Abstract
Levo-dihydroxyphenylalanine (L-DOPA) therapy for Parkinson's disease (PD) patients improves motor symptoms, but long-term treatment induces side effects such as abnormal involuntary movements (levodopa-induced dyskinesia – LID). Increasing evidence has linked LID with neuroinflammatory reaction in the striatum involving microglia and astrocytes. The nerve/glial antigen-2 cells (NG2-glia) are recognized as a glial cell whose function in the adult brain is poorly understood. Here, we used immunohistochemistry, confocal microscopy, and western blot to characterize the cellular distribution of NG2-glia in the dorsal striatum of unilaterally 6OHDA-lesioned rats presenting LID. The effect of doxycycline on NG2-glia cells was determined. Dopamine depletion revealed a minor density increase of NG2-glia in the dorsolateral striatum and an activated phenotype. LID development promotes a decrease in NG2-glia cell density and protein level, maintaining the cell-activated feature. Doxycycline antidyskinetic therapy restores the cells to the control density profile. These results indicated the cellular distribution of NG2-glia in the striatum, but the interaction between these cells and other glial types in this model was unanswered and therefore we carried out a double labeling of NG2 and OX-42 (microglia) and GFAP (astrocyte). In LID, there was a decrease in the possible NG2 and OX-42 positive cells interaction, with no effect in the astrocytes. Doxycycline treatment restored the control characteristic. The ability of NG2-glia to regulate their dynamics in response to PD and LID is the first indication of an active link between them. The results suggest that NG2 cells may play important roles other than serving as oligodendrocyte precursor cells.
Summary Statement
NG2-glia alters its dynamics in response to L-DOPA-induced dyskinesia. In these animals, striatal NG2-glia density was reduced with cells presenting activated phenotype while doxycycline antidyskinetic therapy promotes a return to NG2-glia cell density and protein to a not activated state.
Introduction
Parkinson's disease (PD) is a neurodegenerative disorder with pronounced progressive loss of dopamine-producing neurons in the substantia nigra compacta (SNc) (Vaillancourt & Lehericy, 2018). The disease is primarily characterized by motor symptom, including resting tremors, bradykinesia, limb rigidity, and defects in gait and balance (Obeso et al., 2017). Symptomatic therapies are available, mainly dopamine replacement with levodopa (L-DOPA: l-3,4-dihydroxyphenylalanine) improving the motor deficits. L-DOPA long-term therapy leads to the gradual development of on-off fluctuations, abnormal involuntary movements, and hallucinations (Obeso et al., 2000). Abnormal involuntary movements termed L-DOPA-induced dyskinesia (LID) are among the main challenges in treating PD.
There are several nigrostriatal circuitry modifications associated with LID manifestation. Imaging of the PD brain patients reveals widespread microglial activation associated with the pathological process (Gerhard et al., 2006; Ouchi et al., 2005). Ohlin et al. (2011) using post-mortem basal ganglia tissue from PD patients and a brain tissue from a rat model of LID revealed increased angiogenesis, up-regulation of vascular endothelial growth factor, and altered brain-blood barrier properties. Other pre-clinical studies have shown an inflammatory environment in the dopamine-depleted striatum, with sustained activation of astrocytes, microglia, cicloxigenase-2 (COX-2), and recruitment of immune elements contributing to the pathophysiology of LID (Bortolanza et al., 2015a, 2021; Del-Bel et al., 2016; Morissette et al., 2022; Mulas et al., 2016; Zhang et al., 2021). Supporting this hypothesis, long after the neurotoxin 6-hydroxydopamine (6OHDA) has been cleared from the brain, the striatum of lesioned rats receiving L-DOPA treatment displayed sustained neuroinflammation signals (Bortolanza et al., 2015a, 2015b, 2021).
Pharmacological therapy with anti-inflammatory compounds reduced the severity of dyskinesia (Aron-Badin et al., 2013; Barnum et al., 2008; Boi et al., 2019; dos-Santos-Pereira et al., 2016; Padovan-Neto et al., 2009, 2015), associated with a reduction in the inflammation (Aron-Badin et al., 2013; Bortolanza et al., 2015a, 2015b, 2021; Teema et al., 2016). Doxycycline (6-Deoxy-5-hydroxytetracycline, DOXY) is a second-generation tetracycline antibiotic largely used in pre-clinical studies of neurodegenerative diseases due to the anti-inflammatory features (Bortolanza et al., 2018; Santa-Cecília et al., 2016). DOXY protects dopaminergic neurons from degeneration induced by 6OHDA (Lazzarini et al., 2013), 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP, Cho et al., 2009) and lipopolysaccharide (Zhang et al., 2015). The drug suppressed the development of dyskinesia and attenuates established one without compromising the motor benefits of L-DOPA (Bortolanza et al., 2021). Thus, treatment with DOXY targeting neuroinflammation might be a strategy to restrain PD and LID (Bortolanza et al., 2021; Del Bel et al., 2016; Koutzoumis et al., 2020).
The glial reactions to injury may show unusual populations of glial cells known neuron-glial antigen 2 (NG2 glia), also known as chondroitin sulfate proteoglycan 4, oligodendrocyte precursor cells and polydendrocytes (Levine et al., 2001; Nishiyama et al., 2009). NG2 glia constitutes a distinct class of glial cells (Adams & Gallo, 2018; Dimou & Gallo, 2015; Dimou & Götz, 2014) that give rise to mature myelinating oligodendrocytes throughout lifetime (Dimou et al., 2008). NG2 cells are mainly known for their role as developing regulators, but they might have several roles in the adult brain (Dimou et al., 2008). It has been reported the cell's potential to generate astrocytes and neurons besides oligodendrocyte (Aguirre and Gallo, 2004; Baracskay et al., 2007). In the gray and white matter, NG2 glia exhibits differences in proliferation, differentiation rate (Dimou et al., 2008), and physiological properties (Chittajallu et al., 2004). The cells express receptors and ion channels involved in the rapid modulation of neuronal activities that might have functional significance in both physiological and pathological states (Dimou & Gallo, 2015; Dimou & Götz, 2014; Eugenin-von Bernhardi & Dimou, 2016). The cells secrete paracrine factors that can regulate neuroinflammation, synaptic efficacy, myelin thickness, angiogenesis, and blood-brain barrier integrity (Nishiyama et al., 2021).
The NG2-glia cells detect their microenvironment through their constant filopodia extension (Bribian et al., 2018; Martín-López et al., 2013; Nishiyama et al., 1997), allowing them to monitor and response to local changes. These cell populations adjust to pathological processes as neurodegenerative or demyelinating conditions and traumatic injury. They react to inflammatory cues acquire a reactive phenotype and a heterogeneous appearance, exhibiting a behavior remarkably like microglial cells (Wang et al., 2017). This activation phenotype includes a shortening of the cell cycle (Simon et al., 2011), the withdrawal of long multibranched processes into shorter thick processes, and the enlargement of the cell body. NG2-expressing cells specific roles are only beginning to be elucidated. Their functioning has been barely analyzed in PD and LID (Steiner et al., 2006; Zhang et al., 2019).
Here we investigated NG2-glia cell density and activation in the dorsal striatum of parkinsonian rats presenting LID. The striatum presents a dorsal-ventral distinction and a mediolateral-oriented functional striatal gradient (Voorn et al., 2004). The dorsomedial and the dorsolateral parts were analyzed individually since there is distinct functional role associated with goal-directed behaviors and locomotion habit formation respectively (Lermer et al., 2015; Voorn et al., 2004; West et al., 1990). Our data demonstrate that in the lesioned striatum in LID occurred a reduction of NG2-glia cell density, but presenting a reactive phenotype. DOXY treatment decreased LID, and promoted an augment in NG2-glia cell density and protein amount, limiting the reactive phenotype.
Methods
Subjects: Adult, male Wistar rats (n = 64, 250–300 g, aged 9–11 weeks) were used in this study. Animals were housed in groups of three per cage, maintained at a temperature of 22–25°C, on a 12-h light/dark cycle, with food and water (autoclaved tap water) available ad libitum. Studies on sex differences, which may produce biological variables, were not investigated in this study. The experiments were performed in compliance with the recommendations of the US National Institutes of Health Guide for Care and Use of Laboratory Animals and were approved by the Institutional Animal Care and Use Committee of the University of Sao Paulo (Approval Number: 2017-0014-02). All efforts were made to minimize animal suffering and the number of animals used.
Drugs: The dose regimen and route of administration of drugs were based on previously published studies (Cenci et al., 1998; Gomes et al., 2008; Lazzarini et al., 2013; Padovan-Neto et al., 2015). L-DOPA (L-3,4-Dihydroxyphenylalanine methyl ester hydrochloride; 20 mg/kg orally- Prolopa dispersive, Hoffman-LaRoche, Rio de Janeiro, RJ, Brazil), and benserazide–HCl (5 mg/kg) were dissolved in water (gavage administration). DOXY (40 mg/kg i. p., Sigma-Aldrich, St. Louis, MO, USA) was dissolved in saline and administered 30 min before L-DOPA. All drugs and their respective vehicles (VEH) were freshly prepared before use and injected in a volume of 1 mL/kg.
Parkinsonian lesion: Microinjection of 6OHDA was delivered into the medial forebrain bundle as previously described (Gomes and Del-Bel, 2003; Gomes et al., 2008; Padovan-Neto et al., 2009). Animals were anesthetized with 2,2,2-tribromoethanol (Sigma-Aldrich, St. Louis, MO, USA) (250 mg kg-1, i.p.) and fixed to the stereotaxic apparatus for performing the surgery (David Kopf, model USA, 9:57). Stereotaxic coordinates (from bregma in mm: AP = −4.3; LL = −1.6; DV = −8.3) were based on Paxinos and Watson (2004). Rats received microinjection of 6OHDA in a volume of 2 μL into the left medial forebrain bundle (6OHDA - 2.5 µg µL-1 in 0.9% NaCl supplemented with 0.02% ascorbic acid, 1 μL min-1). After the microinjection, the cannula was left in place for two additional minutes to prevent reflux of the injected solution. At the end of the surgical procedure, the animals were kept warm by a 60 W light bulb until full recovery from anesthesia. Dopamine lesion was confirmed by analysis of tyrosine hydroxylase immunoreactivity (TH-ir) as described before (Padovan-Neto et al., 2015) in the striatum and substantia nigra compacta (Figure 1).
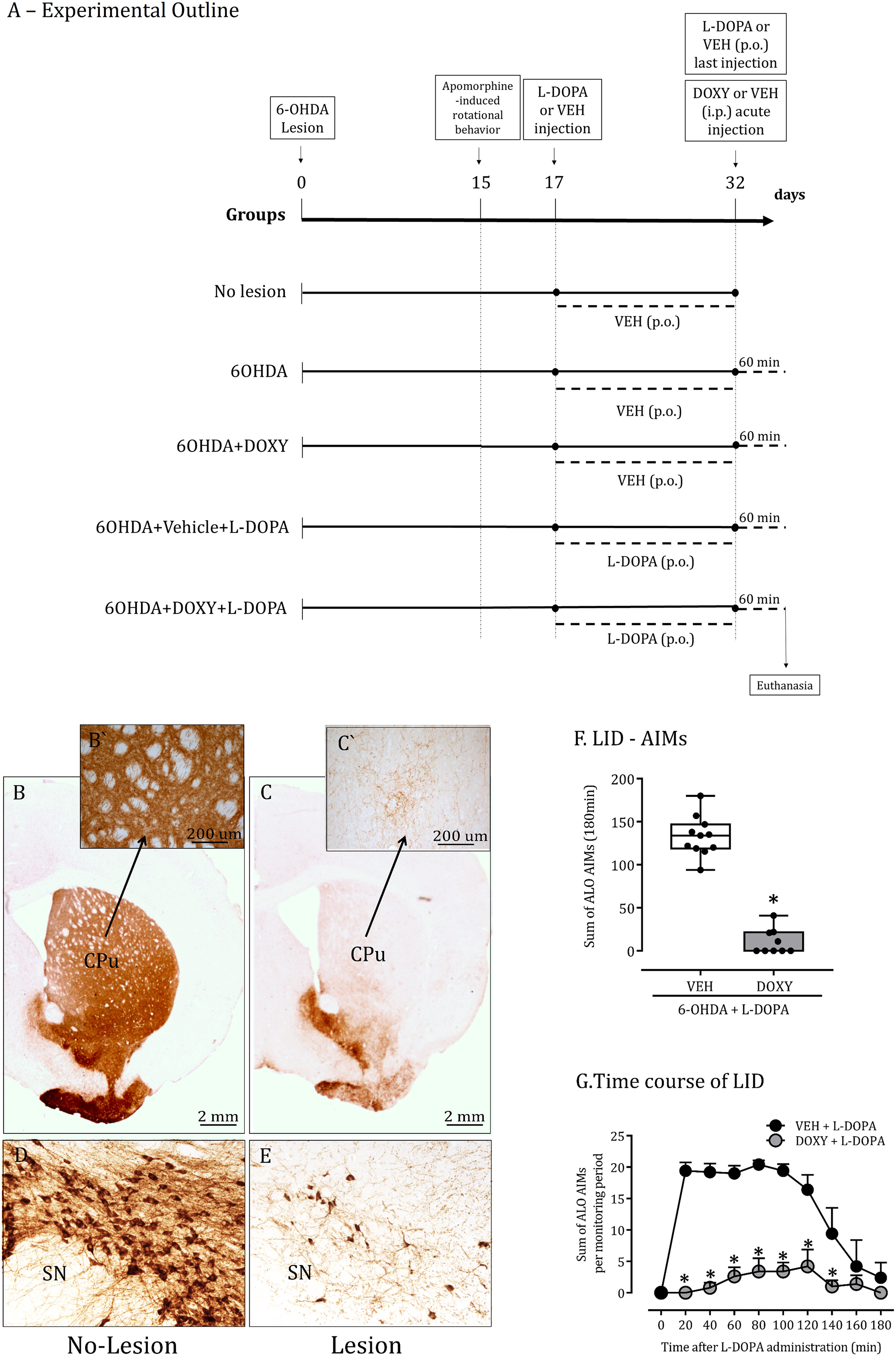
6OHDA nigrostriatal lesion characterization and AIMs scores. A: Schematic drawing summarizing the experimental design. All experimental groups after 15 days of the lesion were submitted to apomorphine test to confirm the lesion (Suppl. Table 1). One group of sham-operated rats was sacrificed 32 days after stereotaxic surgery (No lesion). Two other groups of 6OHDA-lesioned rats treated with Doxycycline (6OHDA+DOXY, 40 mg/kg i.p.) or its vehicle (6OHDA, i.p.) from day 17 until day 32 were sacrificed 90 min after the last DOXY injection. One group of 6OHDA-lesioned rats received daily p.o. injections of L-DOPA (20 mg/kg) + benserazide (5 mg/kg) from day 17 until day 32 and were sacrificed 60 min after the last L-DOPA injection (6OHDA+Vehicle+L-DOPA). The last group of 6OHDA-lesioned rats received L-DOPA in combination with DOXY (6OHDA+DOXY+L-DOPA, 40 mg/kg i.p, 30 min before L-DOPA) from day 17 until day 32 and were sacrificed 60 min after the last L-DOPA injection. Panel
Behavioral Tests
Rotational Behavior: After 2 weeks from surgery, rats were tested for apomorphine-induced rotational behavior (0.5 mg/kg s.c. for 45 min). Only animals with individual means greater than two full turns per min towards the side contralateral to the lesion were selected for the study (Padovan-Neto et al. 2009). Two days later, chronic L-DOPA treatment was initiated.
L-DOPA-Induced dyskinesia: Chronic L-DOPA treatment consisted of daily oral administration of L-DOPA for 14 days. Rats were monitored for LID using a rat dyskinesia scale (Anderson et al., 1999; Cenci and Lundblad, 2007; and modified by Padovan-Neto et al., 2009). Briefly, each rat was scored on a severity scale from 0 to 4 (where 0 = absent, 1 = occasional, 2 = frequent, 3 = continuous but interrupted by sensory distraction, and 4 = continuous, severe and not suppressible) on each of the three subtypes of abnormal involuntary movements (AIMs) – axial, limb, and orofacial (ALO) AIMs (once every 20 min during 180 min after L-DOPA administration. Results are presented as the sum of the ALO AIMs scores and analyzed as one item. Also, a dyskinesia time curve was generated by plotting the L-DOPA induced AIMs score against each monitoring time (20, 40, 60, 80, 100, 120, 140, 160, and 180 min). Animals with AIMs score of >10 and severity grading of >2 were selected for the behavioral analysis (Lundblad et al., 2002; Winkler et al., 2002).
Doxycycline 40 mg/kg (intraperitoneal) was dissolved in saline. Doxycycline or vehicle in all experiments was administered intraperitoneally 30 min before L-DOPA or vehicle. The dosage regimen of doxycycline was based on previously published study by Bortolanza et al. (2021). This doxycycline dose was chosen because it produces a plasma sub-antibiotic concentration in rats (Chtarto et al., 2016).
Immunohistochemical Analysis of the rat Post-Mortem Tissue
Tissue preparation: Rats were anesthetized with tribromoethanol (1.5 g/kg, Sigma-Aldrich, St. Louis, MO, USA) euthanized 60 min after the last L-DOPA administration. The brain was fixed by transcardiac perfusion with 100 mL of Kreb's-ringer buffer and 200 mL of buffered picric acid-paraformaldehyde fixative at room temperature (Somogyi & Takagi, 1982 - 500 mL of 0.2 M sodium phosphate buffer (pH 7.4), 150 mL of saturated picric acid in distilled water; 348 mL of paraformaldehyde solution containing 40 g of depolymerized paraformaldehyde and 2 mL of 25% glutaraldehyde). Brains were dissected and post-fixed in the same fixative for 60 min at 4 °C. The tissues were equilibrated with 30% sucrose in 0.1 M phosphate buffer, frozen in isopentane cooled in liquid nitrogen (−40°C, Sigma-Aldrich, St. Louis, MO, USA) and stored at −80°C until histological processing.
Serial coronal sections throughout the rostrocaudal extent of the striatum (Bregma +2.76 mm, Interaural 11.76 mm – Bregma −2.28 mm, Interaural 6.72 mm) and the SNc (Bregma −4.44 mm, Interaural 4.56 mm – Bregma −6.24 mm, Interaural 2.76 mm) were cut (25 µm) using a freezing microtome (Leica, model CM1850).
Immunoperoxydase labeling of tyrosine hydroxylase (TH): TH immunolabeling to confirm 6OHDA lesion was performed following a standard peroxidase-based method (Gomes et al., 2008; Padovan-Neto et al., 2009). Sections were incubated overnight at room temperature with the primary antibody: rabbit anti-TH (1:4000, PelFreez, 01,229, USA for rats), followed by 2 h of incubation with anti-rabbit biotinylated secondary antibody (1:250, Vectastain, Vector Laboratories, USA). Sections were then incubated with the avidin-biotin-peroxidase complex for 2 h (Vectastain ABC kit, Vector Lab, Burlingame, CA, USA). Immunoreactivity was revealed by a peroxidase reaction using 3,3′-diaminobenzidine diaminobenzidine (DAB; Sigma-Aldrich, St. Louis, MO, USA) as the chromogen. The slices were mounted on slides and coverslipped for microscopic observations.
In-depth analysis of NG2-glia by immunoconfocal morphometry: Immunofluorescent labeling of NG2-glia was performed by incubating brain sections in blocking buffer solution (3% normal goat serum, 0.05% Triton X-100 and 1% bovine serum albumin in phosphate-buffered saline), overnight at 4°C with a rabbit polyclonal antibody to NG2 (1:500; Merck Millipore AB5320, Temecula, CA, USA, Merck Millipore AB5320). After thorough washing, sections were incubated for 90 min at room temperature in secondary antibodies conjugated to Alexa 488 against rabbit (fluorochrome, 1:1000 Invitrogen, A32723). Specificity of primary antibodies was confirmed by the omission of the primary or secondary antibody and absence of immunohistochemical staining in these sections. Brain sections were scanned and photographed by slide scanner (SCN400, Leica Microsystems Ltd., Mannheim, Germany) of a multi-channel confocal microscopy (Leica TCS-SP5, Leica Microsystems Ltd., Mannheim, Germany). Digital images were obtained at a wavelength of 488 nm (green), and the images were converted to TIFF format. Contrast levels were adjusted using Adobe Photoshop v. 10.0 (Adobe Systems, San Jose, CA, USA).
Quantitative analysis of cells was carried out on the medial striatum in the dorsolateral and dorsomedial portion - Bregma +0.96 mm, Interaural 9.96 mm – Bregma −0.24 mm, Interaural 8.76 mm - ×20 objective) of each animal. Results were expressed as the mean density of NG2 glia in pixels. Five areas (160 × 160 μm per field) were chosen in the lesion core of each section and the corresponding striatum. Only glial cells that were on the surface of the sections, where antibody penetration was guaranteed, were counted. Images were digitized with a video camera (Leica DFC420), captured real magnification in gray scale, and evaluated with ImageJ (http:// rsb.info.nih.gov). Integrated optical density (product of area and mean gray value) was averaged over the selected area in each section. For this procedure, the mean gray value was expressed in arbitrary gray scale units, with a scale from 0 to 255 (where 0 represents the most intense labeling), integrated optical density was calculated by multiplying the selected area with the mean gray value. Selected areas consisted of regions of interest measuring 0.1 mm2 . Mean gray value of unstained areas was subtracted from each section to correct for background immunoreactivity. All analyses were performed on the lesioned hemisphere of the brain (right) by a blind investigator.
Quantitative analysis of the ramification index (RI), number of branches, number of junctions, average branch length, and soma area of NG2-glia was carried out in the dorsolateral (DL) and dorsomedial striatum (D) (Bregma +0.96 mm, Interaural 9.96 mm – Bregma −0.24 mm, Interaural 8.76 mm- ×40 objective), according to previous studies (Eder et al., 1999; Heppner et al., 1998). Contrast and brightness of images were uniformly adjusted. A Sholl analysis was performed to quantify NG2-glia morphology in immunofluorescent images of the striatum, according to the following: images were converted into a binary signal and analyzed using the Simple Neurite Tracer plugin of the Fiji software, ImageJ (https://imagej.net/Fiji/Downloads). Cell branches were manually traced, and the centroid of the nucleus was defined. Concentric circles were drawn, starting at 2 μm from the center and increasing diameters of 2 μm. The Sholl analysis plugin was then applied to all traced cells to collect data on the intersections between branches and each increasing circle to create a Sholl plot. For each rat, a mean Sholl plot was generated. Ramification index (RI) is a measure of the ratio of the cell's perimeter to its area (normalized to that of a circle of the same area) and depends on the cell's shape, but not on its overall size. RI was calculated using the formula: R = (perimeter/area)/[2.(π/area)1/2]. The more ramified the cell, the larger R is (Madry et al., 2018). RI was presented as an average of RI of confocal stacks (50 images separated by a 0.25-lm z-axis step size) of NG2-immunolabelled tissue, acquired using a Nikon Plan VC 63x objective (oil immersion). Figure 3 briefly illustrates the reconstruction process. The length of NG2-glia processes was defined as the distance between the soma and the detectable end of an extended process identified by NG2 staining. NG2-glia diameter (soma area) was determined by measuring the cell body's longest axis through the nucleus (µm2).
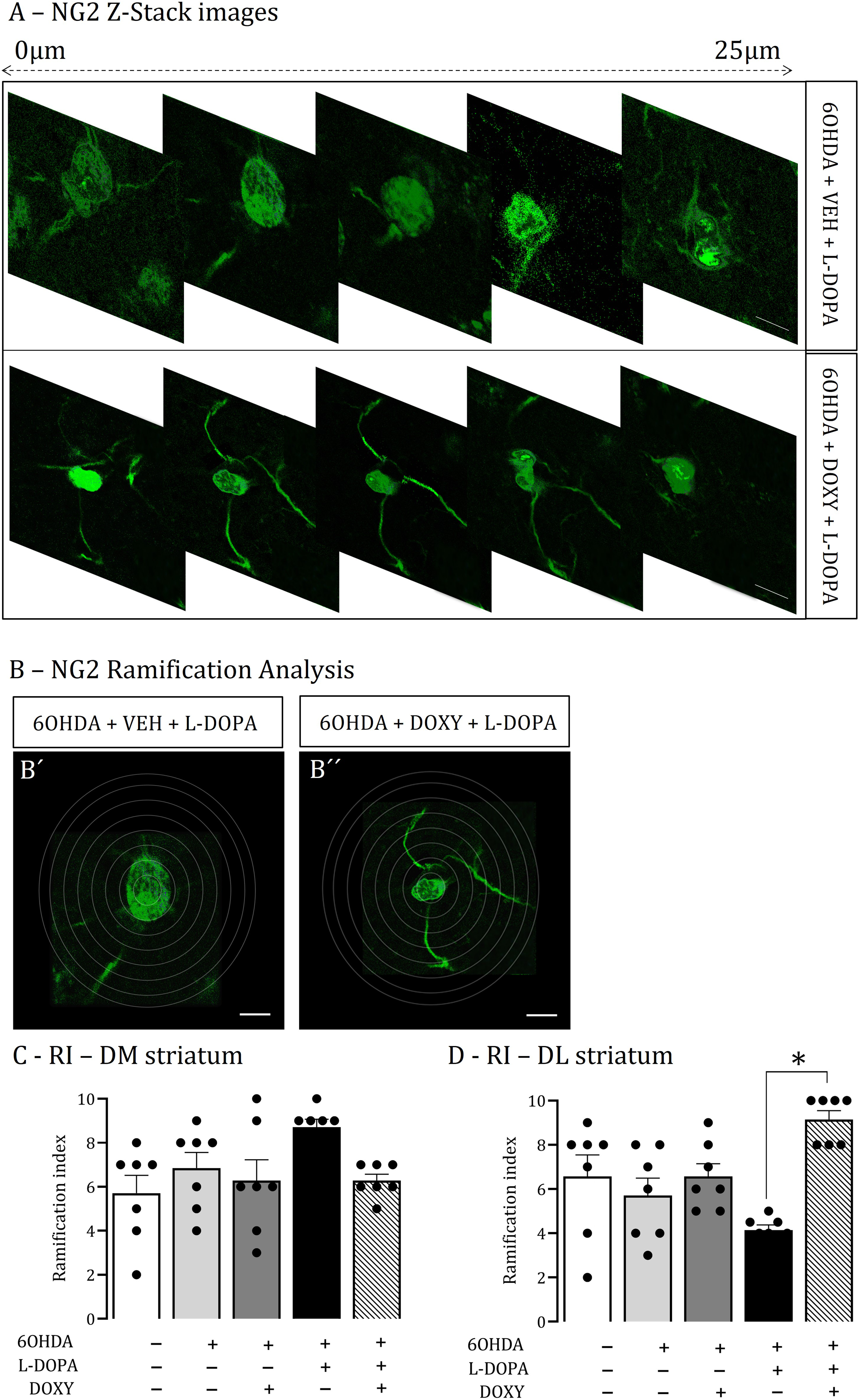
NG2-glia ramification in the striata of rats presenting LID. A: Representative Z-stack projections of confocal photomicrographs showing NG2-glia (green) in the dorsolateral striatum of rats exhibiting LID, either receiving vehicle (Upper panel) or DOXY (Lower panel). Confocal stacks (50 images separated by a 0.25 z step size; 63x magnification; scale bar 20 µm). B: Ramification analysis: representative image of a NG2-glia cell (green, scale bar 20 µm) used for the morphological analysis - detection of the cell with the surrounding Sholl rings in the dorsolateral striatum of rat exhibiting LID treated with vehicle
Dual labeling of NG2-glia and glial fibrillary acidic protein (GFAP, to reveal astrocytes) or OX-42 (CD11b/c equivalent protein of microglia to reveal microglia) was achieved incubating brain sections in blocking buffer solution (3% normal goat serum, 0.05% Triton X-100 and 1% bovine serum albumin in phosphate-buffered saline) overnight at 4°C with a rabbit polyclonal antibody to NG2 (1:500; Millipore, Temecula, CA, USA) and mouse monoclonal antibody to GFAP (1:700; MAB360 Millipore, Temecula, CA, USA) or rabbit polyclonal antibody to NG2 and mouse monoclonal antibody to OX-42 (1:600; Abcam AB33827, Branford, CT, USA). After thorough washing, sections were incubated for 90 min at room temperature in secondary antibodies conjugated to Alexa488 against rabbit and Alexa568 against mouse (fluorochromes, 1:1000 Invitrogen Z25006). The specificity of primary antibodies was confirmed by the omission of the primary or secondary antibody and the absence of immunohistochemical staining in these sections.
The striatum was unilaterally evaluated in the dorsolateral and dorsomedial quadrants. The regions were located according to the coordinates of the Paxinos and Watson (2004) rat brain atlas. Immunofluorescent digital images were obtained using a Leica microscope (Leica Microsystems Launches Leica FW4000 - Cambridge, UK), and were acquired separately for each wavelength [488 nm (green) and 568 nm (red)] and then merged.
For spatial relationships between NG2-positive cells and GFAP and/or OX-42-positive cells analysis, results of GFAP and OX-42 were analyzed as optic density (pixels). The yellow pseudo-color structures visible in the digital immunofluorescent images of the double labeled sections might represent potential points of interaction of NG2-glia immunoreactivity (green) on GFAP or OX-42 immunoreactivity (red) and vice versa.
The striatal area's quantification was measured using the ImageJ system (National Institutes of Health - NIH; Schneider et al., 2012). Striatal NG2, GFAP, and OX-42 immunostaining (optical density) were quantified at 20× magnification.
Western Blotting (WB) Analysis
Tissue preparation
An independent group of animals was decapitated and the dorsal striatum was dissected on an ice-cooled plate, with the help of magnifying lens (Leica Zoom 2000), and immediately frozen in dry ice. Tissue samples were stored at −80°C until use. The sample (200 µL of tissue) was homogenized on ice with sterile saline using a Polytron®PT 1200 handheld homogenizer (KinematicaInc; NY, USA). The homogenate was used for WB measurement.
Immunoblotting analysis
Protein was isolated from the dorsal striatum of both controls and experimental lesioned rats. Samples were treated with boiling lysis buffer (1% sodium dodecyl sulfate, 1.0 mM sodium orthovanadate, 10 mM Tris, pH 7.4). Equal amounts (30 μg) of total protein were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (10%) and transferred to polyvinylidene difluoride membranes. Immunostaining of the blots was performed using two primary antibodies, rabbit polyclonal antibody to NG2 (1:1.000; Millipore) and mouse monoclonal antibody to anti-β-actin (1:10.000; Sigma-Aldrich 49409). Membranes were then incubated with peroxidase-coupled secondary antibodies (1:2.000; Millipore) for 1 h at room temperature. Blots were developed using the Amersham ECL Prime western blotting detection reagent (GE healthcare, Little Chalfont, UK). Densitometric analysis was performed using the Eagle Eye TMII Still VideoSystem (Stratagene, La Jolla, CA, USA).
Experimental design: The experimental groups were described at Figure 1 (Figure 1A). A control experimental group received saline microinjection in the MFB, No-lesion group (n = 7). Experimental 6OHDA-lesioned rats were randomized to one of the following treatment groups: 6OHDA+Vehicle+Vehicle (n = 7), 6OHDA+DOXY+Vehicle (n = 8), 6OHDA+Vehicle+L-DOPA (n = 8), and 6OHDA+DOXY+L-DOPA (n = 8). All the animals were submitted to analysis of the AIMs as an index of LID. Animals were sacrificed one hour after the last L-DOPA treatment to obtain the striatum for immunohistochemistry assessment. Rats treated with DOXY or vehicle received the drug 30 min before the last L-DOPA dose. Independent groups of rats submitted to the same treatments and euthanasia were conducted to obtain the striatum for Western blotting methodology.
Statistical analysis: For sample size calculation, we used population standard deviation of the variable; maximum estimated difference between the sample mean and the true population mean (it is the margin of error or maximum error of estimate), and 95% confidence interval.
Dyskinesia data failed the Kolmogorov–Smirnov test for normality. Therefore, these results were analyzed by a non-parametric approach. The Wilcoxon or Friedman tests were used to verify the effects of time. The Mann–Whitney test investigated the differences between treatments.
A one-way ANOVA, followed by Bonferroni's test, was performed to analyze the immunohistochemical and western blotting results. The statistical analyses were handled using GraphPad Prism software 6 or the Statistical Package for the Social Sciences (SPSS) v.20.0. Data were expressed as the mean ± standard error of the mean (SEM). Values of p < 0.05 were considered statistically significant.
Spearman's rank correlation coefficient was calculated to investigate associations between the global ALO AIMS and NG2 expression (Supplementary material).
Supplementary Table 1 indicate the statistical details performed in this study.
Results
Nigrostriatal Lesion Characterization and AIMs Scores
We examined TH-immunoreactivity in the striatum coronal sections of 6OHDA-lesioned and control animals. TH is the rate-limiting enzyme in dopamine synthesis, widely accepted as a marker of dopaminergic neurons.
Two weeks after neurotoxin microinjection, lesioned rats presented contralateral turns following the apomorphine challenge demonstrating >90% dopaminergic loss (Table 1). It was confirmed by more than 90% reduction in the TH-immunostaining in the striatum and SNc compared to No-lesion group (controls; Figure 1B–E).
Apomorphine Rotational Test Data a .
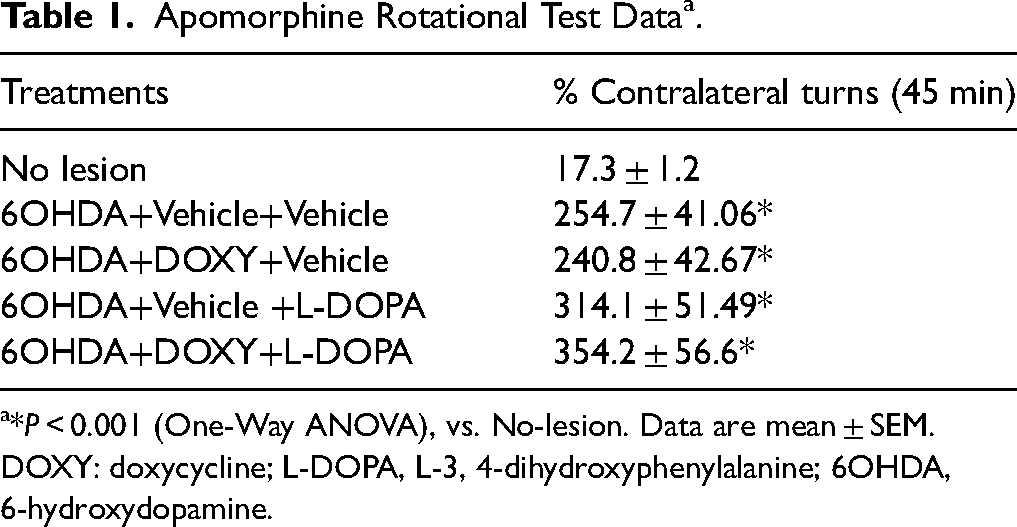
*P < 0.001 (One-Way ANOVA), vs. No-lesion. Data are mean ± SEM. DOXY: doxycycline; L-DOPA, L-3, 4-dihydroxyphenylalanine; 6OHDA, 6-hydroxydopamine.
Dopamine-depleted animals developed AIMs associated to L-DOPA treatment (two weeks), impacting the contralateral forelimb, orofacial movements and axial dystonia (Figure 1F). The maximum total AIMs score was reached 20–120 min after L-DOPA administration and gradually declined to the baseline level over 180 min (Figure 1G).
Administration of DOXY (40 mg/kg, single dose) 30 min before L-DOPA decreased AIMs (79%; Figure 1F–G, 6OHDA+Vehicle+L-DOPA ALO AIMs score = 122.41 ± 24.5 vs. 6OHDA+DOXY+L-DOPA ALO AIMs score = 26.77 ± 10.79, p < 0.001).
AIMs were not observed in both the non-lesioned rats and lesioned rats treated with vehicle or DOXY.
Analysis of Striatal NG2-glia Density in 6OHDA-Lesioned Rats
Animals brain striata were analyzed 32 days after the neurotoxin microinjection. It was determined spatial distribution of NG2-glia in the rat dorsal striatum subdivision of all the experimental groups (Figure 2A–D; see methods).
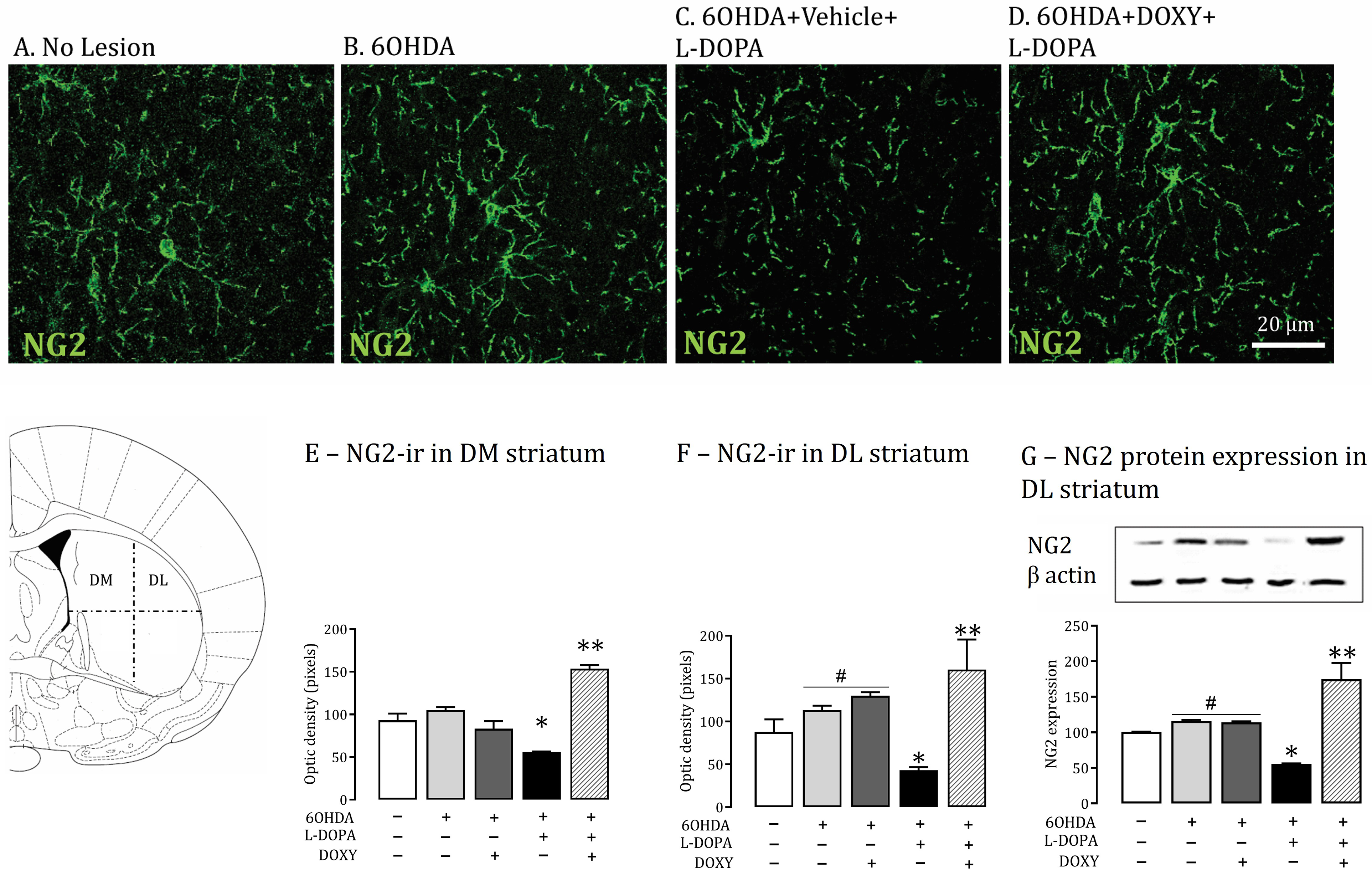
Striatal NG2-glia density and protein levels in 6OHDA-lesioned rats treated or not with L-DOPA. A-D: Representative sections showing NG2-glia immunostaining in the dorsolateral (DL) striatum from rats without lesion (A-No lesion); lesioned with 6OHDA toxin (B-6OHDA); lesioned/dyskinetic rats treated with vehicle (
The dorsolateral striatal region (DL) from the experimental groups 6OHDA+vehicle+vehicle and 6OHDA+DOXY+vehicle showed a slight increase (about 15%) in NG2-glia cell optical density when compared to No-lesion group (controls, p < 0.05; Figure 2F–G); the dorsomedial striatum (DM) presented no change (Figure 2E).
Parkinsonian rats developed LID after L-DOPA treatment and revealed NG2-glia cell optical density reduction in both dorsolateral and the dorsomedial striatum compared to all experimental groups (p < 0.001; Figure 2E–F). DOXY inhibition of L-DOPA dyskinesia has shown an increase in the NG2-glia cells's optical density in both the dorsolateral and dorsomedial striata compared to all groups (p < 0.001; Figure 2E–F).
There was a negative correlation of NG2-glia density with the dyskinesia score (dorsomedial r = −0.83, dorsolateral r = −0.88, Supplementary Figure 1).
Quantitative Analysis of NG2-glia Protein Levels by Western Blot
Immunoblotting demonstrated in dorsolateral striata extract a protein band of about 26 kDa (Figure 2G). Similar to immunohistochemical optical density measurement, lesioned rats treated or not with DOXY showed a slight increase (17%) in NG2-glia protein in the dopamine-depleted striatum when compared to No-lesion animals (p < 0.05, Figure 2G). In contrast, NG2-glia protein levels decreased 50–60% in the lesioned striatum of L-DOPA-treated dyskinetic rats compared to all other groups (p < 0.05, Figure 2G). DOXY treatment increased NG2-glia protein amount by around 76% compared to animals presenting LID (p < 0.05, Figure 2G).
Analysis of NG2-glia Morphology in the striatum in 6OHDA-Lesioned Parkinsonian Rats
The effect of LID on the phenotype of NG2-glia cells has not yet been investigated. Typical quiescent NG2-glia cells in the central nervous system present multibranched processes and small cell bodies. Inflammation or brain injury triggers produce changes in the cell size and the number/length of cellular processes (Kitamura et al., 2010; Nielsen et al., 2013; Nishiyama et al., 1997; Xiang et al., 2015).
It was analyzed the number/length of branches and the soma area (µm2) of NG2-glia in the dorsal striatum of all experimental groups. The NG2-immunolabelled cells ramification index (RI) was determined using a Nikon Plan VC 63x objective (Figure 3A and B), is the average of the 50 confocal stacks images, separated by a 0.25-lm z-axis step size. The dyskinetic rats presented a decrease of NG2-glia RI in dorsolateral striatum (DL), prevented by DOXY treatment (Figure 3D). There is no identified change in the dorsomedial part (DM, p < 0.05, Figure 3C). Parkinsonian rats and other experimental group presented no striata cells RI modifications. Likewise, dorsolateral striatum of rats presenting LID, acquired NG2 cells with fewer branches, shorter branch lengths, and an increase in the soma area, features of a reactive state (Figure 4D–F). DOXY treatment reduced LID and, concurrently, NG2 cells's morphological features of reactivity (p < 0.05, Figures 4D–F and 3A–B). LID development induced no changes in the dorsomedial striatum (Figure 4A–C). In both dorsomedial and dorsolateral striata of parkinsonian rats there was a significative reduction of branch lengths compared to No-lesion group (controls, p < 0.05, Figure 4B–E).
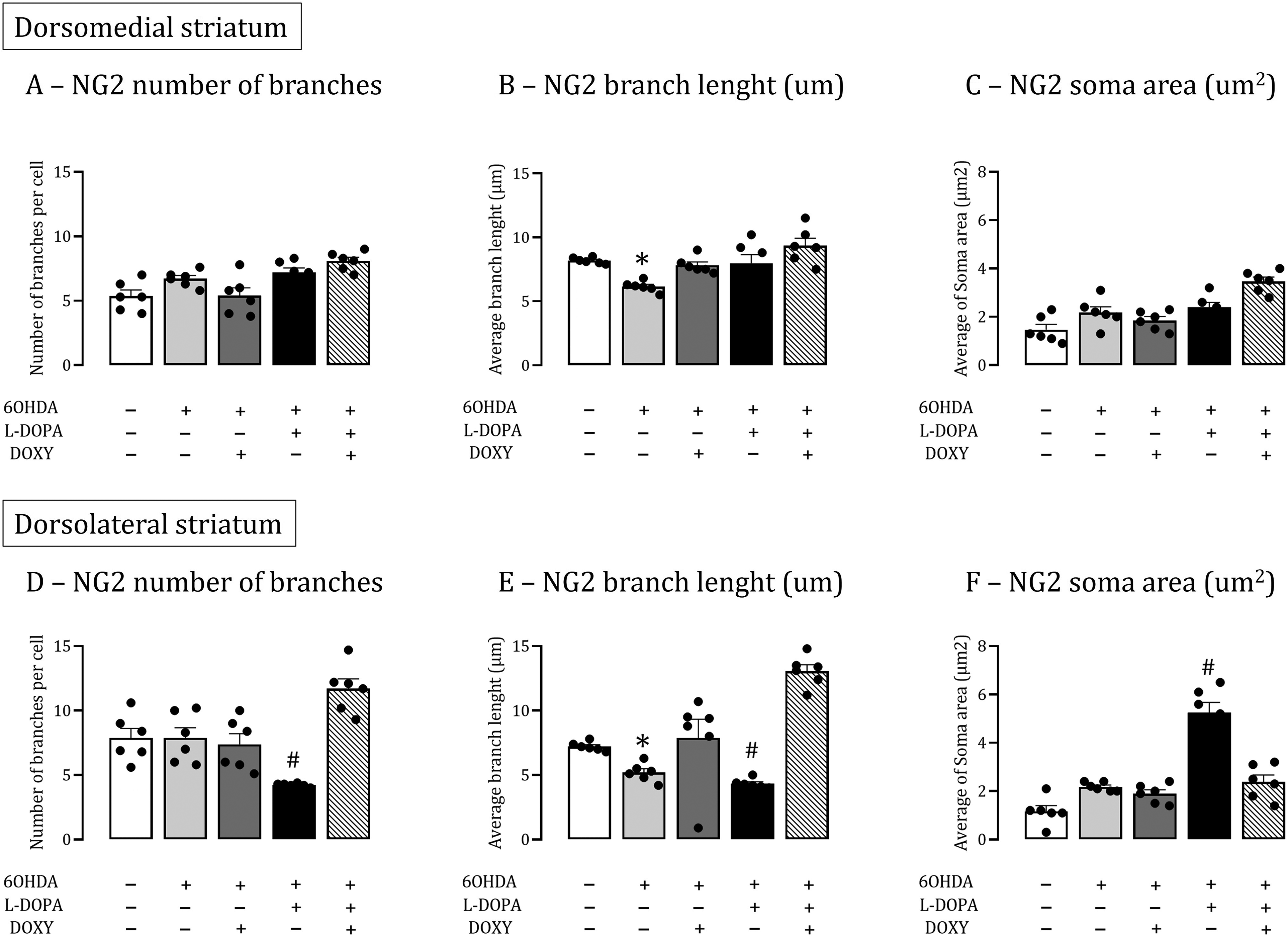
NG2-glia undergoes morphological changes in the striata of rats presenting LID. NG2-glia cells morphometric analysis data summary for DM (A-C) and DL (D-F) striatal areas including number of branches, average branch length and average soma area analyzed. #P < 0.001 (One-Way ANOVA), vs. 6OHDA+DOXY+LDOPA. *P < 0.001, vs. all groups. Data are presented as mean ± SEM. DOXY: doxycycline; L-DOPA, L-3, 4-dihydroxyphenylalanine; 6OHDA, 6-hydroxydopamine. DL = dorsolateral, DM = dorsomedial.
NG2-glia and Astrocyte or Microglia Structures Proximity Analysis
The distribution of the astrocyte marker GFAP and the microglial marker OX-42 immunoreactivity in the lesioned striata in response to LID was in agreement with data published by Bortolanza et al. (2015a). GFAP and OX-42 immunoreactivity increased in response to LID, in the lesioned striata (p < 0.05, Table 2) compared to 6OHDA lesioned animals. DOXY administration reduced the GFAP and OX42 density (p < 0.001, Table 2) compared to rats presenting LID.
GFAP and OX-42 Immunoreactivity in the Dorsolateral Striatum a .

*P < 0.001 (One-Way ANOVA), vs. No-lesion. #P < 0.001 (One-Way ANOVA), vs. 6OHDA+Vehicle+Vehicle. +P < 0.05 (One-Way ANOVA), vs. 6OHDA+Vehicle+L-DOPA. Data are mean ± SEM. DOXY: doxycycline; L-DOPA, L-3, 4-dihydroxyphenylalanine; 6OHDA, 6-hydroxydopamine.
Sections double-stained for NG2-glia and GFAP or OX-42 in the rat brain striata of animals from all 6OHDA experimental groups revealed scarce overlap of their topographies. There were punctate structures with yellow pseudo-color that might represent a superimposition of NG2-glia immunoreactivity (green, Figures 5 and 6) on GFAP(+) (Figure 5B–F) or OX-42(+) immunoreactivity (Figure 6B–F).
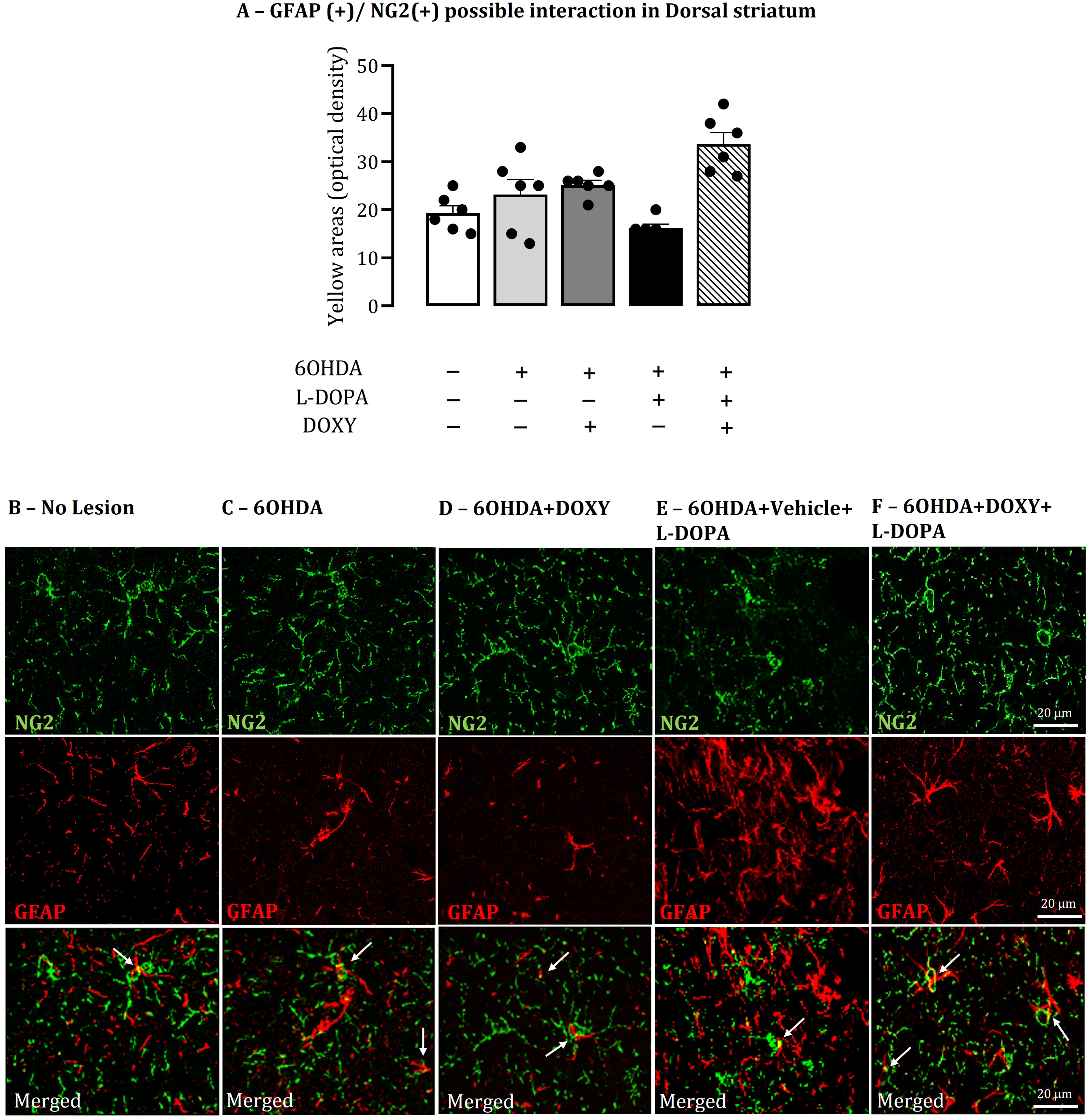
Double-stained of NG2-glia and astrocyte immunohistochemistry markers in the striatum. A: Graph shows the optic density of yellow areas in the total of GFAP in total dorsal striatum (sum of dorsolateral and dorsomedial areas). The yellow pseudo-color areas (white arrows) visible in optical images could represent the superimposition of NG2(+) (green) immunoreactivity on GFAP(+) (red) immunoreactivity and vice versa. B-F: Representative sections showing NG2-glia (green), GFAP (red) staining and a merged image in DL striatum from No lesion, 6OHDA, 6OHDA+DOXY, 6OHDA+Vehicle+L-DOPA and 6OHDA+DOXY+L-DOPA experimental groups of rats (n = 6 per group). Bar represents 20 µm. Data are mean ± SEM. DOXY: doxycycline; L-DOPA, L-3, 4-dihydroxyphenylalanine; 6OHDA, 6-hydroxydopamine. *P < 0.05, 6OHDA+LDOPA vs. all other groups. **P < 0.001, 6OHDA+DOXY and 6OHDA+DOXY+L-DOPA vs. all other groups.
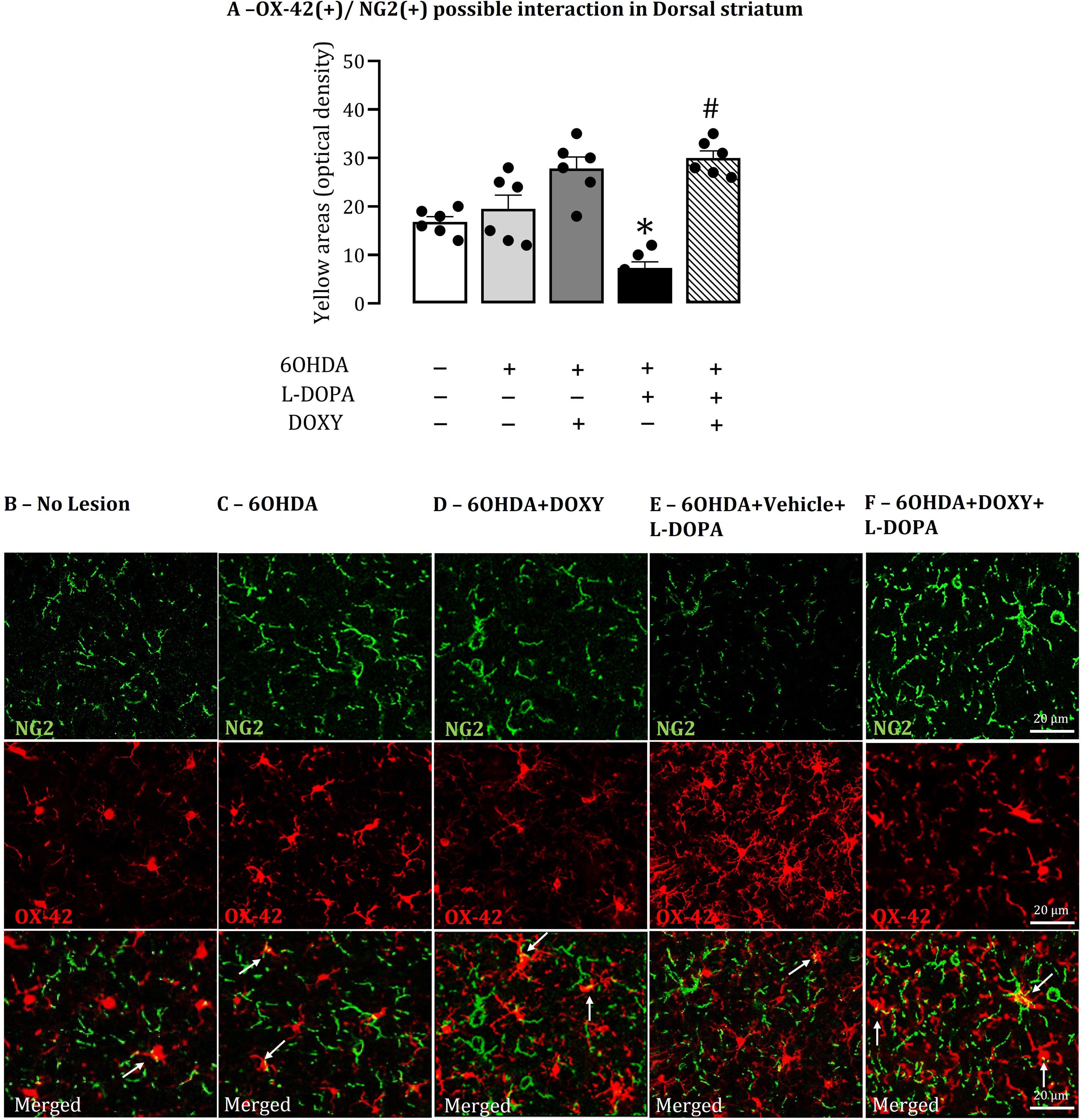
Double-stained of NG2-glia and microglia immunohistochemistry markers in the striatum. A: Graph shows the optic density of yellow areas of OX-42 in the total dorsal striatum (sum of dorsolateral and dorsomedial areas). The yellow pseudo-color areas (white arrows) visible in optical images could represent the superimposition of NG2(+) (green) immunoreactivity on OX-42(+) (red) immunoreactivity and vice versa. B-F: Representative sections showing NG2-glia (green), OX-42 (red) staining and a merged image in DL striatum from No lesion, 6OHDA, 6OHDA+DOXY, 6OHDA+Vehicle+L-DOPA and 6OHDA+DOXY+L-DOPA experimental groups of rats (n = 6 per group). Bar represents 20 µm. Data are mean ± SEM. DOXY: doxycycline; L-DOPA, L-3, 4-dihydroxyphenylalanine; 6OHDA, 6-hydroxydopamine. *P < 0.05, 6OHDA+Vehicle+LDOPA vs. all other groups.
Rats expressing LID exhibit a reduction of yellow pseudo-color punctate structures (p < 0.001; Figure 6A) compared to the other experimental groups, for OX-42 (p < 0.001; Figure 6A) immunostaining, while DOXY treatment induced an increase in the yellow punctate formations in these groups (p < 0.001; Figure 6A). For GFAP immunostaining, there were no statistical differences between the groups.
Discussion
There is little knowledge about the mechanisms that regulate the expression of NG2 in the adult central nervous system. This study is the first to evaluate the effect of LDOPA treatment in parkinsonian rats on the density and the activation state of NG2 cells in the dorsal striata using immunohystochemistry techniques. In evidence, dorsal striata is pathological loci of movement disorders. It controls the motor and cognitive functions, adjusting voluntary movements severely compromised in PD (Flaherty and Graybiel, 1994; Mehlman et al., 2019).
We herein originally report (i) a reduction in the NG2-glia cells’s density and NG2-glia protein level in the dorsal striata of parkinsonian rats presenting LID. The cell morphology indicates a reactive state. DOXY antidyskinetic therapy concurrently promotes NG2-glia cell density and protein increase to values similar to control levels. DOXY treatment restored morphology to the resting state shape. DOXY treatment did not alter the L-DOPA treatment motor improvement. (ii) Small but significant increase in NG2-glia protein level and cell density in the dorsolateral striatum of 6OHDA lesioned rats receiving or not DOXY treatment, compared to No-lesion ones. The cells presented a tiny reduction of process length. (iii) It revealed in LID an inverse amount of the NG2-glia density/activation/protein measure when compared to microglial (OX-42) or astrocyte (GFAP) labeling. In the double-stained sections, it was detected yellow pseudo-color punctate dots. The dots decreased in the lesioned striata of dyskinetic animals in OX-42/NG2 double-labeled sections, and the reduction was reversed by DOXY treatment. The reports discussed here provide the first evidence that NG2 glia regulation in LID.
The exact nature of what regulates NG2-glia density in homeostasis and responsiveness is unclear. As predicted by the dorsal striatal distinct functions our work revealed a distinct recruitment of NG2-glia in dorsomedial and dorsolateral striatum. There was a small but significant increase in NG2-glia protein level and cell density in the dorsolateral striatum of 6OHDA lesioned rats receiving or not DOXY treatment, not observed in dorsomedial striatum, compared to No-lesion one. A NG2-glia reactive phenotype was detectable in dorsolateral striatum from dyskinetic animals that was reversed by DOXY treatment. Reactive NG2 cells displayed characteristic thick and ramified processes reinforcing the possibility that NG2 cells, like other glia, likely undergo changes that coincide with the observed morphological changes following neuroinflammatory reaction. Through elegantly performed two-photon imaging of NG2-glia, Hughes et al. (2013) showed a tight autoregulation of NG2 glial density in the adult cerebral cortex of mice (Hughes et al., 2013). Zhang et al. (2019) reported a decrease of NG2-glia immunoreactivity in the substantia nigra of PD patient brain compared with healthy subjects. Chronic social stress causes NG2 glia density to decrease in areas critical to major depressive disorder pathophysiology (Birey et al., 2015). In mice, depletion of NG2-glia in prefrontal cortex changes glutamatergic signaling and outcomes in depressive-like behavior without significant demyelination (Birey et al., 2015). In the post-mortem brain specimens from patients with Alzheimer's disease, proximity to amyloid-β plaques was associated with retracted processes in NG2 cells (Nielsen et al., 2013). Also, NG2-glia cells are susceptible to various perturbations within their microenvironments. Desu et al. (2021) described that the neuro-inflammation leads NG2-glia to shift toward an immunomodulatory phenotype while reducing their ability to proliferate (Desu et al., 2021). Pro-inflammatory cytokines interleukin-6 and interleukin-1β and corticosterone (Wennstrom et al., 2014) reduce the proliferation of NG2 cells in vivo and in vitro. Mice exposed to chronic stress show decreased NG2 glial cells number by the upregulation of death receptor 6 (DR6), a member of TNF receptor superfamily (Yang et al., 2016). The appropriate potential of the NG2-glia response thus depends on the type of pathological stimulus, the degree of tissue damage, and the milieu changes (Valny et al., 2017), which agrees with the current results.
The importance of neuroinflammation from microglia and astrocyte has recently emerged as a critical component of LID. In the adult brain, NG2-glia, similar to microglia, represents a cell population that responds to inflammation/injury. Reactive NG2 cells displayed characteristic thick and ramified processes reinforcing the possibility that NG2 cells undergo altered gene expression and physiological changes that coincide with the observed morphological changes following neuroinflammatory reaction. DOXY, a tetracyclic antibiotic shows protective effects, proposed to be due to its anti-inflammatory (Bortolanza et al., 2021; Lazzarini et al., 2013; Santa-Cecília et al., 2016), anti-aggregation/accumulation of misfolded proteins (González-Lizárraga et al., 2017; see Bortolanza et al., 2018; Reglodi et al., 2017; Socias et al., 2018) actions. Bortolanza et al. (2015a, 2021) and dos-Santos-Pereira (2016) described in LID an increase in the striatal glial cells, pro-inflammatory cytokines, reactive oxygen species and metalloproteinase activity. Those observations corroborate the presence of an inflammatory state in the striata, attenuated by the DOXY treatment (Bortolanza et al., 2021; Santa-Cecília et al., 2016). In models of synucleinopathies like PD, NG2 cells have been shown to accumulate endogenous α-synuclein and internalize exogenous α-synuclein, leading to a reduction of trophic and metabolic support of neurons (Kaji et al. 2018). González-Lizárraga et al. (2017) reported that DOXY reshapes α-synuclein oligomers, inhibits α-synuclein aggregation and the seeding of new oligomers, attenuate the production of mitochondrial-derived reactive oxygen species thus preventing cytotoxicity in dopaminergic cell lines. In a C.elegans animal PD model, DOXY induces a cellular redistribution of α-synuclein aggregates, an effect associated with a recovery of dopaminergic function (Dominguez-Meijide et al., 2021). The phenotypic diversity of NG2-glia in LID indicates a potential role which needs additional investigation.
NG2-glia did not express markers of mature glial cells, such as the GFAP of astrocytes, the myelin basic protein of myelin-producing oligodendrocytes, or the OX-42 of microglia (Butt et al., 1999; Levine et al., 1993). It indicates that NG2 antibody positive cells are dissimilar from the typically defined types of neuroglial cells, as well as neurons (Levine et al., 2001; Nishiyama et al., 1999). The assessment of sections double-stained for NG2 and GFAP or OX-42 revealed the presence of yellow spots, potential points of interaction between the two markers in the striatum. It might be suggestive of a functional interaction. Interestingly, the yellow spots were almost absent in rats expressing LID, but DOXY antidyskinetic treatment increased their number. Interaction between NG2-glia and astrocytes (Hamilton et al., 2010; Xu et al., 2014), or microglia (McKenzie et al., 2014), and neurons (Du et al., 2021; Maldonado and Angulo, 2015) was previously demonstrated. Often the processes of NG2 cells are intertwined with those of microglia and astrocytes, suggestive of a functional interaction (Nishiyama et al., 1997). The changes in the NG2 glia, directly and indirectly, influence populations of neurons and other glial cells (Jakel & Dimou, 2017; Rodriguez et al., 2014). In Alzheimer disease model, Liu and Aguzzi (2020) demonstrated that NG2-glia and microglia interaction played pivotal roles in regulating microglia states in the adult mouse brain (Sánchez-González et al., 2020). These cells have been shown to be involved in multiple functions in physiological and pathological states in the central nervous system (Dimou and Gallo, 2015; Dimou and Gotz, 2014). Whether all distinct NG2 glia cells can participate in dissimilar functions, and the relevance to human disease is a question to pursue further on.
Conclusion
We herein originally report NG2-glia cells alterations in the striatum in LID in parkinsonian rats. The cells substantially decreased in LID assuming an activated form. DOXY concurrently promoted a return to NG2-glia cell to the resting form, with a diminution of the cell density/protein. It revealed in LID an inverse amount of the NG2-glia density/activation/protein measures when compared to microglia and astrocyte. Our results may have translational significance. Foster advancing our understanding of the cellular and molecular targets regulated by NG2-glia may provide therapeutic targets for PD and LID.
The present study is not without limitations. Our results have been obtained by immunohistochemical staining of NG2 (chondroitin sulphate proteoglycan 4) as a marker of oligodendrocyte precursor cell. Further evaluation of PDGFRα (platelet-derived growth factor receptor alpha) may be considered (Dimou and Götz, 2014; Zhang et al., 2021). Similar, the identification of other astrocytes markers may be used (Chang et al., 2000; Sofroniew & Vinters, 2010). The molecular mechanisms involved in the DOXY upregulation of the NG2-glia have not been determined. Future studies should examine potential sex differences.
Supplemental Material
sj-docx-1-asn-10.1177_17590914231155976 - Supplemental material for Dynamic Involvement of Striatal NG2-glia in L-DOPA Induced Dyskinesia in Parkinsonian Rats: Effects of Doxycycline
Supplemental material, sj-docx-1-asn-10.1177_17590914231155976 for Dynamic Involvement of Striatal NG2-glia in L-DOPA Induced Dyskinesia in Parkinsonian Rats: Effects of Doxycycline by G.C. Nascimento, M. Bortolanza, A. Bribian, G.C. Leal-Luiz, R. Raisman-Vozari, L. López-Mascaraque and E. Del-Bel in ASN Neuro
Footnotes
Acknowledgments
The authors would like to thank Célia A. da-Silva, Sara Saltareli, and Vitor Castania for their technical assistance. We would like to thanks Dr. R. Raisman-Vozari for the incentive and our test experiment realization.
Author Contributions
GCN, MB, AB, GCLL performed the experiments. EDB, LLM, GCN, MB AB contributed to the experimental design. EDB, LLM contributed with resources. GCN, MB, AB, EDB, LLM wrote and edited the manuscript. GCN, MB performed data acquisition and analysis. All authors have approved the final manuscript.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo, Conselho Nacional de Desenvolvimento Científico e Tecnológico, Ministerio de Ciencia e Innovación, (grant number 2014/25029-4, 2017/24304-0, 2015/03053-3 and 2018/, 88882.317597/2019-01, PID2019-105218RB-I00).
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
