Abstract
Background:
The underlying disease mechanisms of Parkinson’s disease (PD) are still unknown and knowledge about risk and prognostic factors is sparse.
Objective:
To examine the association between intelligence, education, body height, and body mass index (BMI) in young adulthood and risk of PD and subsequent survival.
Methods:
In total, 656,751 men born 1939–1959 with information from conscription examinations around age 19 years were followed for PD and mortality from 1977–2018 in Danish registries. Cox proportional hazard regression was used to conduct the analyses.
Results:
During follow-up, 5,264 (0.8%) men were diagnosed with PD. Higher intelligence, education, and body height conferred a higher hazard of PD, independent of age at disease onset. BMI above compared to below the mean (22.8 kg/m2) was associated with slightly higher hazard of late-onset PD (>60 years). During follow-up, 2,125 (40.5%) men with PD died, corresponding to a 2.55 (95% confidence interval:2.44–2.66) times higher mortality compared to men without PD. Intelligence was inversely associated with mortality in men with and without PD. Higher education and body height were also inversely associated with mortality in men without PD, whereas the estimates were less pronounced and imprecisely estimated for men with PD. Having an obese BMI was associated with higher mortality in men with PD.
Conclusion:
Intelligence, education, and body height in young adulthood are positively associated with risk of PD later in life among men. BMI above the mean only confer a higher risk for late-onset PD. For men diagnosed with PD, high intelligence is the only early life indicator associated with better survival, whereas obese BMI predicts poorer survival.
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disorder after Alzheimer’s disease. In 2016, an estimated 6.1 million individuals suffered from PD worldwide and the incidence is increasing [1–3]. Prevention of PD has high priority due to the lack of a curative treatment. The existing treatment modalities are symptomatic and cannot prevent nor stall the progression of the disease. The treatment often has significant side effects, for instance, anorexia, hypotension, psychosis, and hallucinations [4, 5]. Elucidating the underlying disease mechanisms and identifying risk and prognostic factors for PD might open new avenues for treatment, rehabilitation, and prevention.
The incidence of PD is about 1.4 times more common in men than in women [2]. Genetic factors moderately predispose to PD, but no genetic cause can be identified in 90–95% of patients with PD [1, 6]. As to behavioral risk factors, smoking, coffee consumption, and alcohol use have been suggested to reduce risk of PD [6–9], yet it is unclear if this association is causal. Mixed findings regarding the association between body weight and PD have been reported. Thus, several case-control studies have shown that patients with PD have a lower body weight than their respective controls, whereas cohort studies, mainly in middle-aged populations, have shown no or a weak positive association between body mass index (BMI) and PD [10]. Moreover, a Swedish cohort study has shown that people with high socioeconomic position, measured by occupational status, had a higher risk of developing PD [11]. Finally, a recent study among Swedish conscripts showed that high intelligence at age 17–20 years was associated with a higher risk of early-onset PD, whereas physical fitness was not [12]. All in all, the risk profile for individuals who develop PD seems to be markedly different from most other chronic diseases, which are typically related to lower socioeconomic position and poor lifestyle. The risk of PD appears to be founded early in life [13, 14], but studies of early-life risk factors for PD are challenging. Since PD is a relatively rare disease, studies of early-life risk factors require large cohorts with information on early-life factors. Furthermore, to capture both early and late onset PD, it is necessary to have a follow-up period that spans nearly the entire life course. Moreover, risk factors for PD in adult life may not be an optimal proxy for early-life factors, since late-life factors may be prone to reverse causation, i.e., preclinical PD may influence the adult risk factors.
Patients with PD experience higher mortality than individuals without PD [15]. Yet, it is unknown whether factors, which are shaped in early life and identified as important predictors of mortality throughout life in the general population [16, 17], impose a different risk of death in individuals with and without PD, respectably. Understanding the importance of early life factors for mortality in individuals with PD will help elucidate why this patient group has higher mortality than the general population.
Against this background, this study aims to investigate the role of early-adult intelligence, education, body height, and BMI for the development of PD with early and late-onset and subsequent survival in a cohort of 728,158 men.
METHODS
Participants
In Denmark, all men are instructed by law to appear before the conscript board for a physical and mental examination between the ages of 18 and 26 years. This study was based on data from the Danish Conscription Database (DCD), which has been described in detail elsewhere [18]. The DCD holds information on body height, weight, intelligence test scores, and educational level from Danish conscription board examinations around the age of 19 years for 728,158 men born predominantly between 1939–1959. We excluded 61,842 (8.5%) men due to missing information on all key variables. Men with no information mainly include those exempted from the draft board examination due to medical conditions such as mental retardation, asthma, epilepsy, or type 1 diabetes. Moreover, 1196 men were excluded because they were born before 1939 or after 1959, or because they were less than 17 or more than 30 years old at the time of examination. Finally, 8,369 men were excluded because they had died (n = 4,027) or emigrated before 1977 (n = 4,342) when follow-up was started in the Danish National Patient Registry. As shown in Supplementary Figure 1, this left 655,230 men for analysis. Moreover, the analysis of BMI was restricted to 374,610 men (57.1%) because 282,141 (43.1%) men in the main study population belonged to conscript districts with no systematic registration of weight [16]. The study has been approved by the Danish Data Protection Agency.
Measures from conscription board examinations
The conscript board intelligence test (the Børge Prien Prøve (BPP)) consists of logical, verbal, numerical, and spatial subtests. The BPP score has a range of 0 to 78 and correlates well with the full-scale Wechsler Adult Intelligence Scale-score (r = 0.82) [19]. Information on the educational level reported at conscription was categorized into low (7–9th grade), medium (vocational training or 10th-11th grade), and high education (12th grade or more advanced). Body height was measured without shoes and weight was measured with only underwear. BMI (kg/m2) was calculated as weight in kilograms divided by the square of height in meters. We initially explored the associations between each of the continuous variables with PD using restricted cubic splines with 4 knots equally spaced over the variable. The knots for intelligence were 17; 33; 43; 57 with mean intelligence (38) as the reference, for body height the knots were 166; 174; 179; 188 with the mean (178) as the reference, and for BMI the knots were 18.3; 20.7; 22.4, and 26.3 kg/m2 with mean BMI (21.7 kg/m2) as the reference. The initial analyses indicated that intelligence and body height could be entered as continuous variables, whereas BMI was best described in two categories (below and above mean) (Supplementary Figure 2). For mortality outcomes, BMI was divided in 1) underweight:<18.5 kg/m2, 2) normal weight: 18.5 to < 25 kg/m2, 3) overweight: 25 to < 30 kg/m2, and 4) obese: ≥30 kg/m2.
Parkinson’s disease and mortality
PD was defined by the first in- or outpatient hospital discharge diagnosis (International Classification of Disease (ICD) version 8 (ICD8):342 and ICD10: G20.9) in the Danish National Patient Registry [20]. The Danish National Patient Register (DNPR) includes data on diagnosis and time of admission for all in-hospital contacts in Denmark since 1977 and information on outpatient and emergency contacts since 1995. The validity of PD diagnoses obtained from the DNPR has been assessed to be 82% when compared to the diagnostic criteria at clinical case review [21]. We supplemented our definition of PD with data on individual patients’ refill of at least one redeemed prescription of an antiparkinson agent identified by the Anatomic Therapeutical Chemical (ATC) classification system codes (N04) in the Danish National Prescription Registry. This register holds information on all prescription drugs sold at Danish pharmacies since 1995 [22]. Some antiparkinson agents, especially some types of dopamine agonists (N04BC), are also approved for the treatment of restless leg syndrome, and consequently, we only included prescriptions with the indication codes for PD.
Information on the date of death was obtained for all cohort members from the Danish Civil Registration System [23]. We defined premature mortality as death before 65 years.
Supplementary analysis
Previous studies have indicated that bipolar disorders are associated with PD [24], and we constructed the following supplementary analyses to explore the influence of bipolar disorder on the associations. First, we explored the association between bipolar disorders and PD, second, we adjusted for bipolar disorder as a potential confounder, and third, we excluded men with bipolar disorder at study entry. Bipolar disorder was defined as the first admission diagnosis to a psychiatric or somatic ward since 1969 or 1977, respectively, in the Danish Psychiatric Central Research Registry (established in 1969) and the DNPR. Bipolar disorder was defined as ICD-8 : 296.19, 296.29, 296.39, 296.89, 296.99.
Statistical analysis
Descriptive statistics were used to report the characteristics of the cohort. The cohort was followed from their age on January 1, 1977, (the date the DNPR was initiated, for men conscripted 1957–1976; age 17–37 years) or at the date of conscription examination (for men conscripted 1957–1984, age 17–21 years), whichever came last, and until their age at the first registration of PD outcome, emigration, death, or end of follow-up (January 1, 2019; age 59–81 years), whichever came first. First, the Aalen-Johansson estimator was applied to calculate cumulative hazard curves of PD by each of the four indicators taking competing risk of death into account as a censoring event. Second, to retrieve hazard ratio estimates, Cox proportional hazard regression also with age as the underlying timescale was applied. These analyses were first conducted with PD diagnosis based on hospital visits as the outcome and secondary analyses were conducted with PD diagnosis based on both hospital visits and/or use of antiparkinson agents. In this second analysis, the cohort was followed from January 1, 1995, when the Danish National Prescription Registry started. Since associations may vary with the age of PD onset, analyses were also split at age 60 and 70 years of follow-up. In the Cox Proportional Hazard model for mortality, men were followed from age on January 1, 1977 or date of conscription examination, whichever came last, and until the age at first registration of emigration, death, or end of follow-up (January 1, 2019), whichever came first. In these models, diagnosis of PD was entered as a time-dependent variable, i.e., individuals contributed with person-years as not having Parkinson’s disease-until the time they were diagnosed. The proportional hazard assumption was examined by inspection of cumulative hazard plots for categorical variables and Schoenfeld residuals for continuous variables. All analyses were carried out in Stata version 15.
RESULTS
Table 1 shows the distribution of young adult intelligence, education, body height, and BMI in the cohort. Mean intelligence, body height, and BMI were 37.8 points (standard deviation (SD) 12), 176.8 cm (SD 6.8), and 21.8 (SD 2.5), respectively. During a follow-up until mean age 66.7 (SD 9.2) years, 5,264 (0.8%) men were diagnosed with PD, and of them, 1,181 had been diagnosed before age 60, and 2593 were diagnosed before age 70 years (Supplementary Table 1). During follow-up, 178,743 (27.2%) men died of which 2,215 had been diagnosed with PD corresponding to 40.3% of the total number of men diagnosed with PD.
Distribution of main variables in relation to diagnosis of Parkinson’s disease and mortality in a cohort of Danish men born 1939–1959
*percent of all men; **percent of men with Parkinson’s disease.
The cumulative incidence of PD was 1 % at age 60 years. Figure 1 shows the cumulative incidence of PD taking the competing risk of death into account and stratified by young adult intelligence, education, body height, and BMI. For intelligence, education, and body height, men in the highest quintiles had the highest cumulative incidence of PD, and the cumulative incidence gradually decreased to the lowest incidence for men in the lower quintiles. The cumulative incidence of PD was slightly higher among men with a BMI above compared to below the mean.
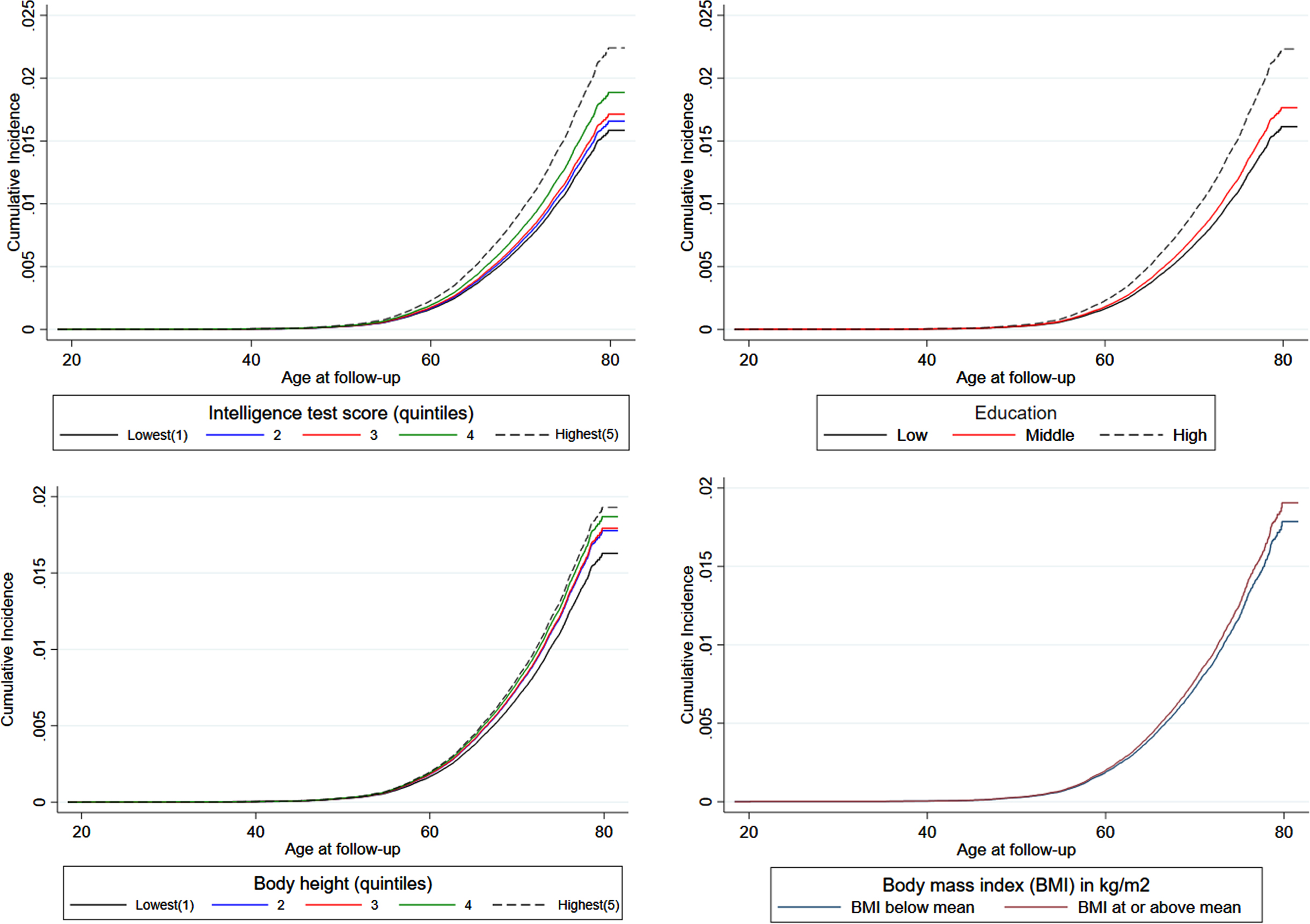
Cumulative incidence of diagnosis of Parkinson’s disease by levels of intelligence, education, body height, and body mass index (BMI).
In the Cox proportional hazard regressions (Table 2), intelligence, education, and body height in early adulthood were positively associated with both early and late-onset of incident PD diagnosis. In general, there was little difference in risk estimates between early and later follow-up. However, a BMI above the mean was only associated with late onset PD and the risk estimates were strongest at age 60–69 years. During follow-up, 7,207 (1.1%) men had either been diagnosed with PD and/or had redeemed an antiparkinson agent. Cox Proportional hazard analyses of PD defined as a registered diagnosis and/or redeemed antiparkinson agent showed overall same association patterns as the analyses with PD defined only be registered diagnosis (Supplementary Table 2).
Hazard Ratios (HR) and 95% Confidence Intervals (CI) for young adult intelligence, education, body height and body mass index (BMI) and later diagnosis of Parkinson’s disease in Danish men born 1939–1959
*Adjusted for age and stratified on birth cohort; body height is also adjusted for education.
Figure 2 shows that survival at age 65 years was 60% and 85% in men with and without PD since diagnosis of PD and conscription or 1977, respectively. The survival curves stratified by intelligence, education, body height, and BMI showed that men with PD had the lowest survival in all strata. Men with a lower level of intelligence, education, body height, and obesity, respectively, had the poorest survival within the two groups of men without and with PD.
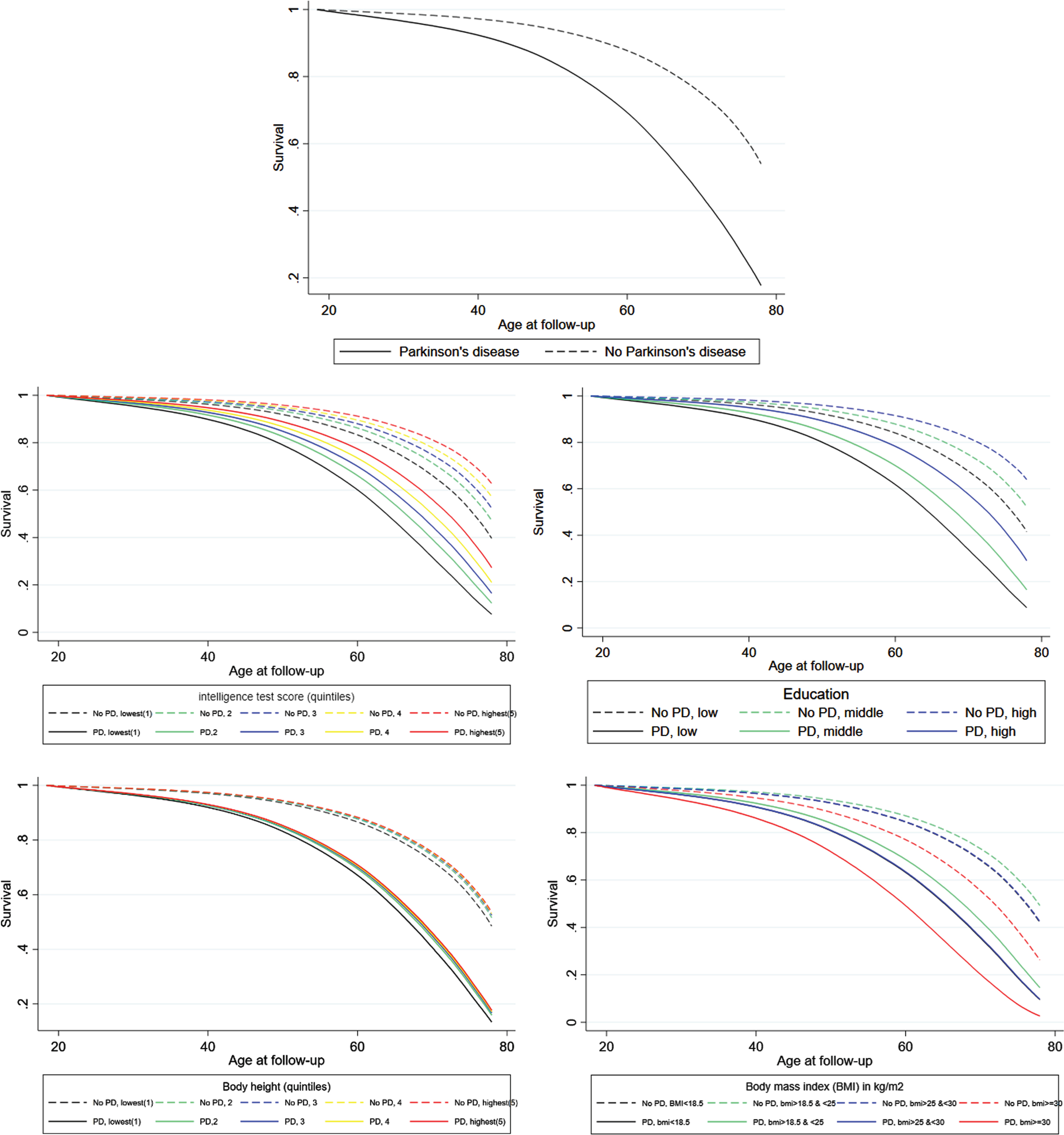
Survival in men with or without Parkinson’s disease and by levels of intelligence, education, body height, and body mass index (BMI). For BMI the blue and black curves are overlapping both in men with and without PD.
The Cox proportional hazard regression showed that men with PD had a 2.55 (CI: 2.44–2.66) times higher mortality than men without and the risk estimate became slightly stronger when the early life factors were included as covariates in the statistical model (HRadjusted = 2.63 (CI: 2.52–2.75)) (Supplementary Table 3). The HRs did not differ for death before (HR = 2.68 (CI:2.41–2.91)) or after (HR = 2.52 (CI:2.40–2.69)) age 65 years. In stratified analyses, intelligence was inversely associated with mortality in men with and without PD. Education and body height were also inversely associated with mortality in men without PD, whereas the corresponding risk estimates were less pronounced and imprecisely estimated in men with PD (Table 3). The association between BMI and mortality was U-shaped in men without PD, whereas only obese BMI was associated with higher mortality in men with PD.
Hazard Ratios (HR) and 95% Confidence Intervals (CI) for young adult intelligence, education, body height and body mass index (BMI) and mortality in Danish men with or without Parkinson’s disease
*Adjusted for age and stratified on birth cohort; body height adjusted for age and education.
The supplementary analysis showed that 614 men were diagnosed with bipolar disorder at study entry and men with bipolar disorder had nearly three times higher risk of PD than men without bipolar disorder at study entry (HR = 2.94 (1.58–5.46)). However, the risk estimates for the other variables reported above were not changed by adjustment for bipolar disorder or nor if we excluded these men from the analyses.
DISCUSSION
This study of 656,751 men followed in nationwide registers from conscription to a maximum age of 82 years showed that high intelligence and education, as well as tall body height in young adulthood, were associated with a higher hazard of developing PD. BMI above compared to below the mean was associated with a slightly higher hazard of late-onset PD. Intelligence was inversely associated with mortality both in men with or without PD, whereas education and body height only were inversely associated with mortality in men without PD. The relationship between BMI and mortality was U-shaped for men without PD, whereas only obese BMI was associated with higher mortality in men with PD.
Comparison with previous studies
A recent Swedish cohort study with follow-up until a mean age of 53 years also found that high intelligence was associated with early-onset PD [12]. Our study confirmed and extended this finding by showing that the association was also present for late-onset PD. The identified relationship between high intelligence and a higher risk of PD is contrary to previous evidence of associations between low intelligence and other somatic and psychiatric diseases [25–27]. An exception is bipolar disorder, which has been associated with high intelligence and excellent school performance [28, 29]. Moreover, large cohort studies have indicated that bipolar disorders are associated with PD [24], and it has been suggested that the two conditions share the mechanisms of dopamine dysregulation, which persons with high intelligence might be particularly sensitive towards. In the present study, bipolar disorder was associated with a higher risk of PD, but bipolar disorder did not explain the found association between the main exposures and PD. We, as well as other studies, found that higher socioeconomic position, previously measured by occupational status and in this study measured by educational level, was associated with increased risk of PD [11]. A higher socioeconomic position is also associated with healthy lifestyle exposures which may be associated with PD [9]. A significant shortcoming of the previous studies about the role of BMI in the development of PD is the fact that bodyweight changes constitute a very early sign of PD. A decrease in body weight several years before PD diagnosis has been reported in some studies, whereas other studies found no association or, contrastingly, weight gain [30]. In the present study, BMI was measured at least 20 years before the onset PD, thereby minimizing the risk of reverse causality. We found that BMI above the mean was associated with a slightly higher risk of late-onset PD, whereas we found no association with early-onset PD.
In agreement with most previous studies, we found that men with PD had higher mortality than men without PD. The adjusted HR of 2.63 (CI: 2.52–2.75) in our study was higher than the pooled estimate of 1.5 reported in a recent meta-analysis based on studies of mortality in PD [15]. This difference might be explained by the present study being based on men, and several studies have suggested that men with PD have higher relative mortality than women with PD [15, 32]. Further, many previous cohort studies have been based on a relatively small number of individuals with PD leading to imprecise effect measures. In keeping with some previous studies [8], we found no difference in the risk of premature or later mortality. No studies have previously examined the influence of early-life risk factors on mortality in individuals with PD. However, a recent study from the UK, which examined mortality risk in 10,104 patients with PD and a matched reference population of 55,664 without PD, showed that PD patients living in the most socially deprived areas had higher mortality. In our study, low intelligence in young adulthood was associated with higher mortality following PD, whereas the estimates for education did not reach significance. Intelligence and education are highly correlated and some studies have indicated that low intelligence is associated with social disadvantage later in life [33, 34]. The observation of an increased risk of PD in higher social groups, but a better subsequent survival subsequent has also been reported for other chronic diseases such as prostate cancer and melanoma [35]. This suggests that even though a healthy lifestyle may not impact the development of the conditions, fewer co-morbidities and better compliance with treatment seen in patients from higher social groups may contribute to their better subsequent survival after receiving the diagnosis.
Strength and limitations
The biggest strength of our study is the large sample size and long follow-up period, which covered earlier birth cohorts (1939–1959) than the comparable Swedish conscription cohort (1950–1987) [12]. Our study covered 21 annual consecutive birth cohorts followed up for a mean of 38 years up until an age of 62 to 82 years. This enabled us to study not only early-onset but also late-onset PD. Furthermore, in the present study, outcome information was retrieved from national registers with sensitivity measures of PD above 80% [21]. The Danish National Patient registry started on January 1, 1977, and cases diagnosed the first years from this date may not represent true incident cases. However, cohort members had a mean age of 27.8 (range 17–37 years) in 1977, and less than five men were diagnosed with PD before 1983. It is, thus, unlikely that excluding these few cases the first five years of the register would change the findings. Unfortunately, we did not have information on lifestyle, such as exercise or smoking from the health registers. This may pose a risk of unmeasured confounding in our study because these factors may influence BMI and later risk of PD as well as mortality. However, these factors may also have acted as mediators for the association and should, thus, not be adjusted for. Yet, it would be relevant for future studies to address the possible mediating role of lifestyle factors for the relationships identified in this study. Finally, our cohort consisted of men only, precluding generalization to women. The relation between the exposures and PD might be different for women, who have a lower incidence and subsequent mortality, later onset, and different symptomatology of PD [3].
In conclusion, intelligence, education, and body height at entry into adulthood are positively associated with the risk of PD later in life among men. BMI above the mean only confer a higher risk for late-onset PD. Higher intelligence, education, and body height are associated with better survival in men without PD. For men diagnosed with PD, high intelligence is the only early life indicator associated with better survival. In contrast to the U-shaped relationship between BMI and mortality in men without PD, only obese BMI is associated with poorer survival in men with PD.
