Abstract
Background:
Inflammation is an integral part of neurodegeneration including in Parkinson’s disease (PD). Ashkenazi Jews have high rates of genetic PD with divergent phenotypes among
Objective:
To assess central and peripheral cytokines among PD patients with mutations in the
Methods:
The following cytokines were assessed from peripheral blood and cerebrospinal fluid (CSF): TNF-α, IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10 and INF-
Results:
Data from 362 participants was collected: 31 idiopathic PD (iPD), 30
Conclusion:
In this study, we could not detect any evidence on dysregulated immune response among
INTRODUCTION
α-synuclein accumulation is the pathological hallmark of Parkinson’s disease (PD). It occurs within the central nervous system (CNS) and in the autonomic nervous system [1]. The neurodegenerative process of PD is associated with an inflammatory response which can be detected in peripheral cytokines, stool samples, and colonic biopsies [2]. Age-dependent spread of α-synuclein from gut to brain has been previously reported [3] raising the possibility that the inflammation and α-synuclein spread are intertwined and represent an early phenomenon in the pathogenesis of PD [4]. However, whether this process is initiated in the CNS or peripheral nervous system (PNS) is still debated [5]. Interestingly, these inflammatory processes occur in patients with a short disease duration [6] and abate with disease progression.
Neuroinflammation in PD is associated with a chronic immune response in the CNS, in which innate immune cells (astrocytes and microglia) are responsible for an inflammatory response and potentially neural death [7]. Whether the microglial response is a consequence of PD related cell death or an independent process is still debated [8]. PD has been associated with the accumulation of pro-inflammatory cytokines including tumor necrosis factor alpha (TNF-α), interferon gamma (INF-
Among Ashkenazi Jews (AJ), mutations in the
As the penetrance of the mutations in the
In this study, we aimed to characterize the inflammatory profile of patients with PD carriers of
METHODS
This study evaluated demographic, laboratory and clinical data from subjects who participated in the BEAT-PD study (TLV-0204–16), a natural history study which set out to characterize
Procedure
Participants were genotyped for the G2019S-
Disease severity was assessed using the MDS-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) during ON medication [32]. The Montreal Cognitive Assessment (MoCA) was used to assess global cognitive functions [33]; mood was assessed using the Beck Depression Inventory (BDI) [34]. The Non-Motor Symptoms Questionnaire (NMSQ) [35], the Scale of Autonomic Function in PD (SCOPA-AUT) [36] and the REM sleep Behavior Disorder Questionnaire (RBDQ) were collected [37]. Olfaction was tested using the University of Pennsylvania Smell Identification Test (UPSIT) [38]. These measures were used to calculate the probability for prodromal PD (Likelihood Ratio Score) for all participants without a diagnosis of PD, based on the updated MDS task force prodromal criteria guidelines [39]. Each non-manifesting subject was allocated a ratio between 0–100%for risk for future development of PD based on risk and prodromal symptoms. Levodopa equivalent daily dose (LEDD) was calculated for all patients [40]. In addition, all participants were assessed for inflammatory disease and use of anti-inflammatory medications including aspirin and antibiotics in the days prior to participation.
Blood tests were collected from all participants without an overnight fast. A complete blood count was drawn with white blood count (WBC), hemoglobin, platelet count, absolute lymphocyte and neutrophil levels recorded, and a ratio of lymphocyte/neutrophil (NLR) calculated. Uric acid, urine microalbumin, and CRP were assessed as well.
Blood samples were centrifuged at 1500×g at 4°C for 15 min, 30 min after blood draw. The serum was separated and aliquoted into 2 ml tubes and placed in a –80°C freezer. For the cytokines panel assay, serum samples were thawed on wet ice and sub aliquoted to 105μl in cryotubes.
Lumbar punctures were performed in a sub-group of participants as detailed previously [41]. CSF was collected in 15 ml conical tubes and centrifuged at 2000×g at 25°C for 10 min. The supernatant was collected to a conical tube, aliquoted into 0.5 ml and frozen at –80°C. For the cytokine measurement, the CSF aliquots were thawed on wet ice and sub-aliquoted to 100μl in cryotubes. The CSF samples were stored in –80°C and shipped in dry ice. CSF and serum were taken at the same day for those who underwent a lumbar puncture.
Human high sensitivity cytokine Luminex custom 8-plex kits were used to assess cytokine levels. The assays were performed in 96-well plates according to the manufacturer’s instructions (R&D Systems, Inc. Minneapolis, MN USA). Standards were run in duplicates, while samples were run in singles. Briefly, the Luminex Assays utilize color-coded superparamagnetic beads coated with analyte-specific antibodies. Beads recognizing different target analytes were mixed together and incubated with the bio-fluid sample. Captured analytes are subsequently detected using a cocktail of biotinylated detection antibodies and a streptavidin-Phycoerythrin conjugate. Magnetic beads were isolated and measured by using the Luminex MAGPIX® Analyzer. The following cytokines were detected in serum and CSF: TNF-α IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, and INF-
For the GCase analysis blood samples were collected in a 10 ml EDTA tube. Fifty microliters of whole blood were dropped on each of ten circles on a filter paper and dried at room temperature for at least 4 hours. The filter paper was then stored in a sealed plastic bag at room temperature until the samples were shipped for processing at Centogene (Rostock, Germany). A specific synthetic substrate was added, and the plates were incubated further at 37°C for 16 h. The reaction was stopped by changing the pH to 10.5. The enzymatic product was quantified by fluorimetry using an external standard curve. The results of the enzymatic activity determination were calculated in μmol/L/h. As quality parameters for the assays, standard blood samples were added to each batch to ensure the accuracy of the determination.
Statistical analysis
Descriptive statistics (means and standard deviations (SD) for continuous variables) were computed for all measures. Missing cytokine variables were treated with two-imputation methods: 1) Taking the panels’ threshold as the value; 2) Imputed them as 0. The two methods allowed us to examine the sensitivity of the analysis in two extreme ends of possible values.
Log transformation was performed to reduce the natural skewness of the following variables: peripheral cytokines: TNF-α, IL-2, IL-1-β, IL-6 and IL-10; CSF cytokines: TNF-α, IL-1β, IL-6 INF-
We first created a linear model based on age, disease duration, body-mass index (BMI), use of anti-inflammatory medication and presence of additional inflammatory diseases for each of the CSF/peripheral cytokines. Next, the residuals of each group were compared using Wilcoxon Test in order to avoid any assumption about the residuals’ distribution. Due to the limited number of observations and natural skewness of the measures, we used the non-parametric Wilcoxon’s test which is appropriate when there is no evidence for normal distribution. This test is known to be more powerful than the
Non-parametric Spearman-rho correlations (used to identify monotonic relationships) between cytokines and additional laboratory and behavioral measures for each study group were assessed. The resulting
Finally, random forest classification was used on the cytokines to classify the participant into genetic groups. This was performed separately for the PD subgroups and the non-manifesting subjects. In this model we used the original cytokine values as explanatory variables in conjunction with the covariates of the linear model detailed above. Since random forest is based on decision trees, it can be used to identify inflammatory profile for each group of participants. Statistical analysis was performed using R version 4.0.3.
RESULTS
Data was collected from 356 participants: 31 idiopathic PD (iPD), 30
Characteristics of the PD cohort
iPD, idiopathic Parkinson’s disease; M, male; F, female; BMI, body mass index; LEDD, LevoDopa Equivalent Daily Dose; MDS-UPDRS, Movement Disorder Society - Unified Parkinson’s Disease Rating Scale; MoCA, Montreal Cognitive Assessment; UPSIT, University of Pennsylvania Smell Identification Test; RBDQ, REM Sleep Behavior Questionnaire; SCOPA-AUT, Scales of Outcomes in PD –autonomic; GCase, beta glucocerebrosidase; WBC, white blood cells. Data is shown as mean±standard deviation or percentage. *
Characteristics of the Non manifesting participants
NMC, non-manifesting carrier; M, male; F, female; BMI, body mass index; MDS-UPDRS, Movement Disorder Society - Unified Parkinson’s Disease Rating Scale; MoCA, Montreal Cognitive Assessment; UPSIT, University of Pennsylvania Smell Identification Tes;, GCase, beta glucocerebrosidase; WBC, white blood cells. Data is shown as mean±standard deviation or percentage. *Significant difference between
CSF samples were collected from a subgroup of participants that underwent lumber puncture (
We compared behavioral and laboratory measures within the three groups of PD participants (iPD,
GCase activity was significantly lower among
Peripheral INF-
No significant difference in CSF or peripheral cytokine levels were detected between any of the PD subgroups or the non-manifesting subgroups when missing levels were computed as “0” or as the specific batch threshold (Figs. 12). Furthermore, assessing cytokine differences between mGBA-PD and sGBA-PD and between mGBA-NMC and sGBA-NMC did not yield any differences.
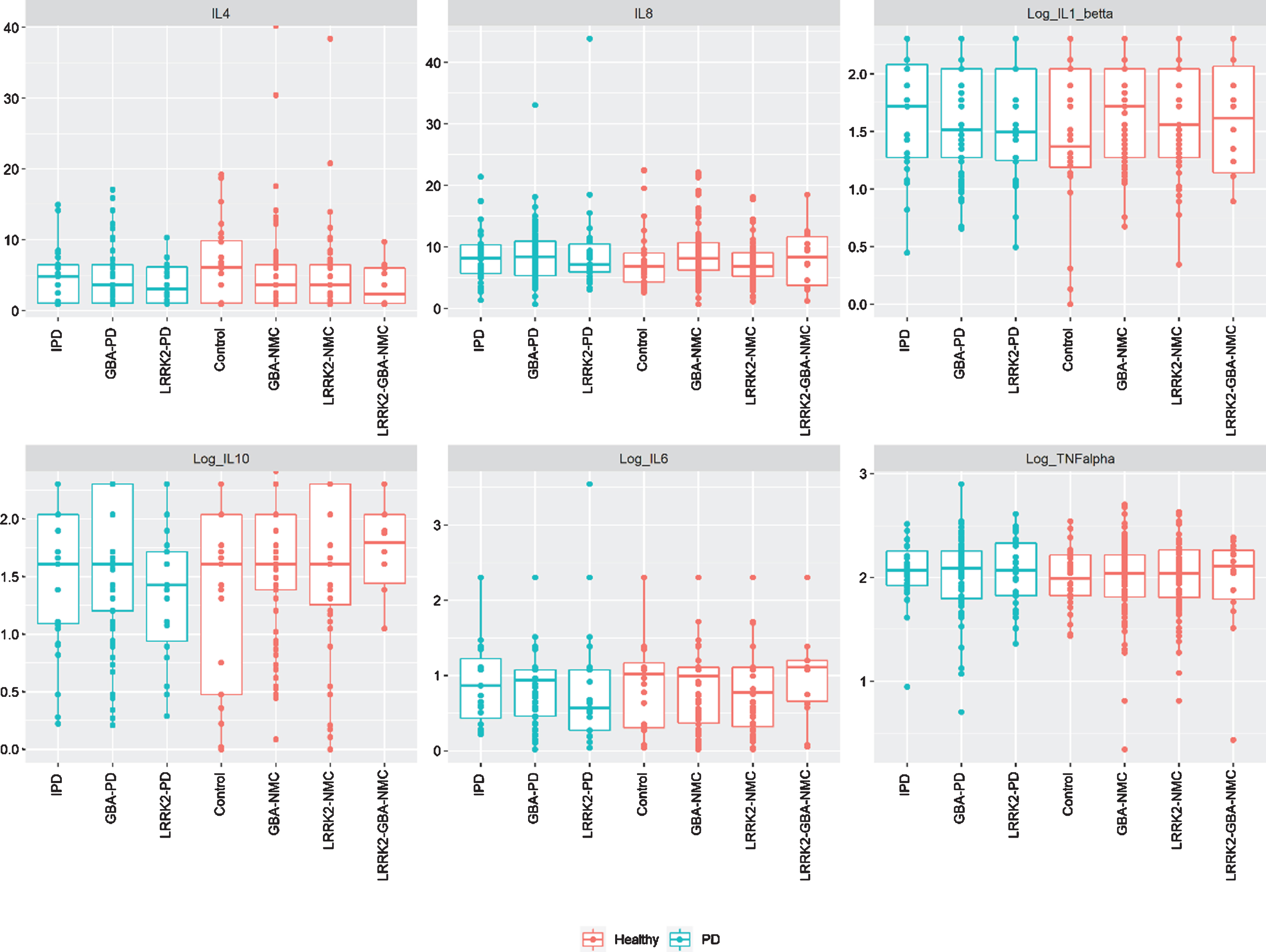
Levels of peripheral cytokines among study participants iPD, idiopathic Parkinson’s disease; NMC, non-manifesting carriers; IL, interleukin; TNF, tumor necrosis factor.
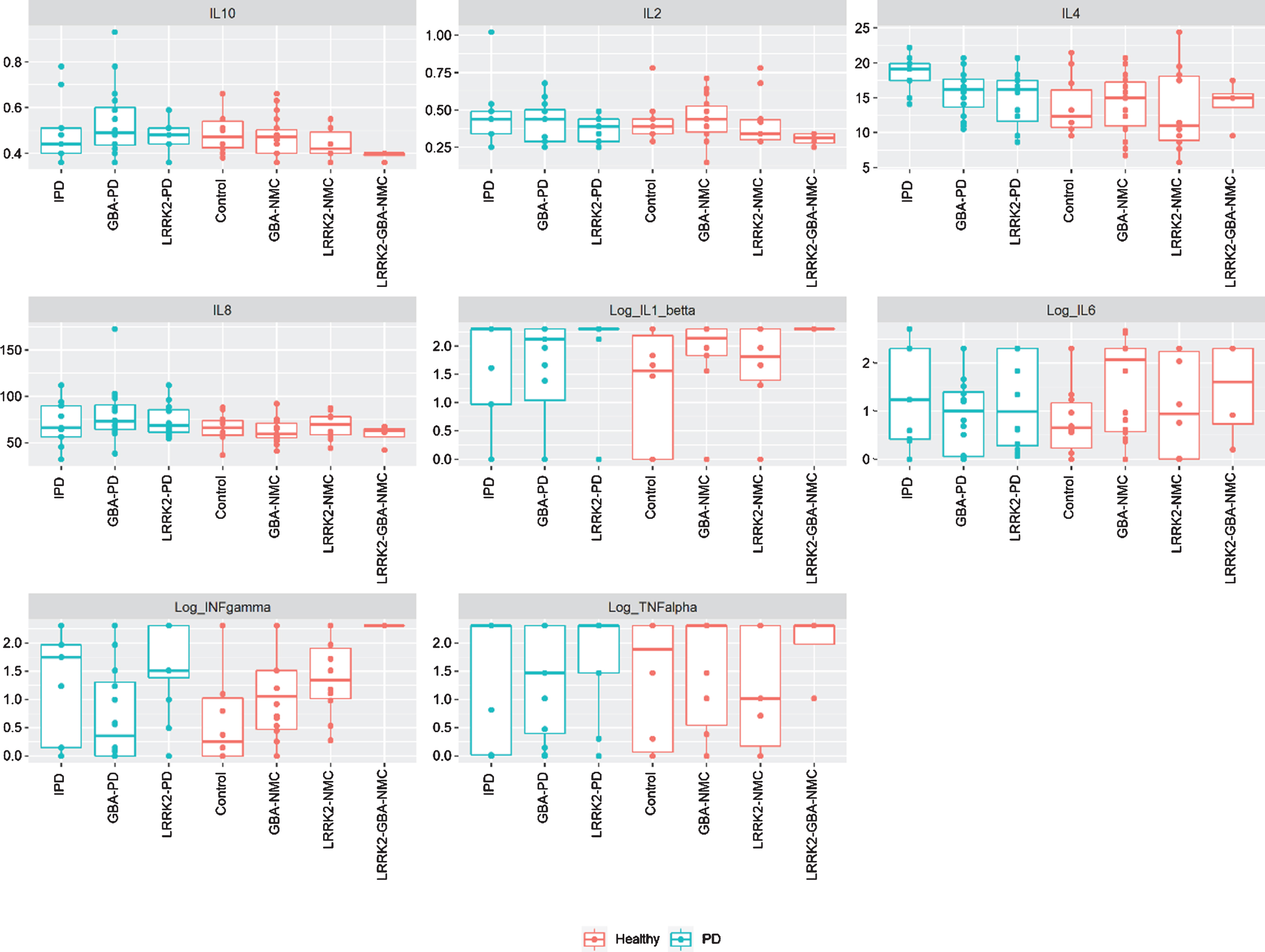
Levels of CSF cytokines among study participants iPD, idiopathic Parkinson’s disease; NMC, non-manifesting carriers; IL, interleukin; INF, interferon; TNF, tumor necrosis factor.
CSF cytokine levels were not correlated with any behavioral or laboratory measures or with risk for prodromal PD among any of the sub-groups.
Weak, but statistically significant correlations were detected among the total cohort of participants between peripheral cytokine levels and the following measures: CRP and IL-6 (r = 0.417,
The probability score for prodromal PD did not correlate with any of the cytokines among all non-manifesting participants or sub-groups assessed separately. GCase activity was not correlated with cytokine levels in any subgroup of participants either.
Performing the same analysis, excluding participants that reported concurrent inflammatory conditions or use of anti-inflammatory medications did not yield any significant findings.
The random forest analysis resulted with high error rates, meaning the explanatory variables alone could not be used to distinguish between the genetic sub-groups.
DISCUSSION
In this study assessing a large group of
In the central nervous system, resident immune system is comprised mostly of innate immune cells, with microglia and astrocytes as the main components. Under certain conditions, peripheral innate and adaptive immune cell can enter the CNS [42]. This reaction can be beneficial or deleterious depending on its severity and duration. Although cytokines participate in the inflammatory process, most have multiple biological targets and activities. The specific role of any cytokine in a clinical setup is dependent on the spectrum of polypeptides that are secreted alongside it.
With accumulating evidence that GI symptoms are present from the earliest stages of PD, it has been proposed that PD pathology may originate in the gut and later spread to the CNS. Immune activity may advance this progression, as intestinal inflammation can promote systemic and also CNS inflammation [43] contributing to PD-related neurodegeneration [44]. Immune activation promotes increased α-synuclein expression and aggregation [45] and α-synuclein in turn stimulates proinflammatory immune responses [46], resulting in a positive feedback loop.
However, data regarding cytokine expression in PD is conflicting: The ICICLE study assessed early PD patients and detected elevation of TNF-α, IL-1β, IL-2 and IL-10 compared with controls [13]. While elevated levels of cytokines including IL-1β, IL-2, TNF-α, IL-6 IL-10 and CRP have been reported among patients with PD compared with controls in a comprehensive meta-analysis [10]. However, the pooling of different studies, included patients in different disease stages, disease duration, use of medication and concomitant medical condition, is a limitation of this meta-analysis.
As CSF is in close contact with the CNS, paired plasma and CSF assessments may provide insight into the relationship between peripheral and central inflammation in PD. However, our study, in line with previous reports, failed to detect any correlation between cytokines analyzed in the CSF and serum of PD patients and NMC [47]. A recent post-mortem study examined mRNA levels of cytokines and mediators of the immune response from PD patients across different disease stages and found increased expression of IL-6 in the substantia nigra of patients in early disease stage while at later stages of the disease the same marker was downregulated [48]. In this work however, we could not detect evidence of central or peripheral inflammation, at least as reflected in ambient cytokine concentrations, among patients with PD or NMC of disease related mutations when correcting for anti-inflammatory medications or additional inflammatory conditions or when excluding participants with these conditions. We did detect correlations between pro-inflammatory cytokines and pro-inflammatory laboratory measures such as CRP, uric acid and microalbumin [49, 50] indicating that states that are associated with increased inflammation were associated with increased pro-inflammatory cytokines.
The pathological hallmark of GD is the accumulation of characteristic macrophages containing glycolipids (Gaucher cells) leading to chronic inflammation [51]. Glycolipid accumulation and lysosomal dysfunction in GD prime macrophages to release pro-inflammatory cytokines [52, 53] and indeed, in GD cell lines, activated microglia and astrocytes were detected prior to cell loss with an increase in pro-inflammatory cytokines [54]. Patients with type 1 GD showed elevated serum IL-6 [53]. Homozygote PD
Data regarding inflammation among
The penetrance of the mutations in the
While this is the largest study to date to assess inflammatory measures in
The role of inflammation in neurodegeneration and PD specifically seems robust with epidemiological evidence of reduced risk for PD among Inflammatory bowel disease patients treated with anti-TNF medication [59], indicating a role for monocyte activation factors and disease progression [60]. There still exists an unmet need for establishing a reliable approach for the study of neuroinflammation in PD patients and prodromal subjects. A combination of blood and CSF cytokines, cellular immunity and brain imaging might help to elucidate the relationship between inflammation, neurodegeneration and α-synuclein, but is extremely difficult to execute.
In this comprehensive analysis of central and peripheral inflammatory measures among
Footnotes
ACKNOWLEDGMENTS
This work was funded by the MJFF grant #16503, the Silverstein Foundation for Parkinson’s with GBA and by Biogen Inc.
CONFLICT OF INTEREST
AT –Receiving honoraria from Abbvie Israel.
NO –Nothing to disclose
NG –Serves on the Editorial Board of the Journal of Parkinson’s Disease. Serves as consultant to Biogen, Genzyme-Sanofi, Sionara, NeuroDerm, Intec Pharma, Pharma2B, Denali Neuron23 and Abbvie. Receives royalties from Lysosomal Therapeutics (LTI) and payment for lectures from Abbvie, Sanofi-Genzyme, Bial and Movement Disorder Society. Received research support from the Michael J Fox Foundation, the National Parkinson Foundation, the European Union 7th Framework Program, the Israel Science Foundation, Teva NNE program, Biogen, LTI, and Ionis.
TG –Advisory board membership with honoraria from Abbvie, Neuroderm, Medison, Allergan, Cytora and Synnerva, research support from Phonetica Ltd., Israeli Innovation Authority, Sagol School of Neuroscience and Parkinson’s Foundation. Receiving travel support from Abbvie, Allergan, Medisson and Medtronic.
ABS –Nothing to disclose
MGW –Nothing to disclose
OG –Nothing to disclose
MK –Payment for lectures from Abbvie and Teva.
JCS –Employee and shareholder in Biogen, Inc.
JMC –A former employee and shareholder in Biogen, Inc.
AOU –Research support from the Michael J Fox Foundation, Chaya Charitable Fund and Biogen and payment for lectures from Sanofi Genzyme and Pfizer.
KR –Lecture fees and travel grants from Teva, BMS, Biogen, Sanofi Genzyme, Roche, Medison, Novartis and Serono.
SST –Nothing to disclose
AM- Serving as advisor to Neuroderm.
