Abstract
Background
The α-synuclein seed amplification assay (αS-SAA) represents a promising strategy for identifying individuals with α-synuclein pathology, empowering development of tailored Parkinson's disease (PD) therapeutics and clinical trial design.
Objective
To assess the αS-SAA in cerebrospinal fluid (CSF) from PD patients, non-manifesting carriers (NMCs) and non-manifesting non-carriers (NMNCs) of pathogenic GBA1 and LRRK2 variants.
Methods
This study collected phenotype data from participants in the single-center, longitudinal, natural history BEAT-PD study (TLV-0204-16), which included PD patients and high-risk individuals for whom CSF samples were collected at baseline and 2 years post baseline. Clinical assessments in high-risk individuals enabled calculation of the International Parkinson and Movement Disorder Society probability scores for prodromal PD.
Results
CSF from 98 participants was evaluated, with no differences in age or sex distribution observed between PD and NMC subgroups. All iPD (14/14) and GBA1-PD (14/14) participants were αS-SAA positive at baseline versus only 5/13 LRRK2-PD participants (p < 0.001); 44/45 participants with longitudinal follow-up-maintained baseline αS-SAA status at year 2.
LRRK2-PD carriers, all who carried the G2019S variant, with and without positive αS-SAA status were similar in all phenotype characteristics, except for younger age at diagnosis among αS-SAA positive individuals (p = 0.04). Prodromal PD probability scores were higher in αS-SAA positive versus negative GBA1-NMCs (p < 0.001) and NMNCs (p < 0.001).
Conclusions
In LRRK2-PD, αS-SAA was associated with younger age of onset but not with motor or non-motor symptoms. In at-risk participants, αS-SAA–positive status was associated with probability scores for prodromal PD. Longitudinal follow up is required to test if αS-SAA-positivity predicts future conversion to clinical PD.
Plain language title
Detecting the pathology at the basis of Parkinson's disease among different genetic groups
Plain language summary
Researchers are working on tests that can help detect early signs of Parkinson's disease by identifying a protein called alpha-synuclein in the fluid around the brain and spinal cord (called cerebrospinal fluid, or CSF). This test is called the alpha-synuclein seed amplification assay (αS-SAA), and it could help identify people who are at risk for Parkinson's disease before they show symptoms. In this study, researchers looked at this test in different groups of people: those who already have Parkinson's disease, people who carry genetic changes (in the GBA1 or LRRK2 genes) that increase their risk for Parkinson's disease but don’t have symptoms yet, and healthy people without these genetic changes.
Key findings
The test was positive in all people with Parkinson's disease who had either no known genetic cause or a GBA1 gene change.
Among those with a LRRK2 gene change and Parkinson's disease, only about one-third had a positive test.
For those with LRRK2-related Parkinson's disease, having a positive test was linked to being diagnosed at a younger age, but it wasn’t linked to having different symptoms.
The test results were stable after two years of follow-up.
Among people at risk for future Parkinson's disease, those who had a positive test were more likely to have additional clinical early signs suggesting they might develop the disease in the future.
The researchers will continue to follow participants over time to see if those who tested positive but didn’t yet have Parkinson's disease eventually develop the disease.
Introduction
Background
The pathological diagnosis of Parkinson's disease (PD) is defined by substantia nigra atrophy and α-synuclein deposition in Lewy bodies (LBs). 1 Seed amplification assays were developed for prion amplification but were adapted to other proteins, including α-synuclein. 2 Published data demonstrate that the α-synuclein seed amplification assay (αS-SAA) has high accuracy in distinguishing PD cases from controls.3,4 Recent postmortem studies have suggested αS-SAA positivity to be correlated with α-synuclein pathology.5–7
Pathogenic variants in LRRK2 and GBA1 are common genetic risk factors for PD 8 and have distinct genotype-phenotype associations. 9 Pathological studies of individuals with PD almost universally associate GBA1 pathogenic variants with α-synuclein pathology. 10 In LRRK2-PD, pathological findings are more heterogeneous and may include α-synuclein, tau, or TDP-43 pathology or atrophy without clear protein depositions. 11 A recent study detected high rates of αS-SAA positivity among GBA1-PD participants (93%) and lower rates among LRRK2-PD participants (78%). 12 Garrido and colleagues found α-synuclein seeds in cerebrospinal fluid (CSF) using an αS-SAA in 40% of LRRK2-PD participants (6 of 15) and 18.8% of LRRK2 carriers without PD (3 of 16). 13 Recently published results from the αS-SAA analysis on the Parkinson's Progression Markers Initiative (PPMI) cohort looking comprehensively at results in idiopathic and mutation carrier individuals at-risk for or with PD observed αS-SAA positivity in 93.3% of sporadic PD participants, 95.9% of GBA1-PD participants, and 67.5% of LRRK2-PD participants. 7 Limited data is available looking at αS-SAA longitudinally in these populations. 12
In preparation for future prevention trials, the International Parkinson and Movement Disorder Society (MDS) task force developed a probability score for defining prodromal PD, to be used as a research tool.14,15 This tool, validated in different at-risk cohorts,16,17 provides an evidence-based, methodological framework to statistically estimate the likelihood that an individual has prodromal PD using a Bayesian classifier approach. A higher likelihood ratio score indicates a higher probability of prodromal PD based on motor and non-motor prodromal symptoms. The understanding of the relationship between the αS-SAA and the prodromal PD score is currently limited.
Objective
Here, we used the αS-SAA to assess the presence of α-synuclein seeds in the CSF of carriers of the LRRK2 p.G2019S and GBA1 pathogenic variants with and without PD as well as non-manifesting non-carriers (NMNCs). Our primary hypotheses were that (1) αS-SAA positivity will be less frequent in LRRK2-PD than in GBA1-PD or idiopathic PD (iPD); (2) among participants with LRRK2-PD, positive αS-SAA results will be associated with more severe disease, particularly the presence of non-motor symptoms; and (3) participants with positive αS-SAA results at baseline will remain positive at year 2. In addition, we hypothesized that at-risk individuals who had positive αS-SAA status compared with those who had negative status would have higher MDS prodromal probability scores.
Methods
Study design and settings
This cohort study assessed participants who were recruited from the BEAT-PD study (TLV-0204-16), a Biogen–Tel Aviv Sourasky Medical Center collaborative natural history study that set out to characterize genetic subgroups of PD participants (GBA1, LRRK2, idiopathic), non-manifesting carriers (NMCs) of these mutations, and NMNCs.
Participants
PD patients were recruited if they were of Ashkenazi Jewish decent, diagnosed with PD by a movement disorders specialist based on the MDS clinical diagnostic criteria for PD within 7 years of enrollment, 18 and had Hoehn and Yahr stage 1-3 PD. Non-manifesting participants were recruited if they were first-degree relatives of a LRRK2 or GBA1 carrier with PD and were older than 40 years of age and were excluded if they were using dopamine-depleting medications. Additional exclusion criteria for all participants included significant non-PD neurological or psychiatric disorders; malignancy; positive HIV, HBV, or HCV tests; and use of medications that prevented lumbar puncture (LP; anticoagulants). The study was approved by the Tel Aviv Sourasky Medical Center ethical committee and the National Institutional Review Board committee, according to the guidelines of the Declaration of Helsinki. All participants provided written informed consent prior to participation. This manuscript includes only participants who underwent a lumbar puncture for CSF testing. 19
Variables and measurements
Participants were genotyped for the G2019S-LRRK2 mutation and nine pathogenic variants in GBA1 as previously described.20,21 Participants with none of these pathogenic variants were considered to have iPD or be NMNCs.
Disease severity was assessed using the MDS-Unified Parkinson's Disease Rating Scale (MDS-UPDRS) during “on” medication. 22 The Montreal Cognitive Assessment was used to assess global cognitive functions 23 ; mood was assessed using the Beck Depression Inventory. 24 Responses on the Non-Motor Symptoms Questionnaire, 25 Scales for Outcomes in Parkinson's Disease–Autonomic Dysfunction, 26 and REM Sleep Behavior Disorder Questionnaire were collected. 27 Olfaction was tested using the University of Pennsylvania Smell Identification Test (UPSIT). 28 Blood pressure in the supine and standing positions, uric acid, and hemoglobin A1C were assessed. Results enabled calculation of the probability for prodromal PD (likelihood ratio score) in non-manifesting participants based on the updated MDS task force guidelines, 15 excluding dopamine transporter imaging and substantia nigra hyperechogenicity. Levodopa equivalent daily dose was calculated for all PD patients. 29
CSF was collected by a certified neurologist through a lumbar puncture when the participants were either in the lateral recumbent position or sitting upright. CSF was extracted with a 21-gauge atraumatic needle, collected in 15-mL conical tubes, and centrifuged at 2000 Xg at 25°C for 10 min. The supernatant was collected into a conical tube, aliquoted into 0.5 mL, and frozen at −80°C. CSF aliquots were shipped with dry ice to Biogen and stored.
αS-SAA analysis
αS-SAA analysis was conducted at Amprion. C-terminal His-tagged recombinant α-synuclein was purified from Escherichia coli DL21(DE3) using immobilized metal affinity chromatography, as previously described. 30 One batch of substrate was used for this study. Each CSF sample was analyzed by the αS-SAA in triplicate (40 µL of CSF per well). Detailed methodology for the αS-SAA and the unbiased probabilistic algorithm for determination of the final result (positive, negative, or inconclusive) have been previously published.30,31 If all three replicates were positive, the CSF sample was considered to contain α-synuclein seeds (positive). If zero or one of the replicates was positive, CSF was considered negative, meaning that α-synuclein seeds were not detected. If two replicates were positive and the average maximum fluorescence (Fmax) of the three replicates was <5000 RFU or if the coefficient of variation for the Fmax was higher than 110, the sample was considered negative. Samples that did not fit positive or negative criteria were considered inconclusive and a retest was suggested. Cohort analyses were blinded to genotype and PD status. Prior to cohort sample testing, the αS-SAA performance characteristics were evaluated and acceptable qualitative (positive or negative) and semi-quantitative (time to 50% maximum fluorescence) metric precision was achieved (Supplemental Figure 1).
Statistical analysis
Prior to analysis, demographic and clinical variables were examined for normality using the Shapiro-Wilk W test. Outliers were excluded when appropriate if values were two standard deviations (SDs) from the mean. Descriptive statistics (mean and SD for continuous variables) were computed for all measures. Generalized linear models were performed to evaluate differences between groups in collected clinical measures based on disease and genetic status. The analysis was adjusted for age and sex in both cohorts and for disease duration among patients. Differences in sex within each cohort were evaluated using chi square tests. Due to a lack of normality, the Fisher exact test was used to compare the LRRK2-PD, GBA1-NMC, and NMNC groups with and without α-synuclein seeds in CSF based on the αS-SAA. All tests adopt a two-sided type I error level of 0.05. Significance was determined at p < 0.05 for descriptive measures, as this was an exploratory pilot study; results were not corrected for multiple comparisons. Statistical analysis was performed using SPSS Statistics version 22 (IBM).
Results
Participants
Demographics, clinical characteristics, and CSF from 42 PD participants (14 iPD, 13 LRRK2-PD, 14 GBA1-PD (11 N370S, 2 R496H, and 1 370Rec), and one LRRK2-GBA1-PD (N370S)) and 56 non-manifesting participants (23 NMNC, 11 LRRK2-NMC, 19 GBA1-NMC (11 N370S, 4 84GG, 1 V394L, 1 370Rec, 1 IVS2, and 1 R496H), and three LRRK2-GBA1-NMC (2 E326 K and 1 N370S)) were collected at baseline. All participants were invited to have a second LP 2 years after baseline; 15 PD participants (five iPD, 5 LRRK2-PD, and 5 GBA1-PD) and 30 non-manifesting participants (15 NMNC, 7 LRRK2-NMC, and 8 GBA1-NMC) (46%) had a second LP. Baseline data from one GBA1-NMC were not included, as their αS-SAA results came back inconclusive with insufficient volume for retesting.
Genetic subgroup clinical characterization
Total MDS-UPDRS scores were lower in LRRK2-PD participants than in GBA1-PD and iPD participants (p = 0.035). No significant between-group differences were detected in clinical measures between the non-manifesting participants (Table 1).
Characteristics of the study cohort.
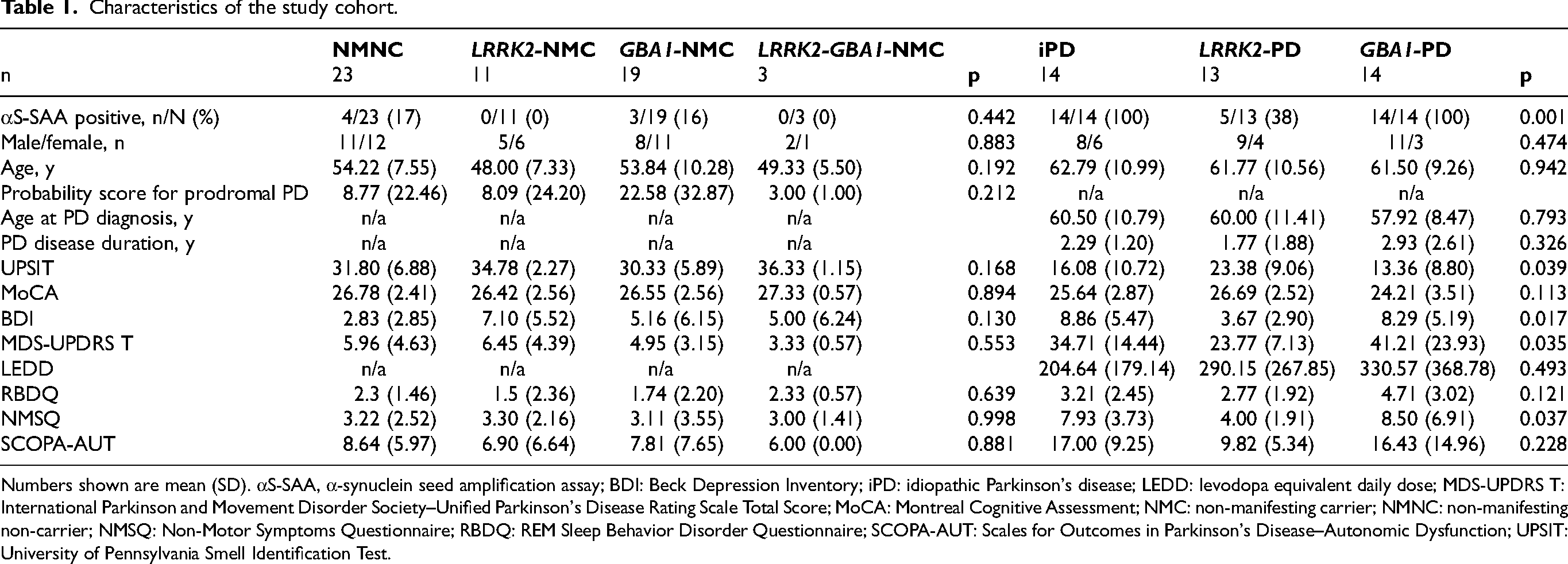
Numbers shown are mean (SD). αS-SAA, α-synuclein seed amplification assay; BDI: Beck Depression Inventory; iPD: idiopathic Parkinson's disease; LEDD: levodopa equivalent daily dose; MDS-UPDRS T: International Parkinson and Movement Disorder Society–Unified Parkinson's Disease Rating Scale Total Score; MoCA: Montreal Cognitive Assessment; NMC: non-manifesting carrier; NMNC: non-manifesting non-carrier; NMSQ: Non-Motor Symptoms Questionnaire; RBDQ: REM Sleep Behavior Disorder Questionnaire; SCOPA-AUT: Scales for Outcomes in Parkinson's Disease–Autonomic Dysfunction; UPSIT: University of Pennsylvania Smell Identification Test.
Genetic subgroup analysis on the αs-SAA
At baseline, all iPD and GBA1-PD participants were αS-SAA positive compared with only 5/13 LRRK2-PD participants (p < 0.001). A single LRRK2-GBA1-PD participant was evaluated and was also αS-SAA positive. All the LRRK2-NMC and all three of the LRRK2-GBA1-NMC participants were αS-SAA negative, while 3/19 GBA1-NMCs (1 N370S, 1 IVS2, and 1 R496H) and 4/23 NMNCs were αS-SAA positive (Figure 1). Sensitivity and specificity for distinguishing clinically diagnosed PD from non-PD for GBA1 variant carriers and non-genetic participants were 100% and 83%, respectively. Sensitivity was reduced to 80%, with a specificity of 87%, when including LRRK2 mutation carriers as a result of the high αS-SAA–negative rate among LRRK2-PD participants.
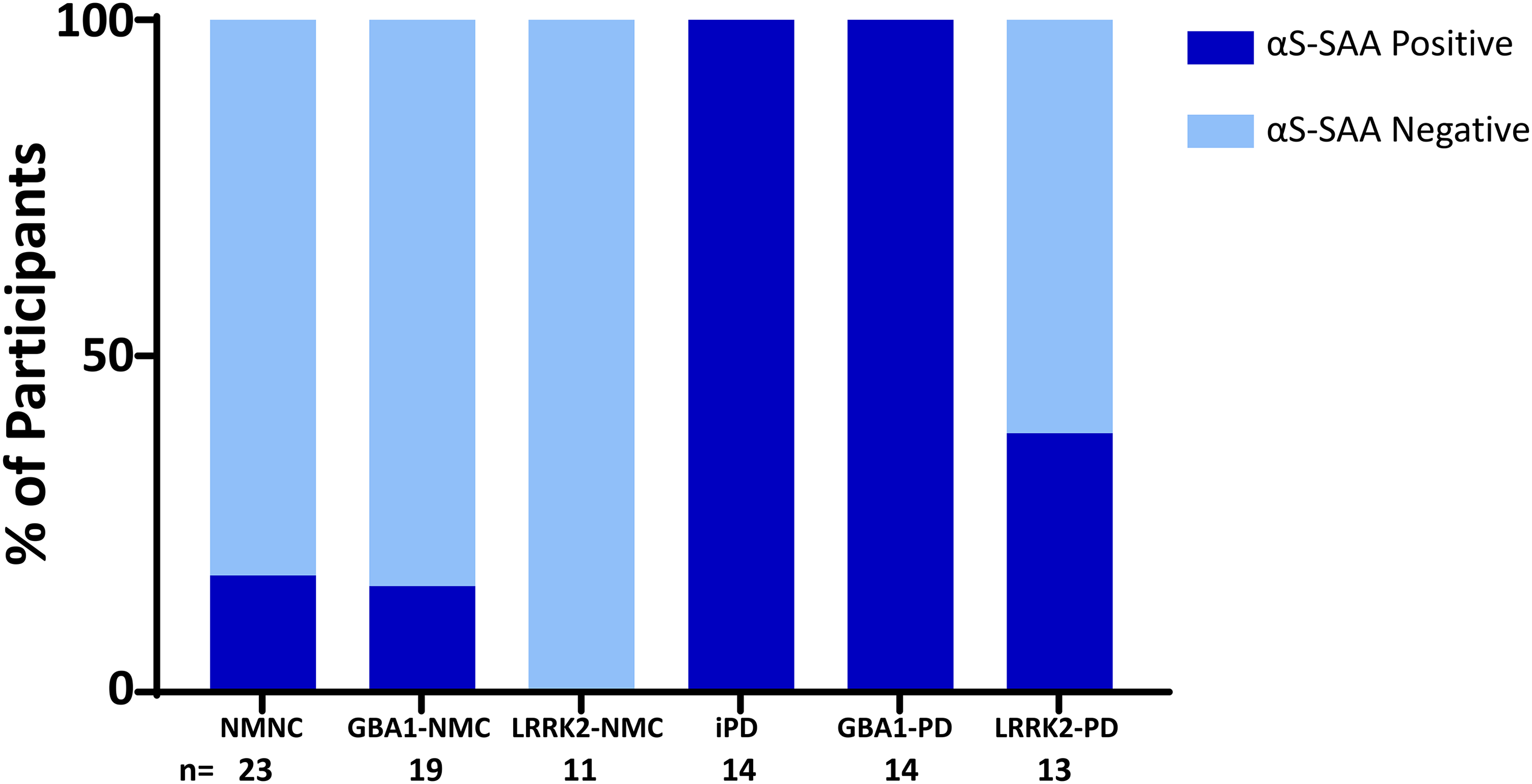
Results from αS-SAA evaluation of baseline CSF from pG2019S-LRRK2 mutation carriers, pathogenic GBA1 mutation carriers, and non-carriers with and without Parkinson's disease. Results from one NMNC and one GBA1-NMC were inconclusive and are not included in this analysis. αS-SAA: α-synuclein seed amplification assay; CSF: cerebrospinal fluid; iPD: idiopathic Parkinson's disease; NMC: non-manifesting carrier; NMNC: non-manifesting non-carrier.
Kinetic metrics of the αS-SAA sigmoidal fluorescence aggregation curve, including time to 50% maximum fluorescence, Fmax, and slope, have been suggested to potentially inform the quantity or types of species of α-synuclein seeds in the CSF. 31 We observed no significant differences in any of these metrics between αS-SAA–positive PD subgroups (Supplemental Figure 2).
LRRK2-PD participants who were αS-SAA positive had a younger age of onset of motor symptoms (52.20 ± 13.86 vs. 66.88 ± 6.55; p = 0.04) and younger age at assessment (54.60 ± 12.68 vs. 66.25 ± 6.29; p = 0.04) than LRRK2-PD participants who were αS-SAA negative. The LRRK2-PD group consisted of 4 females and 9 males; all females were SAA-negative while 5/9 males were SAA-positive. No differences were observed in other demographic or phenotypic features between αS-SAA positive–and αS-SAA–negative LRRK2-PD participants (Table 2).
Comparison of clinical features by αs-SAA status within αs-SAA heterogeneous subgroups.
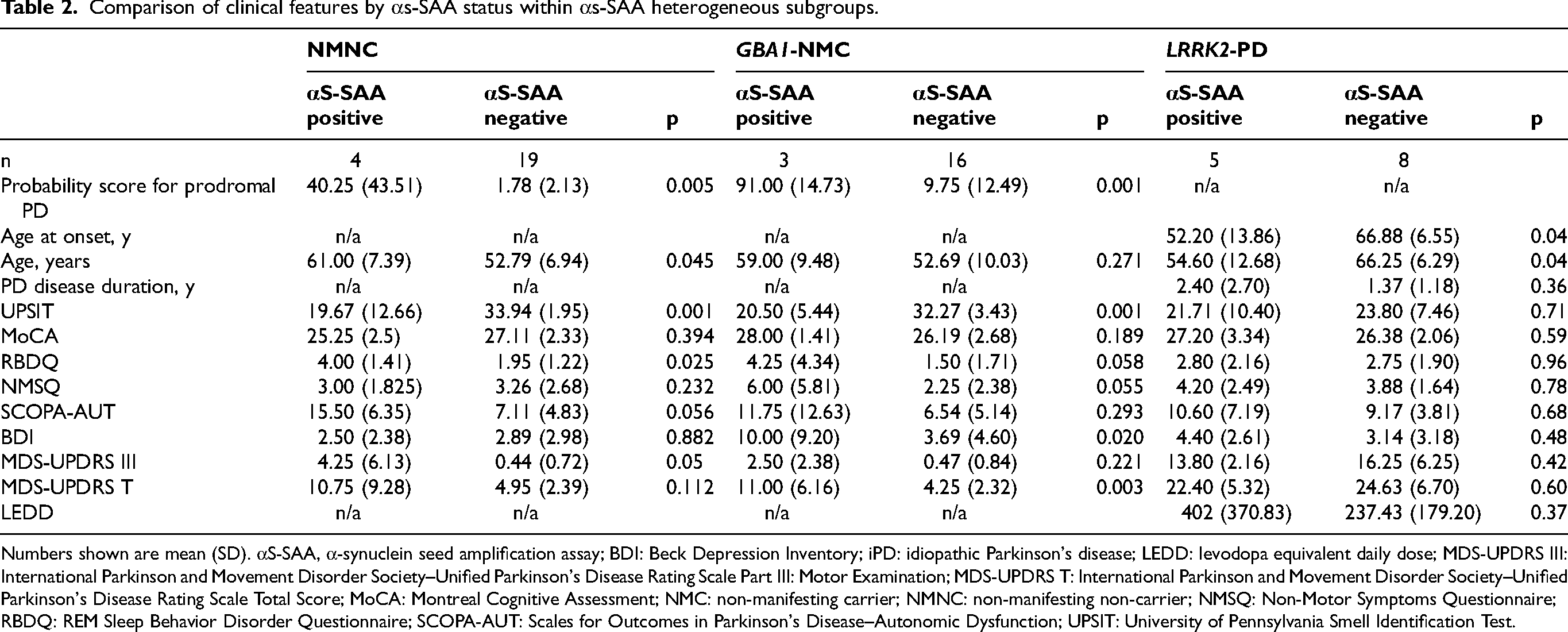
Numbers shown are mean (SD). αS-SAA, α-synuclein seed amplification assay; BDI: Beck Depression Inventory; iPD: idiopathic Parkinson's disease; LEDD: levodopa equivalent daily dose; MDS-UPDRS III: International Parkinson and Movement Disorder Society–Unified Parkinson's Disease Rating Scale Part III: Motor Examination; MDS-UPDRS T: International Parkinson and Movement Disorder Society–Unified Parkinson's Disease Rating Scale Total Score; MoCA: Montreal Cognitive Assessment; NMC: non-manifesting carrier; NMNC: non-manifesting non-carrier; NMSQ: Non-Motor Symptoms Questionnaire; RBDQ: REM Sleep Behavior Disorder Questionnaire; SCOPA-AUT: Scales for Outcomes in Parkinson's Disease–Autonomic Dysfunction; UPSIT: University of Pennsylvania Smell Identification Test.
Probability scores for prodromal PD were higher in αS-SAA–positive participants than in αS-SAA–negative participants in both GBA1-NMC (91.00 ± 14.73 vs. 9.75 ± 12.49; p < 0.001) and NMNC (40.25 ± 43.51 vs. 1.78 ± 2.13; p < 0.001) subgroups (Table 2; Figure 2).
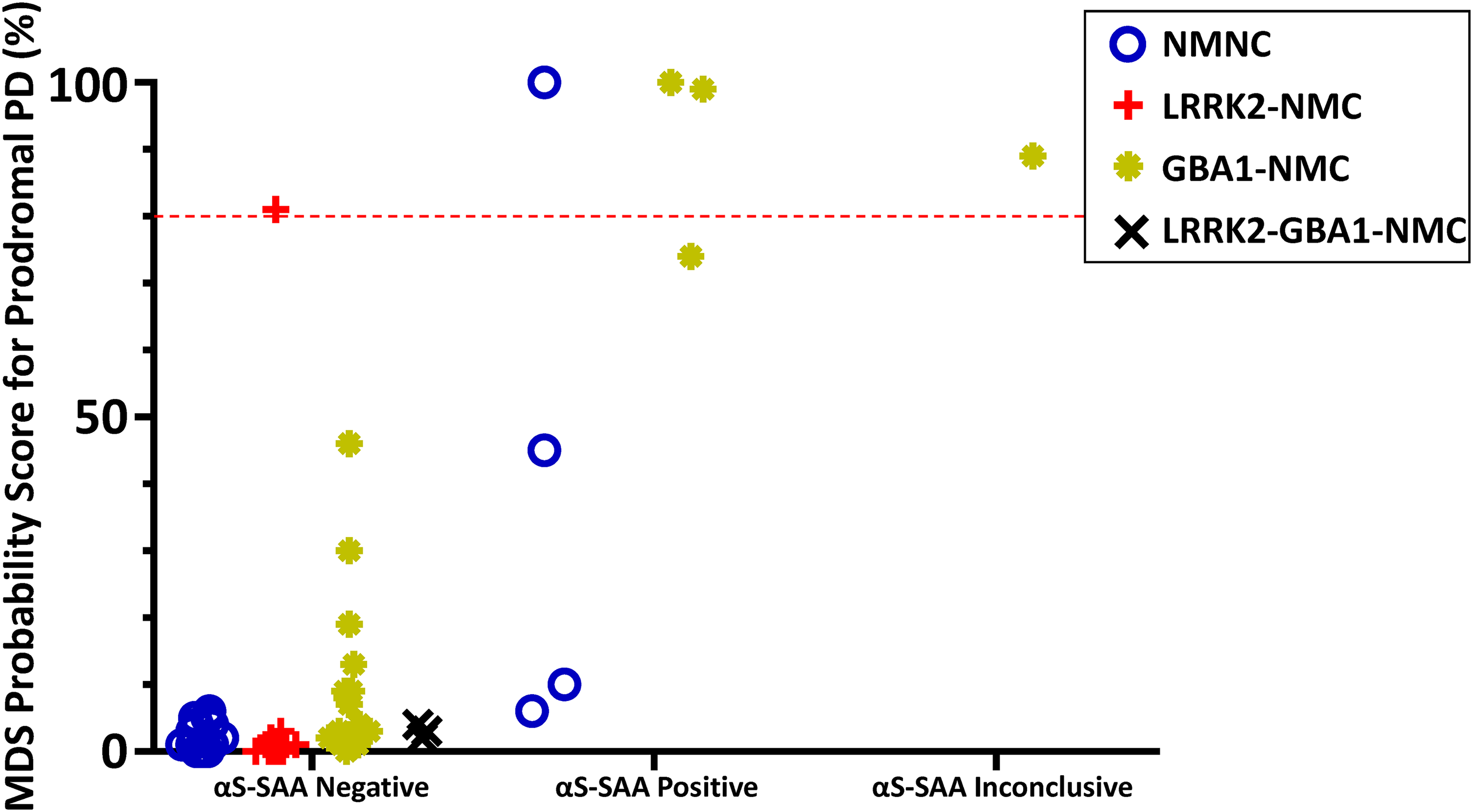
Results from the MDS task force probability scoring for prodromal PD as stratified by αS-SAA status and genetic status. The dotted red line represents the cutoff for prodromal PD as defined in Berg et al. (2015). 14 αS-SAA: α-synuclein seed amplification assay; MDS: International Parkinson and Movement Disorder Society; NMC: non-manifesting carrier; NMNC: non-manifesting non-carrier; PD: Parkinson's disease.
Among participants with a probability score above 50%, 4/5 were αS-SAA positive (three GBA1-NMC and one NMNC) and one was αS-SAA negative (LRRK2-NMC). UPSIT scores were lower for αS-SAA–positive versus αS-SAA–negative participants in both GBA1-NMC (20.50 ± 5.44 vs. 32.27 ± 3.43; p < 0.001) and NMNC (19.67 ± 12.66 vs. 33.94 ± 1.95; p < 0.001) subgroups (Table 2).
Two years after baseline collection, CSF was collected from a subgroup of participants (n = 45); all but one maintained their baseline αS-SAA status. One LRRK2-PD participant who was αS-SAA negative at baseline converted to αS-SAA positive at the year 2 follow-up (Figure 3).
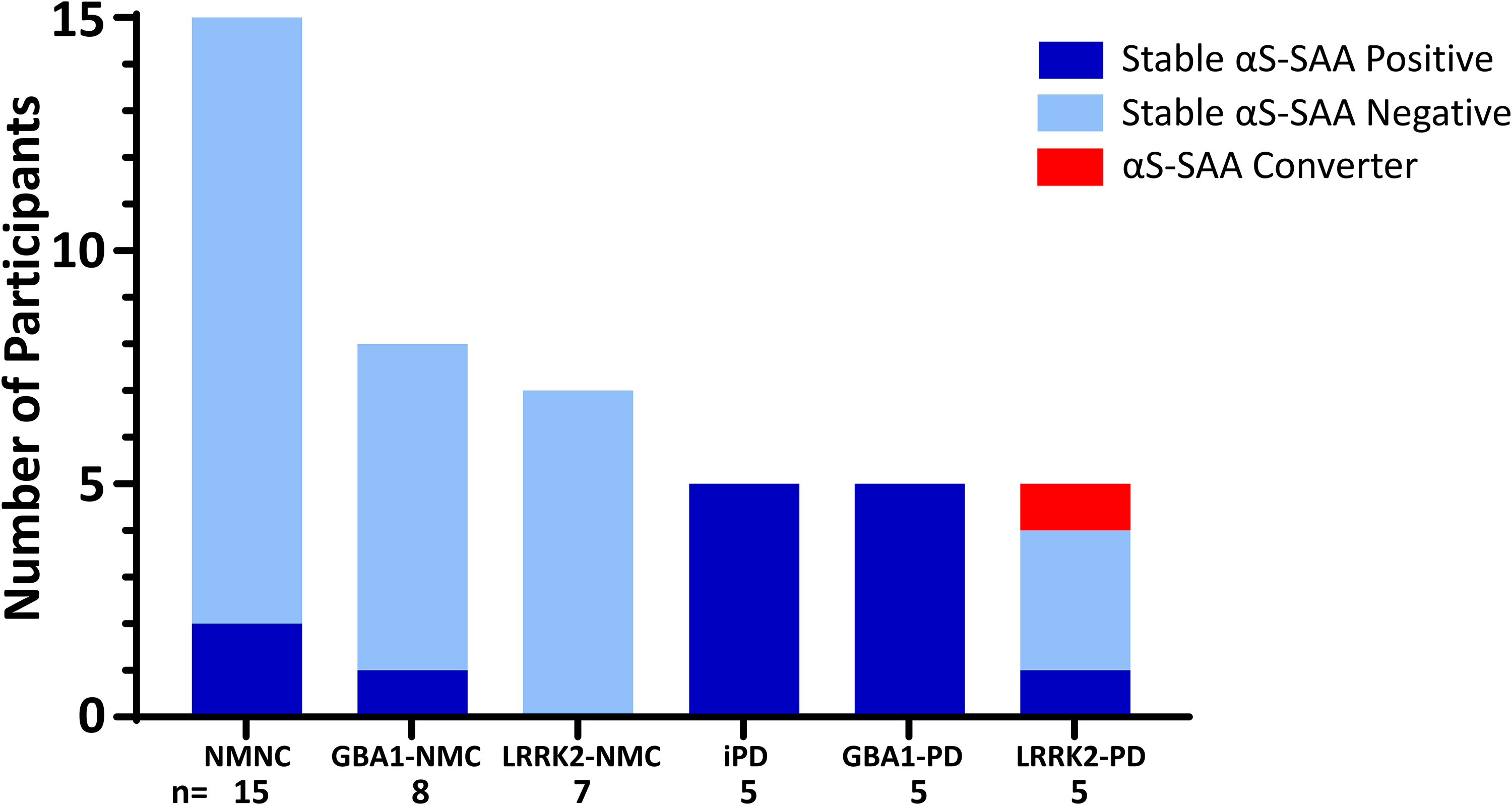
Results from 2-year longitudinal αS-SAA evaluation of CSF from pG2019S-LRRK2 mutation carriers, pathogenic GBA1 variant carriers, and non-carriers with and without Parkinson's disease at year 2 compared with baseline. “Stable αS-SAA positive” indicates positive αS-SAA at baseline and year 2; “stable αS-SAA negative” indicates negative αS-SAA at baseline and year 2; “αS-SAA converter” indicates negative αS-SAA at baseline and converted to positive αS-SAA at year 2. 44 of 45 participants had the same αS-SAA status at year 2 as their baseline measurement, and one LRRK2-PD participant converted from αS-SAA negative at baseline to αS-SAA positive at year 2. αS-SAA: α-synuclein seed amplification assay; iPD: idiopathic Parkinson's disease; NMC: non-manifesting carrier; NMNC: non-manifesting non-carrier.
Discussion
Key points
α-Synuclein SAAs have demonstrated promising sensitivity and specificity in iPD cohorts, 4 but recent studies have suggested differential findings in genetic cohorts.7,13 In this study, CSF was collected from a single-center genetic cohort and evaluated with the αS-SAA. The key findings in our study are that (1) all GBA1-PD and iPD participants had a positive αS-SAA status at baseline, while only 5/13 (38%) LRRK2-PD participants were αS-SAA positive, consistent with previous reports7,13; (2) all LRRK2-PD females were SAA-negative; (3) all αS-SAA–positive participants who provided year 2 longitudinal CSF collections remained αS-SAA positive, while one (1/45) αS-SAA–negative LRRK2-PD participant converted to αS-SAA positive at year 2; (4) in the LRRK2-PD group, age of onset of motor symptoms was younger in those who were αS-SAA positive; and (5) in the GBA1-NMC and NMNC subgroups, MDS probability scores for prodromal PD were higher and UPSIT scores were lower among those who were αS-SAA positive. Hereafter, we discuss these key findings.
Amplification assay in genetic PD
The differential presence of α-synuclein pathology in LRRK2-PD versus GBA1-PD observed in previous αS-SAA studies7,13 is supported by these results and is consistent with autopsy results perhaps partially explaining the differences in phenotype, as GBA1-PD is associated with more severe disease compared with LRRK2-PD. 9 A limitation of previous αS-SAA analyses in genetic PD cohorts is a lack of longitudinal data in these populations. Within this study longitudinal follow-up at year 2 was evaluated and all participants who were αS-SAA positive at baseline remained positive at follow-up. This suggests that upon manifestation of a disease state in which these α-synuclein seeds emerge, they remain a stable feature of PD for both genetic and non-genetic individuals. During the duration of the study, one LRRK2-PD participant converted from αS-SAA negative to positive. All 3 technical replicates for this subject were negative at baseline adding confidence to the likelihood of a true baseline negative. This result might suggest that detectable α-synuclein seeds may emerge after the onset of motor symptoms for some LRRK2-PD individuals. This may indicate that emergence of α-synuclein seeds in LRRK2-PD is a potentially downstream result of the core pathogenic driver of PD in these individuals and not a predominant driver itself. Longitudinal studies with repeated SAA measurements would be useful to address these hypotheses of αS-SAA emergence patterns in LRRK2-PD populations, and longer studies may also allow observation of αS-SAA conversion among NMNCs and GBA1-NMCs.
Recently, a large cross-sectional PPMI study assessing SAA among LRRK2 participants was published. 31% of the LRRK2-PD were SAA-negative and had an older age at assessment, with similar disease duration and motor scores on MDS-UPDRS compared to the SAA-positive LRRK2-PD group. SAA-negative LRRK2-PD had a larger proportion of females compared to SAA positive participants. Longitudinally, the SAA-negative group remained stable in functional rating scale scores in contrast to the positive group who had a significant increase (worsening) in MDS-UPDRS part II. 32
Among NMNCs, we observed an αS-SAA positivity rate higher than previously reported for controls (17% vs. 10%). 5 This result is not surprising because most NMNCs in this study had an increased risk for PD (a high probability score) not only due to family history 33 but also due to prodromal markers such as REM sleep behavior disorder and hyposmia. 15 This is attested by the relatively high probability scores for prodromal PD among NMNC. Additionally, PD patients were recruited to the study if they were of Ashkenazi Jewish decent, and observations from this study may differ from non-Ashkenazi Jewish populations.
Amplification assay and PD phenotype in LRRK2-PD
A neuropathological study comparing LRRK2-PD individuals with and without LB pathology detected higher rates of non-motor symptoms among those with LB pathology. 34 Therefore, we hypothesized that αS-SAA positive status would be associated with the presence of non-motor symptoms among LRRK2-PD participants. Similar to results from the PPMI αS-SAA analysis,7,32 limited associations, however, were observed, and the only phenotypic difference we observed was in age of onset of symptoms. The 13-year difference in age of onset between αS-SAA positive and negative participants suggests a true biological difference between the groups. Similarly, in the aforementioned pathological study, 34 LRRK2-PD individuals with LB pathology had a 5-year, albeit not statistically significant, younger age of onset of symptoms than those without LB pathology (56 vs. 61 years). The causes of differential pathology observed among LRRK2-PD are unknown and present a challenge in the interpretation of natural history and clinical study data in this population. Stratification of LRRK2-PD by αS-SAA status may provide extra insight into analyses of data from clinical trials in this population and provide a new lens for how we might approach LRRK2-PD data sets.
Amplification assay and relationship to prodromal features of PD
The PPMI cohort study observed high αS-SAA positivity rates in non-PD participants with features of prodromal PD (RBD, hyposmia). 7 Within our study probability scores for prodromal PD were calculated for high-risk individuals and a strong correlation was observed between αS-SAA status and the probability score for prodromal PD.14,15 This data supports the hypothesis that αS-SAA positivity is associated with prodromal features of the disease.
Previous reports have indicated that individuals with LRRK2-PD had better overall olfaction scores and were less likely to be hyposmic than individuals with iPD. 35 We anticipated that this phenotypic heterogeneity may be driven by heterogeneity of pathology, and differences in hyposmia were observed between αS-SAA positive and negative LRRK2-PD participants of the PPMI study. 7 We did not, however, observe any differences in UPSIT scores between αS-SAA positive and negative LRRK2-PD participants in this study potentially due to limited sample size. Overall, in the full population of the study UPSIT scores closely corresponded to αS-SAA status. Identification of predictive measures of αS-SAA status that can potentially be used as pre-screening tools for inclusion/exclusion criteria frameworks in clinical studies implementing the αS-SAA for patient selection could greatly reduce the number of patients requiring LP. Further research into the potential for UPSIT to serve this purpose is warranted.
In total, these results suggest that αS-SAA positivity may reveal individuals who are phenotypically characteristic of prodromal PD. Future studies are warranted in order to understand the ability of the αS-SAA, in combination with such tools as the MDS probability score, to refine the identification of individuals with prodromal PD and potentially inform the design of prodromal PD or α-synuclein–targeted therapeutic trials in the future.
Limitations
The strengths of our study are the broad availability of GBA1 and LRRK2 mutation carriers with and without PD, detailed phenotyping available for participants, and longitudinal characterization of αS-SAA results. The major limitation of the study is the sample size, especially for the longitudinal αS-SAA analysis group. This limits the ability to derive high statistical confidence and is unpowered to generate statistical findings that would withstand adjustment for multiple comparisons. While previous larger studies on αS-SAA have been published, this work is one of the only studies evaluating the longitudinal stability of αS-SAA. In addition, the findings confirm previously published studies in an independent cohort which is crucial for validating methodology, strengthening reported findings and highlighting limitations of the measurements. Such process of replication of results solidifies potential use and increases generalizability across studies.
Results from this study provide descriptive observations and replicating these findings in additional cohorts, with sufficient study duration and size to monitor these features as well as PD phenoconversion, is important to confirm the predictive value of such amplification assays.
An additional limitation is that the duration of this study only captures a fraction of the time course of disease in these individuals. The study duration limits our ability to determine if positive αS-SAA results are associated with future development of motor PD and with LB pathology in autopsies of PD participants.
The NMNC group did not carry mutations in the GBA1 and LRRK2 genes yet demonstrated relatively high probability scores and αS-SAA positivity, indicating increased risk for future PD. This is in line with the proposed biological stratification methods which do not rely solely on genetic status but rather on SAA status.36,37 Future studies should carefully choose control participants with no prodromal symptoms of PD. Another limitation of the current assay is the lack of capability to inform quantitative changes in α-synuclein seeds. Further investigation into quantitative measures of α-synuclein seeds is needed to empower monitoring of treatment response and a more nuanced understanding of patient heterogeneity.
Finaly, we used targeted GBA1 mutation assessment that has been previously demonstrated to capture more than 96% of the GBA1 mutations in the Ashkenazy Jewish population. 38 Interestingly, full sequencing of the GBA1 gene performed on the general Israeli population in the ROPAD study reached comparable results. 39 While full GBA1 sequencing would be preferable, we believe that the current method does not reduce the validity of the findings keeping in mind that there could be other GBA1 mutation carriers that were erroneously classified as “non-carriers” due to the current genotyping method.
Conclusions
The data presented in this manuscript demonstrate that as expected, αS-SAA positivity rates are higher in iPD and GBA1-PD compared with LRRK2-PD, which highlights the heterogeneity of PD subgroups. The results provide additional evidence that the αS-SAA is a promising strategy for identifying individuals with alpha-synuclein pathology in vivo. αS-SAA correlations with probability scores for prodromal PD and with UPSIT scores for GBA1 carriers and NMNCs suggest that the αS-SAA may also be associated with prodromal features of PD. Taken together, these data provide additional context regarding the utility of the αS-SAA for enriching populations in anti–α-synuclein clinical trials.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251392799 - Supplemental material for Analysis of α-synuclein seed amplification assay in carriers of GBA1 and LRRK2 pathogenic variants
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251392799 for Analysis of α-synuclein seed amplification assay in carriers of GBA1 and LRRK2 pathogenic variants by Kyle B Fraser, Anat Mirelman, Omar S Mabrouk, Nurit Omer, Luis Concha-Marambio, Tanya Gurevich, Anat Bar-Shira, Mali Gana-Weisz, Orly Goldstein, Avi Orr-Urtreger, Meir Kestenbaum, Jesse M Cedarbaum, Tien Dam, Julia C Shirvan, Nir Giladi, Danielle Graham, Roy N Alcalay and Avner Thaler in Journal of Parkinson's Disease
Footnotes
Acknowledgements
This paper is dedicated to Prof. Nir Giladi, a leading neurologist and researcher with tremendous contributions to Parkinson's disease research and care. His vision and collaborative spirit made this scientific partnership possible. His untimely death is a great loss to his family, to his friends and colleagues, to his patients, and to medicine and science.
ORCID iDs
Ethical considerations
The study was approved by the Tel Aviv Sourasky Medical Center ethical committee and the National Institutional Review Board committee, according to the guidelines of the Declaration of Helsinki.
Consent to participate
All participants provided written informed consent prior to participation.
Funding
This study was financed by Biogen Inc.
Declaration of conflicting interests
KBF, OSM, DG: employees of Biogen, and may hold stock.
TD, JCS: former Biogen employees, and may hold stock
AM: received research grant funding from MJFF, the US department of defense, and JNPD, Is an Associate editor at the Journal of Parkinson's disease.
LCM: is an inventor on several patents related to the SAA (PMCA) technology and is affiliated to Amprion Inc., a biotech company focusing on the commercial utilization of SAA (PMCA) for diagnosis. Has also received funding from MJFF.
TG: has served as advisor to Teva, Abbvie, Medison, Truemed and Neuroderm, received honoraria from Abbvie, Medison, Neuroderm and Teva, and received grants from International Parkinson's disease and Movement Disorders society.
ABS, MGW, OG: No disclosures to report.
MK: Has received lecture fees from Teva and AbbVie, and has received travel grants from Medison and AbbVie.
AOU: Has received funding from MJFF.
JMC: Has received consulting fees from VanquaBio, and has received funding from MJFF.
NG: Has received educational grant from Movement Disorders society, has received research grants from Biogen, MJFF, American Parkinson Foundation, and Aufzien Center at Tel Aviv University.
RNA: Has received consultation feed from Genzyme/Sanofi, Gain Therapeutics, and Takeda. Has received research funding from MJFF and the Parkinson's Foundation, Is an Associate editor at the Journal of Parkinson's disease.
AT: Has received grants from MJFF and has received honoraria from AbbVie.
Data availability statement
De-identified data supporting the findings reported in this work can be made available by the corresponding author upon reasonable request by researchers who meet criteria for access to the confidential data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
