Abstract
Background:
The longitudinal association between dynamic changes in the metabolic syndrome (MS) status and Parkinson’s disease (PD) has been poorly studied.
Objective:
We examined whether dynamic changes in MS status are associated with altered risk for PD.
Methods:
This study was a nationwide retrospective cohort study. We enrolled 5,522,813 individuals aged≥40 years who had undergone health examinations under the National Health Insurance Service between 2009 and 2010 (two health examinations with a 2-year interval). Participants were followed up until the end of 2017. The participants were categorized into four groups according to MS status changes over 2 years: non-MS, improved MS, incident MS, and persistent MS groups. Multivariable Cox hazard regression was performed.
Results:
During the 7-year median follow-up, there were 20,524 cases of newly developed PD. Compared with non-MS group, improved, incident, and persistent MS groups for 2 years were significantly associated with higher risks of PD (model 3; hazard ratio: 1.12, 95%confidence interval: 1.06–1.19 [improved MS]; 1.15, 1.09–1.22 [incident MS]; and 1.25, 1.20–1.30 [persistent MS]). Individuals with incident and persistent abdominal obesity, low levels of high-density lipoprotein cholesterol, hypertriglyceridemia, and hyperglycemia had a significantly increased risks of PD compared with those without either condition over 2 years.
Conclusion:
Persistent and incident MS and its components may be risk factors for incident PD. Ever exposure to MS may also be associated with PD risk. Appropriate intervention for preventing and improving MS may be crucial in decreasing the PD incidence.
INTRODUCTION
Parkinson’s disease (PD) is a complex disabling disorder related to the progressive loss of dopaminergic neurons in the substantia nigra [1]. PD is known to be the second most common neurodegenerative disease after Alzheimer’s disease [2]. It reportedly affects up to 2%of the elderly population worldwide [2], and its prevalence is expected to double by 2030 among people aged≥50 years owing to the global increase in life expectancy [3]. Higher rates of mortality as well as disability and dementia have been noted in individuals with PD than in controls [4, 5]. This increasing prevalence and poor outcome of PD constitute a health-care burden to society. Genetic and environmental factors have been reported to contribute to PD development [6]. Particularly, recent evidence suggests the possible role of metabolic syndrome (MS) in PD development [7].
MS refers to a cluster of several interrelated metabolic abnormalities and is associated with increased risks of type 2 diabetes, cardiovascular diseases, and mortality. As the MS prevalence has been recently increasing according to the obesity epidemic, it imposes a huge burden on health-care costs [8, 9]. Increasing evidence indicates that features of systemic metabolic dysfunction, including oxidative stress, altered lipid pathway, inflammation, and insulin resistance observed in MS-related diseases, seem to be partly shared by neurodegenerative diseases, including Alzheimer’s disease and PD [10, 11]. Thus, the role of MS in the development of PD has been recently receiving attention. However, there is limited epidemiologic evidence on the association between MS and PD development. To our knowledge, only two longitudinal cohort studies have reported on the association between MS and the risk of incident PD [12, 13]. However, the results were conflicting and methodological differences, concerning follow-up duration, sample size, and ethnicity of the study population, were present. Moreover, previous studies evaluated the association between MS at a single time point and the risk of incident PD. We hypothesized that the impact of longitudinal changes in MS status on incident PD might strongly support the previously reported associations between MS and PD. In this study, we sought to examine whether dynamic alterations in MS status over a 2-year period is associated with the risk of incident PD during a 7-year follow-up, by using nationwide population-based cohort data in South Korea.
MATERIALS AND METHODS
Data source and study population
This nationwide, population-based cohort study is based on the dataset from the South Korean National Health Insurance Service (NHIS). The NHIS as a single and mandatory insurer managed by the South Korean government. It covers nearly the entire South Korean population, including employee and regional insurance subscribers. It provides at least biannual health examinations for all insured Koreans. Thus, the NHIS possesses the health information data of the whole South Korean population, including demographics, disease diagnosis based on International Classification of Diseases-Tenth Revision-Clinical Modification (ICD-10-CM) codes, and medical treatment and procedures. This nationwide dataset has been released for researchers whose study protocols are approved by an official review committee since 2015.
For our study, we recruited 12,724,418 individuals aged≥40 years who had undergone health examinations between January 1, 2009 and December 31, 2010. We excluded individuals with any missing data (n = 60,882) and those who did not undergo a follow-up examination after about 2 years (n = 7,125,224). We also excluded individuals with a previous diagnosis of PD within 5 years before enrollment (n = 13,762) and those who developed PD within a 1-year lag period from enrollment (n = 1,737). Finally, we enrolled 5,522,813 individuals with two health examinations with a 2-year interval. Participants were followed up until December 31, 2017, and the median follow-up period was 7.04 (interquartile range: 6.46–7.46) years. This study adhered to the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of Korea University Anam Hospital (no. 2019AN0038). As anonymous and de-identified information was used, written informed consent was waived.
Study outcome
The study endpoint was newly developed PD. We diagnosed PD based on the ICD-10-CM code (G20) and the registration code (V124) for PD, which are assigned by neurologists or neurosurgeons. Since 2006, the South Korean NHIS is running a registration program for rare intractable diseases for a copayment reduction.
Data collection and definitions
The participants’ demographic and lifestyle information was collected from standardized self-report questionnaires. Income level was divided into quartiles, and participants were categorized as 2 income groups: lowest quartile (Q1) and the second lowest to the highest quartiles (Q2-Q4). Participants were categorized as non-smokers, former smokers, and current smokers based on their smoking history. Concerning alcohol consumption, individuals who reported drinking≥30 g of alcohol per day were defined as heavy alcohol drinkers [14]. Physical activity was assessed using the participants’ answers on intensity and weekly frequency, and regular exercisers were defined as those who engaged in≥1 sessions of strenuous exercise for at least 20 min per week. The NHIS health examination included anthropometric and blood pressure (BP) measurements and laboratory tests. The participants’ height, weight, and waist circumference (WC) were measured, and body mass index (BMI) was calculated by dividing weight (kg) by height squared (m2). BP was measured in a seated position after at least 5 min of rest. Serum levels of glucose, total cholesterol, triglycerides, high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol were measured from blood samples obtained after overnight fasting. We defined baseline comorbidities based on the combination of health examination results and ICD-10-CM codes for disease diagnosis and prescription. Depressive disorder was defined as at least 1 claim per year under ICD-10-CM code F32 or F33. Stroke was defined as a record of ICD-10-CM code I63 or I64 during hospitalization with claims for brain magnetic resonance imaging or brain computerized tomography.
Definition of MS and change in MS status
According to the modified criteria of the National Cholesterol Education Program Adult Treatment Panel III and the Asian-specific WC cutoff for abdominal obesity [15, 16], we defined MS as the presence of≥3 of the following components: (i) abdominal obesity (WC≥90 cm for men and≥85 cm for women), (ii) high triglycerides (serum triglycerides level≥150 mg/dL or treatment with lipid-lowering medication), (iii) low HDL-C (serum HDL-C level < 40 mg/dL for men or < 50 mg/dL for women, or treatment with lipid- lowering medication), (iv) high BP (systolic BP≥130 mmHg, diastolic BP≥85 mmHg, or treatment with antihypertensive medications), and (v) high fasting plasma glucose (FPG) (FPG level≥100 mg/dL or treatment with antidiabetic medication). We defined lipid-lowering medication use as at least 1 claim per year for lipid-lowering medication prescription under ICD-10-CM code E78, antihypertensive medication use as at least 1 claim per year for antihypertensive medication prescription under ICD-10-CM code I10-13 or I15, and antidiabetic medication use as at least 1 claim per year for antidiabetic medication prescription under ICD-10-CM code E11-14. The participants were categorized into 4 groups according to MS status changes over 2 years. Individuals without an MS diagnosis at either of two screenings were defined as the non-MS group, those who were diagnosed with MS at both screenings as the persistent MS group, those who were diagnosed with MS in the first screening but not in the second screening as the improved MS group, and those who were not diagnosed with MS in the first screening but were diagnosed with MS in the second screening as the incident MS group.
Statistical analysis
SAS version 9.4 (SAS Institute, Cary, NC, USA) was used for statistical analysis. The baseline characteristics according to MS status changes over 2 years were presented as mean±standard deviation for continuous variables or as number (%) for categorical variables. The values were compared using analysis of variance for continuous variables and with the chi-squared test for categorical variables. Kaplan-Meier curves were plotted for incidence probabilities of PD according to MS status changes, and log-rank p was calculated. The PD incidence rate was calculated by dividing the number of PD events by 1000 person-years. Multivariable Cox proportional hazard regression analysis was performed to evaluate the association of MS status changes with the risk of incident PD. The hazard ratios (HRs) and 95%confidence intervals (CIs) of PD were also calculated. Model 1 was adjusted for age and sex, and model 2 was further adjusted for smoking status, alcohol consumption, physical activity, income, depressive disorder, and stroke. In model 3, variables from model 2 were further adjusted for baseline BMI. We performed subgroup analyses according to age, sex, and obesity status. p for interaction was calculated using Cox regression analysis. A p-value < 0.05 was considered statistically significant.
RESULTS
Characteristics according to MS status change over 2 years
Table 1 presents the baseline characteristics of the study population according to MS status change over 2 years. Among 5,522,813 participants, the distribution of MS status changes was as follows: 3,021,648 had no MS, 516,715 had improved MS, 663,157 had incident MS, and 1,321,293 had persistent MS. PD developed in 20,524 participants during the 7-year follow-up. The mean age and the proportions of male sex and lifestyles were significantly different among MS status change groups. Cardiometabolic parameters were significantly different among the 4 groups, with the mean values of BMI, WC, systolic BP, fasting glucose, and triglycerides being the highest in the persistent MS group. Depressive disorder and stroke were also most prevalent in the persistent MS group.
Characteristics of the study population according to status changes in MS over 2 years
MS, metabolic syndrome; BP, blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol. Values are presented as mean±standard deviation or number (percentage). † Values are presented as median (interquartile range) using the Wilcoxon rank-sum test.
Association of 2-year status changes in MS and its components with the PD incidence and risk
Figure 1 and Table 2 show the longitudinal associations between the 2-year status changes in MS and its components and the PD incidence and risk. The incidence probabilities of PD significantly increased when participants shifted from non-MS, incident MS, or improved MS to persistent MS (log-rank p < 0.001) (Fig. 1). Compared with the non-MS group, the improved MS, incident MS, and persistent MS groups exhibited significantly increased HRs for PD irrespective of adjustments for confounding variables. After adjusting for all potential confounding variables (model 3), the HR of PD was 1.12 (95%CI: 1.06–1.19) in the improved MS group, 1.15 (1.09–1.22) in the incident MS group, and 1.25 (1.20–1.30) in the persistent MS group. With respect to each MS component, individuals with improved, incident, and persistent abdominal obesity had significantly increased PD risk compared with those without abdominal obesity (HR: 1.14, [95%CI: 1.08–1.20] for improved abdominal obesity, 1.14 [1.08–1.21] for incident abdominal obesity, and 1.14 [1.08–1.20] for persistent abdominal obesity). Compared with the non-high triglycerides group, individuals with incident and persistent hypertriglyceridemia had an increased risk of PD (HR [95%CI]: 1.10 [1.04–1.15] and 1.10 [1.06–1.14], respectively) after adjusting for all potential confounding variables.
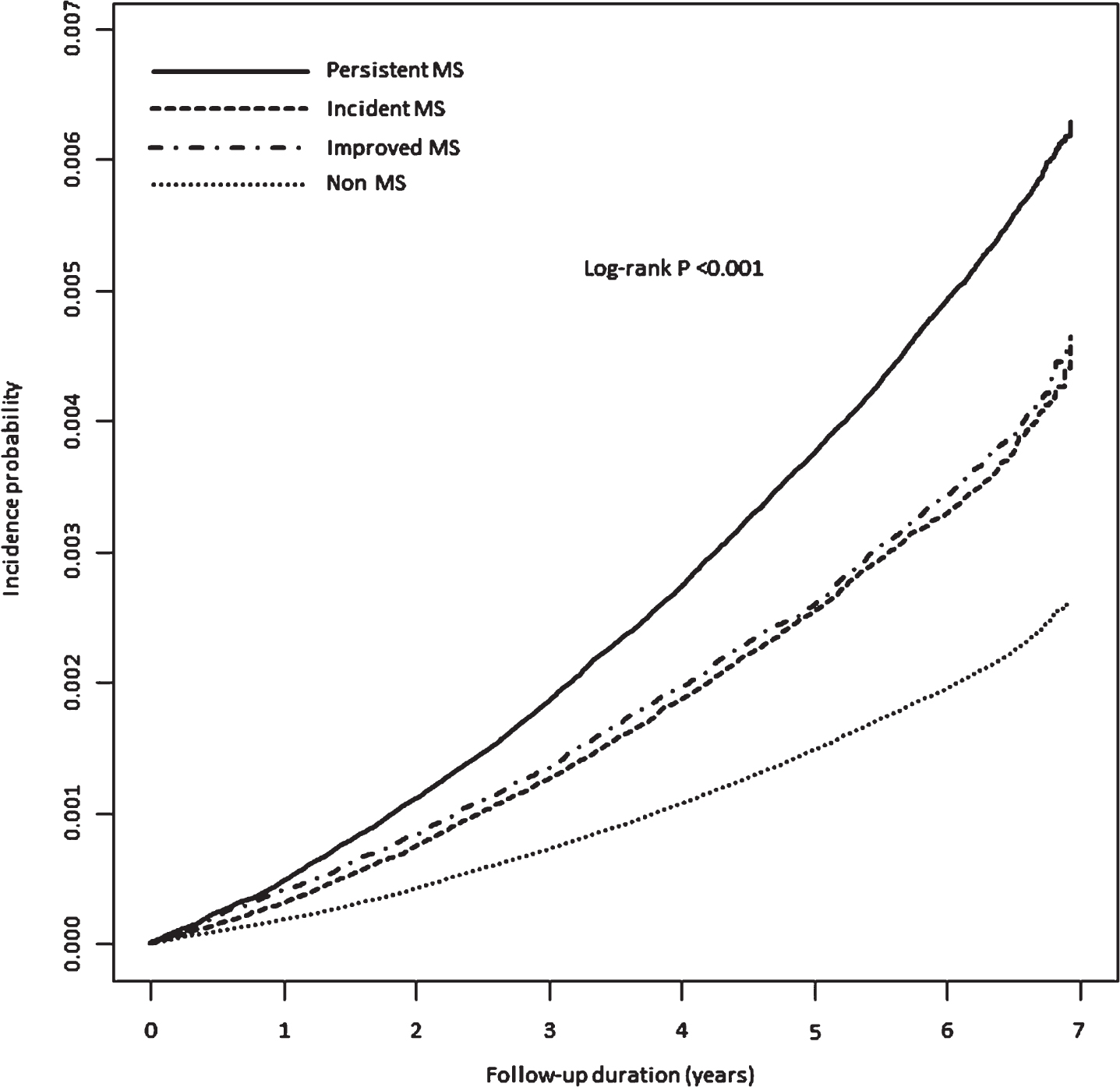
Longitudinal associations between 2-year status changes in MS and incidence probabilities of PD. The incidence probabilities of PD significantly increased when participants shifted from non-MS, incident MS, or improved MS to persistent MS (log-rank p < 0.001). MS, metabolic syndrome; PD, Parkinson’s disease.
Longitudinal association of status changes in MS and its components with PD incidence and risk
MS, metabolic syndrome; PD, Parkinson’s disease; IR, incidence rate; HR, hazard ratio; CI, confidence interval; HDL-C, high-density lipoprotein cholesterol; BP, blood pressure. †PD incidence per 1000 person-years. ‡Model 1 was adjusted for age and sex. §Model 2 was adjusted for age, sex, smoking status, alcohol consumption, physical activity, income, depressive disorder, and stroke. ¶Model 3 was adjusted for age, sex, smoking status, alcohol consumption, physical activity, income, depressive disorder, stroke, and body mass index.
Those with improved, incident, and persistently low HDL-C had higher HR for PD than those without low HDL-C in model 3 (HR [95%CI]: 1.11 [1.05–1.17], 1.20 [1.14–1.26], and 1.24 [1.20–1.29], respectively). Individuals with improved BP for 2 years had an increased risk of PD (HR [95%CI]: 1.09 [1.02–1.16]), but incident and persistently high BP were not significantly associated with PD risk. Incident and persistent hyperglycemia for 2 years were significantly associated with PD risk compared with the absence of hyperglycemia (HR [95%CI]: 1.10 [1.04–1.15] and 1.27 [1.22–1.31], respectively) in model 3.
Subgroup analyses
Table 3 shows the HRs (95%CIs) of PD in the MS status change groups compared with the non-MS group in subgroups by age, sex, and BMI. The longitudinal association between MS status changes and incident PD was stronger among individuals older than 65 years, women, and those with BMI < 25 kg/m2 (p for interaction = 0.004, 0.004, and 0.012, respectively).
Longitudinal association of status changes in MS with PD incidence and risk among subgroups
MS, metabolic syndrome; PD, Parkinson’s disease; IR, incidence rate; HR, hazard ratio; CI, confidence interval. † PD incidence per 1000 person-years. ‡ HR (95%CI) was calculated after adjusting for age, sex, smoking status, alcohol consumption, physical activity, income, depressive disorder, stroke, and body mass index.
DISCUSSION
In this large-scale cohort study of the South Korean population, dynamic status changes in MS and its components over 2 years were independently associated with 7-year incident PD. Individuals with incident and persistent MS for 2 years exhibited 15%and 25%increased HR for PD during the median follow-up of 7 years. With respect to each MS component, incident and persistent status of abdominal obesity, low HDL-C, hypertriglyceridemia, and hyperglycemia was associated with an increased PD risk compared with the absence of either component. Persistent status of MS, abdominal obesity, low HDL-C, hypertriglyceridemia, and hyperglycemia was associated with the highest risk of PD, and a worsened status of both MS and these components was also associated with an increased PD risk. Further, even a resolved status of MS, abdominal obesity, low HDL-C, and high BP was associated with a higher PD risk. Our results suggest that persistent and incident MS and its components may be risk factors for incident PD. Moreover, ever exposure to MS may also be associated with the PD risk. Our findings imply that appropriate intervention for preventing and improving MS may have crucial role in decreasing the incidence of PD.
To date, no study has evaluated the association between dynamic alterations in MS status and incident PD. As previously mentioned, very few epidemiologic studies have evaluated the association between MS and PD, with conflicting findings. A 30-year follow-up cohort study of 6,641 Finnish adults reported that the relative risk (RR) of PD was 0.50 (95%CI: 0.30–0.83) in individuals with MS compared with those without MS [12]. This study attributed this association to hypertriglyceridemia (hypertriglyceridemia vs. non-hypertriglyceridemia; RR: 0.52, 95%CI: 0.30–0.89) and hyperglycemia (hyperglycemia vs. non-hyperglycemia; RR: 0.56, 95%CI: 0.32–0.98). Conversely, in our previous nationwide cohort study of about 17 million South Koreans with 5.3 years of follow-up, MS was associated with 24%increased PD incidence (HR: 1.24, 95%CI: 1.21–1.27) [13]. This study reported consistently positive associations between all five components of MS and the incident PD risk. The findings from these two previous studies appear to be very conflicting. Moreover, the methodology also differed between the two studies. To our knowledge, ours is the first study on the association of status changes of MS and its components with the incident PD risk. Our current findings seem to support the reported positive association between MS and PD, and provide more concrete evidence because we considered the effect of dynamic alterations in MS status on PD.
Previous studies have inconsistently reported the associations of MS components, such as diabetes mellitus (DM), dyslipidemia, obesity, and hypertension, with PD incidence. Many studies support the positive association between DM and incident PD, demonstrating that even prediabetes and insulin resistance have a negative influence on the course of PD [17–19]. However, some studies reported discrepancies in the association, and a recent meta-analysis on the association between DM and PD risk reported completely opposite findings [20–22]. The current results showed an impact of incident and persistent hyperglycemia on the PD risk, supporting the positive association between the two diseases. Studies on obesity (mainly based on BMI) and incident PD have reported contrasting findings [21, 23]. In our study, improved, incident, and persistent abdominal obesity was consistently associated with increased PD risk even after adjusting for confounders including BMI, supporting the positive association between abdominal obesity and incident PD. Abdominal obesity may be a more reliable marker reflecting insulin resistance and inflammation than BMI [24, 25]. Further, even an animal study has shown that exposure to dopamine-specific toxin accompanied by a high-fat diet lowered the threshold for PD development [26]. The association between serum triglycerides and HDL-C and the risk of PD is scarcely reported, whereas the association between total cholesterol and the risk of PD has been inconsistently described [20, 27]. However, our study revealed that incident and persistent hypertriglyceridemia, and improved, incident, and persistently low HDL-C were significantly associated with an increased PD risk, supporting the associations between these types of dyslipidemia closely related to insulin resistance and the PD risk. Defects in the insulin signaling pathway may play a role in the pathogenesis of PD [28]. With respect to hypertension, it seems to have the most conflicting evidence with respect to the association with incident PD, among the other MS components [20, 29]. Our findings have not demonstrated any association between incident and persistent hypertension and PD risk. More studies are warranted to confirm the association between hypertension and PD risk.
The mechanisms linking MS with PD are unclear. MS and PD may have common pathophysiologic pathways. Insulin resistance and inflammation underlying MS may contribute to the progressive loss of dopaminergic cells, resulting in PD [30, 31]. Further, the insulin-related abnormalities of the central nervous system observed in PD have been suggested to increase the sensitivity to neurotoxins and the accumulation of α-synuclein, because the activities of insulin growth factors support neuronal growth and survival [28, 32]. Systemic inflammation induced by MS and its components can damage the blood-brain barrier, decrease waste removal, and increase immune cell infiltration, leading to disruption of glial and neuronal cells, hormonal dysregulation, and increased immune sensitivity linked to neurodegenerative diseases [33]. Cerebral ischemia due to hypertension, dyslipidemia, and diabetes, which are the components of MS, may damage dopaminergic neurons in the substantia nigra [34]. This may induce oxidative stress and affect the loss of dopaminergic neurons in the striatum [35].
Since the effect of MS on PD incidence has not been fully elucidated, our study aimed to examine whether the status changes in MS and its components are associated with incident PD. Our findings strongly support our previous study results showing the positive associations of MS and its components with incident PD, allowing us to suggest that appropriate prevention and management of MS may be a novel approach to reduce the PD incidence. Interestingly, the longitudinal association between MS status changes and incident PD was stronger among elderly individuals, women, and individuals without obesity. The results of subgroup analyses showed that detection and control of metabolic abnormality may be important in these groups. Further studies are needed to confirm the association.
This study has some limitations. First, there may be reverse causality owing to the retrospective study design. Because prodromal symptoms in PD patients could affect weight loss and metabolic features, it may be helpful to exclude individuals with symptoms of pre-motor PD before enrollment to reduce the possibility of reverse causation. We considered a washout and lag period for PD and observed the MS status changes over 2 years to minimize this issue. Second, the diagnosis based on claims may over- or under-estimate PD; moreover, we did not perform serial and repetitive examinations for PD diagnosis. To overcome this, we considered a registration code of PD from the Korean NHIS registration program for rare intractable diseases in addition to ICD-10 code for PD. This operational definition of PD could enhance the accuracy of PD diagnoses. Third, there may be selection bias because we selected individuals who participated in two health examinations with a 2-year interval. As these individuals are more careful about their health than those who participate less in health examinations, we may have underestimated the impact of MS status change on incident PD. Finally, there may be a limitation in generalizing our results to other ethnicities because of possible ethnic or racial differences in MS or PD. Nonetheless, the major strength of our study is the very large sample size based on a nationwide cohort with verified health examination data. This enabled us to consider a wide range of confounding variables related to MS and PD. The novelty of our study lies in the analysis of the dynamic alterations in the status of MS and its components over 2 years and the longitudinally observation of the PD incidence.
In conclusion, our study revealed that status changes in MS are independently associated with PD development. Persistent and incident MS and its components may be risk factors for incident PD; and ever exposure to them may also be associated with incident PD. Our findings suggest that proper management for both improvement and prevention of MS may play a crucial role in decreasing the incidence of PD.
Footnotes
ACKNOWLEDGMENTS
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2019R1I1A1A01061757) and this funding source had no role in the design of this study and will not have any role during its execution, analyses, interpretation of the data, or decision to submit results.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
