Abstract
Background:
A link between diabetes mellitus (DM) and Parkinson’s disease (PD) have been proposed but evidence are sparse and inconsistent.
Objective:
Perform a systematic review of all evidence that link DM and PD characterising the prevalence of DM in PD patients, the risk of developing PD in DM patients and the influence of DM on PD severity and progression.
Methods:
MEDLINE, Scopus, and Cochrane Library from inception to June 30, 2021 were searched. Studies reporting prevalence, incidence, severity and disease progression of DM and PD were included. Prevalence of DM in PD and incidence of PD in DM patients, and characteristics of PD.
Results:
A total of 21 studies (n = 11,396) included data on DM prevalence in PD patients, 12 studies (n = 17,797,221) included data on incidence of PD in DM patients, and 10 studies (n = 2,482) included data on DM impact on PD severity and disease progression. The prevalence of DM in PD patients was 10.02 %, (95%C.I. 7.88 –12.16), DM patients showed a higher risk of developing PD (OR: 1.34 95%CI 1.26–1.43 p < 0.0001) compared to non-DM, and PD patients with DM showed a greater severity of motor symptoms, with higher Hoehn and Yahr stage (SMD: 0.36 95%CI 0.12–0.60; p < 0.001) and higher UPDRS (SMD 0.60 95%CI 0.28–0.92; p < 0.001) compared with PD patients without DM.
Conclusion:
Although the prevalence of DM in PD patients is similar to the general population, patients with DM have a higher risk of developing PD, and the presence of DM is associated with greater PD severity and faster progression, which suggests that DM may be a facilitating factor of neurodegeneration.
INTRODUCTION
Potential relations between Parkinson’s disease (PD) and diabetes mellitus (DM) have been described during the last decades [1]. Although pathophysiological mechanisms, such as inflammation and oxidative stress, have been suggested as shared common pathways in DM and PD, it is unclear how and whether the two diseases are linked. Experimental studies showed that hyperglycemia impairs nigrostriatal dopaminergic transmission and insulin signaling is involved in the regulation of brain dopaminergic activity [2, 3]. In animal model of insulin resistance, decreased nigrostriatal dopamine release was associated with peripheral insulin-resistance and dopamine uptake was impaired [4]. Epidemiological studies have not consistently defined an association between PD and DM, since different case-control or cohort studies have reported conflicting results [5, 6]. Furthermore, even available systematic reviews yielded inconsistent results, suggesting either a bidirectional increased risk or no association at all [7, 8]. In particular, DM has been associated with postural instability, gait difficulty [9, 10], and cognitive decline [11] in PD patients, further onset and progression of motor abnormalities and cognitive decline have been described in older adults with DM and without PD diagnosis [12]. The concomitant presence of DM in PD patients has been described to increase motor symptom severity and to reduce levodopa therapeutic efficacy [13, 14]. Recent articles have summarized the role of DM on PD clinical presentation [7, 15]; however, the impact of DM on PD phenotype and progression, should be better clarified. Thus, the purpose of the present systematic review and meta-analysis was to explore the association between DM and PD with the following specific aims: To assess the prevalence of DM in PD patients; To assess the risk of developing PD in DM patients; To assess the impact of DM on PD severity and disease progression.
MATERIALS AND METHODS
This systematic review was performed according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) [16] criteria and the recommendations in the Preferred Reporting Items for Statistic Reviews and Meta-Analyses (PRISMA) statement [17]. This study followed a pre-determined unpublished protocol available upon request.
Search strategy and selection criteria
Studies were identified and evaluated independently by two authors (KK, GP) in different databases including: MEDLINE/PubMed, Cochrane CENTRAL, and Scopus until June 30, 2020. The following free text terms and or MeSH terms according to the requirements of each database were used for the search strategy: “Parkinson’s Disease”, “Diabetes Mellitus”, “Insulin Resistance”, “Glucose Intolerance”. Additional eligible studies were identified by screening the reference lists of studies included in our analysis. Only articles published in English language were considered.
All selected titles and abstracts were independently reviewed by two authors (KK, GP). Studies were considered eligible if they fulfilled the above criteria: (a) they reported prevalence of DM in patients with PD; (b) they reported the incidence or the occurrence of PD in patients with and without DM; (c) they reported relevant analysis of PD phenotype and disease progression in patients with and without DM; (d) case-control, cross-sectional and cohort design were considered. Studies where DM diagnosis was based on clinical evaluations, self-reported, ICD codes or antidiabetic therapy were considered for inclusion. Reviews, case-reports, non-human studies, book chapters, overlapping studies and abstracts or conference proceedings were excluded.
Risk of bias and quality assessment
Using the guidelines in the Newcastle-Ottawa Scale (NOS) [18], included studies were graded by two independent reviewers (KK, GP) based on selection, comparability, exposure (case-controls design) or endpoint (cohort design). These items were categorized into three major components, containing eight items. For studies where cross-sectional design was clearly stated in methodology description, Joanna Briggs Institute (JBI) checklist for cross-sectional studies was used [19]. Disagreement was resolved by consensus of a third reviewer (GR). Presence of publication bias was explored visually performing the test for asymmetry of the funnel plot by Egger test [20].
Data extraction
Two reviewers (KK, GP), independently using a standardized form, completed data extraction. Disagreement was resolved by consensus and by the opinion of a third reviewer (GR) when necessary. Information was recorded on: study year, author first name, data regarding prevalence/incidence of PD or DM, setting, diagnosis criteria of PD and DM, sample size and characteristics of patients.
Statistical analysis
The prevalence of DM was summarized using descriptive statistics. Pooled prevalence rates accounting for inter-study variation were analysed using a non-linear random effects model and statistical uncertainties were expressed in 95%Confidence Intervals (CI). Continuous outcomes, mean difference (MD) or standardized mean difference (SMD) were calculated for each outcome. The results were pooled using the inverse variance method. Odds ratios (OR) and 95%CI were calculated for the risk of PD outcome by using the intention-to-treat principle. The choice to use OR was driven by the retrospective design of the meta-analysis based on published studies that varied in design, subjects’ population, treatment regimen, primary outcome measure and quality. Heterogeneity was assessed using I2 statistic that accounts of between-study (or inter-study) variability as opposed to within-study (or intra-study) variability. Because of latent clinical heterogeneity, random effects model was used to synthesize data than the fixed effects model, independently of statistical evidence for heterogeneity. Heterogeneity has been considered substantial if I2 value was greater than 25%.
To explore the influence of potential effect modifiers on endpoints, a meta-regression analysis was performed to test age, sex (male %), body mass index (BMI), smoking, hypertension (%), and disease duration in years. For all meta-regression analyses, random effects model was used to take into account the mean of a distribution of effects across studies. Subgroup analysis was performed for studies where DM diagnostic criteria were based on clinical evaluations vs medical records and or self-reported and for studies where all the included population presented type 2 DM population vs studies without clear type 2 DM frequency. All reported test results were two-tailed and a p value≤0.05 was considered significant. Data analyses were performed with STATA version 16.
RESULTS
A total of 3,829 articles have been identified by the initial search (Fig. 1). Ninety manuscripts have been retrieved for more detailed evaluation and the studies finally included in the systematic review were: 21 for the assessment of the prevalence of type 2 DM in PD patients (aim 1, Supplementary Table 1); 12 for the evaluation of the risk of PD in DM patients (aim 2, Supplementary Table 2); 10 for the impact of type 2 DM on PD severity and disease progression (aim 3, Table 1).
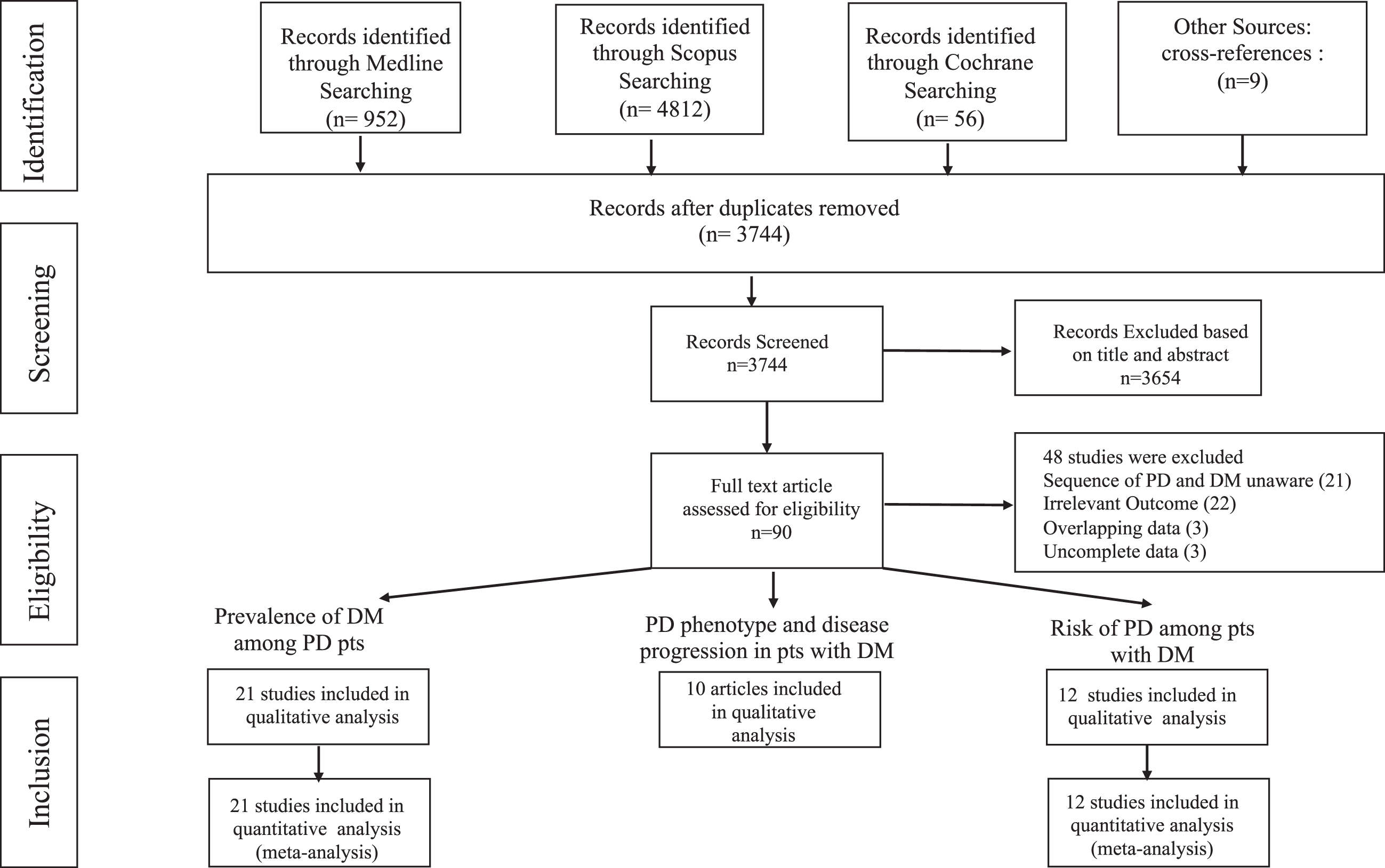
Flowchart indicating search results.
Characteristics of the included studies for aim 3: PD phenotype and type 2 DM
T2DM, type 2 DM; PD, Parkinson’s disease; HY, Hoeg and Yarh; N/R, not reported.
Prevalence of DM in patients with PD
A total of 21 studies reporting the prevalence of DM in patients with PD have been identified [5, 21–38]. A total number of 11,396 patients was identified with an average age ranging from 58.1 to 79 years. The population presented a not negligible variability in rate of male gender (ranging from 37.4%to 100%), BMI (from 22.3 to 27.3 Kg/m2), disease duration (from 0.1 years to 9.25), Hoen and Yarh (H&Y) Stage (from 1.5 to 2.73), rates of active smokers (from 17.6 %to 41.6%), and hypertensive patients (from 15.6%to 43.4%). The weighted prevalence of DM in patients with PD was 10.02%, (95%[C.I.] 7.88 –12.16; I2 = 92.9%, Fig. 2). The potential effect modifiers tested to explain this high heterogeneity (I2) were age, gender (%men), BMI, disease duration, rates of active smokers, and hypertension (%of hypertensive patients). Meta-regression analysis revealed that age (beta coefficient 0.81 95%CI 0.286–1.32 p = 0.005) and hypertension were significant moderators (beta coefficient 0.53 95%CI 0.359–0.721 p < 0.001), while gender, disease duration, BMI and smoking were not relevant moderators.
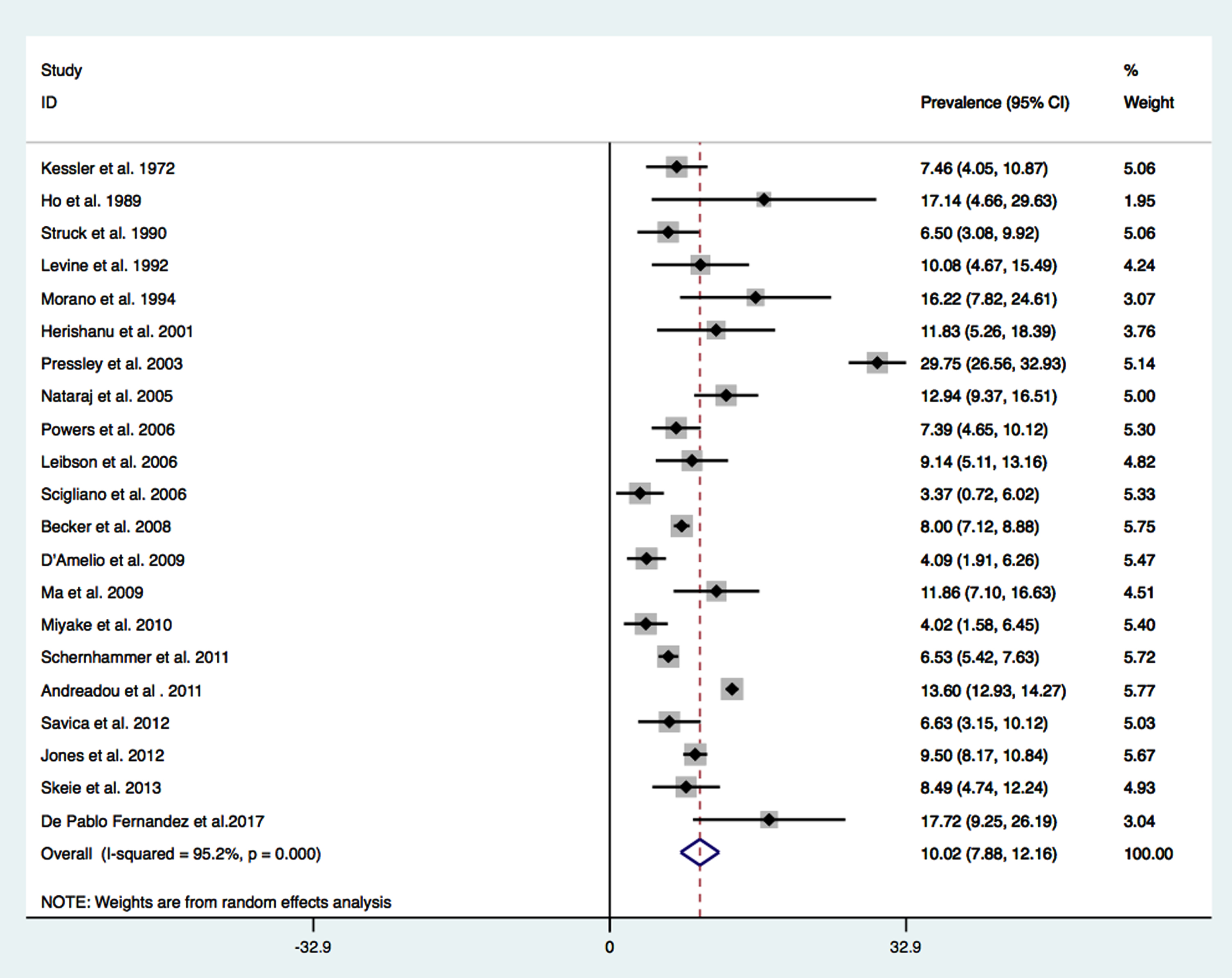
Type 2 DM prevalence in PD.
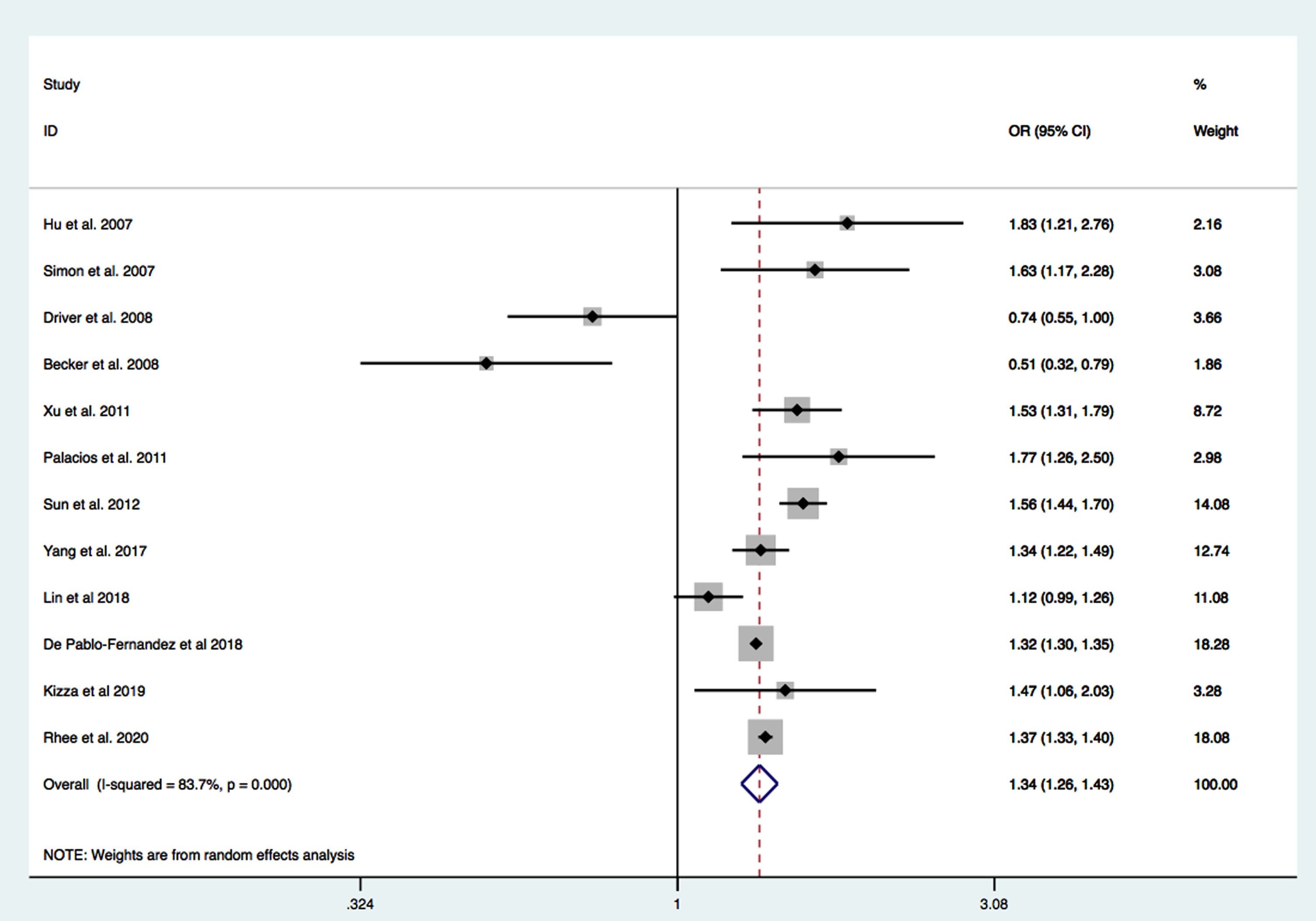
PD risk in type 2 DM.
Risk of developing PD in patients with DM
A total number of 17,797,221 participants from 12 cohort studies were included in this analysis [30, 39–49]. The average age at inclusion ranged from 43 to 63 years, while male gender range was from 30 to 100%(Supplementary Table 2). In most cohorts diagnoses of DM and PD were based on health questioners, medical records, whereas only two studies provided diagnosis based on patient’s clinical evaluation. The pooled summary OR for developing PD among patients with type 2 DM compared to non-diabetic (non-DM) patients with PD was 1.34 95%CI 1.26–1.43 p < 0.0001 (Fig. 4). The heterogeneity was high (I2 = 83.7%) Based on the available data we tested as possible variables influencing the high heterogeneity: age, gender, BMI, follow-up duration, hypertension, and smoking status. Meta-regression analysis did not reveal any significant moderator.
Subgroup analysis
Regardless differences in diagnosis criteria for DM clinical evaluations vs medical records or self-reported, a positive relationship between DM and risk of developing PD was observed: OR 1.70 95%CI 1.30–2.20 p < 0.0001 I2 = 0%clinical evolutions and OR 1.33 95%CI 1.25–1.41 p < 0.0001 I2 = 84.6%self-reported and or medical records (Supplementary Figures 1 and 2).
For both patients where only type 2 DM were considered and those where type 1 DM could also have been included, DM presence was associated with increased risk for PD development: OR 1.17 95%CI 1.01–1.36 p < 0.03 I2 = 87.5%and OR 1.47 95%CI 1.34–1.64 p < 0.0001 I2 = 65.6%(Supplementary Figures 3 and 4).
Impact of DM on PD severity and disease progression
Ten studies have been included for evaluating the impact of DM on PD severity and disease progression, including 603 patients with concomitant PD and DM, and 1,879 non-DM patients with PD. In the overall population, mean age ranged from 62.9 to 80.7 years, while PD duration from 3.9 to 9.1 years (Table 1). Data on motor symptoms in PD patients, with a specific focus on the presence of bradykinesia, rigidity, tremor or postural impairment, were not available in all studies; therefore, we performed a meta-analysis considering H&Y Stage, Unified Parkinson Disease Rating Scale (UPDRS), and cognitive impairment. Hedge’s Test indicated that patients with concomitant PD and type 2 DM presented a worse H&Y stage (SMD 0.36 95%CI 0.12–0.60; p < 0.001), and higher UPDRS (SMD 0.60 95%CI 0.28–0.92 p < 0.001) (Supplementary Figures 5 and 6). Since cognitive performance was evaluated with different tools, data could not be compared. However, in seven studies the presence of DM in PD was associated to a worse cognitive performance [14, 50–52]. Moderate heterogeneity among studies was found for UPDRS (I2 = 49.38%) and cognitive impairment (I2 = 42.26%). Meta-regression analysis, including age, gender, disease’s duration, BMI, and hypertension, did not find relevant moderators.
Study quality
The quality of the included studies, evaluated by NOS criteria was moderate or good, ranging from 4 to 8 points. JBI checklist for cross-sectional studies, indicated a good overall appraisal. Results of the NOS quality assessment and JBI checklist of the included studies are reported in Supplementary Tables 3 and 4.
Publication bias
Asymmetry was assessed by visual inspection of funnel plots. However, Egger’s regression test did not indicate significant publication bias among the included studies. Respectively for aim 1: p = 0.167; aim 2: p = 738; aim 3: considering H&Y Stage p = 0.866 considering UPDRS score p = 0.392 (funnel plots in Supplementary Figure 7A-D).
DISCUSSION
The main results of the present study include: a) an overall prevalence of DM in patients with PD of 10.02%; b) an higher risk of developing PD in patients with DM compared to non-DM patients with PD (OR = 1.34, 95%CI 1.26–1.43; p < 0.0001); c) a greater severity and a faster disease progression in patients with concomitant PD and DM.
The global DM prevalence reported in 2019 was 9.3%, with Type 2 DM representing approximately 90%of the total cases of DM [53]. Data on DM prevalence in PD mainly derive from monocentric studies frequently obtained on small number of patients, and the results are widely heterogenous ranging from 3.4 to 9.1%[7]. In our meta-analysis, performed on 21 studies, we have described a similar global prevalence of type 2 DM among PD patients compared to the prevalence described in the overall population. Data from a study including 3,069 participants aged 70–96 years, focusing on the role of DM on cognitive decline, also report that the overall prevalence of DM is about 9%[54]. Whereas in a study based on the neuropathological diagnosis of dementia the prevalence was 12%in Alzheimer’s disease and up to 31%in vascular dementia [55].
Aging is widely recognized as a significant factor influencing diabetes onset and progression, indeed, the prevalence of DM among elderly is reported to rise up to 30%in US [56]; consistently, we have identified age as a factor influencing DM prevalence in our study. Age-dependent impairment in insulin sensitivity and pancreatic islet function, together with physical inactivity, represent pathophysiological mechanisms explaining the higher prevalence of DM in the elderly [57] and in PD patients, usually presenting a higher degree of physical impairment. Furthermore, age-related increase of visceral adiposity and reduction of skeletal muscle mass are associated to insulin resistance, which may be undetected in elderly population affected by PD, and may influence independently of DM the development and progression of PD [58, 59].
In addition, we have identified DM as a risk factor for PD development, confirming the results of two previous meta-analyses [8, 60]. However, both previous meta-analysis included a limited number of studies, whereas we conducted a meta-analysis on 12 cohort studies, characterized by an overall good quality and including a very large number of patients. Of note, a recent observational cohort study [48] with a long-term observational period (49076148.74 person-years) suggested that DM is a potential risk factor for PD, independently from other main comorbidities such as cardiovascular, cerebrovascular, and renal diseases. These results suggest that the risk of developing PD is higher in DM patients and that DM might facilitate the neurodegenerative process and drugs that improve DM might have a potential neuroprotective effect. Furthermore, a recent meta-analysis study found that DM conferred a 1.25 to 1.91 fold excess risk for cognitive impairment and dementia, strengthening the role of DM in the neurodegenerative process [61]. Future studies are needed to test the hypothesis that antidiabetic drugs might reduce the risk of PD in DM patients. As expected, the heterogeneity in our study was high, probably as a consequent of the observational nature of the included studies.
Regarding clinical manifestation and disease progression, patients with concomitant PD and DM experience a higher impairment in motor signs. We were not able to perform a quantitative analysis regarding specific clinical presentation of PD motor signs; nonetheless, the global PD signs were significantly worse among PD patients with type 2 DM. Two studies reported a worse DM-dependent postural instability without influence on bradykinesia, tremor, or rigidity [10, 12]. Giuntini and colleagues [14] have found a worse disease progression regarding gait domains and cognitive performance. Worse semantic fluency score was also observed by Jones et al. [11]. Ong and Petrou [50, 51] reported that the presence of DM in PD patients was associated with greater white matter atrophy, even if initially not associated with a decline in cognitive performance. However, the longitudinal progression of cognitive performance was accelerated in DM-PD patients compared to non-diabetic PD patients. Of note, other two studies [52, 62] have also reported a faster progression of PD’s severity regarding motor phenotype and cognitive performance. Increased postural instability has been described [10, 14], even if dopaminergic impairment did not explain this feature, since PET, MRI imaging techniques and vibratory detection analysis did not reveal significant differences regarding striatal dopaminergic denervation, leukoaraiosis, or large fiber polyneuropathy. Alternative pathways may be linked to insulin resistance and mitochondrial dysfunction, accordingly several experimental models of PD have demonstrated that stimulation of Glucagon Like Peptide 1 Receptors (GLP1 R) may provide neuroprotective effects with amelioration of motor and non-motor signs [63]. Promising data from a randomized double-blind placebo-controlled trial on the GLP1 R agonist exenatide patients suffering from PD reported improvement in motor scores and persistence of positive effects [64]. Following these promising results, a phase III clinical trial was initiated in which exentaide is administered on a weekly basis over two years (NCT04232969). Two phase II double-blind placebo-controlled trials in PD patients are ongoing, one evaluating the safety and efficacy of daily subcutaneous administration of liraglutide on motor and non-motor function at 28 and 54 weeks of follow-up (NCT02953665) and another evaluating the efficacy of lixisenatide on the MDS-UPDRS III motor examination score (evaluated in the best ON condition) at 12-month follow-up (NCT03439943).
We performed an observational study focusing on early-stage PD patients which demonstrated bradykinesia to be more pronounced in patients affected by PD and DM, DM associated with greater striatal dopaminergic deficits and increased cerebrospinal fluid tau protein [62]. We also showed that the presence of diabetes mellitus was associated with faster motor progression and cognitive decline at 36-month follow-up. Modification of tau protein level has been well described in patients with Alzheimer’s disease and DM [65], supporting the hypothesis of a cross-link between tau, DM, and cognitive impairment. The overall analysis of the included studies showed a trend toward a more pronounced cognitive decline in DM and PD patients. Therefore, particular attention should be paid to progression of motor and cognitive symptoms in diabetic PD, and additional studies are needed in order to better define the clinical phenotype of PD-DM patients and explore the role of antidiabetic drugs on PD progression.
The mechanisms underlying the connection between PD and DM are not fully understood, however studies on experimental models have allowed to speculate on presumable shared pathophysiological pathways: a) chronic hyperglycaemia leads to degeneration of the nigrostriatal pathway and dopaminergic neurons loss, through the activation of oxidative stress [66]; b) overproduction of reactive oxygen species in PD may accelerate mitochondrial dysfunction and insulin resistance onset and progression [67]; c) insulin signalling mediates alpha-synuclein expression and aggregation, resulting in dopaminergic neuronal dysfunction [63]; d) alpha-synuclein has been suggested as a key modulator of insulin release and glycaemic metabolism [68, 69]; e) impairment of sympathetic nervous system may effect both PD and DM onset [69].
Insulin resistance is considered a precursor of diabetes and correlation between PD symptoms and the presence of insulin resistance has been described [70]. In 62%of PD patients with dementia, characterized by a longer disease duration and motor impairment, insulin resistance was found [70], while almost two-thirds of PD patients despite normal fasting glucose and normal glycated haemoglobin, is present insulin resistance [59]. Overlapping mechanisms between insulin resistance, mitochondrial disfunction, oxidative stress, alpha-synuclein expression could influence the development of neurodegeneration process. In animal model of peripheral insulin resistance, together with impairment of dopamine turnover, increased iron deposition in substantia nigra was also detected [4]. High mitochondrial iron content has been associated to oxidative damage, and modification of mitochondrial function has been linked to PD [71]. Data from a recent in vivo and in vitro experimental study of high fat diet animals, reveal that insulin resistance promotes the development and progression of PD through overactivation of polo-like kinase-2 (PLK2) signalling, which mediates mitochondrial dysfunction and modification of alpha-synuclein expression and signalling [72].
Previous studies have also demonstrated that macroscopic and microscopic infarcts and atherosclerosis are common in elderly with parkinsonian gait [73]. Vascular comorbidity as DM, hypertension, smoking, and dyslipidaemia were significantly associated with a gait and cognitive impairment in early PD [74]. Cerebrovascular diseases are characterized by fragmentation, injury and remodeling of capillary network in different brain areas. DM as a potential vascular risk factor may play a crucial role in the capillary brain remodeling process localized in brain stem nuclei, substantia nigra, and middle frontal cortex.
Activation of nucleotide-binding oligomerization domain, leucine rich repeat and pyrin domain-containing (NLRP) 3 inflammasome, is another important mechanism linked to PD pathogenesis. NLRP3 binds to apoptosis-associated speck-like protein CARD (PYCARD/ASC) and activates pro-caspase 1, transforms inactive pro-IL-1β and pro-IL-18 into their biologically active secreted forms and triggers pyroptosis and cell death [75, 76]. The evidence has demonstrated that chronic upregulation of IL-1β and IL-18 leads to increased insulin levels, which may be important for DM development [77]. In addition, alpha-synuclein deposition activates NLRP3 inflammation, which in turns may enhance PD development but also interfere with glucose metabolism [78].
This study has some limitations that should be mentioned. First, in many studies, type 2 DM and even PD diagnostic criteria were only based on medical records, or self-reported health questionnaires; confirmation, through laboratory or clinical/instrumental findings, was rarely performed. Second, not all the studies clearly stated in their method that the included population presented type 2 DM, other studies based on the prevalent elderly population, the rare prevalence of type 1 DM among elderly in the years when studies were conducted, and use of oral antidiabetic drugs very probably included only type 2 DM population. Third, patients affected by DM may be characterized by an increased risk of cardiovascular death and this may influence, follow-up regarding PD development. Moreover, important heterogeneity was registered, and publication bias may be present, but it should be expected in meta-analysis of observational studies. Finally, due to the unavailability of the information regarding type 2 DM duration, medication history for type 2 DM or PD, identification of cases with vascular-type or not-idiopathic PD in some included papers, data analyses on these characteristics was not performed and identification bias cannot be excluded.
CONCLUSIONS
About 10%of PD patients are affected by DM and diabetic patients suffer from higher risk of developing PD. Notably, patients with concomitant PD and DM are burdened by a more severe form of PD, characterized by more frequent postural instability, gait abnormalities, cognitive decline, and faster disease progression. Based on these results, DM should be considered a facilitating factor of neurodegeneration and PD development. Accordingly, the screening of PD among diabetic patients should be accurately performed and potentially include DM in the identification of prodromal PD.
Footnotes
ACKNOWLEDGMENTS
Dr. Leonardo Bencivenga has been supported by a research grant provided by the Cardiopath PhD program.
CONFLICT OF INTEREST
Dr. Gennaro Pagano is a full-time employee and a shareholder of F.Hoffmann-La Roche Ltd. All authors declare no conflict of interest.
