Abstract
Background:
First-in-human studies to test the efficacy and safety of human embryonic stem cells (hESC)-derived dopaminergic cells in the treatment of Parkinson’s disease (PD) are imminent. Pre-clinical studies using hESC-derived dopamine neuron transplants in rat models have indicated that the benefits parallel those shown with fetal tissue but have thus far failed to consider how ongoing L-DOPA administration might impact on the graft.
Objective:
To determine whether L-DOPA impacts on survival and functional recovery following grafting of hESC-derived dopaminergic neurons.
Methods:
Unilateral 6-OHDA lesioned rats were administered with either saline or L-DOPA prior to, and for 18 weeks following surgical implantation of dopaminergic neural progenitors derived from RC17 hESCs according to two distinct protocols in independent laboratories.
Results:
Grafts from both protocols elicited reduction in amphetamine-induced rotations. Reduced L-DOPA-induced dyskinesia preceded the improvement in amphetamine-induced rotations. Furthermore, L-DOPA had no effect on overall survival (HuNu) or dopaminergic neuron content of the graft (TH positive cells) but did lead to an increase in the number of GIRK2 positive neurons.
Conclusion:
Critically, we found that L-DOPA was not detrimental to graft function, potentially enhancing graft maturation and promoting an A9 phenotype. Early improvement of L-DOPA-induced dyskinesia suggests that grafts may support the handling of exogenously supplied dopamine earlier than improvements in amphetamine-induced behaviours indicate. Given that one of the protocols will be employed in the production of cells for the European STEM-PD clinical trial, this is vital information for the management of patients and achieving optimal outcomes following transplantation of hESC-derived grafts for PD.
Keywords
INTRODUCTION
Cell transplantation therapy is a promising emerging therapy for Parkinson’s disease (PD), a neurodegenerative motor disorder for which there is currently no cure. The principle of cell transplantation is that dopamine producing neurons can be ectopically transplanted into the putamen, restoring the lost dopaminergic supply previously provided by the nigrostriatal pathway. Proof-of-principle studies using dopaminergic progenitors isolated from the developing fetal ventral mesencephalon show that cells can both survive and re-innervate the striatum to produce functional benefit to patients, reducing their medication burden and improving motor function [1–4]. The significant ethical and logistical limitations of using human fetal tissue as a source for the cells provided major challenges to this approach, but other key considerations also include the highly variable outcomes in these studies and generation of graft-induced dyskinesia (GID: a motor side effect of the grafts) in a significant number of patients [5–8]. Parallel advances in the understanding of human embryonic and induced pluripotent stem cells (hESC and iPSC respectively) have produced solutions to some of the ethical and logistical hurdles. The development of robust differentiation protocols has evolved rapidly to the point of imminent clinical trials [9, 10]; a trial of iPSCs has commenced in Japan [11–13], a human parthenogenetic neural stem cells study in Australia [14], and trials are being planned with hESC-derived cells in the EU [15] and US [16]. Two differentiation protocols are employed in this study, one of which has been validated for use in the imminent European STEM-PD trial.
In trying to understand the variability of graft success and evaluate the potential for GIDs occurring, some preclinical studies have explored how ongoing medication may impact on primary neuron graft survival or treatment outcomes [17–26] but as yet, have not considered hESC or iPSC cell sources. The vast majority of patients will have been receiving the current gold standard dopamine replacement treatment, levodopa (L-DOPA), for an extended period of time prior to transplantation and will continue on their pre-surgical drug regimen following transplantation until the graft reaches functional maturity [27]. L-DOPA usage alongside transplantation has several potential impacts, creating a vastly different environment for the graft compared to the complete striatal dopamine denervation achieved in the standard animal models of transplantation (typically the 6-hydroxdopamine (6-OHDA) lesioned rat) [28]. Beyond the functional benefit derived from restored striatal dopamine, prolonged L-DOPA can also lead to dysfunctional cortico-striatal plasticity, neuroanatomical reorganization and blood flow alterations [18, 29–34] leading to the manifestation of L-DOPA-induced dyskinesia, abnormal involuntary movements which have consequently been indicated as a risk factor for GID [17, 35]. In addition, biochemical and in vitro evidence demonstrates that by elevating levels of oxidative stress L-DOPA could have toxic effects on cells [36], supported by in vivo data suggesting a toxic effect on endogenous nigral and exogenously transplanted dopaminergic neurons or neural stem cells [37–40, 41]. Contrastingly, other studies, including from our own group, support clinical post-mortem studies in which there is clear survival of grafts, despite L-DOPA administration [18–20, 40–42], but it remains unclear how hESC-derived transplanted cells might behave in this environment.
Of the three midbrain dopaminergic populations of neurons, it is the A9, largely GIRK2+, nigrostriatal neurons which are the predominant cell type lost in early-stage PD, the others being relatively spared [43, 44]. Residing in the periphery of the graft, GIRK2+ neuronal outgrowth innervates the striatum and is fundamental to graft-induced motor recovery [45, 46]. Importantly this subpopulation appears to be influenced most heavily by the environment they are in [47]. Belinsky et al. (2013) identified dopamine receptors on H9 hESCs throughout their differentiation and demonstrated the role of the D2 receptor in inducing a GIRK2+ phenotype in the resulting dopaminergic population [48]. Dopamine receptors are also expressed on a wide number of inflammatory cells including those in the brain [49]. In a recent detailed study, we demonstrated that post-transplantation L-DOPA treatment increased the immune response to xenogeneic mouse fetal tissue grafts into rat hosts [42]; however, there has been no similar study with hESC-derived transplants.
Based on the findings described above, whilst the effect of L-DOPA on the survival, function of hES or iPSC-derived neurons has yet to be examined, previous data suggests it is unlikely to be wholly detrimental. Nevertheless, we hypothesized that the increase dopamine turnover produced by supplementation with L-DOPA could stimulate the D2 receptors of the developing dopaminergic neurons and induce a greater proportion of dopaminergic neurons of a GIRK2+ phenotype which could be advantageous, producing superior graft function. Importantly, the ability of hESC-derived dopaminergic neurons to take up exogenous L-DOPA, convert it to dopamine and modify its release in vivo, alongside any ability to reduce L-DOPA-induced dyskinesia, remain to be determined. The aim of the current study was therefore to characterize the effect of ongoing L-DOPA-treatment on striatal hESC-derived grafts. To ensure a robust and reliable outcome, we transplanted dopamine neural progenitors from a clinical-grade hESC line (RC17), differentiated in two labs using two different protocols, into the same 6-OHDA rat model of PD. Rats were pre-treated with L-DOPA to establish L-DOPA-induced dyskinesias (LIDs) prior to transplantation, and L-DOPA was then continued throughout the post-transplantation period, allowing determination of the timeline of graft-induced functional recovery, amelioration of any pre-existing LIDs, any development of GIDs post-transplantation and any subsequent effects on histological indices of graft function.
MATERIALS AND METHODS
Experimental design
The experimental timeline is summarized in Fig. 1. Unilateral striatal dopamine depletion was achieved by a 6-OHDA lesion in the right medial forebrain bundle [50]. Inclusion in the study was based on amphetamine induced rotations evaluated three weeks post-lesion. Other motor function tests were then conducted (details below). Behavioral data were then used to allocate the animals to one of two matched experimental cohorts (DE - Differentiation 1 or DL –Differentiation 2). Each of these cohorts was then subdivided into three groups: a lesion-only group (DL: n = 9; DE: n = 10), a graft-only group (DL: n = 9; DE: n = 10), and a graft +L-DOPA group (DL: n = 9; DE: n = 10). In the L-DOPA-treated groups, daily drug administration started four weeks prior to transplantation and was maintained every day for 15 weeks post-grafting (with the exception of days upon which behavior testing occurred). L-DOPA-induced abnormal involuntary movements (AIMs) and on-drug rotations were evaluated every two weeks. Motor function was evaluated in the absence of L-DOPA (24 h after the last dose was administered) at 4-, 8-, 12- and 16-weeks post-transplantation. At the end of the experiment all the animals were intracardially perfused and their brains extracted for histological analysis.
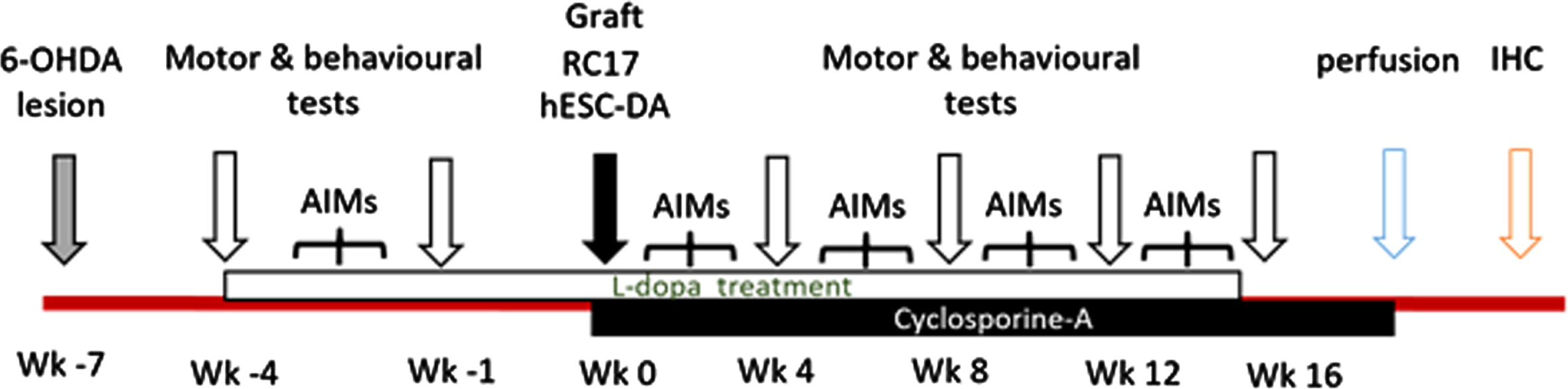
Experimental timeline. Surgical procedures are identified by grey and black arrows, behavioral testing periods in white arrows. Animals were perfused at the end of the study followed by immunohistochemistry. L-DOPA treatment (white bar) daily commenced 4 weeks prior to cell transplantation. Cyclosporine A treatment (black bar) commenced the day before transplantation until perfusion.
Subjects
The experiment involved two cohorts of 27 and 30 female Sprague Dawley rats respectively (Envigo, Bicester, UK). Animals were housed under a 12 h light cycle and had ad libitum access to food (14% protein, Harlan, Bicester, UK) and water. All procedures were conducted in compliance with the UK Animals (Scientific Procedures) Act 1986 and EU directive (2010/63/EU), as well as following approval from local ethical committees at Cardiff University.
Surgical procedures
All the surgical procedures were carried out under aseptic conditions. Rats were anaesthetized with 4-5% isoflurane in a carrier of medical oxygen and anesthesia was maintained at 2-3% isoflurane and 4% nitrous oxide for the duration of surgery. At the start of surgery animals were secured in a stereotactic frame (David Kopf Instruments, Tujunga, CA, USA) with the nose bar set at –4.5 mm For lesion surgeries 3 μl of a 6-OHDA solution (25 mM 6-OHDA + 0.025% ascorbic acid, Sigma) was infused into the medial forebrain bundle of the right hemisphere at the following coordinates from brega AP: –4 mm, ML: –1.3 mm, DV: –7 mm below dura [28, 50]. Following surgery, scalp incisions were sutured, and all rats received 30μl of Metacam (5 mg/ml; Buehringer Ingelheim Lid, Bracknell, UK) and 5 ml 0.9% saline administered via subcutaneous injection (s.c.).
Cell preparation and transplantation
For transplantation, the same surgical procedure was used as described above but the infusion of cells was conducted at the following coordinates from bregma: AP: +0.5 mm; ML: –3 mm; at 2 depths, DV –5 mm and –4 mm, with the nose bar at –4.5 mm. All transplanted animals received daily cyclosporine injection (i.p., Sandimmun® 250 mg/5 ml Novartis, UK) to prevent graft rejection.
Differentiation 1 (DE)
RC17 hESC-DA neurons were generated at University of Edinburgh using the protocol previously described [51, 52] and in detail on protocols.io (dx.doi.org/10.17504/protocols.io.bddpi25n). Cells from day 11 of differentiation were frozen at 24x106 cells per vial in 500μl and stored in a STEM-CELLBANKER (amsbio, 11890) containing ROCK inhibitor and shipped to Cardiff. The cells were then thawed in the presence of ROCK inhibitor and cultured up to day 16 before dissociation with Accutase into a single cell suspension, with a final cell suspension of 250,000 cells/μl in DMEM vehicle. 500,000 cells in total were infused into the right striatum in two deposits.
Differentiation 2 (DL)
RC17 hESC-DA neurons were generated at Lund University as previously described [53]. Cells were used for transplantation on day 16 of differentiation. Cells were dissociated by treatment with Accutase and dissociated cells were prepared as a single cell suspension of 75,000 cells/μl in a vehicle of HBSS with 0.5% dornase alfa (as described in [53]). 300,000 cells in total were infused into the right striatum in two deposits. Different quantities of cells were transplanted from the DE versus the DL batches based on prior experience of these protocols in our lab.
L-DOPA administration and induced behavior
L-3,4-Dihydroxyphenylalanine methylester (L-DOPA; s.c., 6 mg/kg; Sigma-Aldrich, UK) was administered daily. To prevent peripheral conversion of LD to dopamine, benserazide hydrochloride (12 mg/kg; Sigma-Aldrich, UK) was co-administered. Immediately following L-DOPA administration rats were placed into clear perspex bowls and harnessed to automated rotometers to record rotational behavior over 3 h sessions. LIDs were evaluated in L-DOPA-treated animals at 20 min intervals through the 3 h observation period, using the AIMs scale described previously by [54, 55]. In brief the scale allocates a score for the duration and amplitude of abnormal movements in four categories: axial torsion, forelimb dyskinesia, hindlimb dystonia, and orolingual movements. The total score was obtained by multiplying the duration and the amplitude score of each subtype and summing them.
Motor tests
All motor tests were conducted 24 h after the last dose of L-DOPA. Drug induced rotations were induced via administration of methamphetamine (2.5 mg/kg i.p., Sigma, UK), and apomorphine hydrochloride (0.5 mg/kg s.c., Sigma, UK). Automated rotometers recorded rotational frequency and direction across 45 min (apomorphine) and 90 min (amphetamine). Amphetamine rotations were assessed post lesioning, with rats performing an average of 6 full turns a minute considered adequately lesioned [50]. These were repeated monthly. Apomorphine-induced rotational behavior was only conduced at week 16 post-transplantation and not used to determine lesion extent to avoid sensitization of dopamine receptors in non-L-DOPA treated animals.
Basic motor tests evaluated function by determining the percentage response of the contralateral compared to ipsilateral forelimb. For the adjusting step test the body weight is maintained on one forepaw, with the experimenter supporting the rest of the body, while the rat was moved forward along 1 m of bench over 10 s. Right (ipsilateral) and left (contralateral) forelimb steps were counted [56]. For the vibrissae test the paw-placing reflex on the ipsilateral and contralateral side to a light whisker touch was counted [57]. For the cylinder test, rats were placed into a Perspex cylinder (33.5 cm x 19 cm, h x d) and video recorded. The percentage of the first 20 weight-bearing touches to the cylinder with the contralateral forepaw was counted [57].
Perfusion and fixation
On study completion, rats were anaesthetized with sodium pentobarbital (150 mg/kg; Merial, UK, i.p.) and transcardially perfused first with 0.9% phosphate buffered saline then 4% paraformaldehyde (PFA). Brains were removed and post-fixed in 4% PFA solution for 4 h then transferred into 25% sucrose solution. Brains were sectioned at 30μm in a 1:12 series using a freezing microtome and the free-floating sections stored in an antifreeze solution (30% glycerol and 30% ethylene glycol in a buffered salt solution).
Immunohistochemistry (IHC)
3,3′-Diaminobenzidine tetrahydrochloride hydrate (DAB) IHC analysis was conducted as previously described [58] for the following proteins: TH (AB152 Millipore, UK), HuNu (MAB1281, Millipore, UK), STEM-121 (Y40410, Takara Bio, France), GIRK2 (APC 006, Alomone Labs, Israel), CD11b (MCA 275GA Serotec), CD45 (MCA 43R, Serotec), CD68 (MCA 341GA, Serotec), and FosB (7203, Santa Cruz). Briefly, sections were incubated for 1 h at room temperature (RT) with agitation in TXTBS with 3% normal serum, then incubated overnight at RT in a TXTBS 1% serum solution with primary antibodies at the following concentrations: TH 1:1000; HuNu 1:2000, STEM121 1:3000, GIRK2 1:1000, CD11b 1:2000, CD45 1:500, CD68 1:1000, FosB 1:2000. Following three TBS washes sections were incubated for 2 h at RT in TBS with appropriate biotinylated secondary antibodies (Vector Laboratories, UK), and then avidin-biotinylated horseradish peroxidase complex (VECTASTAIN Elite ABC Kit, Vector Laboratories, UK) in TBS for a further 2 h. DAB (Sigma, UK) was used to chromogenically visualize the reaction. Sections were then mounted on gelatinized slides, air dried overnight then dehydrated in graded concentration of alcohol and delipidated with xylene before cover-slipping with DPX solution.
Graft analysis and cell counting
DE grafts
Quantification of TH+ and HuNu+ cells in the striatum was achieved by manual counting, using a Leica light® microscope (20x objective) and corrected with the Abercrombie equation (N =Σ {n x F x T/(T + H)} in which: N = Total corrected number, n = number of the counted cells, F = frequency of the sections (1/12), T = thickness of the sections (30μm), and H = mean diameter of the cells; [61]. The volume of striatal TH and stem-121 was determined by summation of the areas containing TH+ cells or Stem-121 in all the striatal sections of the series, multiplied by section thickness (30μm) and series frequency (x 12). The surface areas were calculated using ImageJ (National Institute of Health, Bethesda, MD, USA) from images captured at 4X magnification.
DL grafts
Estimates of TH+ cell and HuNu+ cell counts were calculated using the optical fractionator method, using an Olympus B 50 Stereology microscope linked to Visopharm software. ROIs were defined under the 4x objective and cell counts were conducted under the 40x objective. The selected dimensions for the frame were 58.7μm on X- axis and 73.46μm on the Y-axis. The number of samples was determined by a step length between samples equal to 283.54μm. The total number of cells was then estimated using the following equation: N = n * A / (a * S) * F * (T / (T + D) in which: N = total estimated number; n = counted number inside sampling boxes; A = region of interest area (graft surface area); a = frame surface area; S = number of samples; F = series frequency; T = section thickness; D = cell diameter. The differing counting methods were necessary due to the difference in graft size between the groups as DE grafts were below the threshold for reliable counting using stereological approaches.
TH fiber outgrowth from the graft border (excluding graft core) was measured in the section located in the middle of the graft to estimate fiber innervation towards the medial or lateral side. Graft images were captured at 10 x magnification using an Olympus B 50. A grid was applied across the image with dimensions of 100*100μm. The percentage of the striatum covered by graft fibers medially and laterally within 600μm distance of the graft border was measured. To selectively focus on the fibers the image thresholding was adjusted in ImageJ, the fiber area was then measured and expressed as a percentage of the total striatal area included in the analysis.
Quantification of GIRK2+ cells in the striatum in both groups was achieved by manual counting, using a Leica light® microscope (20x objective) and corrected with the Abercrombie equation as described above. Striatal FosB immunoreactivity was assessed using Leica light® microscope at 20X magnification on a bright field. Three images were taken in each region of the striatum at the level of the graft and averaged: dorsomedial, dorsolateral, and ventrolateral striatum. FosB+ nuclei were counted using ImageJ 1.51. Optical density of CD11b, CD68, and CD45 was evaluated at one level of the striatum per rat, the striatal area was delineated and the optical density (OD) of the region measured with the corpus callosum used as a control region. Images were obtained with the Leica® light microscope at 4X magnification on a bright field. For all of the inflammatory markers, the evaluation of OD around the graft was expressed as a ratio relative to the OD of the corresponding area of the intact striatum.
Statistical analysis
All statistical analysis was conducted in SPSS 25.0.0.1. Data from the two cell line cohorts were analyzed separately. Normality for all data was assessed using Shapiro-Wilks test, and in instances of violation appropriate non-parametric tests were used. Most behavioral data were analyzed using mixed ANOVA with Session as the within-subjects factor and Treatment (-L-DOPA and +L-DOPA) as the between-subjects factor. Where sphericity was violated degrees of freedom were adjusted using the Greenhouse-Geisser correction. Bonferroni corrected pairwise comparisons were used to investigate significant interactions between factors. AIMS scores were analyzed as RM ANOVA, with Time as the within-subjects factor. Apomorphine rotation data, and all histological data, were analyzed using unpaired student t- tests. Outliers were analyzed through boxplot examination (outliers defined as beyond 1.5xIQR) and retained by default unless removal altered significance, with such instances being detailed. In the DE group one outlier (+L-DOPA), consistent across all behavioral testing, was removed from all analysis.
RESULTS
Postmortem histological analysis confirmed that the MFB lesion resulted in a 98% reduction in TH+ cells in the substantia nigra in the right hemisphere of all animals compared to the intact side. Cell counts in the intact substantia nigra were unaffected by long term L-DOPA administration (Supplementary Table 1 and Fig. 1).
L-DOPA treatment affected graft-induced improvements in motor function
All animals were impaired in the cylinder task with less than 20% of touches being on the paw contralateral to the lesion. In DE grafted rats there was a small reduction in contralateral touches over time, which was comparable between groups treated with saline or L-DOPA (Fig. 2: TIME: F (1,17) 11.731, p = 0.003, DRUG: F (1,17) 0.323, p = 0.577, TIME X DRUG: F (1,17) 0.587, p = 0.454). Animals with DL grafts were impaired prior to transplantation (TIME: F (1,15) 7.517, p = 0.015, DRUG: F (1,15) 11.935, p = 0.004, TIME X DRUG: F (1,15) 8.792, p = 0.010) and did not improve, although a further decrease was observed in the saline treated group at Week 16 (p = 0.003), and at Week 16 there was a difference between the treatment groups (p ≥ 0.001).
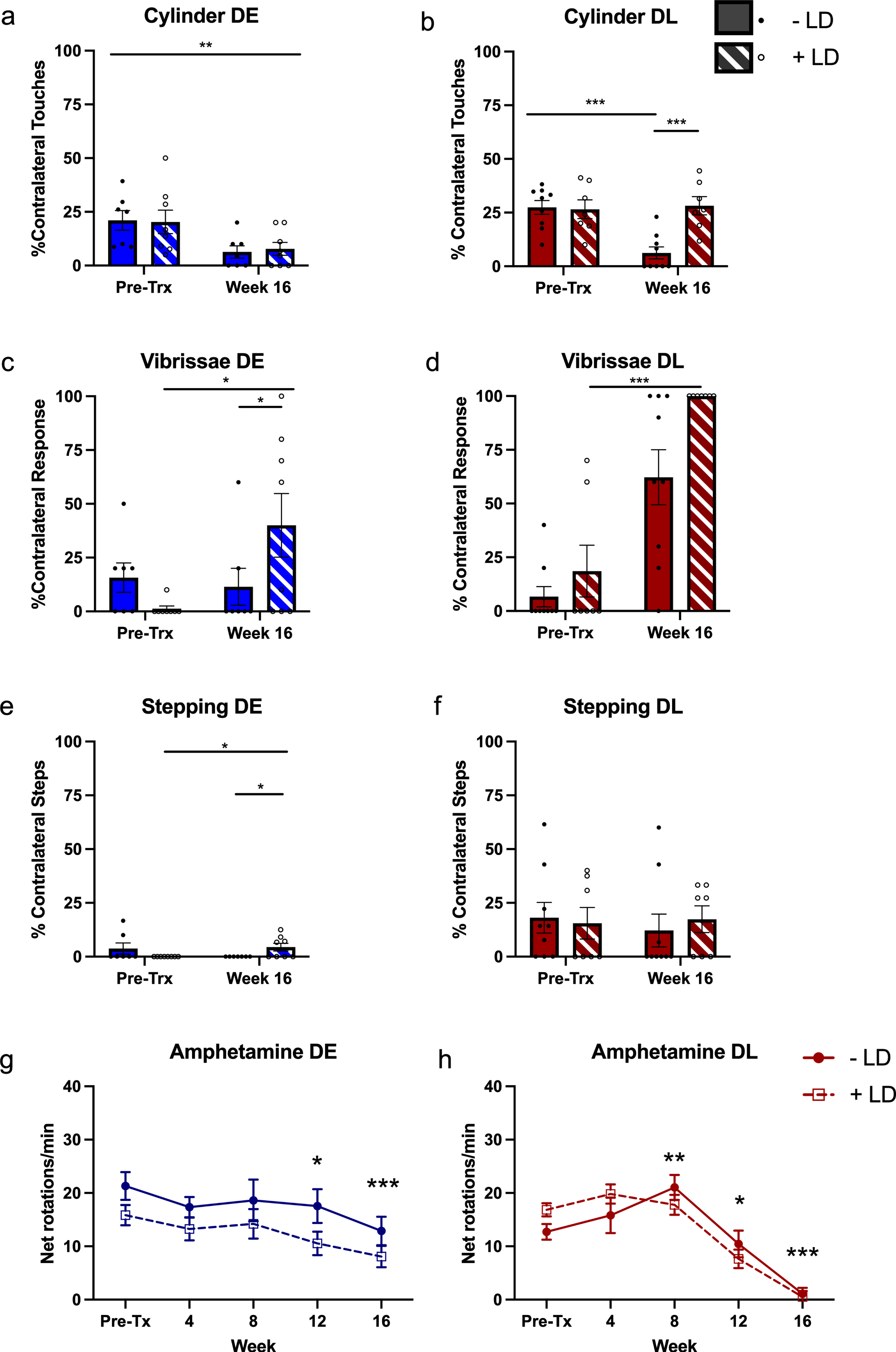
Effects of transplants on behavior. The impact of the grafts at 16 weeks post-transplant, in the presence and absence of L-dopa, was compared to pre-transplant (pre-tx) scores for cylinder (a & b), stepping (c & d), and vibrissae tests (e & f). The effect of L-DOPA treatment (+LD) on amphetamine-induced rotations was analyzed over time (g & h). The two RC17-derived progenitors lines (DE and DL) were analyzed separately. (a) For DE grafts there was an improvement on the cylinder test with time, but no effect of L-DOPA. For both stepping (c) and vibrissae tests (e), there was in improvement in scores of L-DOPA treated rats over time. The stepping +L-DOPA pre-tx and –L-DOPA week 16 scores were zero and so not depicted as bars. For DL grafts there was no improvement in performance in the stepping task (d), but there was an effect of time for the cylinder test (b). The increase at week 16 for the vibrissae test (f), was greater in +L-DOPA treated rats. Amphetamine-induced rotations for both cell lines was unaffected by L-DOPA treatment (g & h). Bonferroni pairwise comparisons determined the effect of time was driven by differences between pre-Tx scores and Weeks 12 (p = 0.015) and 16 (p≤0.001) (DE; g) and Weeks 8 (p = 0.008), 12 (p = 0.011) and 16 (p≤0.001) (DL; h). n = - L-DOPA 10, +L-DOPA 9 (10 for amphetamine) (DE), - L-DOPA 9,+L-DOPA 8 (DL). *p≤0.05, **p≤0.01, ***p≤0.001. Error bars are SEM.
In DE grafted animals motor function in the stepping test was improved in the L-DOPA treated group between pre-transplant and week 16 post transplant (Fig. 2, ANOVA TIME: F (1,17) 0.020, p = 0.891, DRUG: F (1,17) 0.02, p = 0.891, TIME X DRUG: F (1,17) 8.605, p = 0.009. The L-DOPA treated group improved over time unlike the saline treated group (p = 0.044), and there was a separation between the two treated groups at week 16 (p = 0.022). Rats with DL grafts showed no improvement over time and no difference between treatment groups (TIME: F (1,15) 0.231, p = 0.638, DRUG: F (1,15) 0.438, p = 0.518, TIME X DRUG: F (1,15) 0.051, p = 0.825).
The vibrissae test revealed a significant improvement in contralateral responses in L-DOPA treated transplanted groups between pre-transplant and Week 16 in the L-DOPA treated group DE transplanted group (ANOVA TIME: F (1,17) 4.878, p = 0.041, DRUG: F (1,15) 2.154, p = 0.160, TIME X DRUG: F (1,17) 6.707, p = 0.019, post hoc p = 0.023 pre-tx vs. week 16), and a difference between treatment groups at Week 16 only (p = 0.041). The L-DOPA treated group were also found to improve over time in DL grafted animals (TIME: F (1,15) 64.179, p≤0.001, DRUG: F (1,15) 7.028, p = 0.018, TIME X DRUG: F (1,15) 2.629, p = 0.126).
Drug-induced rotations and GID
Both grafts decreased amphetamine-induced rotations over time but there was no effect of L-DOPA treatment (Fig. 2g, h DE: TIME: F (4,72) 9.857, p≤0.001, DRUG: F (1,18) 2.841, p = 0.109, TIME X DRUG: F (4,72) 0.279, p = 0.891; DL: TIME: F (4,60) 59.219, p≤0.001, DRUG: F (1,15) 0.364, p = 0.555, TIME X DRUG: F (4,60) 2.516, p = 0.074). Neither amphetamine-induced dyskinesia, nor spontaneous dyskinesia in the absence of drugs were observed in these animals. Apomorphine-induced contralateral rotations conducted at the end of the experiment did not differ between the two experimental groups (Supplementary Figure 2).
L-dopa-induced dyskinesia and ΔFosB expression were reduced in hESC grafted rats
The total AIMs score for each cell line was analyzed separately and compared between pre-grafting and the final week post-transplantation (Fig. 3). AIMs scores for both DE and DL grafted animals decreased between Pre-Trx and final AIMs scoring sessions (Friedman’s test, χ2 (1) 6.400, p = 0.011) and (χ2 (1) 7.000, p = 0.008) respectively. There was no difference in L-DOPA induced rotations between Pre-Trx and Week 16 for DE grafts (F1,9) 2.131, p = 0.178). By contrast there was a reduction in L-DOPA induced rotations in DL grafted rats (F (1,6) 7.942, p = 0.030).
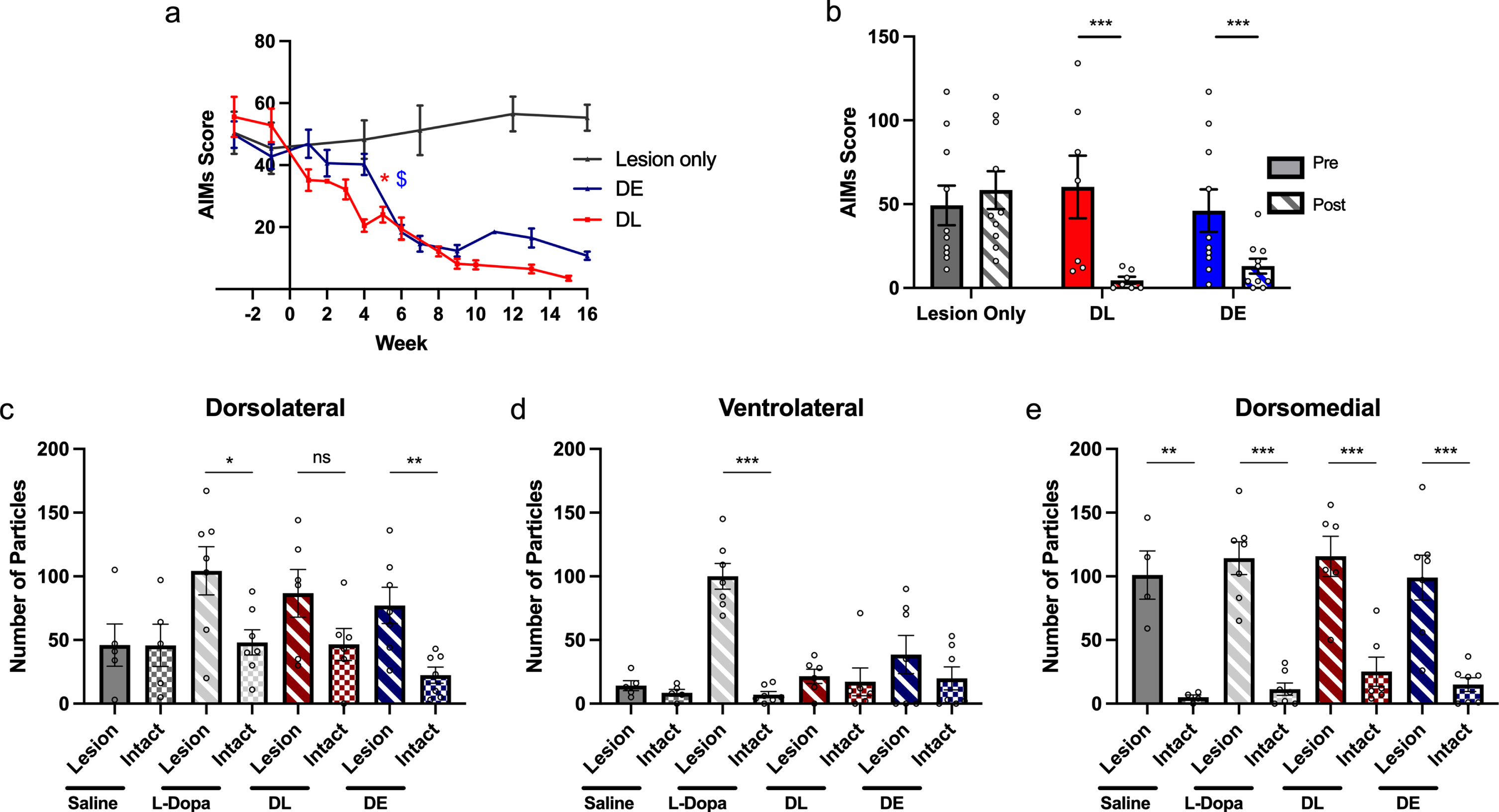
A reduction in AIMS and FosB IHC. Top: a) Representative graph of AIMS scores across time for each cell line. *$ indicates the first timepoint at which each line reaches statistically significant reduction in AIMs compared to pre graft values. Significance in maintained from this point onwards as determined by repeated measures ANOVA. b) The total AIMs scores for both cell lines decrease significantly over time (between Pre-Tx and Week 15/16) despite chronic L-DOPA treatment. The cell lines were analyzed separately. The lesion only group is comparable representative data from a parallel study conducted in the same manner. These animals only received lesioning surgery and L-DOPA treatment at the same dose but without grafting. Unlike the AIMs of the grafted rats the AIMs scores for this group remain high over time. n = 8 (Lesion), 10 (DE), 7 (DL). Expression of ΔFosB was increased in the lesioned hemisphere of the L-DOPA treated controls (from a parallel study) and DE grafted rats in the dorsolateral striatum (c). In the ventrolateral striatum only in the lesioned hemisphere of L-DOPA treated control rats was there an increase in expression (d). In the dorsomedial striatum ΔFosB expression was increased in the lesioned hemisphere of all groups (e). n = 5 (Lesion+Saline) (Dorso/Ventrolateral) 4 (Lesion+Saline) (Dorsomedial), 7 (Lesion+L-DOPA), 6 (DL), 7 (DE). *p≤0.05, **p≤0.01, ***p≤0.001. Error bars are SEM.
ΔFosB expression was analyzed across three regions of the striatum (dorsolateral, ventrolateral, and dorsomedial) and compared between lesioned and intact hemispheres for the DL and DE groups separately using t-tests. In addition, the hemisphere comparison was also conducted for two control groups from a parallel study (lesion+saline - L + S, and lesion + L-DOPA - L + LD) (Fig. 3c-e).
In the dorsolateral striatum, there was increased ΔFosB expression in the lesioned hemisphere of L-DOPA treated rats without grafts which persisted in DE grafted rats but was not seen in DL grafted rats (2-tailed T-test, L + LD: t (12) 2.629, p = 0.0220; DE: t (12) 3.505, p = 0.0043; L + S: t (8) 0.00825, p = 0.9934; DL: t (10) 1.782, p = 0.1051). In the ventrolateral striatum, there was increased ΔFosB expression in the lesioned hemisphere of L-DOPA treated animals only, not evident in those with transplants (L + LD: t (12) 8.996, p = < 0.001; L + S: t (8) 1.181, p = 0.2717; DL: t (12) 0.3535, p = 0.7311; DE: t (10) 1.070, p = 0.3055). In the dorsomedial striatum, ΔFosB expression was increased in the lesioned hemisphere of all groups regardless of treatment (L + LD: t (12) 7.510, p≤0.001; L + S: t (6) 5.060, p = 0.0023; DL: t (12) 4.565, p = 0.0006; DE: t (10) 4.671, p = 0.0009).
No effect of L-dopa on dopaminergic cell number or fiber outgrowth but increased GIRK2 expression
TH+ grafted cells were quantified in the striatum (Figs. 45). Regardless of the smaller grafts in the DE transplants compared to DL, overall L-DOPA treatment had no effect on graft survival (as determined by staining for human nuclei, the number of TH positive cells indicating dopaminergic neurons within the graft, or their innervation (Supplementary Table 1 for statistics). For both cell lines there was significantly more fiber outgrowth medially from the graft compared to laterally, but there was no impact of L-DOPA treatment (DE: REGION: F (1,14) 12.817, p = 0.003; TREATMENT: F (1,14) 0.618, p = 0.445; REGION*TREATMENT: F (1,14) 0.916, p = 0.355; DL: REGION: F (1,16) 24.952, p≤0.001; TREATMENT: F (1,16) 0.293, p = 0.596; REGION*TREATMENT: F (1,16) 0.081, p = 0.780). However, the number of A9-like dopaminergic neurons in the graft (identified with GIRK2, compared separately for each graft line using one-tailed ttest) was affected by L-DOPA treatment (Fig. 6). L-DOPA treatment increased the total number of GIRK2+ neurons in DE grafts (DE: t (13) –2.1192, p = 0.047; DL: t (14) –1.736, p = 0.105), whilst in DL grafts the increase in GIRK2+ cells was not significant but the ratio of GIRK2:TH was significantly increased (DE: t (13) 0.7784, p = 0.2251; DL: t (14) 2.453, p = 0.0139; Fig. 6).
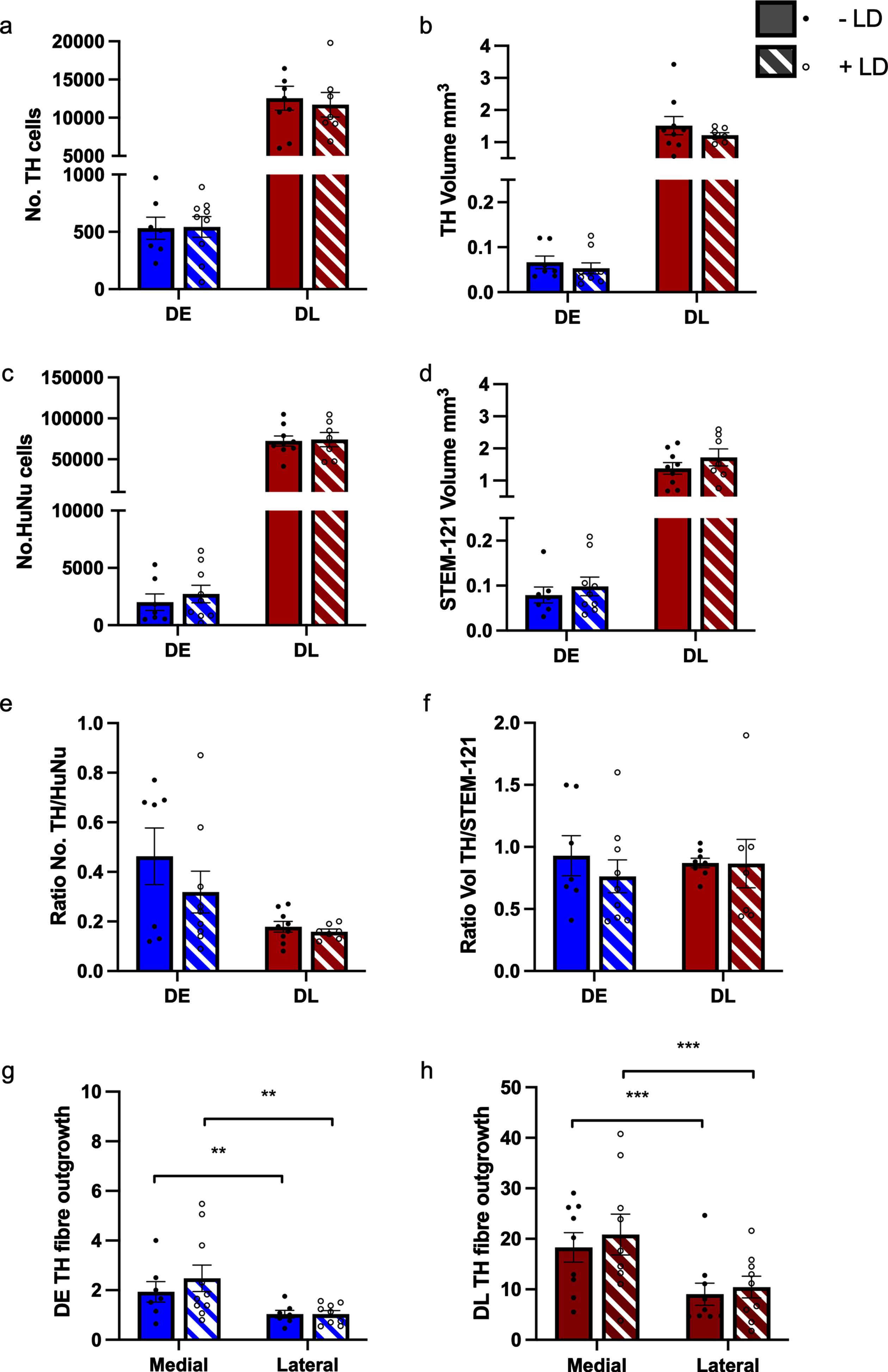
Histological characteristics of grafted striatum. Chronic treatment with L-DOPA had no effect on multiple histological characteristics on grafts derived from DE and DL hESCs compared to untreated grafted animals. a) TH count and b) TH volume; c) HuNu count; d) Stem-121 volume; e) TH/HuNu count ratio; f) TH/Stem-121 volume ratio; g) DE medial and lateral TH fiber outgrowth from the graft and h: DL medial and lateral fiber outgrowth from the graft. n = - L-DOPA 7, +L-DOPA 9 (DE), - L-DOPA 9, +L-DOPA 7 (DL). The cell lines were analyzed separately.
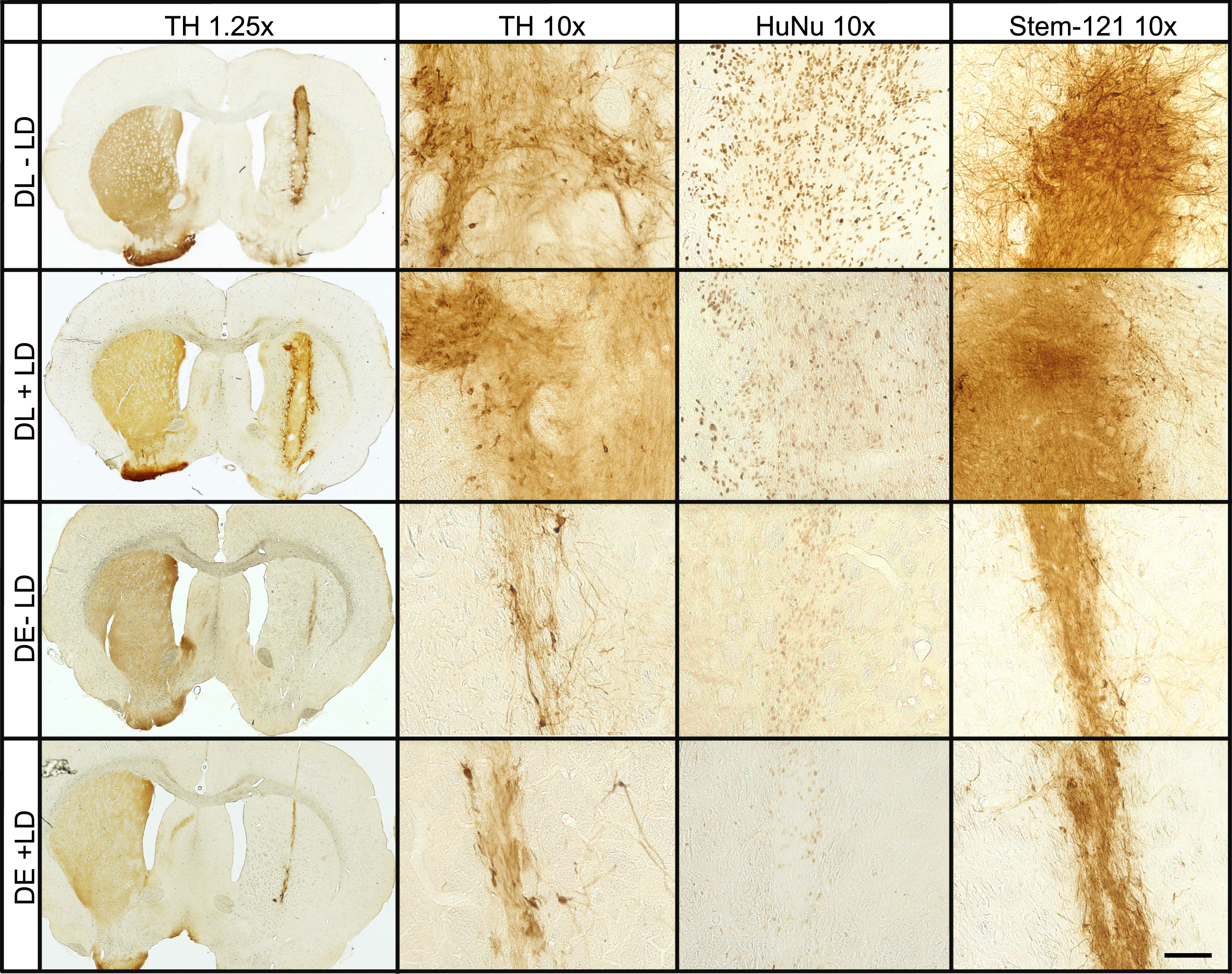
Representative images of grafts. Representative images of striatal grafts from DE and DL derived grafts, with and without L-DOPA treatment, at 1.25x and 10x magnification which have been DAB stained for TH, HuNu, and Stem-121. Each stain was conducted on a separate tissue series. Scale bar represents 50μm.
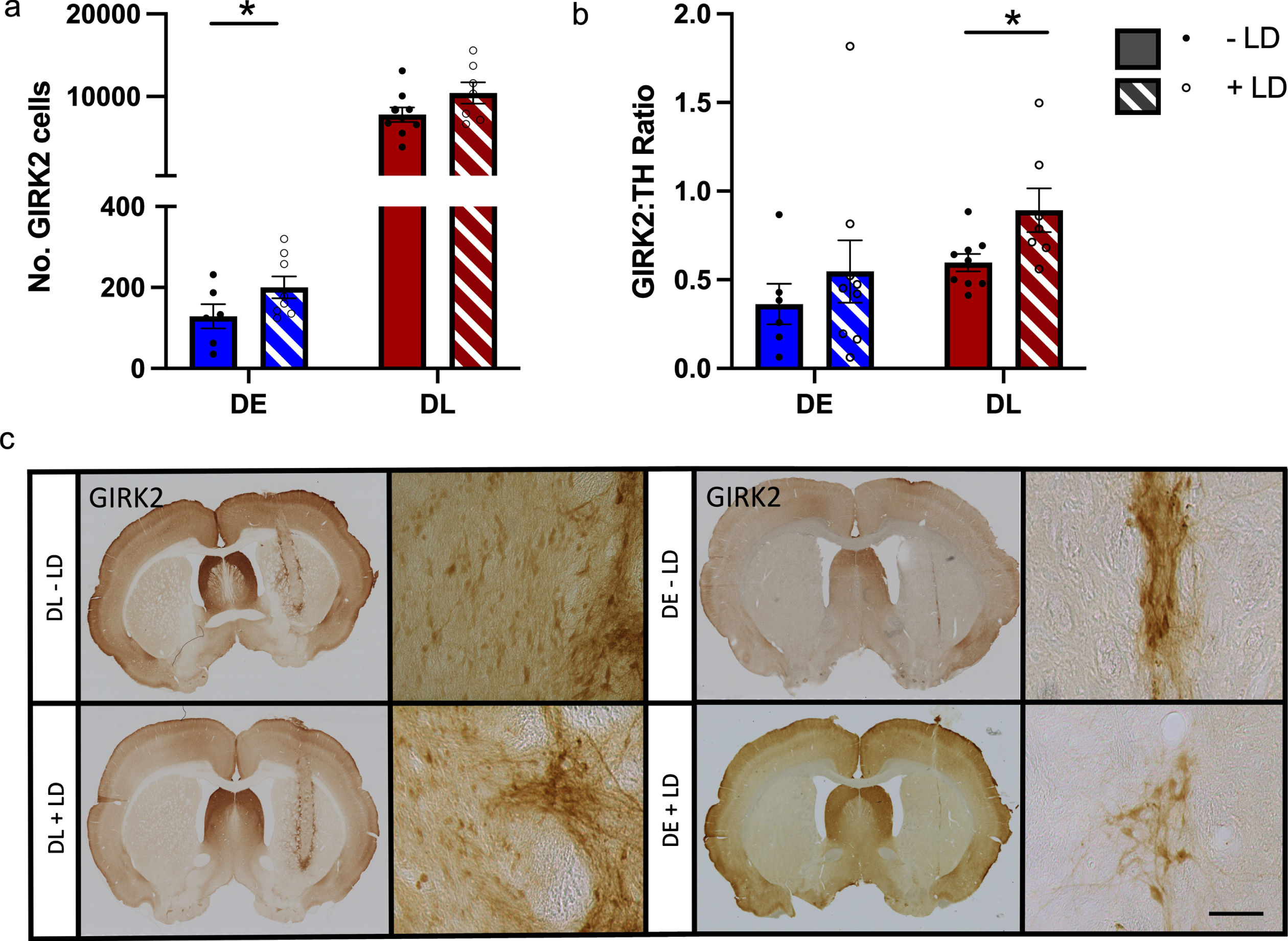
Phenotypic characterization of grafts. Top: Chronic treatment with L-DOPA increased the number of GIRK2+ A9 dopaminergic neurons in the DE derived grafts (a), but had no effect on the GIRK2:TH ratio (b). By contrast there was no effect of L-DOPA treatment on the number of GIRK2+ neurons in the DL derived grafts (a) but there was an increase in the GIRK2:TH ratio (b). n = - L-DOPA 6, +L-DOPA 9 (DE), - L-DOPA 9, +L-DOPA 7 (DL, Count), - L-DOPA 9, +L-DOPA (DL. Ratio). The cell lines were analyzed separately. Bottom: Representative images of striatal grafts from DE and DL derived grafts, with and without L-DOPA treatment, at 1.25x and 10x magnification which have been DAB stained for GIRK2. *p≤0.05. Error bars are SEM. Scale bar represents 50μm.
L-dopa treatment had minimal effect on host immune responses to the grafts
The optical density of three immune markers, CD11b (microglia), CD45 (leukocytes), and CD68 (macrophages), was analyzed to investigate the immune response in the whole lesioned striatum of grafted rats with and without chronic L-DOPA treatment. L-DOPA treatment increased CD11b expression in DE but not DL grafts (2-tailed T-test, DE: t (16) 2.318, p = 0.034; DL: t (12) 0.4649, p = 0.650), but had no effect on CD45 or CD68 expression in either graft group compared to saline treated grafted animals (Fig. 7).
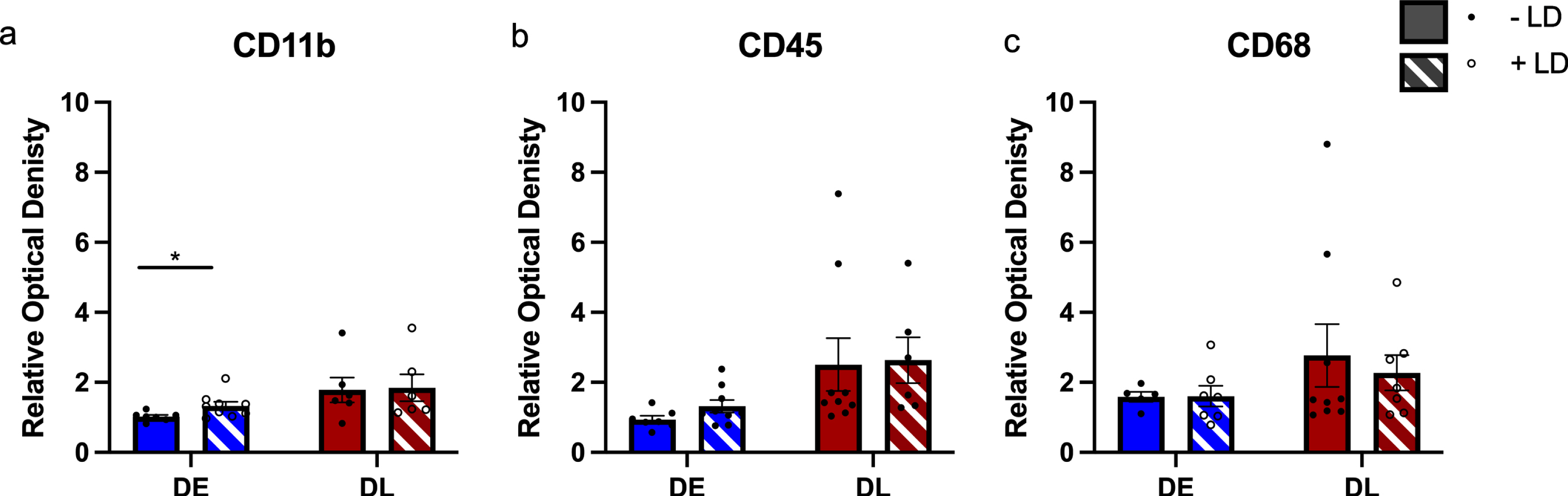
Inflammation in the grafted striatum: The relative optical density of three inflammation markers with and without L-DOPA treatment was investigated. Only in the DE derived grafts was there any change following chronic L-DOPA treatment, and only for microglia marker CD11b (a), but not leucocyte marker CD45 (b) or macrophage marker CD68 (c). The cell lines were analyzed separately. *p≤0.05. n = 7–10. Error bars are SEM.
DISCUSSION
In two parallel experiments we have explored the effect of chronic pre- and post-transplantation L-DOPA treatment on the survival, function, and histological characterization of dopaminergic neuron grafts. Both experiment utilized cells derived from the RC-17 hESC line differentiated using different protocols in a 6-OHDA rodent model of PD with both independent experiments supporting the key findings: 1) L-DOPA has no apparent negative effect on the graft and, consistent with our hypothesis, may support development of the GIRK2 positive A9-like phenotype; and 2) we show for the first time that hESC-derived grafts are capable of ameliorating L-DOPA-induced dyskinesia and that they do so relatively early after transplantation in rodents compared to the manifestation of other functional benefits. Full reversal of L-DOPA-induced dyskinesias was observed even in the DE groups which contained relatively few TH + neurons and which resulted in only partial reduction of amphetamine-induced rotations. These findings indicate that hESC transplantation in a clinical setting may provide amelioration of dyskinesias in L-DOPA treated patients before the onset of any measurable effect on off-state motoric function. Importantly, the DL protocol will soon be employed in the European STEM-PD clinical trial, and this adds further weight to the promising functionality of these cells in clinical scenarios.
Based on the principle that L-DOPA is a fundamental part of a patients’ treatment regime, both prior to and following cell transplantation strategies, our current findings are in excellent alignment with previous in vivo study demonstrating that fetal allogeneic and xenogeneic DA grafts can survive in the host brain following priming with, and ongoing administration of, L-DOPA [19, 37]. The pharmacological challenges applied here enables us to obtain greater insight into the evolution of dopamine handling capability of the striatum as the graft matures. Previous transplantation studies have focused on changes to amphetamine-induced rotations which are an excellent indicator of the ability of the grafted cells to release endogenous dopamine. A reduction, and even reversal, in the overall rotation response occurs within a few weeks with syngeneic rat grafts, takes around 12 weeks with human-to-rat fetal xenogeneic grafts [60] but often in excess of 18 weeks in hESC grafts, which may be due to the earlier developmental stage of the cells at the time of transplantation. Apomorphine, a non-selective dopamine receptor agonist, at low doses selectively targets the supersensitive receptors in the lesioned striatum, inducing contralateral rotations. Reductions in the rotational response provide an indicator of the level of restoration of striatal sensitivity which often occurs later in the post-graft period even in rat-to-rat fetal grafts. In this instance no differences were observed on the basis of L-DOPA exposure.
By tracking the behavioral responses to L-DOPA and postmortem analysis of FosB expression we show both a reduction in the expression of the immediate early gene (often correlated with LID severity [61] consistent with the finding of reduction in L-DOPA-induced AIMs. This suggests that some element of the dyskinetic drive is reduced, whether that is the improved regulation of dopamine derived from the exogenous L-DOPA or normalization of the corticostriatal plasticity associated with these behaviors. Further studies will need to clarify this but as with rodent-to-rodent fetal transplant studies, established L-DOPA-induced AIMS steadily declined following transplantation [17, 62–64]. Interestingly improvements were evident from as early as 5 weeks post transplantation, much earlier than restoration of amphetamine-induced rotation. This indicates that at relatively early stages post-transplantation, prior to the production and release of significant levels of endogenous dopamine, implanted neurons are capable of handling exogenously supplied L-DOPA and managing the release of the consequential dopamine, preventing the dopamine surges that contribute to AIMs manifestation. This is the first evidence, to our knowledge, of an improvement in LID expression following hESC derived dopaminergic grafts and it is worthy of comment that even the relatively sparse innervation of the DE grafts achieved good levels of alleviation of LID much earlier than evidence of reduced amphetamine-induced rotations. This is unexpected and further studies are required to understand how grafts may function to alleviate LIDs. These studies, however, are not without their challenges: with an absolute minimum of 16 weeks required to consistently observed functional improvement following stem cell derived neuronal transplantation into rats (more commonly 18–24 weeks) [12, 46], this contrasts with human fetal tissue dopaminergic transplants which begin to show efficacy at around 12 weeks [60]. Chronic cyclosporin treatment can lead to weight loss and other complications in rodents, adding daily L-DOPA to this regime meant a very cautious approach of daily administration of L-DOPA added to the immunocompromised animals led to the decision to terminate the study at 16 weeks post-graft, shorter than some other hESC studies. Nevertheless, despite this shorter post-graft observation period, graft functionality was found to either be unaffected or enhanced by the administration of L-DOPA. We are confident that we avoided any confounding pharmacological impact of the L-DOPA as, although it improves motor performance in most of these tasks [65, 66], tests were conducted 24 h after the last administration of L-DOPA. L-DOPA has a short plasma half-life and there is no evidence of a long duration response in rodents, unlike in humans [66, 67], allowing the conclusion that the positive effects are indeed driven through the graft rather than residual L-DOPA activity.
At the histological level, in line with the behavioral data there was good survival of both grafts, with no impact of chronic L-DOPA treatment on either total cells surviving, the number of dopaminergic neurons or the area of innervation, critically showing no negative impact of this partially dopaminergic environment on graft development. The slightly later pattern of recovery in DE line, mirrored the DL Lund protocol and is likely explained by the smaller grafts, resulting from the differences in the protocol. While both cell lines were transplanted of d16 of differentiation, the DL cells were transported in culture while the DE line were frozen at d11 for transport and differentiation continued following thawing to d16 prior to transplantation. The differences in protocol and cell number transplanted were based on prior experiences in-house of obtaining good functional grafts with cells from each source. A key aim was not to directly compare the two lines but to determine the consistency of the impact of L-DOPA in both, so the grafts were not intended to be large. Importantly, in both grafts there was evidence of an improved quantity or ratio of GIRK2 + positive neurons following L-DOPA administration. GIRK2 is indicative of the A9 phenotype known to be critical to the restoration of motor function [46], L-DOPA, or the dopamine that is produced, may be having a direct impact on the maturation state of the neurons, again in favor of supporting graft function and echoing in vitro findings [48]. Understanding dopamine flux in the striatum in the presence and absence of a graft would further aid understanding of the impact of the graft of striatal handling of L-DOPA and dopamine. Striatal dopamine levels are negligible following a full medial forebrain bundle 6-OHDA lesion and both LID generation and functional recovery following transplantation have been shown to be closely linked to striatal dopamine efflux. Attenuation of LID is consistent with reduced dopamine levels following L-DOPA administration [68, 69] and functional recovery from grafts conversely related to the increased dopamine release from the grafted cells [70, 71]. Although it is important to note that other brain regions and neurotransmitters such as 5-HT are differentially affected and also linked to LID generation in particular [72, 73].
In addition to the impact of dopamine on the development of the transplanted cells, L-DOPA treatment alone can alter the host environment, triggering inflammatory responses in the striatum [74] making the environment potentially less hospitable for newly transplanted cells. The increased microglial response (CD11b immunoreactivity) observed here, is in line with previous work which demonstrated a heightened immune response around a xenogeneic graft (mouse-to-rat) in L-DOPA treated animals [42]. However, it is worth noting that this was only a small increase in the larger Lund (DL) graft group, in contrast to the immune response focused on leucocytic infiltration of predominantly CD4 + TH cells [42]. It is particularly challenging to explore the effects of the immune system in this model, as the animals are immunosuppressed with CsA for the duration of the experiment to ensure graft survival. Patients receiving transplants will also be immunosuppressed for a period of time but will likely have that immunosuppression withdrawn as was the case for many (although not all) of the previous clinical trials [2, 75]. Further studies are needed to determine the impact of L-DOPA in fully immunocompetent animals, the challenge experimentally will be to have surviving grafts against which we can explore this factor. Whilst extrapolating the exact nature of the immune response may not be clinically meaningful, understanding whether L-DOPA treatment could impact on the likelihood of the transplanted cells to develop into a fully functioning graft is highly relevant to upcoming clinical trials and establishing an understanding of the best therapeutic management of PD in patients with transplants.
Part of the premise of this study was an attempt to understand more about the graft-induced dyskinesia that had been observed in patient trials, specifically in the context of hESC derived grafts. Previous studies suggested that L-DOPA and in particular the induction of LID, might pre-dispose patients with fetal cell transplants to the development of this graft-mediated motor side effect [17, 35]. Despite observation of both spontaneous behaviors and amphetamine induced movements, both of which have been reported as potential models of GID and seen with one of the cell lines described here [19], none of these were observed in this study. The difference may be because of the time point at which the study terminated which was earlier in the development of the graft than other studies. Nevertheless, the lack of overt GID again suggest that these cells have a good safety record ahead of clinical trials and that GID risk appears low.
L-DOPA remains a mainstay treatment for people with PD and, administered upwards of 4 times a day, its ability to regulate the immune system, induce LID and long-term plasticity could interfere with the optimal functioning of novel therapeutic approaches. Our study clearly demonstrates that there is likely to be no negative impact of L-DOPA on transplanted cells but the risk of GID with hESC remains unclear. Post-transplantation medication regimes have not been the subject of much discussion other than to encourage a reduction in medication if motor function improves, but we present evidence here to indicate that there may in fact be a beneficial effect of maintaining some L-DOPA treatment throughout the post-transplantation period. The impact of other dopaminergic or PD medications such as selective dopamine agonists or monoamine oxidase inhibitors remain unknown and should be explored. Patients may be returned to the care of their regular neurologist or care of the elderly physician post-transplantation and clear guidance on how to manage the medication post-transplantation could be key to optimizing the functional recovery that can be achieved. As we move towards clinical trials of both cell transplantation and other forms of restorative or reparative neurosurgical interventions, staying alert to the potential role of ongoing medication (positive or negative) is vital to determining how to achieve the best possible functional outcomes for patients.
Footnotes
ACKNOWLEDGMENTS
The authors would also like to thank the Iraqi Government for their sponsorship of the study. EL was funded by the MRC Regenerative Medicine board, (MR/R00630X/1). TK and ND are funded by the MRC Regenerative Medicine Board Grant (MR/K017276/1).
CONFLICT OF INTEREST
There are no conflicts of interest to declare.
