Abstract
Background:
Freezing of gait (FOG) is a common symptom of Parkinson’s disease (PD) which can result in falls and fall related injuries, poor quality of life and reduced functional independence. It is a heterogeneous phenomenon that is difficult to quantify and eludes a unified pathophysiological framework.
Objective:
Our aim was to document the occurrence and nature of freezing, cognitive stops and stumbles in people with PD during walks with varying cognitive loads and conditions designed to elicit FOG.
Methods:
130 people with PD walked under four conditions (normal walking, walking plus easy and hard dual-tasks, and a FOG elicitation condition. Video and accelerometry recordings were examined to document freezes and other gait disruptions.
Results:
Participants experienced 391 freezes, 97 cognitive stops and 73 stumbles in the trial walks; with total gait disruptions increasing with task complexity. Most freezes in the FOG elicitation condition occurred during turning and approach destination. People who experienced freezing during the walks were more likely to have Postural Instability and Gait Difficulty (PIGD) subtype, longer disease duration and more severe UPDRS part II and part III sub-scores than people who did not freeze. They also took higher doses of levodopa, reported freezing in the past month, more prior falls, had poorer executive function, poorer proprioception, slower reaction time, poorer standing and leaning balance, more depressive symptoms, lower quality of life and greater fear of falling. PD disease duration, reduced controlled leaning balance and poor proprioception were identified as independent and significant determinants of freezing in logistic regression analysis.
Conclusion:
The multiple motor and cognitive factors identified as being associated with freezing, including poor proprioception and impaired controlled leaning balance provide new insights into this debilitating PD symptom and may contribute to potential new targets for rehabilitation.
INTRODUCTION
Freezing of gait (FOG) is a paroxysmal gait disturbance whereby a person has an inability to initiate or resume walking [1]. FOG is a common symptom of Parkinson’s disease (PD), with freezing episodes often lasting several seconds or longer and people commonly reporting they feel their feet are ‘glued to the floor’ [2, 3]. It results in falls and fall related injuries, disability, depression, poor quality of life [2, 4] and reduced functional independence [5].
The phenomenon currently eludes a unified pathophysiological framework. The paroxysmal and heterogeneous nature of the phenomenon and the wide-ranging factors associated with FOG span cognitive, behavioural and environmental domains. The combination of locomotor network vulnerability coupled with modulating factors converge to yield a failure of neuronal integration [6]. Failure of the final pathway determines the ultimate clinical manifestation of a freeze [7]. STN activation is a critical area for this ultimate convergence whereby subsequent (inhibitory) internal Globus Pallidus activation inhibits the GABAergic innervation of the glutamatergic pedunculopontine nucleus. This inhibition in the brainstem then inhibits the successful execution of central pattern generator coordinated agonist and antagonist muscle function elicits an episode of FOG [8].
Better, understanding of the neuropsychological correlates as well as environmental and patient related factors provides insight into the potential mechanisms. Higher level cortical modulators include motor areas such as the prefrontal cortex, supplementary motor area, premotor cortex, and motor cortex as well as non-motor factors [6]. FOG may be triggered by gait initiation, turning, navigating narrow or confined spaces and nearing a destination [3] whereby modulation and flexibility of gait response is required. Loss of gait automaticity in these circumstances necessitates the recruitment of additional cortical cognitive resources. When the required cognitive resource in diverted to other tasks in dual tasking experiments, or during affective states including fatigue and anxiety, the risk of FOG is increased [9–11]. This dual task phenomenon is postulated to result from dysfunction of concurrent information processing across neuronal networks [12]. Motor blocks can be overcome by utilising visual, tactile, emotional or auditory cues. This transfer from habitual motor control to goal directed behaviour thus diminishes reliance on the dysfunctional neuronal circuits thereby also overcoming loss of gait automaticity [13]. Furthermore, the controlled processing of information is impaired with the finding of association between FOG and cognitive dysfunction. PD freezers have greater neuropsychological deficits in executive function, cognitive flexibility and inhibition compared to PD non-freezers [14]. The loss of gait automaticity and dysfunction in controlled processing of tasks can result in the demand for cognitive resources outstripping supply, or where cognitive resources are deficient, FOG episodes occurring [15].
The episodic nature of the phenomenon, coupled with the strong association with environmental, neuropsychological, environmental and disease factors makes quantification of FOG immensely challenging [16]. In clinical and home settings the utilisation of behavioural adaption and external (e.g., lines on the floor) or internal (e.g., counting) cueing techniques may confound assessment. Reliable patient and/or carer report requires appropriate understanding and recognition of the phenomenon as well as reliable recollection and report. Many studies of FOG have used self-reported measures, e.g., the new FOG-Questionnaire (FOG-Q) [17]. These questionnaires categorize people as ‘freezers’ and ‘non freezers’ and provide general characteristics of freezing episodes. However, in addition to the limitations of self-report such as recall bias and possible inclusion of other gait disturbances, FOG questionnaires are unidimensional in nature and do not incorporate medication status or environmental triggers [16]. Despite the confines of a more artificial, laboratory environment, objective analysis of videoed walks is considered the gold standard for assessing FOG as it overcomes some of the limitations of self-report [16–19].
We conducted a detailed video annotation study to document the occurrence and nature of freezes and other gait interruptions in a large sample of people with PD “on” medication. The walks were conducted in an outside setting and comprised walks of varying difficulty including a condition encompassing transfers, gait initiation, narrow gaps, turning and gait termination designed to elicit FOG episodes. Our aims were to: a) document the occurrence and nature of freezes, cognitive stops and stumbles in people with PD when completing simple, dual-task (DT) and a FOG eliciting condition; and b) identify disease- and medication-related factors associated with objectively measured freezing episodes. We hope the collection of multiple freezing events and relevant ancillary data from a large sample can further inform strategies for addressing FOG and other gait disruptions in people with PD.
METHODS
Participants and setting
The present study was a secondary analysis of baseline data from the Rivastigmine to Stablise Gait in Parkinson’s disease (ReSPonD) randomized controlled trial conducted in South West England, which investigated the effect of the drug rivastigmine on gait stability [20, 21]. Participants were included if they: a) had been on stable PD medication for 2 weeks prior to enrolment, b) were able to walk 18 m without a walking aid and c) had at least 1 fall in the previous year. A history of freezing was not an exclusion criterion and a small number (not formally captured) were using (or had used) apomorphine and had had deep brain stimulation. People were excluded if they a) had an absolute contradiction to, or had previously taken acetylcholinesterase inhibitors, b) had any other neurological, visual or orthopaedic problems that significantly interfered with gait or c) had dementia. All participants provided informed consent prior to study participation. Ethical approval was obtained from the South West Research Ethics Committee and a Clinical Trial Authorisation (CTA) was granted from the Medicines and the UK Healthcare Regulatory Agency (MHRA).
Gait assessment
The walking trials were conducted on a 22 m level outdoor covered flat concrete walkway (with no regular markings that could provide external cues) with participants assessed in the “on” medication state as indicated subjectively by participants during daytime hours. Participants walked in their normal outdoor footwear and completed three walking trials in three conditions (blocks), the order of which was block randomised. The three conditions comprised: a) walking with no additional cognitive task, b) walking while naming words beginning with a single letter (easy Dual-Task), and c) walking while naming words alternating between two letters of the alphabet (hard Dual-Task). The number of trials was selected to capture the maximum potential data whilst balancing the fatigue and performance abilities of the participants. Participants were given standardised instructions before every walk that indicated they should walk at their usual pace. For the dual task walks the standardised instructions were to commence both tasks on ‘go’ and no prioritisation instructions were given as to walking or the cognitive task. Participants were not instructed to tell the researcher if they were pausing to think about the words. We included the two secondary task conditions, because it has been shown harder secondary cognitive tasks have greater influences on gait than easier secondary cognitive tasks [22] The verbal fluency tasks were based on the controlled oral word association test (COWAT) [23]. Following these nine walks, participants then completed two walks in a specific paradigm designed to elicit FOG. This comprised rising from a chair, walking 9 m, passing between two chairs placed 50 cm apart, continuing for a further 9 m before turning 360° to the right, then 540° to the left and then walking back along the walkway, passing between the chairs, and finally sitting down on the chair (total distance for each walk: 36 m). Figure 1 shows the set-ups for the dual task and FOG elicitation conditions.
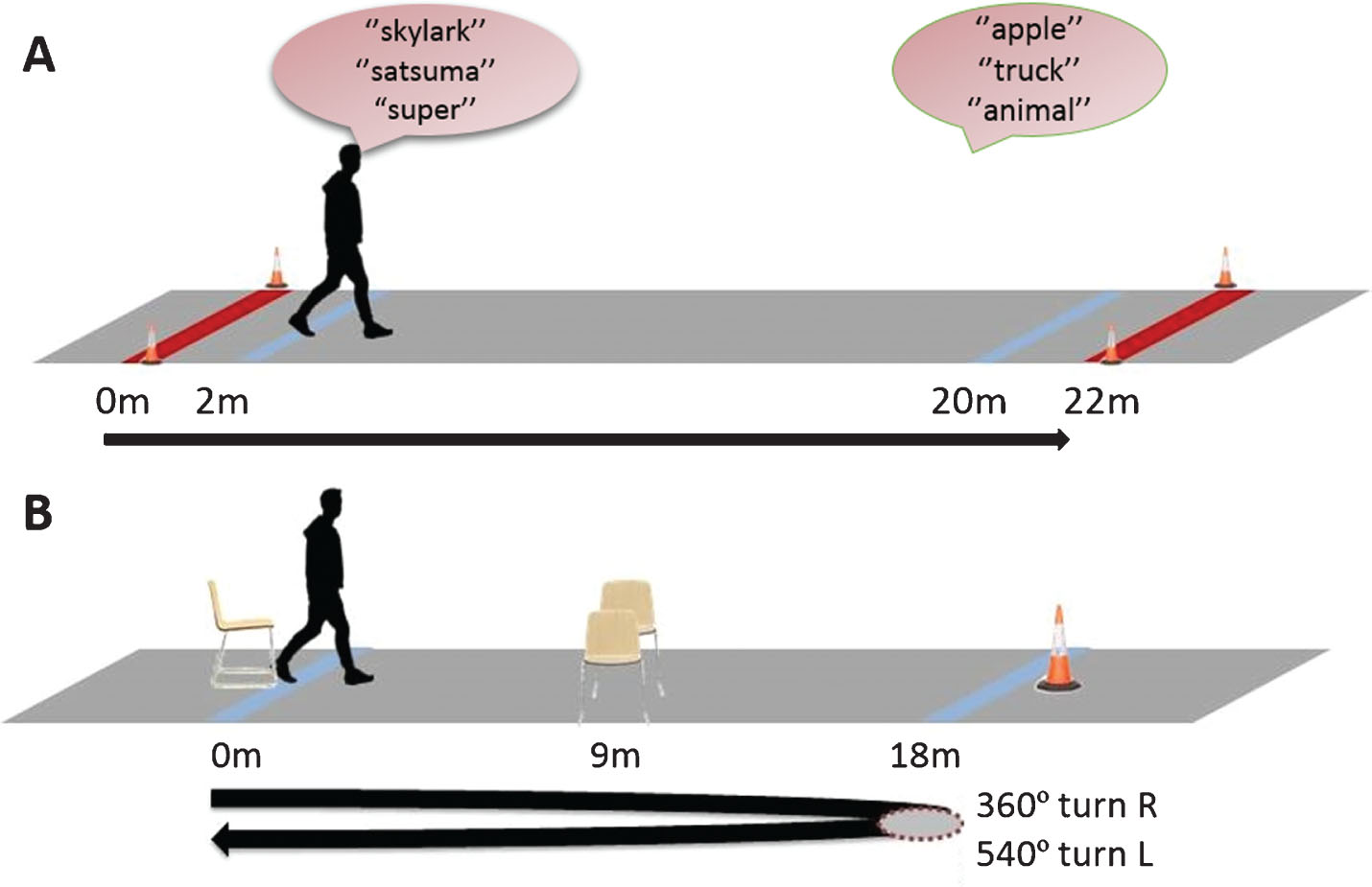
The set-ups for the dual task and FOG elicitation conditions.
Gait disruption annotation and classification
Each walk was videotaped (Panasonic HDC-SD800; frame rate of 50 Hz) by a researcher walking behind the participant. FOG was classified on these videos by a researcher trained in the recognition of FOG and the sub-classification used in this study by a Parkinson’s clinician. Custom software was built in MATLAB version 7.12.0 R2011a (Mathworks, Inc., MA, USA) to facilitate the synchronisation of visual recording of the walk with the triaxial accelerometry captured data. Using this software, a researcher could annotate events that occurred during the walking paradigms. Alterations in gait pattern were annotated with respect to type of freeze, location and duration by a researcher. Events were coded according to consistent, mutually exclusive criteria (see Supplementary Table 1) and where they occurred on the trajectory (see Supplementary Table 2). The researcher used both the video and the wearable device data to precisely annotate the initiation and termination of each event such as turning, walking and FOG according to the standardized instructions. The DynaPort Hybrid (McRoberts, Netherlands) system was attached to the waist with an elastic strap between the posterior superior iliac spines, sampling at 100 Hz. Based on the recorded data, the accelerometer settings were +/–2 g and gyroscope +–229 deg/sec.
FOG was defined as an episode where participants were unable to initiate or continue locomotion for no apparent reason. In some trials participants stopped walking during the tasks because they had forgotten the motor or cognitive task required and sought further instructions; these events were termed ‘cognitive stops’. Other gait disruptions such as tripping or near falls were classified as stumbles. Stumbles were timed from the onset of the gait disturbance until the resumption of normal “steady state” walking which often took several recovery steps and/or a pause. For the no-additional task, easy Dual-Task and hard Dual-Task conditions, gait was evaluated over the full length of the 22 m walkway with gait disruptions occurring in the initial and final 2 m labelled as gait initiation and destination hesitation disruptions respectively.
Freeze severity was classified as a) markedly smaller steps/shuffling (mild); b) trembling in place, associated with no forward motion (moderate); and c) total akinesia (severe) as defined by Schaafsma et al. [3]. For events with mixed manifestation (e.g., complete akinesia followed by a few small steps) we documented only a single event using a hierarchy for which complete akinesia was recorded in precedence to trembling in place and trembling in place was recorded in precedence to small steps. Freezes during the FOG elicitation condition were classified according to the activity being undertaken: a) start hesitation/initiation; b) turning; c) walking in the narrow gap between the chairs; d) destination hesitation: approaching the half way point before the turn and the chair at the end of the walk; and e) open space. The definitions of all gait disruptions are provided in Supplementary Table 1.
Additional assessments
Participants were assessed with respect to demographic and disease-related factors including body mass index (BMI), falls in the previous year and self-reported FOG episodes in the month prior to enrolment. PD characteristics were defined by the Unified Parkinson’s Disease Rating Scale part II and III (MDS-UPDRS), Postural Instability/Gait Difficulty-Score (PIGD; derived from UPDRS part III) and Levodopa Equivalent Dose (LED) [24]. Cognition was assessed with the Montreal Cognitive Assessment (MoCA) [25], executive function with the Frontal Assessment Battery (FAB) [26], quality of life (QoL) with the short version of Health Related Quality of Life Questionnaire (EuroQoL’s EQ-5D-5 L) [27], depressive symptoms with the Geriatric Depression Scale (GDS) [28], and falls efficacy with the Iconographical Falls Efficacy Scale (Icon-FES) [29]. The Physiological Profile Assessment (PPA) [30] was used to assess five parameters of physiologic performance as a measure of physiologic fall risk: visual contrast sensitivity (assessed using the Melbourne Edge Test); proprioception (measured using a lower limb-matching task, with errors in degrees recorded using a protractor inscribed on a vertical clear acrylic sheet placed between the legs), quadriceps strength (measured isometrically in the dominant leg with participants seated with the hip and knee flexed 90°), simple reaction time (measured using a light as the stimulus and a finger-press as the response), and postural sway (path length, measured using a sway meter recording displacements of the body at the level of the pelvis with participants standing on a 15 cm thick foam rubber mat with eyes open). Additionally, participants performed the coordinated stability test (COSTAB), an assessment of controlled leaning balance [31]. The MoCA was selected because it provides good assessment of visuospatial and executive function [25]. The PPA, coordinated stability test and the FAB were selected as they have previously been shown to be risk factors for falls in people with Parkinson’s disease [32]. All assessments were performed by one researcher (EH).
Statistical analysis
The sample size was chosen to detect a treatment effect difference of 0.6 standardised (Z score) units for step time variability (the primary outcome measure in the randomised controlled trial) with 80% power and at a two-sided 5% significance [20]. The occurrences of gait interruptions for the walking conditions are presented in tabular and graphical forms. Medical, physical and cognitive measures were contrasted between those who experienced one or more freezes across the walking trials (freezers), and those who experienced no freezes (non-freezers); with Pearson’s Chi-Square tests, Student’s t-tests and Mann-Whitney-U tests as appropriate. Binary logistic regression analysis was performed to identify significant and independent predictors of freezing status, i.e., freezers vs. non-freezers as defined above. The model derived was considered “explanatory” in nature, given the inclusion only of variables that could elucidate why freezes occur. The significance level was set at p < 0.05. Statistical analyses were performed using IBM SPSS Statistics 24 (SPSS Inc., Chicago, IL).
RESULTS
One hundred and thirty people with idiopathic PD aged between 46 and 90 years, with a disease duration ranging from 5 to 13 years completed the assessments. Demographic, medical, physical and cognitive characteristics of the sample are outlined in Table 1.
Participant characteristics for the total sample and those who did and did not experience one or more freezes in the trial walks
1High score reflects poor performance; 2High score reflects better performance; 3High score reflects higher depression level/greater fear of falling. PIGD (Postural Instability/Gait Difficulty) subtype compared to Tremor Dominant/Indeterminate subtype. at-test t(130)/two-tailed unless otherwise marked; bChi2-test; cMann Whitney U test, *significant at p < 0.05, **significant at p < 0.01, ***significant at p < 0.001. UPDRS, Unified Parkinson’s Disease Rating Scale; PIGD, Postural Instability/Gait Difficulty; MoCA, Montreal Cognitive Assessment; Icon-FES, Iconographical Falls Efficacy Scale; PPA, Physiological Profile Assessment.
Gait interruptions
Thirteen people were unable to complete all 3 attempts at the simple (no-additional task) walk (12 people completed 2 walks, 1 completed 1 walk). Fourteen people were unable to complete all three attempts at the easy Dual-Task walk (13 people completed 2 attempts, 1 completed 1 walk). Fifteen people were unable to complete all three attempts at the hard Dual-Task walk (14 people completed 2 attempts, 1 completed 1 attempt). Eighteen people were unable to complete both attempts at the FOG elicitation condition (10 people completed 1 attempt, 8 people completed no attempts).
Fifty-four participants (42%) experienced a total of 391 freezes throughout the simple and DT walks and the FOG elicitation condition. Seventy-six participants experienced no freezes (58.5%), 25 (19.2%) experienced between one and three freezes, 17 (13.1%) experienced between 4–10 freezes, eight (6.2%) experienced between 11–20 freezes, 3 (2.3%) experienced between 21 to 30 freezes, and one (0.8%) experienced >30 freezes (Fig. 2).
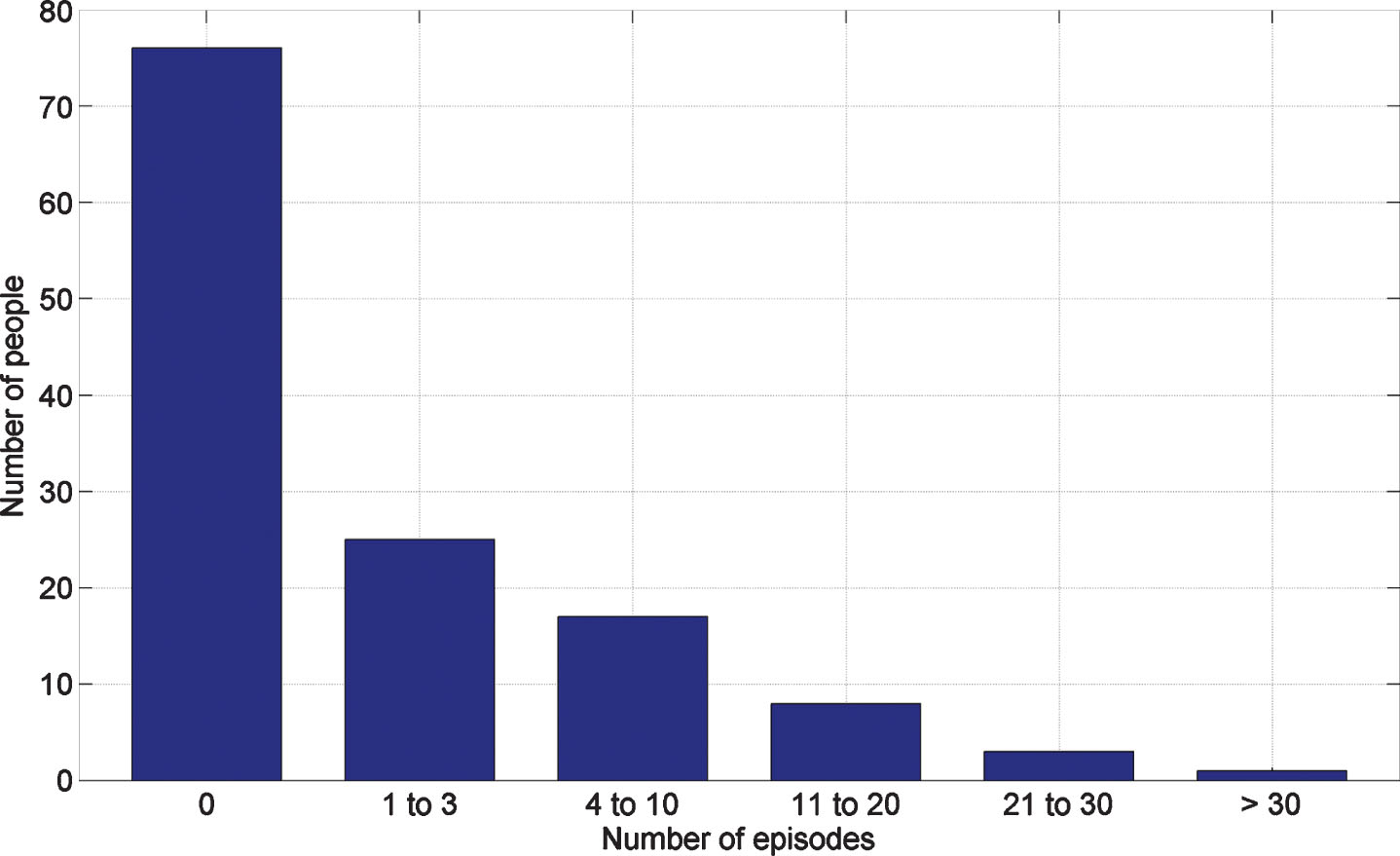
Number of freezes by participants across the four trial conditions.
Of those who experienced FOG, 44 (81.5%) had multiple episodes. No sudden motor ‘offs’ were witnessed. Twenty-four FOG episodes occurred during the simple walk, 67 during the easy Dual-Task walk, 86 during the hard Dual-Task walk and 214 during the FOG elicitation condition (Fig. 3).
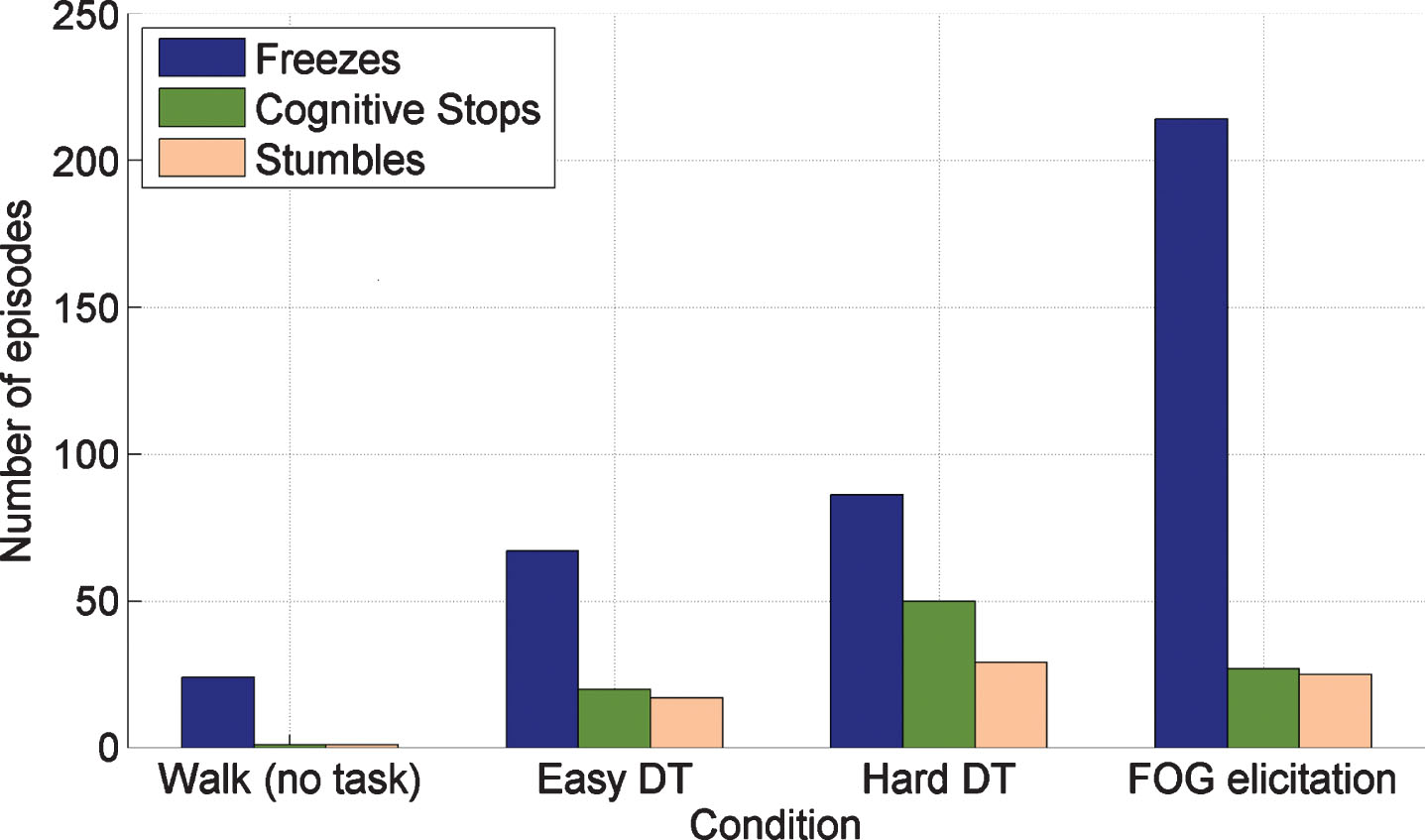
Number of freezes, cognitive stops and stumbles per person across the four trial conditions.
The median (inter-quartile range) duration of freezes were 5.0 s (2.8 – 12.7 s), 3.8 s (2.3–35.2 s), 7.1 s (3.6 – 26.3 s) and 4.9 s (2.3 – 13.4 s) in the simple, easy Dual-Task, hard Dual-Task and FOG elicitation condition walks respectively. Seventy freezes (17.9%) were classified by type as small steps, 188 (48.1%) were trembling legs and 133 (34.0%) were total akinesia. Most freezes in the FOG elicitation condition occurred when turning (66.7%), approaching the destination (11.7%) on initiation of walking (9.4%) (Fig. 4). Only 3.3% of freezes from four participants occurred when walking between the two chairs.
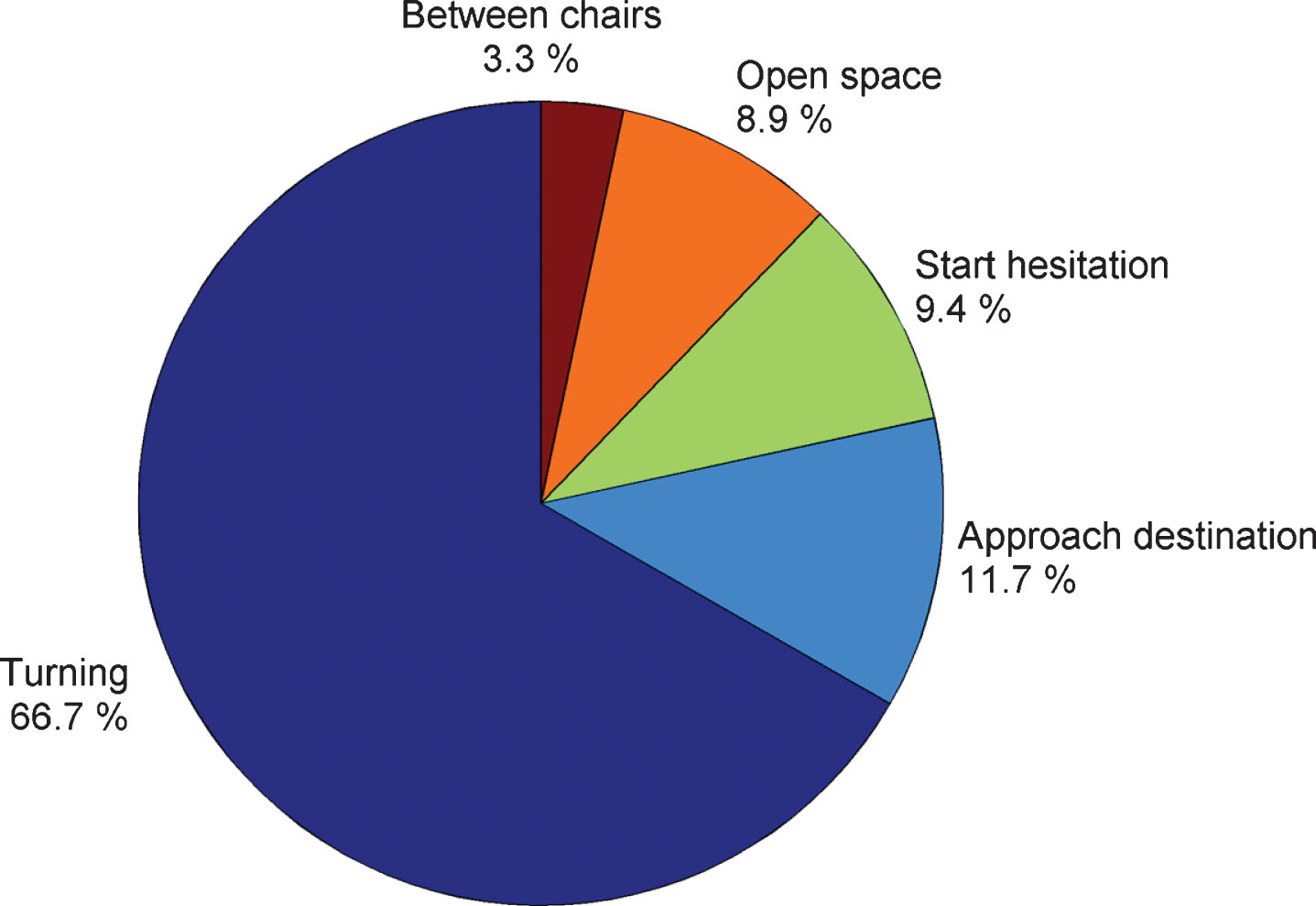
Location of freezing episodes in the freezing of gait elicitation condition.
Other interruptions to gait included 97 cognitive stops experienced by 49 participants and 73 stumbles experienced by 23 participants. No cognitive stops occurred in the simple walk, 20 in the easy DT walk, 50 in the hard DT walk and 27 in the FOG condition walk. Two stumbles occurred during the simple walk, 17 during the easy DT walk, 29 during the hard DT walk and 25 in the FOG elicitation condition. Cognitive stops and stumbles had median (inter-quartile range) durations of 4.5 s (3.6–7.7 s) and 2.4 s (2.0–3.6 s), respectively. No falls occurred during the walking trials.
Freezer versus non-freezer comparisons
Table 1 shows the medical, physical, and psychological measures for the freezers, non-freezers and total sample. Participants who self-reported freezing in the previous month were significantly more likely to freeze during the walking trials. Sensitivity was high (0.85) but specificity was low (0.42) indicating that many of those who reported freezing did not exhibit FOG during the test walks. Compared with non-freezers, the freezers were more likely to have PIGD subtype, longer disease duration, higher UPDRS part II and part III scores and have a higher LED. Freezers also experienced more falls in the previous year and had poorer executive function (lower frontal assessment battery score), increased sway, slower reaction times, poorer coordinated stability, more depressive symptoms, lower QoL and greater fear of falling. Freezers and non-freezers did not differ with respect to age, sex, BMI, vision, quadriceps strength and overall cognitive function (MoCA scores). No significant difference was detected between those who did and did not exhibit cognitive stops for any measures.
Stepwise logistic regression identified PD disease duration, reduced coordinated stability and poor proprioception as independent and significant determinants of freezing status; adjusted odds ratios for these variables are presented in Table 2.
Adjusted odds ratios and 95% confidence intervals for variables included in the stepwise logistic regression model
1Increased odds for each year of PD. 2Increased odds for each degree of error in the lower limb matching task. 3Increased odds for each error made in the coordinated stability test.
DISCUSSION
This video annotation study objectively documented the nature and occurrence of freezes, cognitive stops and stumbles during simple and complex walks in a large sample of people with PD in the “on” medication state. By capturing over 550 gait interruptions, we documented the frequency and duration of FOG episodes, cognitive stops and stumbles across walks with different cognitive and motor stressors, as well as the activity that precipitated the freezing episode. We identified several disease-related measures associated with objectively measured freezing that may assist in understanding and managing this debilitating symptom of PD.
Characteristics of gait disruptions
Across the standard walks, the incidence of freezing increased with increasing complexity of cognitive dual-tasks. Of those who experienced freezing during walking, the majority had multiple episodes and notably a quarter froze on 10 or more occasions. The number of freezes during the FOG condition was more than twice the number that occurred during hard DT walks, despite the total distance walked being only marginally longer: 72 m vs. 66 m respectively. This suggests that the execution of more complex motor activity (gait initiation, gait termination and turning) induces freezing episodes more frequently than the execution of a secondary cognitive task and supports the hypothesis that both automatic and controlled processes are more severely impaired in freezers [33, 34]. The relatively small number of freezes that occurred in narrow quarters between chairs contrasts with previous findings [3]. This might have resulted from differing definitions of freeze circumstance whereby previous studies have reported freezes that occur before, rather than between constrictions [3]. It may also be that two chairs positioned closely together to form a gap have less visual impact than larger frames such as doorways. Trembling legs was documented as the most common type of freezing, a finding consistent with previous studies that have used accelerometers to categorise FOG episodes [16, 35]. However, we acknowledge the relative occurrence of trembling leg and akinetic FOG episodes is unclear due to difficulties in reliably detecting akinetic gait with accelerometers and the potential misclassifications of akinetic FOG episodes and cognitive stops.
Factors associated with FOG
Freezers were significantly more likely to experience cognitive stops and stumbles in the walking trials, which emphasizes their instability and associated elevated risk of falls [5, 36]. Many measures related to PD disease severity (duration of disease, UPDRS II and III scores, LEDs) were identified as risk factors for freezing in bivariate analyses. Self-reported freezing episodes and PD disease severity (as measured with UPDRS that includes freezing as a component), had only moderate predictive value for freezing events in the walking trials. The low specificity of the FOG questionnaire item for freezing episodes most likely relates to participants being assessed in the “on” state when their balance and mobility was optimal, whereas the FOG questionnaire item reflects a one-month time period that would have included end of levodopa dose motor fluctuations. Thus, our test paradigm most likely identifies those more severely afflicted with this phenomenon.
Several physical measures were associated with objectively measured freezing. In the physical profile assessment, which was developed to determine fall risk in older people [30], freezers performed significantly worse in the reaction time and sway tests and as a consequence, had higher composite fall risk scores. Freezers also had poorer controlled leaning balance, and this measure, along with poor proprioception and PD disease duration, were identified as significant and independent risk factors for freezing in the multivariate logistic regression analysis. The controlled leaning balance test used here requires continuous balance adjustments, sustained attention, visual processing and is dependent on processing speed as well as lower limb strength and compact sway [33]. It thus represents a composite measure that involves both automaticity and higher level functioning [36]. The findings complement studies addressing gait in PD freezers that show cognitive load impacts planning of the final steps needed to avoid an obstacle, shifting from more automatic to more consciously controlled balance [37], and that freezing of gait is associated with asymmetric, less rhythmic and uncoordinated gait patterns [38].
The significant association between freezing and impaired proprioceptive processing is consistent with previous studies that proprioceptive loss is associated with impaired motor performance in people with PD [39] and that people with PD who freeze demonstrate an inability to adapt to changes in normal sensory availability [40]. Enhancing proprioceptive input may offer a feasible strategy to address FOG. Pereirra et al. showed that vibration stimulation applied to the triceps surae tendon of the lower affected leg could reduce the severity of FOG episodes [41]. This effect may be mediated via enhanced proprioceptive information processing insofar as steps were more frequently reinitiated following a FOG episode in the contralateral limb.
Freezers did not differ to non-freezers with respect to overall cognition assessed with the MoCA but had significantly lower FAB scores indicating executive dysfunction. Fasano et al. reported in their neuroimaging review that freezers had significant grey matter atrophy in multiple brain areas, especially in the frontal gyrus/supplementary motor area [42] and the link between freezing and executive function has been previously demonstrated [33, 37]. Similar to previous work in this area, freezing was associated with more depressive symptoms, greater fear of falling and lower QoL [4, 39]. The multiple associations uncovered lend further support to the notion that FOG is a multidimensional phenomenon that involves complex motor and cognitive impairments and interactions directly and indirectly related to the neuronal disease [4, 40].
Clinical implications
Our findings have clinical implications in that the sensorimotor, balance and cognitive impairments found to be associated with freezing. These impairments may be ameliorated by current or emergent behavioural and / or rehabilitative strategies and their potential to reduce the occurrence of freezing warrants investigation. These could comprise exercise to improve controlled leaning balance, step initiation and inhibition, proprioceptive cueing as well as counselling programs that emphasise greater concentration in the performance of motor tasks [43].
Strengths and limitations
The main strength of this study is the volume of captured gait interruptions that included over 550 freezes, cognitive stops and stumbles from a series of walks that varied in cognitive and motor complexity. We used definitions and terminology similar to that described in recent study that also used video recording to document FOG episodes [44]. One difference relates to the commencement of FOG where Gilat defined FOG starting when the foot no longer takes an effective step, whereas we took the last heel strike of the last full step as the FOG commencement point as it is easier to precisely identify. A further strength is the use of the gold standard method of video recordings with ancillary reference to synchronised accelerometry data to classify freezing episodes [19, 45]. Thus, given the assessments included cognitive and motor factors known to induce FOG episodes, the incidence and nature of the freezes and other gait disturbances observed should generalise well to those experienced in everyday life. Further research contrasting the determinants of objectively measured and self-reported FOG as well as the determinants FOG circumstances and FOG severity would build on our findings [19].
Our study also has limitations. First, it lacks an inter-rater reliability assessment of the gait disruption categorization, and whilst the rater was trained and strict definitions were adhered to, misclassification of some freezes could, however, have occurred. For example, we may have misclassified a gait interruption as a freeze if the patient did not verbally articulate a difficulty. Second, the sample comprised people with PD who had suffered at least one fall in the previous year, so the findings may not generalise to people with PD who do not fall. Third, participants were tested in a practically defined motoric “on” state, but the average time after medication intake was not recorded. Finally, we acknowledge that the cross-sectional nature of this study means that it is not possible to establish causal relationships between predictor and outcome variables.
Conclusions
This video annotation study captured over 550 gait interruptions and documented the frequency and duration of freezes, cognitive stops and stumbles across walks of variable difficulty. A specific FOG elicitation condition elicited freezes far more frequently than a standard walk whilst performing a concurrent cognitive task. The multiple motor and cognitive factors identified as being associated with freezing, including the inclusion of poor proprioception and impaired controlled leaning balance in the logistic regression model provide new insights into this debilitating PD symptom and may contribute to potential new targets for rehabilitation.
CONFLICT OF INTEREST
All authors declare no competing interests.
Footnotes
ACKNOWLEDGMENTS
We thank Dr Lauren Kark for her supervision of Mira Ketheeswaran research project that contributed to this paper.
Parkinson’s UK funded the trial as part of a fellowship awarded to EJH. British Geriatrics Society and North Bristol NHS Trust awarded EJH a small grant to support pilot work. The funders had no other input into the study or preparation of this manuscript.
