Abstract
Background:
The performance on a visual Go/NoGo (VGNG) task during walking has been used to evaluate the effect of gait on response inhibition in young and older adults; however, no work has yet included Parkinson’s disease (PD) patients for whom such changes may be even more enhanced.
Objective:
In this study, we aimed to explore the effect of gait on automatic and cognitive inhibitory control phases in PD patients and the associated changes in neural activity and compared them with young and older adults.
Methods:
30 PD patients, 30 older adults, and 11 young adults performed a visual Go/NoGo task in a sitting position and during walking on a treadmill while their EEG activity and gait were recorded. Brain electrical activity was evaluated by the amplitude, latency, and scalp distribution of N2 and P300 event related potentials. Mix model analysis was used to examine group and condition effects on task performance and brain activity.
Results:
The VGNG accuracy rates in PD patients during walking were lower than in young and older adults (F = 5.619, p = 0.006). For all groups, N2 latency during walking was significantly longer than during sitting (p = 0.013). In addition, P300 latency was significantly longer in PD patients (p < 0.001) and older adults (p = 0.032) during walking compared to sitting and during ‘NoGo’ trials compared with ‘Go’ trials. Moreover, the young adults showed the smallest number of electrodes for which a significant differential activation between sit to walk was observed, while PD patients showed the largest with N2 being more strongly manifested in bilateral parietal electrodes during walking and in frontocentral electrodes while seated.
Conclusion:
The results show that response inhibition during walking is impaired in older subjects and PD patients and that increased cognitive load during dual-task walking relates to significant change in scalp electrical activity, mainly in parietal and frontocentral channels.
INTRODUCTION
Walking in everyday life relies on the ability to effectively follow internally generated goals and manipulate competing demands from the environment while simultaneously maintaining motor performance. Engaging in additional attention-demanding tasks while walking is frequently required, resulting in recruitment of various motor and cognitive cortical resources that can become overburdened and impair either the cognitive task, gait performance, or both [1–3]. Inadequate motor and cognitive resources in older adults and patients with neurodegenerative diseases, such as Parkinson’s disease (PD) have been shown to impact ambulation [1, 4]. A common way to investigate the interaction between motor and cognitive function and evaluate the behavioral costs associated with either task is by using a dual task walking paradigm [3].
Recent studies have considered the interaction between gait and visual and cognitive functions and emphasized the crucial role of visual attention during gait as a means for selectively tending to specific stimuli while suppressing others [2, 5]. When the inhibitory mechanism is affected, such as in PD or ageing, subjects show reduced ability to refrain from responding to irrelevant environmental cues, which leads to poor executive control [6]. Visual attention is modulated by involuntarily bottom-up and voluntary top-down mechanisms within a brainstem parieto-temporal and basal ganglia-frontal neuronal network [7]. Executive processes at the pre-frontal cortex (PFC) estimate the priority of visual inputs based on the task goal and projects information via attentional circuits back to the temporal cortex, which integrates all the information to successfully complete the task [8]. Alterations in networks that involve the basal ganglia-frontal motor areas in older adults and PD patients may impair the ability to ignore distracting stimuli and result in poor response selection [5, 9].
The Go/NoGo task has been widely used in psychology to measure participants’ capacity for sustained attention and response control, as it requires overcoming a potent response tendency generated by frequent ‘Go’ stimuli to successfully inhibit response execution to ‘NoGo’ stimuli. Studies have shown longer response times but comparable accuracy on ‘Go’ and ‘NoGo’ trials in older adults compared with young controls [6, 11]. On the other hand, PD patients demonstrated higher error rates compared with healthy older adults but with no significant difference in response times [12]. It has been suggested that the similar response time shown by PD patients results from higher rate of false responses, which reflects the trade-off between response time and commission error [9].
Neurophysiological studies that investigated the underlying neural mechanisms of inhibitory control using evoked responses (ERPs) have identified two phases of inhibitory network activity: N2–a relatively early automatic process (200– 350 ms) followed by P300, a later cognitive control process (250– 500 ms) [13, 14]. The functional dissociation of the two components is further supported by neuroanatomical segregation in which N2 generators are primarily located in prefrontal areas, whereas P300 is mainly produced by parietal and inferior temporal areas for the visual modality [10, 16]. For both aging and PD, the amplitude of N2 and P300 components decreased and the latency of P300 increased, especially for the patients with PD [9– 11, 15].
To date, only a single study evaluated changes in P300 characteristics during walking and auditory oddball in PD patients [17]. This study demonstrated no change in P300 latency but reduced P300 amplitude. Given the gait deficits, impaired inhibitory control, and high motor-cognitive interference associated with PD, in depth analysis of EEG data collected during dual task walking may unravel the mechanism underlying normal and abnormal gait. Thus, the objective of the current study is to explore age and PD related differences in the recruitment of cortical circuits underlying inhibitory control using a dual task paradigm involving motor (walking) and cognitive (visual Go/NoGo) tasks. We hypothesize that walking would compromise inhibitory control abilities and that the effect will be pronounced in aging, and more so in PD. Specifically, we hypothesize that the effect of dual-task walking on inhibitory control will be mostly manifested in centrofrontal channels whose N2 and P300 ERP components were altered already during sitting go/no-go task [18–20].
MATERIALS AND METHODS
Study participants
We enrolled 30 patients with relatively mild PD, 11 young adults and 30 older adults. Older adults and PD patients were of similar age (t = – 1.6855, p = 0.119). The PD patients were recruited through the Movement Disorders clinic in Sourasky Medical Center in Tel-Aviv and the social media. The inclusion criteria for the PD patients were: 1) diagnosis within the last five years and, 2) a score of < 2 in the Hoehn and Yahr rating scale for Parkinson’s disease [21]. PD severity was assessed using the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) [22]. All the medications related to PD were annotated using the levodopa equivalent dose calculation (LED score) [23]. Only two PD patients were treated with antidepressants. For PD patients, the timing of last levodopa dose was an hour before the experiment. All PD patients were examined and determined to be in the ‘on’ state at the time of the experiment.
Participants were excluded if they scored less than 21 on the Montreal Cognitive Assessment (MoCA) [24], had a history of neurological disorder other than PD, were unable to walk independently for at least 5 min, had any unstable medical conditions, vision problems or significant psychiatric co-morbidity. The subjects’ demographics is described in Table 1. The study was approved by the local ethical committee and was performed according to the principles of the Declaration of Helsinki. All participants gave their informed written consent prior to participation.
Participants’ characteristics
M, male; F, female; MoCA, Montreal Cognitive Assessment; CTT, Color trial test; LED, Levodopa equivalent doses; n/a, not applicable. Bold values significant difference (p < 0.05), *significant difference between older and young adults, **significant difference between PD patients and young adults, #significant difference between PD patients and older adults. PD Patients showed lower gait speed over ground during dual task (F = 6.604, p = 0.003) and higher Unified Parkinson’s Disease Rating Scale (UPDRS) motor score (F = 58.469, p < 0.001) compared to young and older adults.
Experimental paradigm and design
Participants performed a visual Go/NoGo (VGNG) task [25] with their right dominant hand while seated (cognitive single-task) and during walking on a treadmill (dual-task) (Fig. 1). In addition, they performed 4 min of walking on the treadmill without the VGNG task (motor single-task). These tasks were performed while recording their brain electrical activity using EEG and gait performance using 3D accelerometers placed on the right and left ankle and on the lower back. During the walking conditions, subjects were instructed to walk at their comfortable speed–a walking speed that feels subjectively natural to the subject. Comfortable walking speed on the treadmill was determined before the beginning of the task. The participants were instructed to press a button ‘as rapidly and as accurately as possible’ whenever they saw an English letter (‘Go’ sign event) and withhold response when the letter X was presented (‘NoGo’ sign event).
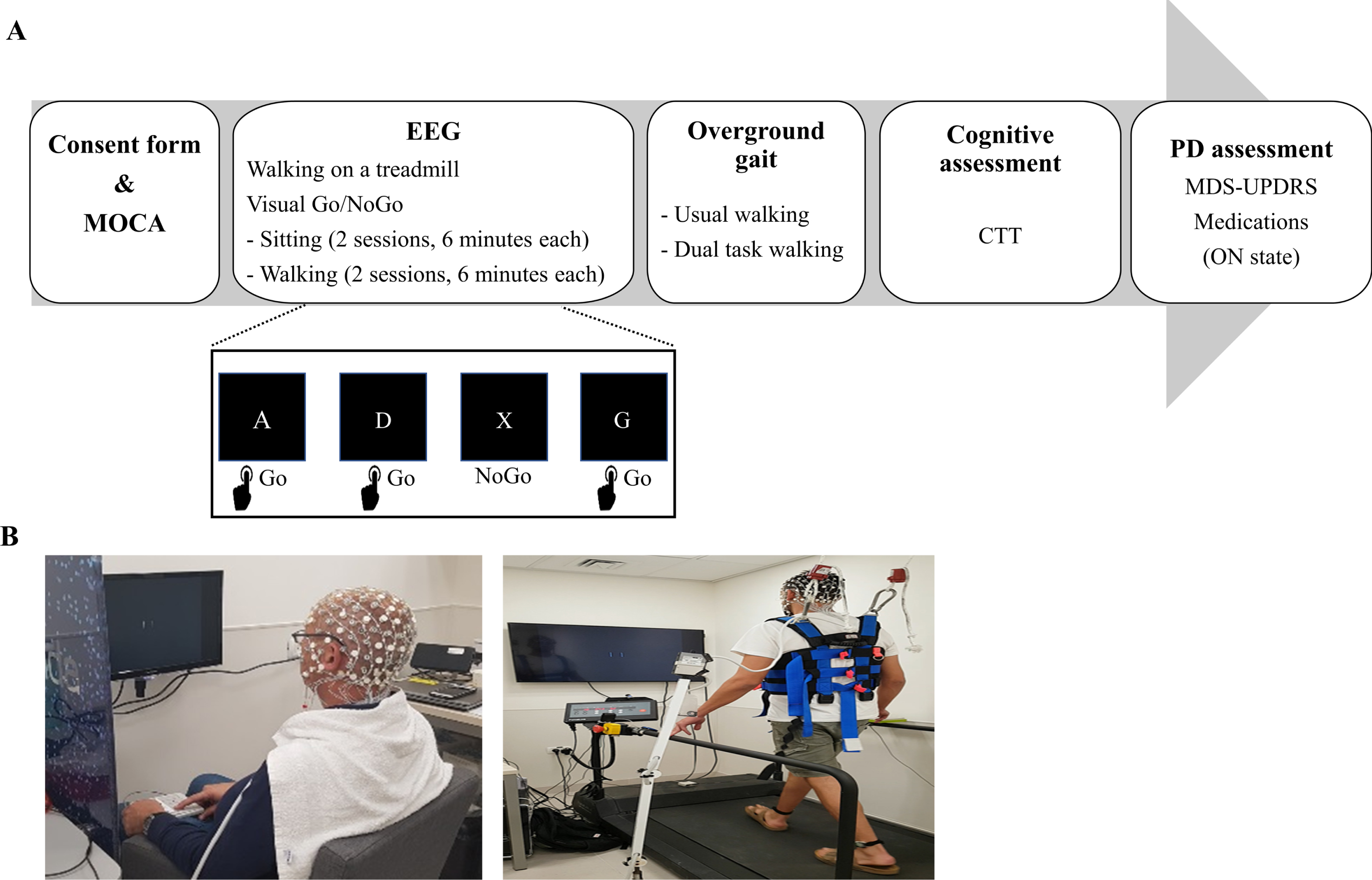
Study protocol and setting. A) Experimental protocol and visual Go/NoGo paradigm. B) EEG assessment performed during sitting and walking on a treadmill. While walking, the subjects were secured by a harness attached to the ceiling.
The experiment comprised of four types of trials: ‘SitGo’, ‘SitNG’, ‘WalkGo’, and ‘WalkNG’. Each condition (Sit or Walk) included two sessions, lasting six minutes each with one-minute inter-session interval. Each session comprised 200 trials, which were presented in a random order. A ratio of 4:1 was set between the frequency of the ‘Go’ and ‘NoGo’ trials to maximize false alarms [26]. The inter stimulus interval was varied randomly between 1000 ms to 3000 ms with steps of 250 ms. The performance in the VGNG task was assessed by the correct percent of ‘Go’ trials, ‘NoGo’ trials, total number of responses and average response time (in milliseconds) in the ‘Go’ trials. The subjects were not instructed to prioritize any task (motor or cognitive) over the other.
After the EEG recording was complete, the participants underwent motor and cognitive assessments. The cognitive assessment included the Color Trail Test (CTT), which evaluates visual scanning, attention and cognitive flexibility [27]. The motor assessment included two walking over-ground tasks, 1 min of normal walking and one minute of walking while performing serial 7s subtraction (dual-task), along a 20-meter corridor. Gait measurements included spatiotemporal features, e.g., gait speed, stride/swing time, and stride/swing regularity to assess gait performance. Stride regularity, defined as the correlation between the original acceleration signal and the acceleration signal phase shifted to the average stride time, was used as a measure of the consistency of the walking pattern: higher values reflect higher stride-to-stride consistency, and vice versa.
EEG acquisition and processing
EEG was recorded using a 64 active channels system (GES 400 EGI, OR, USA). The position of the electrodes on the cap fitted the international 10– 10 system. Electrooculogram (EOG) signals were recorded using four channels—two channels placed above and below the right eye (vertical EOG) and two at the outer canthus of both eyes (horizontal EOG). EEG and EOG Channels were recorded with a sampling rate of 250 Hz. The EEG reference electrodes were positioned bilaterally on the mastoid bones behind the ear. The EEG data were preprocessed by the EEGLAB [28] open source MATLAB software package. First, the data were passband filtered using a zero-phase hamming windowed sync FIR filter between 0.1– 40 Hz in order to discard low band (e.g., baseline drift and motion artefacts) and high band (e.g., EMG) artefacts. Channels with remaining prominent artefacts (excessive eye blinks and/or movement artefacts) were removed based on visual inspection. Next, the data were referenced to the average of all scalp electrodes and ICA was performed on the dataset to remove EOG and electromyogram (EMG) artefacts. The number of removed independent components varied between two and six across subjects. Finally, for each of the four trial types (‘SitGo’, ‘SitNG’, ‘WalkGo’, ‘WalkNG’), the signal was extracted into 0.75 second epochs (trials) of 0.25 s pre-event and 0.5 s post-event relative to a 0.25-s pre-stimulus baseline.
‘Go’ trials (‘SitGo’ and ‘WalkGo’) for which no press button occurred or a press button occurred too early (< 50 ms post stimulus) or too late (> 450 ms post stimulus) were discarded from further analysis. ‘NoGo’ trials (‘SitNG’ and ‘WalkNG’), for which a press button occurred were also excluded from the dataset. Overall, 6.96%of all trials were discarded from further analysis. A complete description of the percent rejected trials for each group, condition and event type is given in Supplementary Table 1. Finally, all electrodes were tested for the presence of residual noise; the standard deviation (SD) of the EEG amplitude was computed for each electrode in a given session and an electrode was marked noisy in a given trial, and that trial was discarded from further analysis, if the electrode amplitude exceeded 6 SD of the mean amplitude. For each subject, condition, and trial type, mean activity was computed, N200 and P300 were determined by visual inspection and their amplitude and latency were recorded.
Statistical analysis
Group and condition effects on task performance and ERPs measures
The mean and standard deviation of all the demographic and behavioral (gait and cognitive) variables were calculated and evaluated for normality and homogeneity using box plots, scatter plots and Kolmogorov-Smirnov tests. Kruskal-Walls and one-way ANOVA, followed by Least Significant Difference (LSD) post-hoc tests, were used to examine differences between groups in demographics and behavioral measures. Linear mixed models were used to examine measures of VGNG performance (correct %and response time), measures of gait performance on the treadmill (stride time and stride regularity), and measures of ERPs (N2 and P300) while controlling for gender and LED. The effect of group (young adults, older adults, PD), condition (sit, walk), event (‘Go’, ‘NoGo’), and their interactions on correct %responses and gait measures were tested.
The correlations between motor-cognitive behavior measures and electrical brain activity measures were examined using Spearman’s tests, setting significance level to p = 0.05. The behavior measures that were included in the correlation analysis encompassed the MoCA score that reflects a general cognitive status, the VGNG performance (response time and correct %) that reflects specific cognitive inhibitory control, and gait speed that reflects motor ability. The electrical brain activity measures that were included in the analysis encompassed the peak amplitude and latency of N2 and P300 during ’SitNG’, ’WalkNG’, ’SitGo’ and ‘WalkGo’ from Fz and Pz electrodes. The statistical analyses were performed using SPSS software for Windows [29].
Dual-task effect on inhibitory processes
In order to assess the effect of dual task on early and late inhibitory processes, the differential activation ‘SitNG’–‘WalkNG’ along the N2 time window (200– 350 ms) and P300 time window (350– 500 ms) was computed. To that end, the EEG activity in the ‘SitNG’ and ‘WalkNG’ trials of all subjects in a given group were pooled resulting in a 60 (electrodes) * 37 (sample points) *N (total number of trials) activation matrix. Next, for each electrode and sample point, a point-wise two-tailed t-test was performed. As multiple comparisons were performed (37 sample points * 60 electrodes) a corrected p-value should be used (0.05/2220 for Bonferroni correction). However, in order to discard sporadic sample points with significant differential activation [30], a significance level of p = 0.05/277 was used, but a differential activation was considered significant only when eight consecutive data points (32 ms) exceeded this criterion, thus, resulting in a corrected p-value of 0.05 / (277 * 8).
Dual-task effect on motor response
In order to assess the effect of dual task on motor response, ‘Go’ trials were analyzed. As the ‘Go’ event (‘SitGo’ and ‘WalkGo’) comprised four times higher number of trials than the ‘NoGo’ event, one fourth of the trials were chosen randomly and used to compute differential activation ‘SitGo’–‘WalkGo’ along the N2 and P300 time window, as described for the ‘NoGo’ event.
RESULTS
Participants’ characteristics
Participants’ characteristics are presented in Table 1. While no significant differences between the young and older adults’ groups were found in the cognitive and gait assessment, the group of patients with PD demonstrated lower MoCA than the young adults’ group and lower gait speed during dual task than both young and older adults’ groups. As shown in Table 1, the patients with PD were at early stages of the disease.
Visual Go/NoGo and gait performance on the treadmill
Response time on ‘Go’ trials (i.e., ‘SitGo’ and ‘WalkGo’) yielded a main effect of group (young adults < (older adults, PD patients)) (F = 9.766, p < 0.001) but no main effect of condition (sit or walk) (F = 1.133, p = 0.291) or interaction between them (F = 2.359, p = 0.103), indicating that young adults responded more rapidly than older adults and PD patients both while seated and during walking (Fig. 2A). The correct %responses on ‘Go’ trials, ‘NoGo’ trials, and total trials showed no main effect of group (‘Go’: F = 2.089, p = 0.132; ‘NoGo’: F = 0.442, p = 0.644; Total: F = 2.333, p = 0.105) or condition (‘Go’: F = 1.266, p = 0.265; ‘NoGo’: F = 2.764, p = 0.101; Total: F = 3.048, p = 0.086); however, there was a significant interaction between these factors (‘Go’: F = 3.854, p = 0.026; ‘NoGo’: F = 4.569, p = 0.014; Total: F = 7.102, p = 0.002) (Fig. 2B). Post-hoc analysis revealed that whereas no significant difference in accuracy rates (F = 1.574, p = 0.215) was found between the groups while seated, the accuracy rates in PD patients during walking were lower than in young and older adults (F = 5.619, p = 0.006). Specifically, PD patients demonstrated lower accuracy rates on ‘NoGo’ trials during walking (dual-task inhibitory control) compared with sitting (single-task inhibitory control) (t = 3.883, p < 0.001).
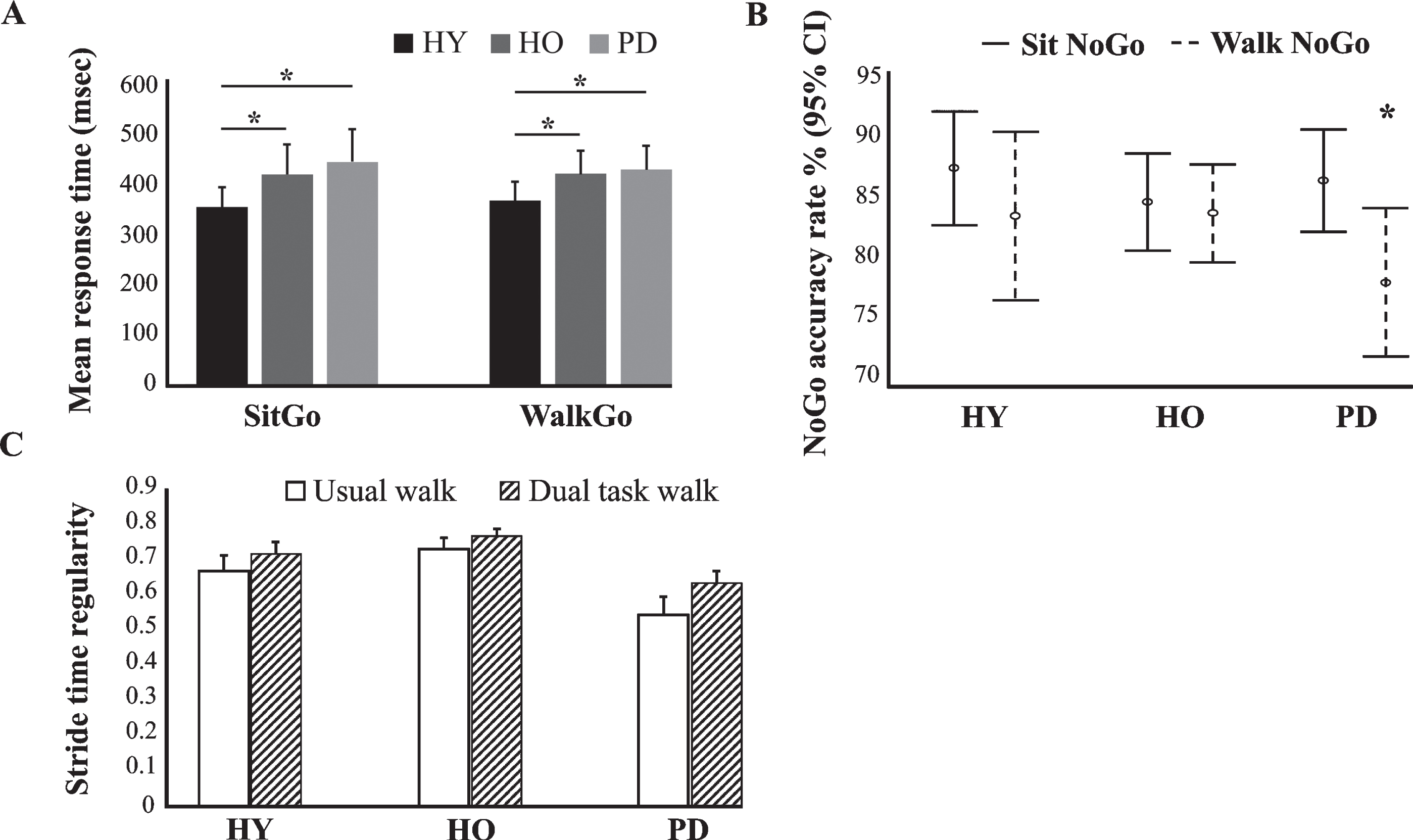
Differences between groups in task and gait performance. A) Mean response time (msec) to ‘Go’ trials while seated and during walking on the treadmill. B) Accuracy rate (%) in ‘NoGo’ trials while seated and during walking on the treadmill. C) Changes in stride regularity between usual and dual task walk in young adults (HY), older adults (HO), and PD patients (PD). *denotes significance (p < 0.05, LSD post-hoc test) between the two conditions.
No main effect of group (F = 0.502, p = 0.608), condition (F = 2.278, p = 0.139) or interaction between them (F = 0.862, p = 0.430) was observed for mean stride time. In contrast, main effect of group (PD patients < (young adults, older adults)) (F = 6.264, p = 0.004) and condition (dual task > usual task) (F = 8.084, p = 0.007) with no interaction between them (F = 0.469, p = 0.629) was found for stride regularity (Fig. 2C). Stride time regularity significantly increased while performing the dual-task walk only in the PD group (t = 2.451, p = 0.025).
ERPs amplitude and latency
‘NoGo’ trials–N2
Figure 3 presents grand average (across subjects and trials) scalp topographies for response inhibition (‘NoGo’ trials) around N2 while seated or during walking in young adults, older adults, and PD patients. For both conditions (sit and walk), N2 amplitude was most prominent (more negative amplitude) and the latency the shortest for the young adults, followed by the older adults and PD patients.
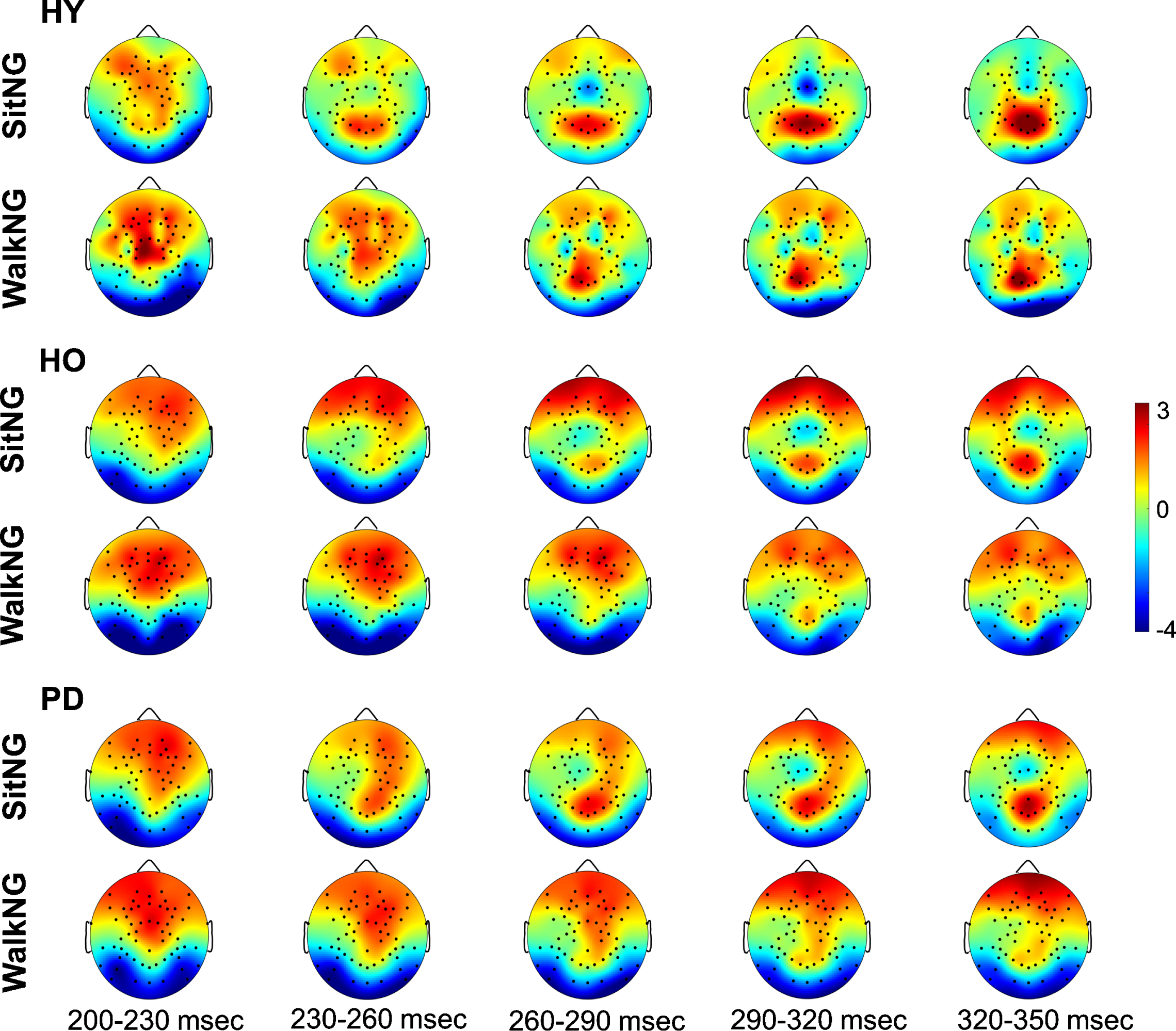
Topographical distribution of global N2 activity (negative values) across the scalp during ‘NoGo’ trials. The upper plots and lower plots present activity in ‘SitNG’ and ‘WalkNG’ trials, respectively in 30msec time intervals for healthy young (HY), healthy old (HO), and PD patients (PD). The color code denotes amplitude (in μV).
In order to study the effect of dual task on the early phase of response inhibition and its relation to age and PD, the differential signal amplitude between the two ‘NoGo’ conditions (‘SitNG’–‘WalkNG’) in N2 time window was assessed (Fig. 4). Overall, there was a gradual increase in the number of electrodes for which a significant differential activation was found from young adults to older adults and onward to PD patients. Moreover, N2 was more demonstrated in central and frontal electrodes while seated, compared to walking, whereas the opposite finding was found for parietal electrodes. Specifically, for the young adults (Fig. 4, left plot), the effect of walking on response inhibition was found only in two frontocentral electrodes (FCz and FC1) and one parietal electrode (P2) and was characterized by more prominent manifestation of N2 (more negative amplitude) while seated in the frontocentral electrodes and reduced manifestation in the parietal electrode. For the older adults (Fig. 4, middle plot), N2 was more manifested while seated not only in FCz and FC1 but also in central electrodes (C1, C2) and bilateral frontocentral electrodes (FC2, FC3, FC4, FC6). Reduced N2 manifestation while seated was found not only in P2 but also in bilateral parietal electrodes (P1, P3, P4, P5, P6, P8) as well as in PO3. For the PD patients (Fig. 4, right plot), the differential activation pattern resembled the pattern that was found for the young and older adults—in FCz and FC1, and older adults—in C1, however, increase in N2 negativity while seated was found also in frontal areas (Fz, F1, F3, AFz) and frontoparietal area (Fp2). Reduced N2 manifestation while seated was found not only in P2, as found for the young and older adults, and P1, P3, P4, P6, and PO3, as found for the older subjects, but also in Pz, POz, and PO4.
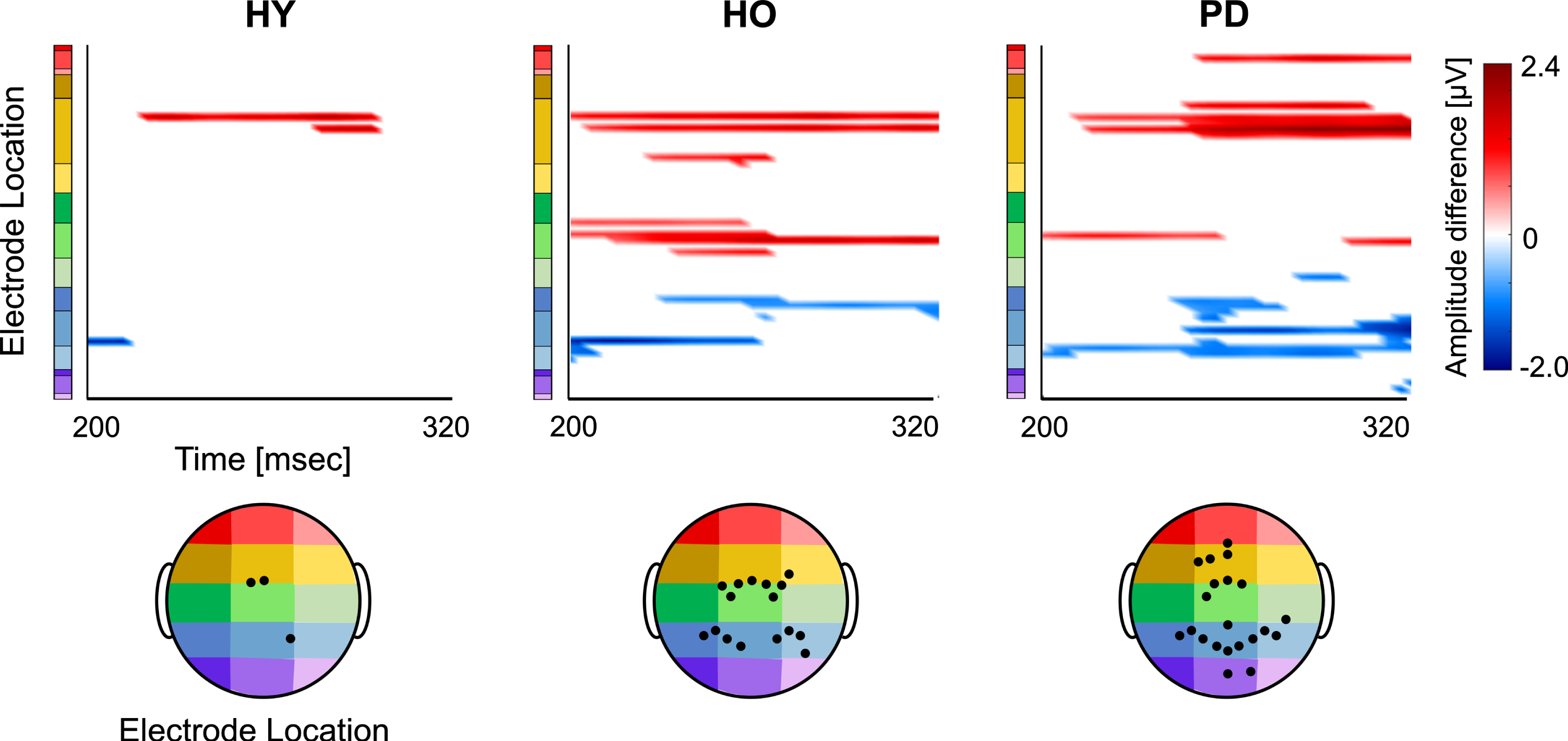
Onset and distribution of global differential N2 responses between ‘SitNG’ and ‘WalkNG’ in young adults (HY), older adults (HO), and PD patients (PD). Point-wise t-test evaluating the differential activation (‘SitNG’–‘WalkNG’) along N2 time epoch (x-axis) and 60 electrode montage (y-axis) was assessed. The differential activation (color code) is shown only electrodes and sample points for which p-value < (0.05/277) and only when eight consecutive data points (32 ms) exceeded this criterion. An amplitude difference was set positive if N2 was more prominent (negative amplitude) while seated than during walking. The black filled circles denote position of electrodes with significant differential activation.
‘NoGo’ trials–P300
The grand average (across subjects and trials) scalp topographies for ‘NoGo’ trials around P300 while seated and during walking in young adults, older adults, and PD patients was computed (Fig. 5). As was found for N2, the most evident difference between groups was in the latency; P300 was first observed in young adults, followed by older adults and lastly in PD patients. However, as opposed to N2, there was no difference across groups in P300 manifestation (positive amplitude).
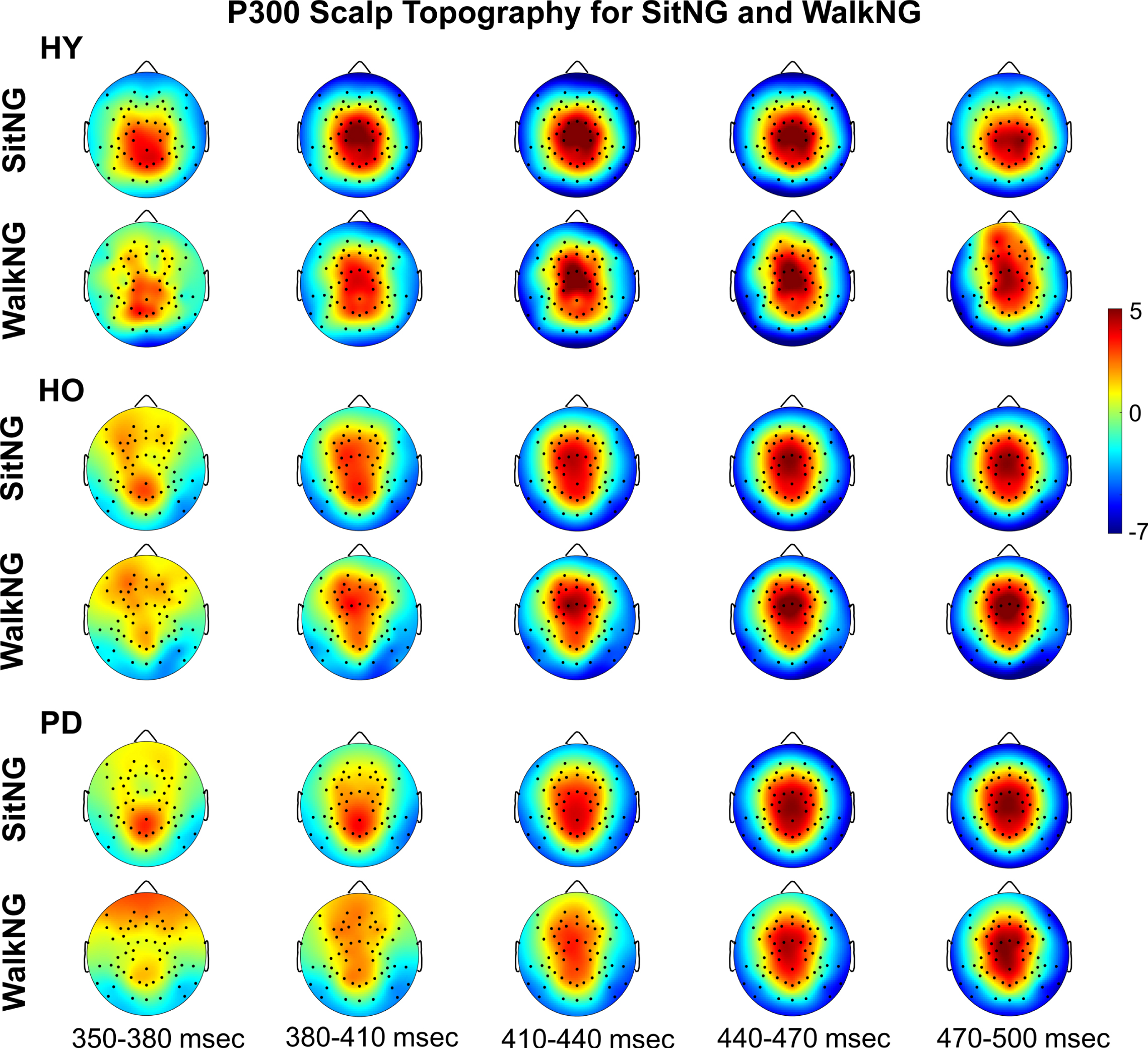
Topographical distribution of global P300 activity across the scalp during ‘NoGo’ trials. The upper plots and lower plots present activity in ‘SitNG’ and ‘WalkNG’ trials, respectively in 30 msec time intervals for healthy young (HY), healthy old (HO), and PD patients (PD). The color code denotes amplitude (in μV).
Assessing significant differential activation between the two ‘NoGo’ conditions (‘SitNG’–‘WalkNG’) has shown that similar to N2, there was a gradual increase in the number of electrodes for which a significant differential activation was found from young adults to older adults and onward to PD patients. The pattern of P300 differential activation over the scalp, however, largely resembled an inverse image of the N2 differential activation pattern; P300 was more demonstrated in central and frontal electrodes and less demonstrated in parietal and occipital electrodes while walking, compared to seating. Specifically, for the young adults (Fig. 6, left plot) P300 was more manifested (more positive) while walking in frontal (F1, F3, Fz, F4, AF3, AFz) and frontocentral (FCz, FC1) electrodes. For the older adults (Fig. 6, middle plot), P300 was more pronounced while walking only in a frontal (AF3) electrode whereas the opposite pattern was found in bilateral parietal electrodes (P1, P2, P3, P4, P5), parietooccipital (PO3) and occipital (O1) electrodes. As was found for N2, the highest number of electrodes with significant differential activation was found for the PD patients (Fig. 6, right plot). More pronounced P300 while walking was found in frontal (AFz, F1, F5, F10) electrodes whereas the opposite finding was found in ipsilateral central (C2, C4, C6) and parietal (Pz, P2, P4) electrodes as well as bilateral parietooccipital (POz, PO3, PO4) electrodes.
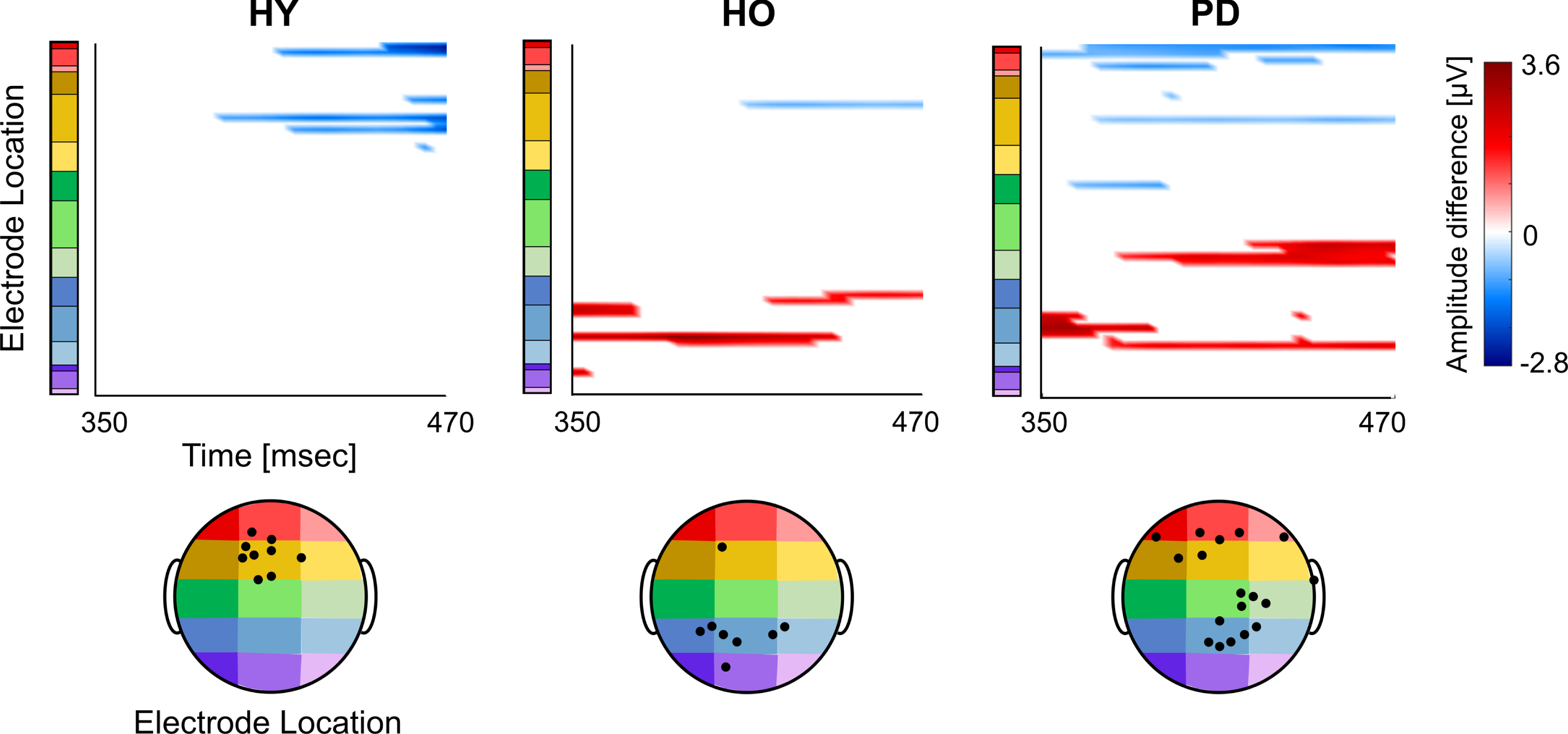
Onset and distribution of global differential P300 responses between ‘SitNG’ and ‘WalkNG’ in young adults (HY), older adults (HO), and PD patients (PD). Point-wise t-test evaluating the differential activation (‘SitNG’–‘WalkNG’) along P300 time epoch (x-axis) and 60 electrode montage (y-axis) was assessed. The differential activation (color code) is shown only electrodes and sample points for which p-value < (0.05/277) and only when eight consecutive data points (32 msec) exceeded this criterion. An amplitude difference was set positive if P300 was more prominent (positive) while seated than during walking. The black filled circles denote position of electrodes with significant differential activation.
‘Go’ trials–N2 and P300
As was observed for ‘NoGo’ trials–N2, young adults demonstrated a more prominent N2 amplitude (more negative amplitude) and a shorter N2 latency compared to older adults and patients with PD in the ‘Go’ trials (Supplementary Figure 2). However, whereas N2 for the ‘NoGo’ trials in young adults was confined to frontocentral electrodes FCz, FC1, and FC2, it is much more widespread in the ‘Go’ trials and included several frontocentral, as well as frontal electrodes. Moreover, the differences between sit-walk ‘Go’ trials included higher number of electrodes also in the young adults’ group although in both conditions (‘Go’ and ‘NoGo’) the same differential activation was found–more pronounced N2 manifestation in central and frontal electrodes while seated and in parietal and occipital electrodes, while walking (Supplementary Figure 3).
For the ‘Go’ trials–P300, the most evident difference between groups was in the latency in the walking condition. The ERP was first observed and was strongly prominent in young adults in frontal electrodes. For the older adults and PD patients, the ERP was less prominent, albeit widely spread over central electrodes, for the older adults, and over centro occipital electrodes, for the PD patients (Supplementary Figure 4). For the older adults and PD patients, no major difference in the number of electrodes for which a differential activation was found, their spatial coverage on the scalp and the polarity of the differential activation was found across the two conditions (‘SitNG-WalkNG’ versus ‘SitGo-WalkGo’). However, as opposed to ‘NoGo’ trials, young subjects exhibited the highest number of electrodes for whom significant differential activation was found (Supplementary Figure 5). P300 in this group was not only more pronounced in central and frontal electrodes while walking, as was found for ‘NoGo’ trials, but was also prominent while seated in bilateral parietal and occipital electrodes.
Correlations between behavioral and ERPs measures
In order to study the effect of group and condition on cognitive (MoCA score) and motor performance (gait speed and response time) in single- and dual- task and their relation with ERP measures, the activity of Fz and Pz, for which a significant differential activation was found was further analyzed. For Fz, longer N2 latency in ‘WalkGo’ dual-task trials was positively correlated with response time (r = 0.358, p = 0.003) (Fig. 7A, left plot). No correlation was found between P300 latency in Fz and any of the behavioral measures (p > 0.05). For Pz, several inverse correlations between the behavioral and ERP measures were found. Longer N2 and P300 latency in ‘WalkGo’ dual-task trials was negatively correlated with MoCA (r = – 0.321, p = 0.025 and r = – 0.302, p = 0.033, respectively) (Fig. 7B, two upper plots). In addition, longer P300 latency in ‘WalkNG’ trials (inhibition in dual-task) was negatively correlated with gait speed over ground (r = – 0.327, p = 0.028) (Fig. 7B, lower left plot). Finally, longer N2 latency in ‘SitGo’ single-task trials was inversely correlated with total correct trials (r = – 0.350, p = 0.005) (Fig. 7B, lower right plot).
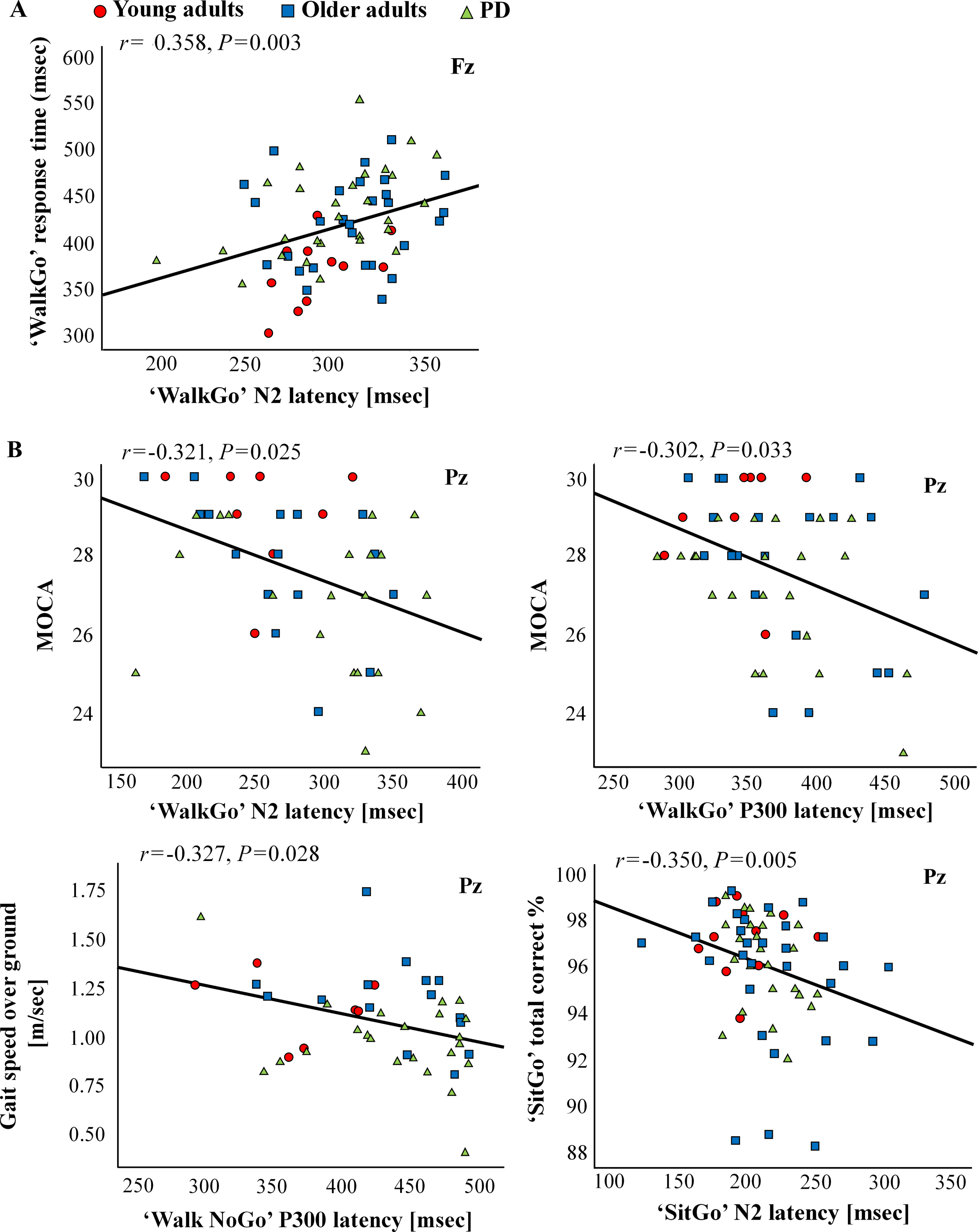
Correlations between behavioral and ERPs measures.)A(Correlation between dual-task walking performance and N2 latency in Fz.)B(Correlations between motor and cognitive assessments and N2 and P300 latency in Pz.
DISCUSSION
Visual Go/Nogo task and gait performance
Comparison between regular (single-task) and VGNG task (dual-task) walking on the treadmill revealed that gait variability significantly decreased while performing the VGNG task only for the PD patients’ group, suggesting that the ‘Go’ and ‘NoGo’ events acted as an external visual (motor automation) cue that improved stride consistency, as previously found for rhythmic auditory cueing [31] and auditory oddball task [32]. Surprisingly, despite motor automation and the recruitment of PD patients that present only slight impairment of gait (a score of < 2 in the Hoehn and Yahr rating scale) that would presumably allow allocating cognitive resources to response inhibition, PD patients showed the most impaired inhibitory control during dual task walking compared with single task on the treadmill (Fig. 2). These results suggest one, or more of the following: 1) cognitive resources allocated for gait, although reduced, were still significant leading to impaired inhibitory control, 2) visual cues were mainly used as a means to regulate stride rather than being regarded as an action cue (‘posture first’ hypothesis), 3) response inhibition and gait in PD patients do not necessarily compete the same cognitive resources. As rates of correct responses in ‘Go’ trials in single- and dual-task were not significantly different for PD patients, the ‘posture first’ hypothesis is likely not the cause for the impaired inhibitory control. Moreover, the altered brain activation is more likely related to inhibition compensatory mechanism rather than to motor performance. To assess the effect of rhythmic external cues and automatic gait pattern on response inhibition, future works will include over-ground walking, simulating a more natural gait setting with no external cues, while performing a visual (or auditory) Go/NoGo task with increased inter-trial time variability.
ERPs characteristics
Similar to Malcolm et al. [33], we found reduced N2 amplitude and longer N2 latency with aging. These findings were extended to PD patients, wherein further reduction in N2 amplitude and longer N2 latency were found in ‘Go’ trials (Supplementary Figure 2), and more noticeably in ‘NoGo’ trials (Fig. 3). Longer delay with age and PD may reflect a general delay in cognitive processing as previously reported in the literature [33, 34]. The topographical distribution of N2 amplitude during dual-task (walking), compared with single-task (sitting) shows an age-related shift, with a more pronounced (negative) N2 in posterior electrodes (Fig. 4), as previously described in Melcolm et al. [33] and less pronounced N2 in centrofrontal electrodes. The PD patients group demonstrated topographical changes in N2 amplitude that resembled the findings found for the older adults but in addition, they showed decreased N2 amplitude during walking in frontal regions. These changes suggest that with aging and PD, impaired automatic inhibition control while walking is underlined by insufficient recruit of neural circuits in frontal cortical regions and possibly a compensation mechanism of increased recruit of parietal cortical circuits. However, whereas the moderate change in electrical activity was accompanied by a successful compensatory mechanism for the older subjects, response inhibition was impaired in PD patients, albeit the substantial change in electrical activity.
For P300, the change in topographical distribution during dual-task walking differed between the groups (Fig. 6); young adults demonstrated increased P300 amplitude in frontal and frontocentral electrodes, older adults in only one frontal electrode, and PD patients in bilateral parietal and parietooccipital electrodes. Although there is some debate as to whether the P300 truly represents late inhibitory control or is driven by underlying latent processes other than inhibition, such as behavioral adaptations in context of performance monitoring operations [35], these results provide direct evidence to the neural costs associated with later phases of inhibitory control due to increased task load with age and PD.
Specific observations at Pz electrode (Supplementary Figure 1) showed that young adults showed higher P300 amplitude than older adults and PD patients, indicating that late control inhibitory processes are modulated with age and disease. However, no differences between sitting and walking were observed, demonstrating that late processing stages of inhibitory control are less sensitive to dual task load. These results are not in line with studies that showed small reduction in P300 amplitude during walking [17, 33]. These discrepancies may relate to the differences in the secondary cognitive task that was performed during walking. On the other hand, we found longer P300 latency with age, disease, walking and ‘NoGo’ trials, indicating that the timing of later control inhibitory process is modulated by these factors.
Correlation between task performance and ERPs
Better performance in the VGNG task, as seen by shorter response time and higher rate of correct responses, was correlated with shorter N2 latency, indicating that the automatic phase of inhibitory control plays an important role in successful task performance. In addition, moderate to strong inverse correlations between ERPs and global cognitive and motor characteristics were found. Longer N2 and P300 latency during walking were correlated with lower MoCA scores, suggesting that slower neural processing reflected by longer latencies in both automatic and cognitive phases of inhibitory control may explain global cognitive deficits. Furthermore, longer P300 latency was correlated with slower gait speed during dual task, suggesting that alterations in the later-cognitive phase of inhibitory control relate to reduced gait performance. These correlations emphasize that increase in task load reveal cognitive alterations that may not be evident during the performance of a simple task.
Limitations of study
Although EEG recording during dual-task walking allows to examine neural activity while subjects are engaged in complex walking situations, walking on a treadmill, as performed in this study, is not natural due to lack of ability to slow down under increasing task load. Moreover, it is well known that the treadmill provides an external cue [31] and therefore, the assessment of gait variability on a treadmill can be misleading and should be carefully interpreted. The improved stride time regularity during the VGNG task further highlights the effect of external cues on motor performance in patients with PD. Rhythmic external cues may, however, also affect inhibition control; as gait is facilitated and becomes more automatic, more cognitive resources can be allocated to response inhibition. Thus, it may be that over-ground walking, simulating a more natural gait setting with no external cues, while performing a visual (or auditory) Go/NoGo task would result in even worse inhibitory control in patients with PD.
CONCLUSIONS
We found robust differences in amplitude, latency and scalp distribution of the ERP components associated with the early (automatic) and late (cognitive) phases of inhibitory control between groups and tasks. These differences were group- and condition- dependent highlighting the involvement of different neural circuits in single- and dual-tasking and the effect of age and PD on response inhibition. A future work will localize, using source localization techniques, the brain areas that underlie normal and impaired inhibitory control and examine the functional and effective connectivity between them.
Footnotes
ACKNOWLEDGMENTS
We thank the research participants and the research assistants for their time and effort. We would also like to thank Gil Issachar and Eran Riva from Elminda company for providing us technical support with the EEG recording system.
The work was funded in part by the Fugelnest foundation–Faculty of medicine resources.
CONFLICT OF INTEREST
The authors declare no commercial or financial relationships that could be construed as a potential conflict of interest.
