Abstract
Background:
Lipopolysaccharide-binding protein (LBP) presents bacterial endotoxin, lipopolysaccharides, to cellular surface pattern receptors for immune responses in the gut-brain axis of Parkinson’s disease (PD).
Objective:
We investigated whether plasma LBP levels were associated with PD severity and progression.
Methods:
This study included 397 participants (248 PD patients and 149 controls). We measured participants’ plasma levels of LBP and pro-inflammatory cytokines, including TNF-α, IL-6, andIL-17A. PD patients underwent motor and cognition evaluations at baseline and at a mean follow-up interval of 4.7±2.3 years. We assessed the progression of motor and cognition symptoms based on changes in the Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part III motor score and Mini-Mental State Examination (MMSE) score, respectively.
Results:
Plasma LBP levels were lower in PD patients than controls (9.08±2.91 vs. 10.10±3.00μg/ml, p < 0.01). A multiple logistic regression model with adjustment for age, sex, and plasma cytokine levels revealed that reduced plasma LBP levels were associated with increased PD risk (odds ratio 0.816, [95% CI 0.717–0.929], p = 0.002). Among PD patients, LBP levels were correlated with MDS-UPDRS part III motor score after adjustment for confounders (coefficient = 0.636, p = 0.017), but not with MMSE score. Adjusted Cox regression analysis showed that higher plasma LBP levels associated with faster motor progression (adjusted hazard ratio 1.084 [95% CI 1.011–1.163], p = 0.024) during follow-up.
Conclusion:
Our results demonstrated that plasma LBP levels reflect risk, motor symptom severity and progression in patients with PD.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a common neurodegenerative disorder, and emerging evidence suggests that the gut-brain axis plays a critical role in PD pathogenesis [1]. The pathological hallmark of PD is Lewy body formation by neuronal accumulation of α-synuclein, which can be identified within the gut enteric nervous system early in the disease process [2, 3]. Moreover, gastrointestinal dysfunction, especially constipation, is consistently the most common and earliest symptom prior to motor disability [4, 5]. Recent studies in PD rodent models show that altered gut microbiota associated with chronic gut inflammation may trigger neuronal aggregation of α-synuclein, and promote motor dysfunction [6, 7]. Additionally, compared to controls, PD patients show more prominent evidence of intestinal hyper-permeability with enteric inflammation, reflected by increased expression of pro-inflammatory cytokines and chemokines in stool and colonic tissues [8]. These findings suggest that leaky gut with intestinal hyper-permeability contributes to the early stage of the disease process [7].
Increased intestinal permeability enables the entrance of bacterial endotoxin, as recently demonstrated by intestinal staining for the gram-negative bacteria Escherichia coli [9]. Lipopolysaccharide (LPS), the outer membrane component of gram-negative bacteria, serves as an endotoxin that can activate surface pattern receptors in intestinal epithelial cells, e.g., toll-like receptors (TLRs), promoting gut hyperpermeability and activating gut and systemic innate immune responses in both PD rodent models and human patients [10–13]. LPS-binding protein (LBP) is a glycoprotein that is produced in the liver and then enters blood circulation, where it can opsonize LPS and facilitate its recognition by macrophages, thus activating monocyte responses that lead to the secretion of inflammatory cytokines [14]. Several cross-sectional studies show reduced plasma LBP levels in PD patients compared to healthy controls [13, 16], but conflicting results have also been reported [17]. It remains largely unknown whether plasma LBP levels reflect PD severity or disease progression.
Our group previously demonstrated altered fecal microbiota in patients with PD compared to healthy controls matched for age, sex, and diet habits [18]. Notably, an abundance of Bacteroides was correlated with motor symptom severity and plasma TNF-α level in patients with PD [18]. Bacteroides is one of the largest genera of gram-negative bacteria in the gastrointestinal tract microbiome, and reportedly activate macrophages and monocytes through LPS-mediated pathways [19]. Thus, in this longitudinal follow-up study we aimed to delineate whether plasma LBP levels were associated with risk, severity and progression of PD, and the concomitant host systemic cytokine responses.
MATERIALS AND METHODS
Study participants
Patients with PD were recruited from the movement disorder clinics of National Taiwan University Hospital, a tertiary medical referral center in Taiwan. Healthy controls were neurologically normal friends or spouses who accompanied the patients with PD to the institute. PD was diagnosed according to the UK PD Society Brain Bank Clinical Diagnostic Criteria [20]. All patients underwent brain magnetic resonance imaging to exclude vascular parkinsonism or other secondary causes of parkinsonism. We also excluded patients with atypical parkinsonism syndromes, including multiple system atrophy, progressive supranuclear palsy, corticobasal syndrome, and neuroleptic agent-related parkinsonism. Blood tests were performed to exclude participants with impaired renal or liver function. All participants provided written informed consent before entering the study. This study was approved by the institutional ethics board committees of National Taiwan University Hospital (institutional review board review number: 201904104RINA), and all methods were performed in accordance with the relevant guidelines and regulations.
Clinical evaluation and follow-up of motor and non-motor symptoms
We measured the severity of motor symptoms using the Movement Disorder Society-Unified Parkinson’s Disease Rating Scale part III motor scores (MDS-UPDRS part III) [21] and the Hoehn-and-Yahr (H-Y) staging scale [22]. Patients with H-Y stage of < 3 were classified as having early-stage PD, while patients with H-Y stage of ≥3 were classified as having advanced-stage PD. Cognition was examined using the Mini-Mental State Examination (MMSE) during the “on” phase of PD [23]. All patients underwent brain MRI studies and routine laboratory tests to exclude non-PD causes of dementia. PD with dementia (PDD) was diagnosed following the criteria proposed by the Movement Disorder Society [24], with an MMSE score of ≤25 operationally applied as the cut-off value for significant cognitive impairment, combined with any impairment in the eight instrumental activities [25].
Patients were prospectively follow-up for motor and cognitive progression, which were assessed based on changes in the scores on MDS-UPDRS part III and MMSE during the follow-up period. Motor progression was defined as a sustained increase of MDS-UPDRS part III by ≥3 points at follow-up. Cognitive progression was defined as a sustained decline in MMSE score by ≥2 points at follow-up.
Measurement of plasma levels of LBP and cytokines
From each participant at enrollment, we collected a 10 ml venous blood sample, which was centrifuged (2500×g for 15 min) within 1 h of collection and stored at –80°C until analysis. Plasma LBP concentration was measured using an ELISA kit (Ray-Biotech Life, Inc. Georgia, USA). Pro-inflammatory cytokines, including tumor necrosis factor alpha (TNF-α), interleukin-6 (IL-6), and interleukin-17A (IL-17A), were assessed using the Simoa platform with the CYTOKINE 3-PLEX B kit (C3PB, Quanterix, Lexington, MA, USA). Each analyte was measured twice from each sample.
Statistical analysis
Continuous variables are expressed as mean±standard deviation, and categorical variables as numbers and percentages. Variables following a Gaussian distribution were compared using two-tailed t-tests or analysis of variance (ANOVA). We tested the homogeneity of variances using Levene’s test. For variables violating the assumptions of normality or homoscedasticity, the groups were compared using the non-parametric Mann-Whitney U test (for two groups) or Kruskal-Wallis test (for more than two groups). The plasma levels of targeted markers (including LBP, TNF-α, IL-6, and IL-17A) were compared between individual severity groups of PD patients and healthy controls. Bonferroni correction was applied for multiple comparisons and the statistical threshold for significance was adjusted to 0.05/6 = 0.0083 in Table 1. We used the Shapiro-Francia test to test the normality of plasma LBP level. We used Spearman’s rank sum test to evaluate the correlations between plasma LBP levels and the targeted pro-inflammatory cytokines, with adjustment for age and sex.
Clinical characteristics of all participants in the current study
IL, interleukin; LBP, lipopolysaccharide binding protein, MDS-UPDRS, Movement Disorder Society Unified PD Rating Scale; MMSE, Mini-Mental State Examination; TNF-α, tumor necrosis factor-α, IL-6, interleukin 6; IL-17A, interleukin-17A; N.A., not available; PD, Parkinson’s disease. Numbers are expressed as mean±standard deviation. *Statistically significant: after Bonferroni correction, the statistical threshold was adjusted to 0.05/6 = 0.0083.
A logistic regression model was applied to examine the relationship between plasma LBP levels and the risk of PD development, with adjustment for variables including age, sex, and plasma levels of cytokines. We used the area under a receiver operating characteristic curve (AUC) to quantify the model’s diagnostic performance for exploring the ability of plasma LBP level, alone or combined with other markers, to predict PD risk. The covariates for the basic model predicting risk of PD or motor symptoms progression are age and sex. Then, we added the plasma LBP level as next covariate into the basic model, followed by plasma TNF-α, IL-6 and then IL-17A individually in a stepwise manner. The predictive ability was determined using Nagelkerke’s R2 index and we tested calibration using the Hosmer-Lemeshow test for goodness of fit. A p value < 0.05 was considered significant deviance from the theoretical perfect calibration.
To examine the correlation between plasma LBP level and PD severity, we first used Spearman’s rank sum test to determine how plasma LBP level was correlated with motor severity (measured by MDS-UPDRS part III score) and cognitive function (evaluated by MMSE). Next, multivariable linear regression analysis was performed to investigate how plasma LBP levels influenced motor symptom severity (MDS-UPDRS part III scores) and cognition symptom severity (MMSE scores). The covariates included age, sex, disease duration, and plasma pro-inflammatory cytokines.
We used a multivariate Cox regression model to examine the association between plasma LBP levels and the occurrence of motor or cognitive progression during follow-up (the outcome of interest). The multivariate model was adjusted for age (continuous), sex (male or female), disease duration (continuous), and plasma pro-inflammatory cytokine levels (continuous). The proportional hazards assumption was tested using Schoenfeld residuals (phtest), revealing no statistically significant violation. Results are presented as adjusted hazard ratios (HRs) with their 95% confidence intervals (CIs) for the associations. Receiver operating characteristic curve (ROC) analysis was performed to assess the diagnostic accuracy of plasma LBP levels for motor progression. The Youden J index was calculated for all points of the ROC curve, and we used the maximum index value to select the optimal LBP cut-off value for predicting motor progression. A Kaplan-Meier curve was used to determine the cumulative probability of risk of motor progression during follow-up in patients with a plasma LBP level at or above versus below the cut-off value determined in ROC analysis. The log-rank test was used to identify statistical differences in the Kaplan-Meier curves between the groups. A p value of < 0.05 was considered significant. All analyses were performed using Prism 9 (GraphPad Software, La Jolla, CA, USA).
RESULTS
A total of 418 participants were screened; 15 did not meet the inclusion criteria due to having signs of vascular parkinsonism or other secondary causes of parkinsonism and 6 were unwilling to participate due to blood sampling (Supplementary Figure 1). We therefore enrolled 397 participants, including 248 PD patients (68.9±10.8 years old, 46.4% male) and 149 healthy controls (67.7±9.9 years old, 46.3% male). Table 1 summarizes the clinical characteristics and baseline plasma levels of LBP and pro-inflammatory cytokines in all participants at enrollment. The PD and control groups did not significantly differ in the basic demographics. Table 1 also presents the mean MDS-UPDRS part III score and MMSE in patients with PD. The results of the Shapiro-Francia test showed the plasma LBP levels were normally distributed (p = 0.089) and the histogram of the plasma LBP levels were shown in Supplementary Figure 2.
Comparison of plasma levels of LBP and pro-inflammatory cytokines between PD patients and controls
Plasma LBP levels were significantly lower in PD patients than controls (9.08±2.91 vs. 10.10 ±3.00μg/ml, p < 0.001, Fig. 1a). For the pro-inflammatory cytokines, plasma levels of TNF-α had a trend to be higher in PD patients than controls (1.76±0.86 vs. 1.59±0.68 pg/ml, p = 0.049, Table 1), while the circulating IL-17A levels tend to be lower in PD patients than controls (0.08±0.12 vs. 0.11±0.14 pg/ml, p = 0.023, Table 1). Plasma IL-6 levels did not significantly differ between groups. Plasma LBP levels showed a modest correlation with TNF-α (ρ= 0.335, 95% CI 0.242–0.422, p < 0.001, R2 = 0.091), IL-6 (ρ= 0.472, 95% CI 0.388–0.547, p < 0.001, R2 = 0.189), and to a lesser extent for IL-17A (ρ= 0.268, 95% CI 0.172–0.360, p < 0.001, R2 = 0.054) (Supplementary Figure 3a-c).
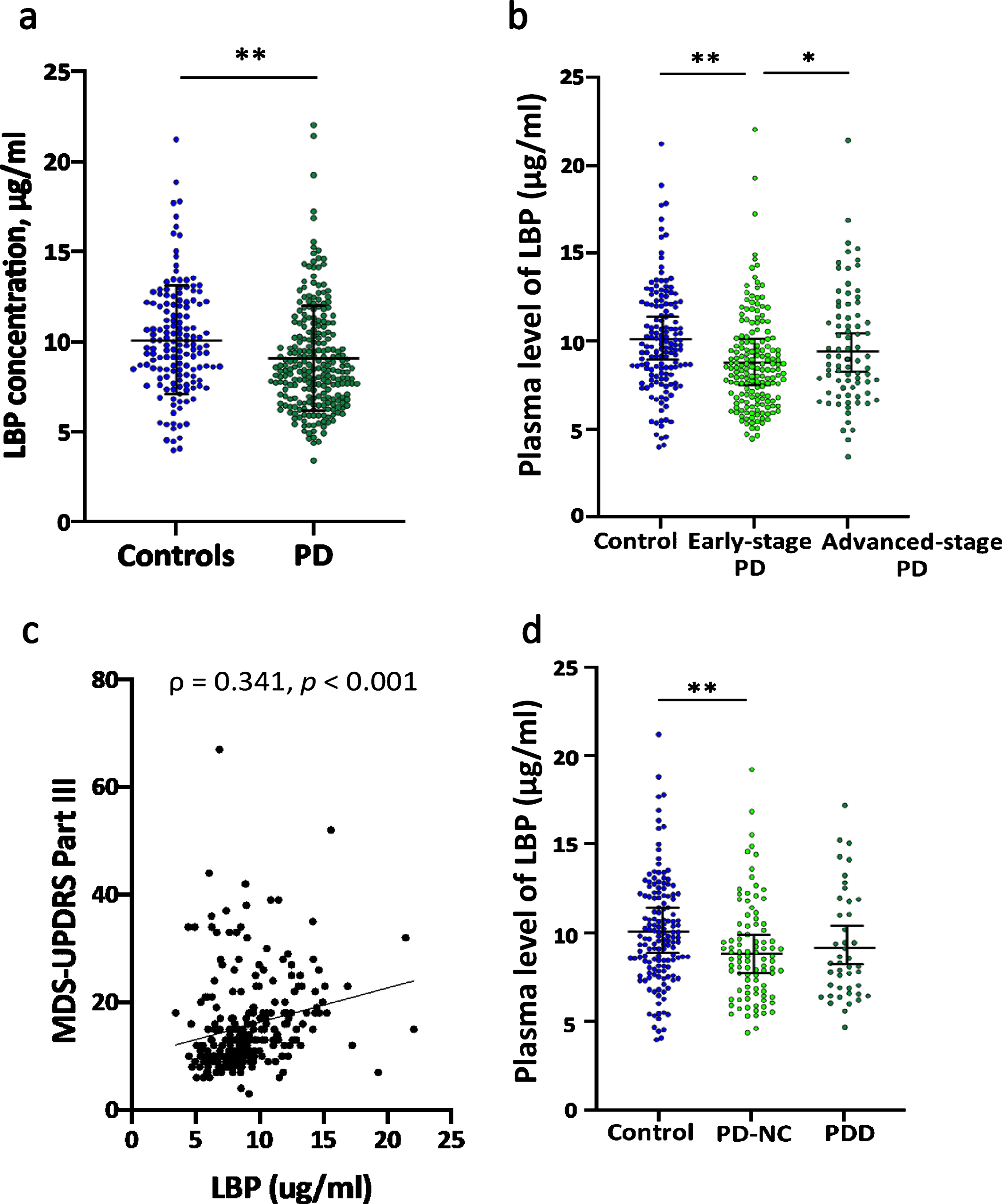
Comparison of plasma LBP levels between PD patients with different severity of symptoms and healthy controls. (a) Compared to healthy controls, PD patients had significantly lower plasma LBP levels (9.08±2.91 vs. 10.10±3.00μg/ml, p < 0.001). (b) Plasma LBP levels were significantly lower in early-stage PD patients than lose in control participants (8.81±2.73 vs. 10.10±3.00μg/ml, p < 0.001). Plasma LBP levels were significantly higher in patients with advanced-stage PD than with early-stage PD (9.64±3.22 vs. 8.81±2.73μg/ml, p = 0.049). (c) Plasma LBP levels were significantly correlated with MDS-UPDRS part III score (ρ= 0.341, 95% CI 0.222–0.450, p < 0.001). (d) Plasma LBP levels were comparable between PD patients with normal cognition (PD-NC) and patients having PD with dementia (PDD) (8.97±2.76 vs. 9.30±2.98μg/ml, p = 0.665).
We then examined the possible influence of age on plasma LBP levels. Spearman correlation analysis revealed that plasma LBP levels were positively correlated with age in all participants (ρ= 0.200, 95% CI 0.100–0.296, p < 0.001, R2 = 0.036) (Supplementary Figure 3d), which is consistent with the findings of a previous study [26].
Reduced plasma LBP levels were associated with increased risk of PD
Multiple logistic regression analysis with adjustment for age, sex, and plasma levels of pro-inflammatory cytokines revealed that reduced plasma LBP levels were associated with increased PD risk (odds ratio 0.816, 95% CI 0.717–0.929, p = 0.002, Table 2). Based on the logistic regression analysis results, we performed ROC curve analysis to predict PD occurrence, with the prediction accuracy expressed as the AUC. The prediction accuracy increased from the basic model (age and sex only, AUC 0.528, 95% CI 0.471–0.585, p = 0.353) to a model with basic characteristics combined with plasma LBP level (AUC 0.626, 95% CI 0.571–0.682, p < 0.01). Prediction accuracy further increased with the full model using basic characteristics combined with the levels of plasma LBP and plasma pro-inflammatory cytokines, including TNF-α, IL-6, and IL-17A (AUC 0.671, 95% CI 0.616–0.726, p < 0.001) (Table 3 and Fig. 2a).
Logistic regression model for risk of PD
CI, confidence interval; LBP, lipopolysaccharide binding protein; TNF-α, tumor necrosis factor-α, IL-6, interleukin 6; IL-17A, interleukin-17A. In this model, presence of Parkinson’s disease was set as dichotomous dependent factor while the independent variables included age (continuous), sex (categorical), plasma le-vels of LBP (continuous) and individual pro-inflammatory cyt-okines (continuous). *p < 0.05; **p < 0.01.
Receiver operating characteristic curve analyses for differentiating between patients with PD and age and sex-matched controls
AUC, area under curve; CI, confidence interval; LBP, lipopolysaccharide binding protein. The independent variables used in this model included continuous variables of age, plasma LBP and proinflammatory cytokines, and categorical variable of sex. Variables in the full model were age, sex, plasma levels of LBP and pro-inflammatory cytokines, including TNF-α, IL-6 and IL-17A. *p < 0.05; **p < 0.01.
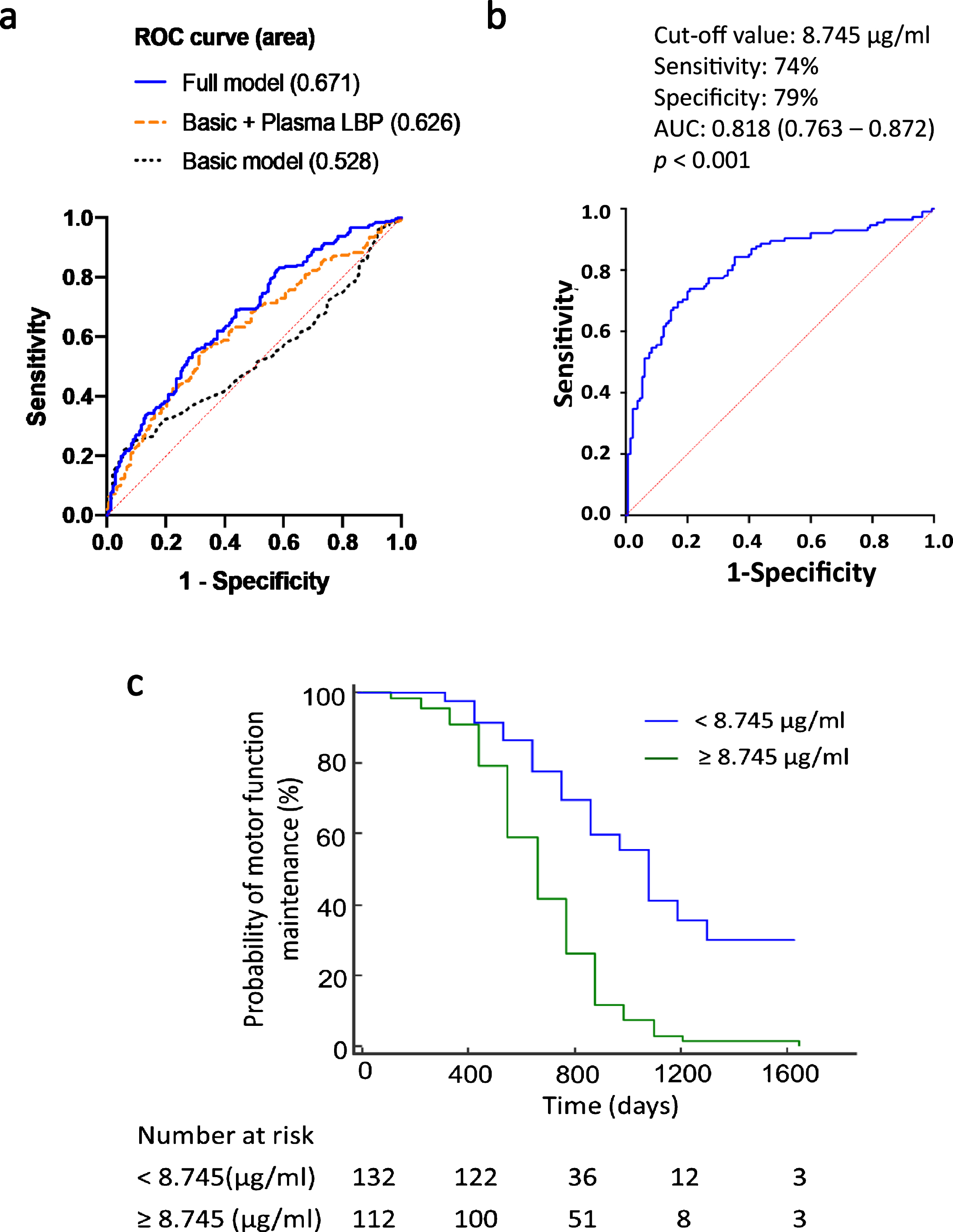
Receiver operating characteristic (ROC) curve analyses for differentiating PD patients from healthy controls, and between PD patients with and without motor symptom progression. (a) The diagnostic accuracy of a model including age and sex alone [area under the curve (AUC) 0.5280 was significantly improved after the addition of plasma LBP and pro-inflammatory cytokines (TNF-α, IL-6, and IL-17A) (AUC 0.671, p < 0.001). (b) ROC curve analysis for differentiating between PD patients with and without motor progression based on LBP level. (c) Kaplan-Meier curve reveals different motor progression outcomes in patients with PD who had plasma LBP levels at or above versus below the cut-off value calculated based on ROC curve analysis.
Correlations between plasma LBP levels and severity of motor and cognitive symptoms
We next examined the correlations between pla-sma LBP levels and disease severity, in terms of either motor or cognitive function. We found that early-stage PD patients had reduced systemic LBP levels compared to controls (8.81±2.73 vs. 10.10±3.00μg/ml, p < 0.001, Fig. 1b). However, among PD patients, plasma LBP levels were higher in PD patients with advanced-stage motor symptoms comp-ared to those with early-stage motor symptoms (9.64±3.22 vs. 8.81±2.73μg/ml, p = 0.029, Fig. 1b). Furthermore, among patients with PD, plasma LBP concentrations were correlated with motor symptom severity as measured by MDS-UPDRS part III scores (ρ= 0.341, 95% CI 0.222–0.450, p < 0.001, Fig. 1c). In the multivariate linear regression analysis, the correlation between plasma LBP levels and MDS-UPDRS part III score remained significant after adjustment for age, sex, disease duration, and pro-inflammatory cytokine levels (coefficient = 0.636, p = 0.017, R2 = 0.200, Table 4).
Multivariate linear regression models for motor symptom severity in PD patients as assessed by MDS-UPDRS part III score
In this model, the MDS-UPDRS part III motor score in the on state was set as the dependent variable, with age, gender, disease duration, and plasma level of LBP and pro-inflammatory cytokines set as independent variables. r: correlation coefficient based on the model of multiple linear regression; t: t value for the coefficient of each parameter in the model; p: for r or t; R2-adjusted = 0.1998, p = 0.01 for the model. *p < 0.05; **p < 0.01. MDS-UPDRS, Movement Disorder Society-Unified Parkinson’s disease rating scale.
On the other hand, plasma LBP levels were com-parable between PD patients with normal cognition versus patients with PDD (8.97±2.76 vs. 9.30±2.98μg/ml, p = 0.665, Fig. 1d). Moreover, plasma LBP levels were not correlated with MMSE score (ρ= –0.117, p = 0.161). Multiple linear regression analysis with adjustment for age, sex, disease duration, plasma LBP levels, and pro-inflammatory cytokine levels did not reveal any association between plasma LBP levels and cognitive severity (coefficient = –0.543, p = 0.700, Supplementary Table 1).
Longitudinal follow-up of motor symptom progression
During the mean follow-up period of 4.7±2.3 years, 27 participants (6 patients with PD and 21 control participants) were dropped out or loss of follow-up. Therefore, 370 participants completed the follow-up study, including 242 patients with PD and 128 controls (Supplementary Figure 1). Of these 242 patients with PD, 115 of 242 (47.5%) PD patients exhibited motor symptom progression that manifested as an increase of ≥3 points in the MDS-UPDRS part III score during the follow-up period while compared to the baseline score at enrollment. The multivariate Cox regression model with adjustment for age, sex, disease duration, and baseline levels of plasma pro-inflammatory cytokines revealed that elevated baseline plasma LBP levels were associated with a higher risk of motor symptom progression (adjusted HR 1.084, 95% CI 1.011–1.163, p = 0.024, Table 5). After calculating the sensitivity and specificity of this association, we performed ROC analysis and identified cut-off values of baseline plasma LBP levels based on the highest Youden index, which showed a 74% sensitivity and 79% specificity in discriminating between PD patients with and without motor deterioration (AUC 0.818, 95% CI 0.763–0.872, p < 0.001, Fig. 2b). Among patients with PD, those with baseline plasma LBP levels of ≥8.745μg/ml had a higher risk of motor symptom progression compared to patients with LBP levels of < 8.745μg/ml. A Kaplan-Meier analysis revealed a clear divergence between patients with baseline plasma LBP levels above and below this cut-off value (p < 0.001, log-rank test) (Fig. 2C).
Multivariate Cox regression analysis of plasma LBP levels and motor symptom progression in patients with PD
CI, confidence interval. Hazard ratio represents the risk of motor function deterioration within follow-up period. *p < 0.05; **p < 0.01.
DISCUSSION
Our present study revealed that plasma LBP levels were significantly lower in PD patients compared to those in age- and sex-matched healthy controls. After adjustment for confounding factors, reduced plasma LBP levels were associated with increased PD risk. We further demonstrated that plasma LBP levels displayed biphasic changes within the disease process of PD. Plasma LBP levels were reduced in PD patients during the early stage of motor dysfunction compared to controls. However, even though the concentrations of LBP increased in the advanced stage of motor symptoms, the LBP levels in patients with advanced stage of the disease were still lower than those in controls. Furthermore, among PD patients, plasma LBP levels correlated with motor symptom severity, as measured by MDS-UPDRS part III motor scores, and with plasma levels of pro-inflammatory cytokines. Our longitudinal follow-up analysis of PD patients revealed that higher LBP levels were associated with greater risk of motor deterioration. Overall, our findings indicate that plasma LBP levels reflected PD motor symptom severity and progression, which supports the concept that bacterial endotoxin-mediated inflammation is involved in PD pathogenesis.
LBP could regulate innate immunity by presenting bacterial LPS to cellular surface pattern receptors, thus prompting immune and inflammatory responses. The LPS recognized by LBP would be transferred to membrane-bound TLR4, and subsequently internalized into cells—triggering the downstream NF-kB pathway, activating expression of the NLR family pyrin domain containing 3 (NLRP3) inflammasome, promoting pro-inflammatory cytokine secretion and cell apoptosis [14]. This mechanism is especially important within the gastrointestinal system since intestinal epithelial cells generally express low levels of membrane forms of TLR4 to create a tolerant environment for commensal pathogens [27, 28]. In this setting, TLR4 could be induced expression by internalized LPS and trigger the downstream pathways [27, 28]. Our present results showed that PD patients had reduced systemic LBP levels compared to controls. This might reflect the internalization of LBP and LPS within intestinal cells, which could elicit subsequent intestinal inflammation and intestinal cell apoptosis, leading to impaired intestinal epithelial barrier function and bacterial pathogen invasion, causing systemic inflammatory responses [29]. Indeed, recent studies show that reduced plasma LBP levels in PD patients are correlated with intestinal epithelial barrier integrity and the degree of intestinal leakage [9, 13]. Moreover, evidence indicates that gastrointestinal inflammation triggers cellular α-synuclein aggregation in the enteric nervous system, and that reduced systemic LBP levels in PD patients are inversely correlated to the abundance of intestinal α-synuclein depositions [9, 30]. These observations suggest that lower plasma LBP levels may reflect the impaired intestinal barrier function, increased gut permeability, and aggravated gut innate immunity observed during the initial stage of PD pathogenesis [31].
Our findings further revealed that plasma LBP levels showed a biphasic change during the disease process of PD. Plasma LBP levels were reduced in early-stage PD and then increased in the later stage with advanced motor symptoms, which were still lower than those in control participants. Previous evidence shows that plasma LBP levels are elevated in the state of chronic inflammation and correlated with the levels of pro-inflammatory cytokines [26, 33], the paradoxically increased LBP levels in advanced-stage PD may be partially caused by systemic chronic inflammatory responses induced during the disease process of PD. However, even though the concentrations of LBP increased in the advanced stage of motor symptoms, the LBP levels in patients with advanced stage of the disease were still lower than those in controls. Moreover, the plasma LBP levels were not increased in patients with PDD. We therefore speculate that chronic inflammation-induced increased production of LBP cannot fully explain our findings in advanced-stage of PD patients. Other factors that regulate the production of systemic LBP concentrations, for example, the changes of gut microbiota or increased risk of medical co-morbidities in advanced stage of PD, may interfere the systemic LBP levels. Recent studies have shown that metabolic syndrome, especially hyperlipidemia, and smoking would also increase the concentrations of LBP [26]. A future large cohort study enrolling more PD patients and serially examining the plasma levels of LBP with more detail information of medical co-morbidities are needed to delineate the changes of plasma LBP levels in the disease course of PD.
The gut microbiota shapes and maintains host immunity homeostasis through metabolites, cyt-okines, and hormones [34]. In PD patients, an altered gut microbiota composition (increased Lactobacillaceae and reduced Prevotellaceae) modulates T-cell differentiation toward T helper 1 (Th1) cells and reduces the production of IL-17-producing T helper (Th17) cells, which are an important intestinal mucosal protector against invading pathogens [34–36]. Upon interaction with bacteria, Th17 cells in the intestinal lamina propria and Paneth cells produce IL-17A, which promotes anti-microbial peptide secretion and neutrophil chemotaxis [34, 37]. Inhibition of IL-17A leads to increased intestinal inflammation, impaired epithelial barrier function, and systemic bacterial invasion [37]. Our present results showed decreased circulating IL-17A in PD patients, which corresponded to the plasma LBP concentrations. In early PD, gut microbial dysbiosis may lead to altered intestinal immunity and reduced IL-17A expression. In turn, this may cause impaired intestinal clearance of invading pathogens, triggering intestinal inflammation, increased local infiltration of endotoxin, and intracellular internalization of LBP along with LPS that further exacerbates gut inflammation.
Although we found that plasma LBP levels correlated with motor symptom severity and progression, there was no significant association between LBP levels and cognitive decline in our PD population. PDD is a heterogenous clinicopathological syndrome, and post-mortem studies of PDD patients have shown formations of amyloid plaque and neurofibrillary tangles in addition to Lewy bodies [38]. While some evidence indicates that endotoxin-mediated inflammation is related to a-synucleinopathy in PD, the mixed neuropathology in PDD may partly explain why LBP levels were not correlated with cognitive decline in our cohort of PD patients. Some studies show that the abundance of LPS-producing bacteria (e.g., Bacteroides) is correlated to motor symptom severity [8, 40]; however, an entirely different non-overlapping spectrum of gut microbiome is reportedly increased in PD patients with cognitive decline [39]. Overall, the finding that plasma LBP level was not associated with cognitive symptom severity in our study may suggest that the progressions of motor versus cognitive symptoms in PD involve different triggering mechanisms or propagation trajectories.
Our study has several limitations. First, we did not simultaneously analyze the gut microbiota structure and, therefore, could not verify the association between plasma LBP levels and altered gut microbiota in our study cohort. However, gut microbial dysbiosis appears to be a reasonable explanation for elevated plasma LBP levels in PD since LBP is an acute phase protein mainly triggered by LPS, and because both increased LPS-producing microbiota and plasma LBP concentrations reportedly enhance the levels of pro-inflammatory cytokines and motor symptom severity [8, 29]. Future studies combining shotgun metagenomic sequencing, and quantification of plasma LBP and other endotoxin-associated inflammatory markers, may provide a better understanding of the altered gut microbiome and host immune responses in PD pathogenesis. Second, we did not adjust for body mass index, and obesity is reportedly associated with a higher plasma LBP level. Third, the blood sampling time of each participant was not recorded at enrollment but all participants had blood drawn at the end of the clinic hours, which were usually in the afternoon. As there is a diurnal rhythmicity of pro-inflammatory cytokine production which peaked in the early morning due to the rhythm of plasma cortisol [40], whether plasma levels of LBP showed similar rhythmicity with pro-inflammatory cytokine changes needs future exploration. Finally, we assessed cognitive function only using MMSE, which is a simple measurement of global cognitive function. The use of detailed neuropsychological tests to evaluate individual cognitive domains is warranted to further assess the correlations between plasma LBP levels and declines in individual cognitive domains among patients with PD.
In conclusion, our results showed the biphasic alteration of circulating LBP levels related to PD risk, and motor symptom severity and progression in PD. Our findings support that altered gut bacterial endotoxin-mediated inflammatory responses in the gut-brain axis of PD. A future large cohort study is needed to confirm our findings.
Footnotes
ACKNOWLEDGMENTS
The authors are grateful to the patients who participated in this study. We are grateful for the funding supports by grants from National Health Research Institutes (NHRI-EX110-10716NC), Ministry of Science and Technology (MOST 109-2320-B-002-073 –), National Taiwan University Hospital (NTUH 109-T18 and 109-EDN02) and National Taiwan University Hospital Bei-Hu Branch (10904). We also thank the staff of the Second Core Lab, Department of Medical Research, National Taiwan University Hospital, for technical support during the study.
CONFLICT OF INTEREST
All authors report no conflict of interests.
DATA AVAILABILITY
All the data are available from the corresponding author on reasonable request.
