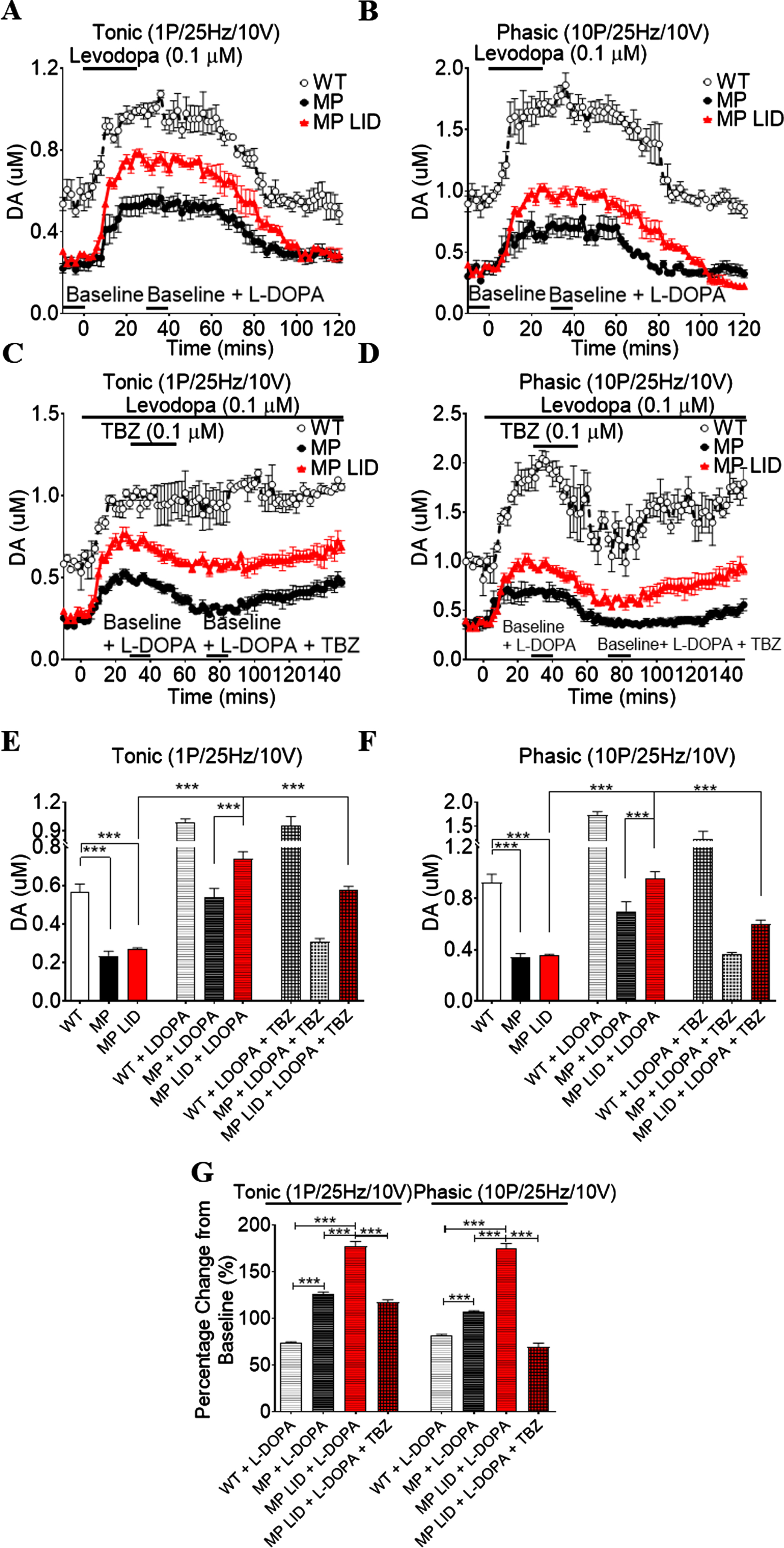
DA release in striatal slices detected by FSCV. The tonic (A) and phasic (B) release increased significantly after levodopa (0.1μM) infusion into brain slices in all groups. Both levodopa-induced increments in tonic (C) and phasic (D) release were suppressed after TBZ (0.1μM) infusion in brain slices in each group. (E) Tonic DA release increased significantly in LID animals when L-DOPA was infused into brain slices, and this was suppressed by TBZ infusion. Interestingly, the increment in DA concentration induced by L-DOPA in LID could be suppressed close to the DA level of WT animals after TBZ infusion (lattice red bar: LID + LDOPA + TBZ vs. Open bar: WT, p = 0.7795). (F) Significant increment in DA phasic release after L-DOPA infusion in LID animal-derived striatal slices could be suppressed after TBZ infusion (horizontal stripes red bar: LID + LDOPA vs lattice red bar: LID + LDOPA + TBZ, p < 0.001). While the increment in phasic DA release induced by L-DOPA in LID animals was suppressed by TBZ, the DA levels in this group were lower than phasic release in WT animals (open bar: WT vs. lattice red bar: LID + LDOPA + TBZ, p < 0.01). (G) Increments of DA release after L-DOPA infusion in both untreated MP and LID animals were higher than in WT animals. Moreover, both increments of tonic and phasic DA release induced by L-DOPA in the LID groups were higher than in untreated MP mice and even higher than in WT animals. The increments in tonic and phasic releases were suppressed by TBZ infusion to near normal levels in WT. Thus, this change in tonic and phasic DA release induced by L-DOPA, suppressed by TBZ, was parallel to the dyskinesia in MP LID mice. This might explain the dyskinesia induced by L-DOPA in LID animals as resulting from abnormal DA release elevations after L-DOPA infusion only in MP mice. Unpaired t-test; ***p < 0.001.
Augmented release of DA may relate to the abnormal behavior of L-DOPA-induced dyskinesia in MP mice. Moreover, the co-administration of L-DOPA and TBZ in the LID group could reverse this excessive DA concentration in the synaptic cleft. This might further explain that dyskinesia induced by L-DOPA in LID animals, resulting from abnormal DA release elevations after L-DOPA infusion, is seen only in MP mice (Fig. 4G). Additional data on DA release dynamics are shown in Supplementary Figures 1-3. After administering 0.1μM TBZ alone for 15 min in striatal slices, DA release was significantly reduced in the dorsal striatum of all mice (Supplementary Figure 4). In the tonic phase, MP mice were most markedly affected by TBZ’s inhibition of DA release, followed by the MP-LID group, and the WT group was affected least. Conversely, in the phasic phase, the DA release in WT and MP-LID mice were decreased more than that in MP mice (Supplementary Figure 4). After the administration of L-DOPA, the slope of the linear regression in striatum in WT, MP, and LID groups all increased. The representative FSCV signals are shown in Supplementary Figure 5.
DA release probability in WT mice showed that L-DOPA induced probability increases and that the increment of release probability resulting from L-DOPA administration (0.1μM) was suppressed by TBZ (0.1μM) (Supplementary Figure 6). Similar changes were found in MP and LID mice. The slope of linear regression for each group was plotted, which revealed that significant increases in slope with L-DOPA injection could be significantly reduced by TBZ administration. The DA release probability was calculated as noted above in Methods with different DA release concentrations using different stimulations (i.e., =2, 5, and 10 p/25 Hz) ([DA] d = [DA]10P-[DA]xP), with linear regression of concentration differences under these different stimulation paradigms. Differences in concentration release by the different stimulation parameters are referred to as releasing probability (slope).
TBZ treatment affects DA and serotonin transporter expressions in striatum of L-DOPA-primed MitoPark mice
DAT is known to localize in the dopaminergic cell bodies striatum. Reduced DAT in the nigrostriatal system accompanies neurodegeneration of dopaminergic neurons in PD. Recently, [18F]FE-PE2I was established as a radioligand for in vivo imaging and quantification of DAT. [18F]FE-PE2I radioactivity was found to be significantly lower in striatum of MitoPark mice than in WT mice (Fig. 5A, B, p < 0.001), showing that there is a significant loss of striatal DAT protein following dopaminergic neuron loss in 22-week old MitoPark mice. Nevertheless, the striatal [18F]FE-PE2I binding in MitoPark mice with L-DOPA/Carbidopa treatment was not different from that of age-matched MitoPark mice with placebo treatment (Fig. 5B, p = 0.5967). Also, TBZ treatment did not affect striatal [18F]FE-PE2I radioactivity in L-DOPA-primed MitoPark mice (Fig. 5B, p = 0.0517).
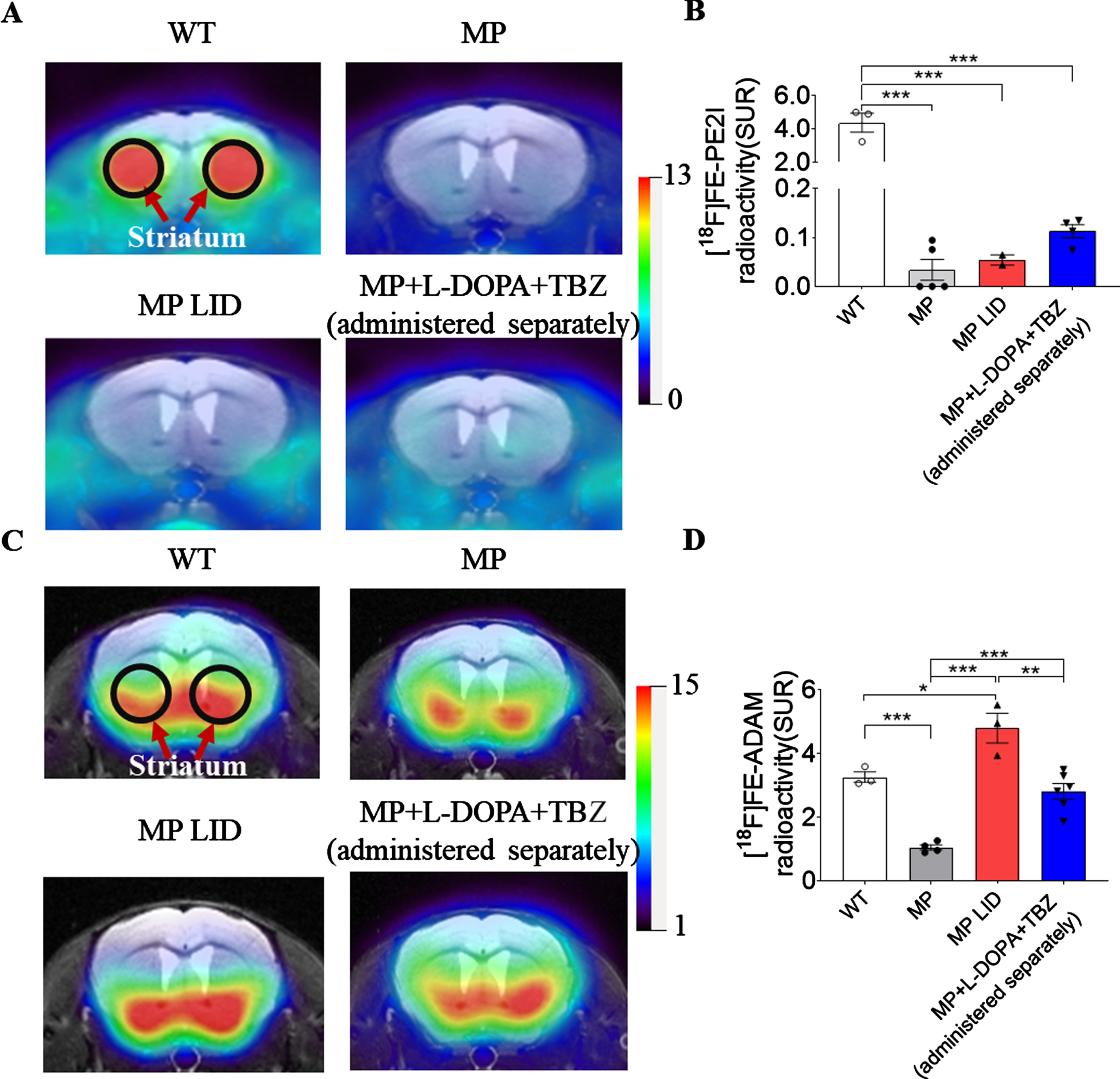
PET scans of functional monoamine transporter activity in WT, MP, and MP LID mice. (A) The [18F] FE-PE2I PET scan shows DA transporter activity in striatum (black circle) in each group of animals given L-DOPA. (B) The radioactivity (SUR) of DA transporters in MP LID and MP + L-DOPA + TBZ (separately administered) groups were significantly lower than in WT mice (p < 0.001). (C) [18F] FE-ADAM scan showed serotonin activity in striatum (black circles) in each group. (D) The radioactivity of the serotonin transporter after L-DOPA administration in MP mice was lower than in WT mice and activity was higher in LID mice than in WT mice (red bar vs. open bar with p < 0.05). After administration of L-DOPA with TBZ to MP mice, the radioactivity in the serotonin transporter was decreased (blue bar vs. Red bar). Unpaired t-test; *p < 0.05, **p < 0.01, ***p < 0.001.
These data suggest that there might be another monoamine transporter, which can transport DA in CNS, particularly when DAT abundance is relatively low. Although the serotonin transporter (SERT) does not play a primary role in the clearance of DA in normal striatum, SERT could participate in DA uptake associated with locomotor activity in sparsely dopaminergic innervated striatum. Therefore, we sought to investigate the role of serotonergic terminal function in the development of LID in MitoPark mice. We used the 18F-labeled SERT imaging agent N, N-dimethyl-2-(2-amino-4-18Ffluorophenylthio)-benzylamine (4-[18F]-ADAM; T1/2 = 110 min) together with micro-PET to determine SERT density in the dorsal striatum of MitoPark mice and measure the effects of L-DOPA or TBZ on the serotonin system in this Parkinsonian mouse model. In MitoPark mice, PET images showed clearly lower striatal 4-[18F]-ADAM uptake compared to the WT group, indicating that SERT decreases in DA-denervated striatum (Fig. 5C, D, p < 0.001). At 2 weeks following L-DOPA/Carbidopa treatment, PET imaging of the MP-LID mice revealed a significantly increased uptake of 4-[18F]-ADAM uptake in striatum compared to WT or MitoPark mice (Fig. 5D, p < 0.05), showing SERT is upregulated from 32% (22-weeks MitoPark mice) to 128% (2-week post-L-DOPA/Carbidopa treatment) of that found in normal striatum of WT mice. However, in MP + L-DOPA + TBZ separate administration group, the uptake of 4-[18F]-ADAM was significantly decreased compared to the MP-LID group (Fig. 5D, p < 0.01). Specifically, the average SUR values of 4-[18F]-ADAM in striatum of MP + L-DOPA + TBZ separate administration group were similar to those in the corresponding region of the WT group, implying that TBZ, by decreasing DA release, attenuates L-DOPA-enhanced SERT expression in DA-denervated striatum.
TBZ treatment alters hyperactivation of the D1R signaling pathway in LID
Since TBZ can inhibit VMAT-2 reversibly to interfere with DA translocation into synaptic vesicles, we investigated if chronic treatment of TBZ could affect striatal VMAT-2 expression in L-DOPA-primed MitoPark mice. As a proof-of-principle, we first examined the expression of VMAT-2 in striatum of WT and MitoPark mice in vehicle- and L-DOPA-treated groups. Interestingly, the relative level of VMAT-2 in striatum of MP-LID mice was increased as compared with untreated MP mice (Fig. 6A, B, p < 0.05), suggesting that exogenous DA derived from L-DOPA replacement could increase VMAT-2 protein expression in the DA-denervated striatum. However, TBZ did not significantly alter L-DOPA-upregulated expression of VMAT-2 in the corresponding area (Fig. 6B, p = 0.5625). Therefore, we sought to examine if TBZ, mitigating LID development in MitoPark mice shown above, would regulate DA D1 receptor and subsequent DARP32 signalings in striatum. Compared to WT and untreated MitoPark mice, the expression of D1 receptor protein (Fig. 6C, p < 0.01), but not D2 receptor protein (Fig. 6D, p = 0.2680), was increased in striatum of MP-LID mice. Moreover, the L-DOPA-enhanced expression of D1 receptor protein was decreased by administration of TBZ (Fig. 6C, p < 0.05). In addition, phosphorylation of DARPP32 was reduced in the striatum of the MP + L-DOPA + TBZ separately administration group compared to the MP-LID group (Fig. 6E, p < 0.01), suggesting that TBZ reduces L-DOPA-induced striatal DARPP32 activation. Since TBZ was shown to affect presynaptic serotonin storage and release, we examined if TBZ treatment altered the expressions of serotonin 1A/B receptors. However, there was no significant differences between groups in the expression of serotonin 1A (5HT1A) (Fig. 6F, p = 0.2670) or serotonin 1B (Fig. 6G, p = 0.2264) receptors in striatum.
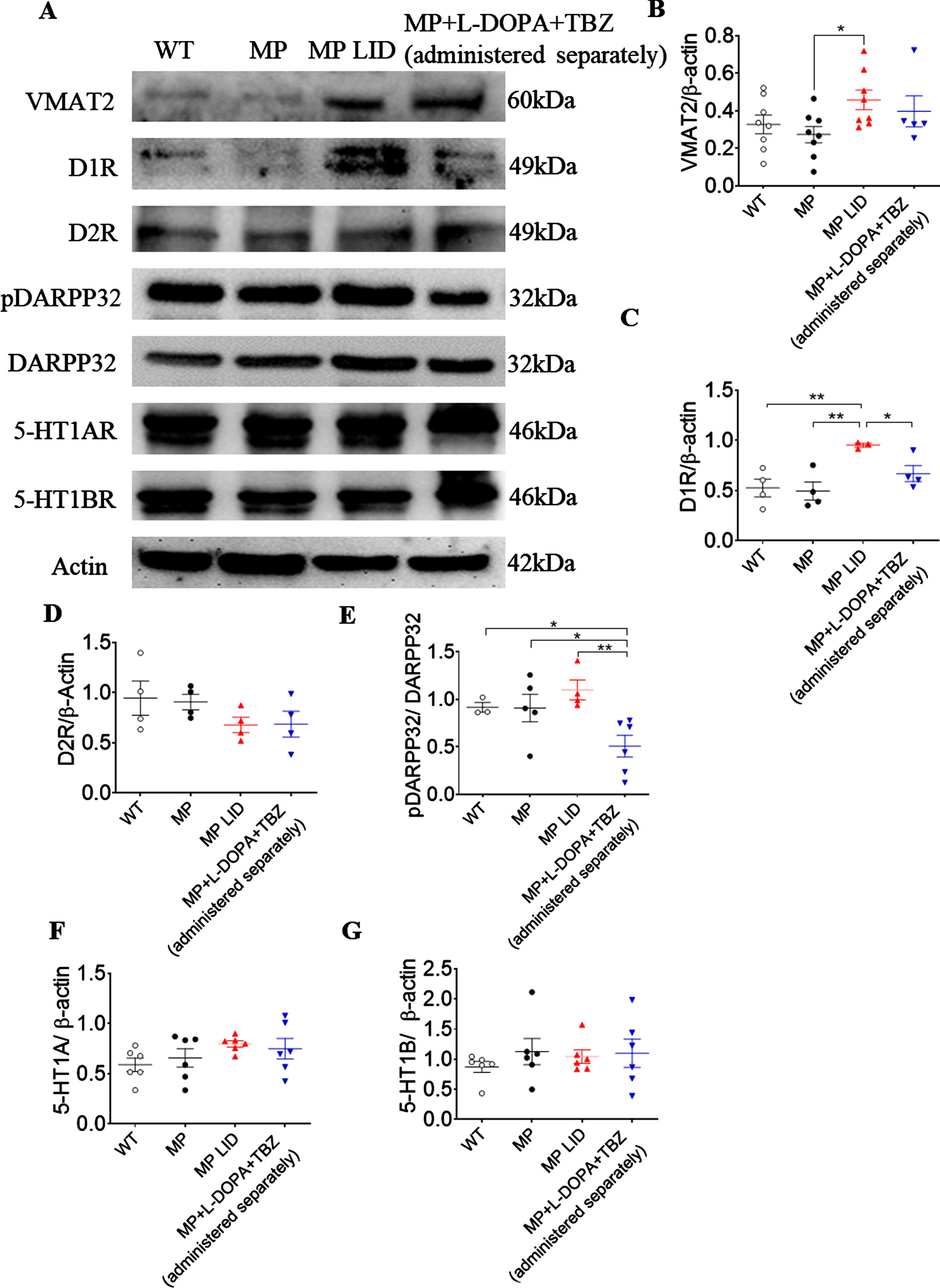
Expression of proteins involved in dopaminergic transmission in striatum after L-DOPA administration. (A) Dopaminergic transmission-related protein expression after L-DOPA administration. (B) The VMAT2 expression ratio in MP LID mice increased significantly compared with untreated MP mice. (p < 0.05). (C) D1 receptor expression was higher in LID mice compared with untreated MP mice and this increment was suppressed when TBZ was administered (Blue dot vs. red dots, p < 0.05). (D) There were no significant differences in D2 receptor expression between groups. (E) The L-DOPA-upregulated phosphorylation of DARPP32 was suppressed in MP + L-DOPA + TBZ (administered separately) (Blue dots vs. red dots, p < 0.01). (F) There were no significant differences in serotonin 1A (5HT1A) or, (G) serotonin 1B receptor expression. Unpaired t-test; *p < 0.05, **p < 0.01.
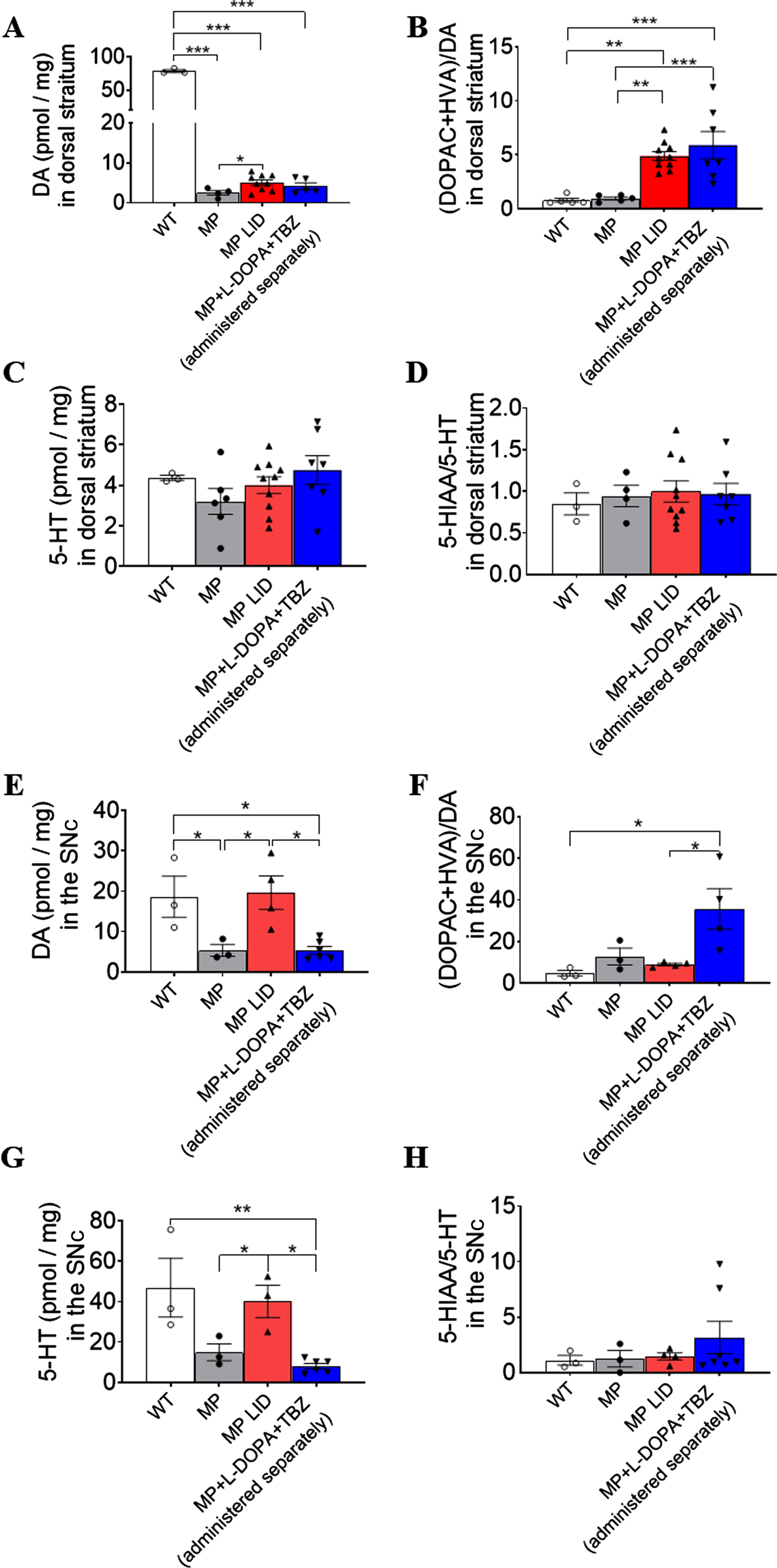
HPLC measurement of DA and serotonin tissue concentrations in striatum and SNc in mice after L-DOPA administration. (A) Both DA concentrations after L-DOPA in MP LID and MP mice were lower than in WT mice and the concentrations in LID mice were higher than those in MP mice. (B) The DA turnover rate increased significantly in LID and LID with TBZ which was higher than in MP and WT mice. (C) Serotonin tissue concentrations in striatum showed no differences between groups. (D) Serotonin turnover was also not different between groups. (E) DA tissue concentration in the SNc increased significantly in LID mice compared with MP mice, and this increment was reduced in the TBZ administration group (Blue bar: MP + L-DOPA + TBZ (separately administered) vs. Red bar: MP LID, p < 0.05). (F) DA turnover rate increased significantly in SNc in LID mice given L-DOPA and TBZ (Blue bar vs. Red bar, p < 0.05). (G) Serotonin concentration in substantia nigra increased in LID mice after L-DOPA administration (Grey bar: MP vs. Red bar: MP LID, p < 0.05) and this increase was suppressed in LID given L-DOPA plus TBZ (Blue bar: MP + L-DOPA + TBZ (separately administered) vs. Red bar: MP LID, p < 0.05). (H) There was no difference in serotonin turnover rate in SNc between groups.
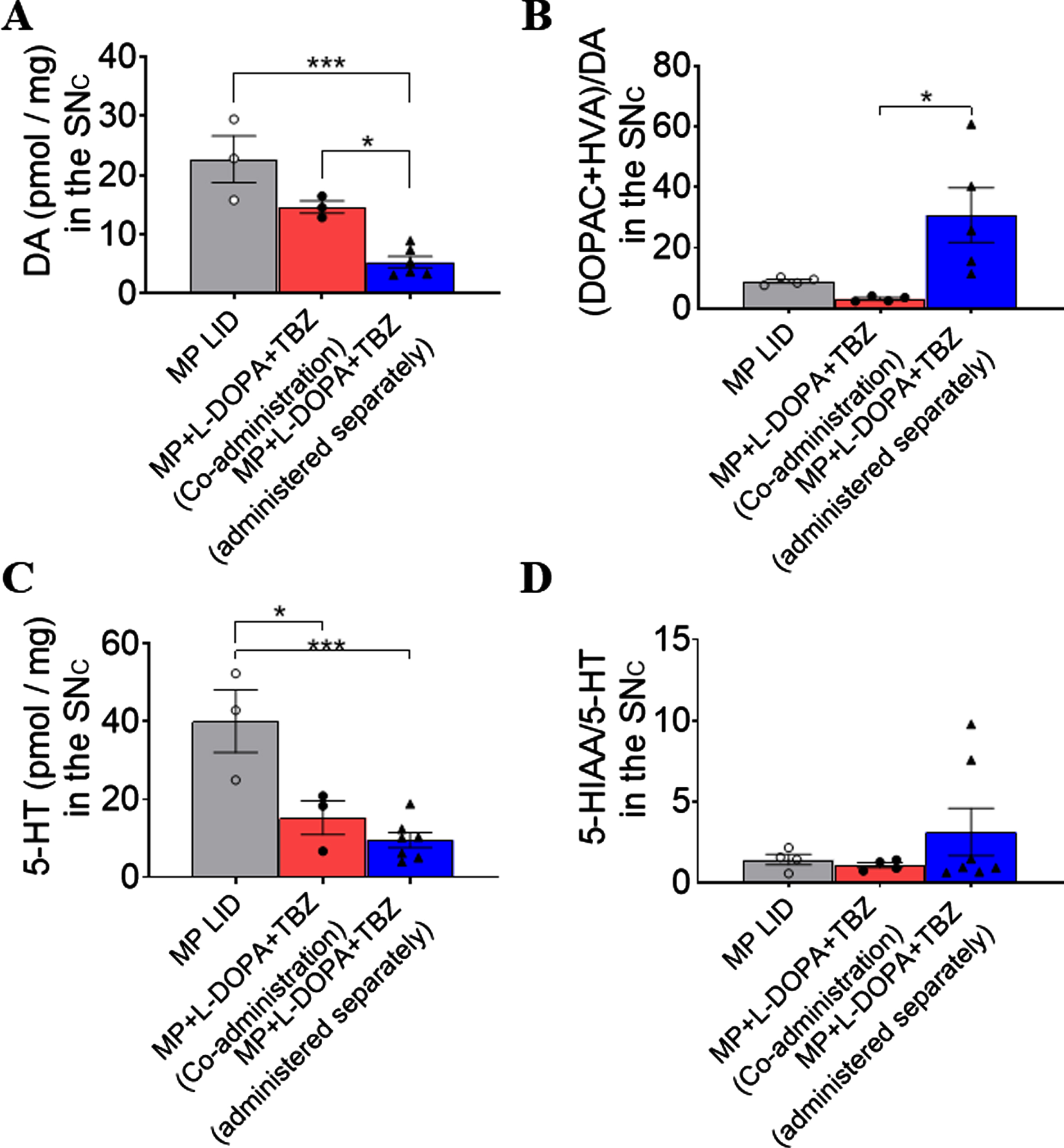
HPLC measurements of DA and serotonin in SNc with different TBZ administration protocols. (A) Tissue concentrations of DA decreased significantly in TBZ and L-DOPA separate administration groups and, (B), turnover rate of DA increased significantly only in this group. (C) The serotonin tissue concentrations were lower in both TBZ administration groups. (D) There was no difference in serotonin turnover between groups.
TBZ administered in the interval of L-DOPA treatment increased DA turnover in the nigrostriatal system of MitoPark mice
Since L-DOPA and TBZ were shown to influence quantal DA release, we determined if chronic treatments with L-DOPA, with or without TBZ, further alter the levels of DA and its metabolites in the nigrostriatal system of MitoPark mice. DA levels were significantly reduced in striatum (Fig. 7A, p < 0.001) and substantia nigra (Fig. 7E, p < 0.05) of MitoPark mice compared to the WT group. Following 2-weeks replacement of L-DOPA/Carbidopa in MitoPark mice (MP-LID group), DA levels were increased slightly in striatum (Fig. 7A, MP vs. MP LID, p < 0.05) and increased considerably in substantia nigra (Fig. 7E, MP vs. MP LID, p < 0.05), respectively. Co- or separately administered TBZ with L-DOPA did not decrease DA levels in striatum, compared to the MP-LID group (Fig. 7A, p = 0.4063). In substantia nigra, L-DOPA-increased DA level was significantly suppressed by TBZ-L-DOPA administered separately (Fig. 7E, p < 0.05), but not co-administered (Fig. 8A, p = 0.1190), suggesting that TBZ specifically given in the interval between L-DOPA replacements can regulate DA levels. In striatum of MitoPark mice, the 3,4-dihydroxyphenylacetic acid (DOPAC) level was decreased (Supplementary Figure 7A p < 0.01), while homovanillic acid (HVA) was increased (Supplementary Figure 7C, p < 0.05), implying that DA denervation disrupts homeostatic control of DA metabolism. L-DOPA/Carbidopa treatment did not significantly alter DOPAC levels in striatum (Supplementary Figure 7A, p = 0.1984) or substantia nigra (SNc) (Supplementary Figure 7E, p = 0.3365) of MitoPark mice. However, an increased level of HVA was found in the substantia nigra of the MP-LID group (Supplementary Figure 7G, p < 0.001), suggesting that chronic DA replacement in MitoPark mice predominantly increases DA and HVA levels in SNc. Similar to the DA levels noted in SNc, separately administered TBZ was shown to suppress L-DOPA-increased HVA levels in the corresponding area (Supplementary Figure 7G, p < 0.001).
DA turnover was examined by comparing DOPAC + HVA with DA. In MP-LID mice there was a significantly increased DA turnover in dorsal striatum (Fig. 7B, WT vs. MP-LID, MP vs. MP-LID, p < 0.01). A similar change was found in animals receiving TBZ administered in the interval of L-DOPA, but not co-administering of TBZ and L-DOPA (Fig. 8B, WT vs. MP + L-DOPA + TBZ (administered separately), MP vs. MP + L-DOPA + TBZ Co-administration, p < 0.001). Importantly, separate administration of TBZ with L-DOPA also significantly increased DA turnover in substantia nigra, compared to WT, MP-LID mice (Fig. 7F, p < 0.05). However, co-administration of TBZ and L-DOPA did not show a similar response (Fig. 8B). Increased HVA/DA ratio was found in striatum of MitoPark mice (Supplementary Figure 7D, p < 0.001), implying that dopaminergic denervation in striatum accelerated DA metabolization into HVA. The HVA/DA ratio in striatum of MitoPark mice was significant reduced by L-DOPA or L-DOPA + TBZ (Supplementary Figure 7D, p < 0.05). Similar to the DA turnover noted above, the DOPAC/DA ratio was increased by TBZ administered separately with L-DOPA (Supplementary Figure 7B, striatum: WT vs. MP administered separately with TBZ + L-DOPA (Supplementary Figure 7F, substantia nigra: WT vs. MP + L-DOPA + TBZ (administered separately); MP LID vs. MP + L-DOPA + TBZ (administered separately), p < 0.05). These data suggest that TBZ, by blocking vesicular storage of DA, promotes DA turnover, especially DA metabolized into DOPAC.
We found that DA concentrations in striatum have a positive correlation with the AIM score at day 14 (r2 = 0.7873) (Supplementary Figure 10A). Linear regression analysis also shows that the DA tissue concentration in striatum has a positive correlation with the AIM score (Supplementary Figure 10B). The DA concentration in SNc also had a positive correlation with AIM score (Supplementary Figure 10C). Linear regression analysis also shows that the DA tissue concentration in SNc has a positive correlation with AIM score (Supplementary Figure 10D).
Finally, we examined if TBZ or L-DOPA also affects serotonin (5-HT) and its metabolite (5-HIAA). There was no difference in 5-HT levels in striatum between groups (Fig. 7C). However, in substantia nigra, chronic treatment with L-DOPA/Carbidopa in MitoPark mice increases the 5-HT level, which was suppressed by TBZ administration (Fig. 7G). There was no difference in 5-HIAA/5-HT ratio in striatum and SNc (Fig. 7D, H), implying that DA denervation, L-DOPA replacement or even TBZ treatment did not markedly affect 5-HT turnover in the nigrostriatal system.
DISCUSSION
Dyskinesia is one of the most problematic complications of advanced PD and the use of high dosage of DA replacement therapy. This is believed to be related to the dopaminergic release “surge” after DA replacement [44]. Here, we used a two-week induction protocol, found sufficient to study AIM in MitoPark mice, and high doses of DA replacement in 22-week-old MitoPark mice to induce dyskinesia. We used FSCV in striatal slices to determine the increment of DA release after L-DOPA administration in parallel with the AIM score. The AIM score, as well as dopaminergic surge, in LID mice could be suppressed by TBZ administration, but only when given separately.
There are several unique properties of the MitoPark model that facilitate this type of study, other than the slow progression of a PD phenotype. First, since this is a genetic model with a strong PD phenotype, there is uniformity between individuals, and lack of variability often seen with toxin models. Second, although controversial, there are reports of little or no behavioral changes upon DA neuron degeneration, and even some spontaneous recovery, seen with both MPTP and PFF alpha synuclein toxin models [45, 46]. Because the loss of TFAM is driven by the DAT promoter, the progressive loss of mitochondrial activity is mostly limited to midbrain DA neurons in MP mice, versus the more generalized toxicity seen with intracerebral administration of neurotoxins.
It has been suggested that gender is an important factor in the development of PD. The disease is more common in men than in woman by an approximate ratio of 1.5–2.1. In addition to prevalence, several other PD-coupled parameters differ between men and women, including onset of symptoms, types of motor and non-motor symptoms, medication use, the effect size of PD risk factors, levodopa bioavailability, neuropsychiatric, and cognitive changes, development of hallucinations, caregiver utilization and reliance, and the quality of life. In women, the age of PD onset has shown a positive correlation with fertility. Sex hormones, especially estrogen, may thus influence PD pathogenesis and be an important gender differentiation factor. We have previously shown that synthesis, release, and reuptake of DA are all reduced in MP mice, prior to loss of the neurons themselves [47]. We showed that progressive loss of these phenotypic properties in MP mice is found earlier in males than females [47]. We also showed that the loss of release, reuptake and TH are all correlated with loss of motivated and spontaneous behaviors that occurs earlier in male than female MP mice. We hypothesized that the sex difference in MP mice relate to female sex hormones. This hypothesis was supported by our previous findings that after ovariectomy, female MP mice showed progressive changes in DA presynaptic dynamics, such as release and reuptake as well as behavioral impairment with time courses similar to those of male MP animals, in contrast to the delayed changes seen in intact MP females [47]. Because of these considerations as to gender differences, we used only male mice in the present study to eliminate the confound of estrogen effects on the time course of midbrain DA changes in MP mice.
In our study, AADC and VMAT2 in the serotonin system may play crucial roles in DA production and higher concentrations of DA release after L-DOPA. This could be shown not only in the FSCV subsecond release patterns but also in HPLC measurements for tissue concentrations. PET scans also showed that SERT activity increased in LID mice rather than in MP mice. Thus, LID may be related to long term elevated DA generation after L-DOPA administration, which could induce neural plasticity in dopaminergic circuits and activity in the D1 pathway shown here in LID mice.
We first characterized the levels of DA/5-HT and their metabolites in the nigrostriatal system of MitoPark mice. Lower DA levels, but a higher HVA/DA ratio in striatum was observed in MitoPark mice, and has also been seen in the 6-OHDA-induced rat model of PD. However, in contrast to 6-OHDA lesioning, there was no increase in DOPAC or DOPAC/DA observed in striatum of MitoPark mice, implying that the increase in HVA and HVA/DA might reflect increased glial COMT expression, or an increase in the number of glial cells expressing COMT in the DA-denervated MP striatum. Of note, we found that an increase in HVA, but not HVA/DA was accompanied by increased DA levels in substantia nigra of MP-LID mice, suggesting that a certain subpopulation of glial cells in substantia nigra might also contribute to clear excess DA derived from L-DOPA replacement. Similar to the findings that PD patients with dyskinesia have higher DA turnover in striatum, the increased ratio of (DOPAC + HVA)/DA was observed in MP-LID mice, supporting the view that increased AIM score correlates with increased DA turnover. Although TBZ given separately with L-DOPA replacement could reduce the AIM scores, it did not suppress L-DOPA-increased DA turnover in striatum. Instead, it increases DA turnover in substantia nigra. These data support that TBZ and L-DOPA given separately could enhance clearance of excess DA in substantia nigra of MitoPark mice receiving 2-weeks of L-DOPA, although the effect of increased DA turnover in substantia nigra on LID development need to be further investigated.
Recently, there has been an increasing interest in the presynaptic mechanisms of LID development as excellently reviewed [44]. Therefore, we utilized FSCV to determine extracellular DA levels with tonic or phasic firing of neurons in DA-denervated and control striatum exposed to L-DOPA. Both increments of tonic or phasic DA release induced by L-DOPA in the MP-LID group was higher than in untreated MP mice and still higher than in WT animals. Moreover, this change in tonic and phasic DA release induced by L-DOPA was parallel to the dyskinesia in MP-LID mice. In the WT group, TBZ was shown to diminish only phasic DA release in striatum, similar to previous data observed in accumbens in free-moving rats. However, in MP-LID mice, the L-DOPA-induced increments in both tonic and phasic releasing of DA were suppressed by TBZ infusion to the basal levels seen in WT mice. Taken together, these data suggest that DA denervation with subsequent L-DOPA treatment leads to unregulated DA release after L-DOPA challenge, which could be normalized by TBZ modulating phasic release of DA related to synaptic activation and by tonic release dependent on the intracellular DA pool.
A number of studies in rodents and primates indicate that neonatal loss of nigrostriatal and mesencephalic DA input, using neurotoxin-induced lesions of the midbrain DA neurons, elicit markedly increased serotonergic input and function in striatum. Near-total DA-depleting brain lesions produced in 3-day-old rats by intracerebroventricular injection of the neurotoxin 6-hydroxydopamine led to pronounced increases in striatal 5-HT and 5-HIAA contents 1–8 months later [48–53]. This effect was associated with an increase in in vitro high affinity 5-HT uptake, suggesting that proliferation of new serotonergic terminals had occurred within striatum. Interestingly, MitoPark mice have a loss of midbrain DA function starting in the perinatal period, and also have a modest upregulation of many 5HT receptors [43]. Following a progressive loss of dopaminergic neurons in advanced PD, most L-DOPA conversion to DA, vesicular uptake and release appear to occur with striatal 5-HT axons [54]. Dysregulated release of DA from serotoninergic terminals might cause peak-dose LID because neurotransmission would be dependent on the availability of exogenous L-DOPA rather than DA neuronal activity [55, 56]. Therefore, aberrant serotonergic neuroplasticity, such as increased striatal 5-HT innervation [57], SERT expression [58, 59] and SERT: DA transporter ratio [60, 61], may be associated with LID development. In addition, LID is associated with increased localization of D1 receptors at the surface of medium spiny neurons, which may be caused by impaired receptor internalization and trafficking [62]. The pathological enhancement of the number of D1 receptors at the plasma membrane may contribute to increases in DARPP-32 phosphorylation, observed in response to the administration of L-DOPA, and the modulation of this signaling pathway could effectively suppress LID in animal models [63]. A previous study showed that AADC and VMAT2 in the serotonin system may play crucial roles in DA production for higher concentrations of DA release after L-DOPA [64]. This may be shown not only in our FSCV sub-second release data, but also in the HPLC measurements of tissue concentrations. PET scans also showed SERT activity increased in MP-LID mice rather than in MP mice. Thus, LID may be related to long-term elevated DA generation after L-DOPA administration, which could induce neural plasticity in dopaminergic circuits and activity in the D1 pathway shown here in LID mice. We found that TBZ given separately with L-DOPA/Carbidopa not only decreases D1 receptor overexpression but also its downstream target protein; it also normalizes SERT expression in the DA-denervated striatum. Taken together, these results suggest that the effects of TBZ on LID are due to down-regulation of the D1 receptor signaling pathway and amelioration of aberrantly released DA from serotonergic terminals.
However, the question arises as to what possible mechanisms underlie the lack of efficacy of TBZ when given simultaneously rather than separately with LDOPA/CB. One possible mechanism involves aromatic stacking. The chemical structures of compounds often contain moieties which lead them to participate in drug–drug interactions (DDI); while most have no adverse effects, reduced efficacy is always a possibility. DDIs are often due to the chemistry of the compounds involved [65].
The bioactive component of both L-DOPA and TBZ is their aromatic ring. Non-covalent interactions between aromatic compounds do take place and is referred to as aromatic (π–π) stacking. It is due to the overlap of the π-orbital of the aromatic electron clouds. The structure of both L-DOPA and TBZ lends itself to π–π stacking, thus resulting in an interaction taking place between these two compounds [66, 67]. Basically, in vivo when they are administered at the same time, this direct chemical interaction interferes with the bioavailability of TBZ. Thus, the chemistry of this direct interaction which results in TBZ sitting on top of the L-DOPA instead of going to VMAT2 to inhibit it, may cause changes in DA dynamics.
In conclusion, we show that TBZ administration in the intervals between L-DOPA replacement can suppress the development of dyskinesia associated with long-term L-DOPA treatment in a mouse model of PD (Supplementary Video 6). Hyperactivation of the D1 receptor signaling pathway as well as increased SERT expression caused by repeated administration of L-DOPA in a DA depletion PD model was attenuated by TBZ administered separately. Moreover, we demonstrated that TBZ separately given with L-DOPA could accelerate excess DA clearance in the SNc of MitoPark mice. Of note, an increasing range of tonic or phasic DA release induced by L-DOPA in the MP-LID group was higher than in the MP group, suggesting that chronic L-DOPA replacement results in dysregulated presynaptic control of vesicular loading and uncontrolled release of DA in the striatum following L-DOPA challenge. This abnormity could be normalized by TBZ administration. Although several questions remain regarding the neurobiological basis of the reported effects here, the present work provides a strong rationale for the continued study of this designed regimen of TBZ treatment as a reposition-ready anti-dyskinetic strategy in PD patients.
Footnotes
ACKNOWLEDGMENTS
Supported by The Ministry of Science and Technology of Taiwan, ROC, MOST- 109-2314-B-016 -019 -MY2, MOST-110-2314-B-016-034, and MOST-108-2314-B-016-058-MY2; Medical Research Project grants TSGH-C04-110027, TSGH-D-110074, TSGH-E-110213, TSGH-D109-100, TSGH-D109-099, TSGH-C03-111029, and TSGH-D-111088 from the Tri-Service General Hospital of Taiwan; Medical Research Project grants MAB-D-111005 from Ministry of National Defense-Medical Affairs Bureau; the National Defense Medical Center, ROC, MND-MAB-110-077 and MND-MAB-C-11102-111004, The Swedish Brain Foundation, and the Swedish Research council VR- 2019-01356.
CONFLICT OF INTEREST
The authors have no conflicts of interest to report.
