Abstract
Background:
Neurofilament light (NfL) can reflect the extent of neuron/axon damage, thus providing an opportunity to examine the severity and progression of the diseases with such damage.
Objective:
Whether serum NfL can be used as an indicator to monitor the cognitive progress of de novo Parkinson’s disease (PD) remains unclear.
Methods:
In this research, 144 healthy controls and 301 de novo PD patients from Parkinson’s Progression Markers Initiative (PPMI) were recruited. Linear mixed effects models were used to examine the associations of baseline/longitudinal serum NfL with cognitive decline. Cox regression was used to detect cognitive progression in PD participants.
Results:
We found PD patients had higher serum NfL than controls at baseline (
Conclusion:
In conclusion, our results indicated that the serum NfL concentration could function as an easily accessible biomarker to monitor the severity and progression of cognitive decline in PD.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a neurodegenerative disease with complex clinical, genetic, and molecular characteristics [1]. Although PD is commonly regarded as a motor disorder, around 25% of patients actually meet the criteria for mild cognitive impairment (PD-MCI) [2, 3] and up to 80% of patients will be diagnosed as dementia (PD-D) [1]. Therefore, identifying a useful biomarker that may predict changes in cognition associated with PD would be an essential means to assist clinical management and results evaluation in clinical trials [4].
Neuroaxonal degeneration remains the pathological mechanism that causes permanent disability in neurological disorders [5]. Reliable quantification of axonal injury is essential for both disease monitoring and prognostication. Neurofilament light (NfL), a cytoskeletal scaffolding protein found exclusively in neurons, is released to cerebrospinal fluid (CSF), interstitial fluid, and serum as a consequence of axonal injury and neurodegeneration [6]. Recently, studies have advanced NfL measurements and discovered a close correlation between NfL in the CSF and serum, which has aroused attention to NfL blood-based biomarker for monitoring disease progression and neurodegeneration [7]. Serum NfL levels are reported to be elevated in PD and significantly increase over time in PD, which is correlated in turn with clinical features of disease progression [8]. Furthermore, NfL concentration is higher in PD-D than in cognitively normal PD patients [9]. As a potential indicator of disease severity or prognosis, its dynamic analysis in blood sample might provide the possibility to detect and measure the accumulation of pathological products in surviving patients over time. However, longitudinal data is rare and comprehensive longitudinal models of longitudinal changes in values are deficient in previous studies.
We hypothesize that the blood derived NfL and the accumulation of NfL during the disease may be a potential biomarker to predict the subsequent cognitive progression of PD. Hence by means of the rich Parkinson’s Progression Markers Initiative (PPMI) dataset with standardized longitudinal data, we assessed the baseline and longitudinal progression of NfL in PD and evaluated the relationship of NfL with CSF biomarkers and clinical cognitive characteristics. The study population was limited to newly diagnosed and untreated PD patients because there is a rising consensus that recruiting individuals at the earliest detectable stage of PD may enhance the potential for identifying progressive biomarkers and provide a better population for the final disease-modified drug trials [10].
MATERIALS AND METHODS
Participants
Data were downloaded from the PPMI database (http://www.ppmi-info.org). The PPMI is an ongoing, observational, longitudinal, prospective, international multicenter study that intends to identify biomarkers for the progression of PD. At enrollment, de novo PD patients must be 30 years or older at the time of diagnosis; have two symptoms among bradykinesia, resting tremor, and rigidity or only asymmetric resting tremor or bradykinesia; within two years after diagnosis; be not treated for PD; absence of treatment; no dementia that determined by examiners; be untreated with medication that might interfere with dopamine transporter imaging or CSF collection. To prevent misdiagnosis, the researchers conducted a longitudinal review of the diagnosis. If PSP or MSA is suspected, the patient is excluded from follow-up. Healthy controls (HC) could be included if: no obvious neurologic impairment; all first-degree relatives without PD; and their Montreal Cognitive Assessment (MoCA) score higher than 26. Participants in this study were regularly evaluated to collect clinical data and to participate in its biomarker studies, including the collection of CSF. Supplementary Table 1 show the clinical and biomarker information of the samples during the follow-up. Data for our analysis was limited to participants that had no missing baseline NfL and had at least one additional monitoring NfL at year one or later. After baseline examinations, serum NfL levels were measured at baseline (BL), 6months, and 12, 24, 36, and 60 months, and a total of 2174 measurements were performed. Longitudinal follow-up including three annual CSF biomarker tests and eight annual clinical assessments after the first lumbar puncture (first NfL measurement).
Analysis of serum NfL
Serum NfL concentrations were measured with the Simoa Human NF-light Advantage kit using a fully automated SIMOA® HD-1 analyzer based on a Single-Molecule Array technology (Quanterix, Lexington, MA, USA) [11]. Quantitative measurements of NfL in blood by Simoa technology have been shown to be reliable [12]. More detailed information on the handling of samples in this cohort can be read in the biologic’s manual for PPMI [13].
Analysis of CSF biomarkers
The methods and analyses for CSF biomarkers have been previously described in detail (details can be read in the biologic’s manual for PPMI) [13]. The electrochemiluminescence (ECL) method was used to measure CSF amyloid-β42 (Aβ42), total tau (T-tau), and phosphorylated tau (P-tau). As mentioned earlier, ECL immunoassays were performed on a completely automated cobas e 601 analyzers (Roche Diagnostics) [14]. Additionally, CSF total
Clinical assessment measures
Clinical results for each clinical visit have been formerly reported [13]. We selected continuous cognitive function measurement indicators in several specific areas, including global functioning (MoCA), episodic memory (Hopkins Verbal Learning Test Total Recall; HVLT Total Recall, HVLT Delayed Recall, HVLT Retention, HVLT Recognition Discrimination), visuospatial functioning (Benton Judgment of Line Orientation Score; JOLO), executive function/working memory (Letter Number Sequencing; LNS), language (Semantic Fluency Test) and processing speed/attention (Symbol Digit Modality Test; SDMT) [13].
The MoCA is the most commonly used cognitive measurement in PD that owns an elevated specificity and sensitivity for distinguishing MCI in PD [16]. MoCA is at least equivalent to the PD-focused Scales for Outcomes in PD–Cognition (SCOPA-COG), and superior to the standardized MMSE (S-MMSE). The advantages of using MoCA in large multicenter PD studies include it assesses primary cognitive domains at risk in PD, has been validated in multiple languages and requires substantially less time and training to properly administer and score than a full neuropsychological assessment [17]. In the present study, cognitive status was defined based on the criteria of the MDS level I guideline: PD with normal cognition (PD-NC) if MoCA scored > 26, PD-MCI if scored between 26 and 22, and PD-D if the MoCA was < 22 [18, 19].
Statistical analysis
Serum NfL levels (Kolmogorov-Smirnov test,
Association of baseline serum NfL with CSF biomarkers and cognitive data were evaluated with a linear regression model. The linear mixed-effects (LME) model was used for modeling the rate of change in the serum NfL, which included random slope and intercepts terms. We extracted the rate of change in the NfL for each individual from the model for subsequent analyses. LME models tested associations of baseline NfL levels or rate of changes with longitudinal data on other measurements. We evaluated models with tertile and continuous variables. Although only 6.5% of PD patients had abnormal levels of Aβ42 and tau in the CSF at the time of initial diagnosis [20], 60% to 80% of PD patients demonstrated the pathology of Alzheimer’s disease (AD) at autopsy change [21]. We, therefore, characterized the AD-related CSF profiles of PD and controls by applying a cut off point for amyloid positivity recognized in AD [14]. To alleviate distinctness in pre-analytical elements between PPMI and AD cohorts that influence CSF Aβ42 levels [22], we used the transformation formula [23] to convert Elecsys values to tantamount values of AlzBio3 [
Kaplan-Meier curves were then used to compare the cumulative probability risk of cognition progression during the follow-up among different groups. Besides, a multivariate Cox regression model was used to analysis the association between NfL levels and the occurrence of cognitive progression during the follow-up.
All statistical analyses were performed using the R 3.5.1. All regression analyses were corrected for age, gender, educational level, Apolipoprotein E (
RESULTS
Study participants
Table 1 listed the demographic information of the study population. We showed that the clinically defined diagnostic groups in this study consisted of de novo PD patients (
Clinical characteristics and serum NfL levels of participants in individual groups in the current study
Categorical variables are reported as numbers and percentages; continuous variables are reported as means±SDs.NfL, Neurofilament light; HC, Healthy control; PD, Parkinson’s disease; F, Female; M, Male;
Cross-sectional serum NfL analyses
Baseline NfL level was higher in the de novo PD group (13.00 pg/mL) than controls (11.71 pg/mL) (
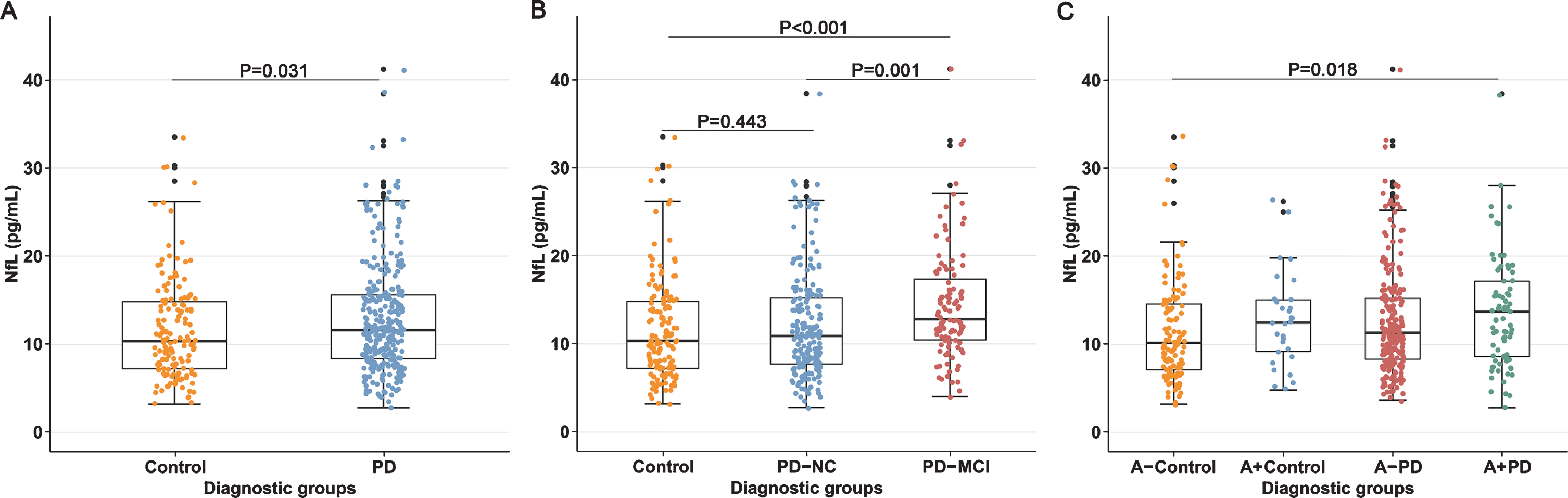
Baseline serum NfL levels of controls and patients with
Following consideration and adjustment of age, gender, educational level,
Prediction of longitudinal changes of CSF biomarkers and cognitive decline using baseline serum NfL
We conducted an exploratory analysis based on previous work on biomarkers [7, 26] to analyze the predictive value of serum biomarker in PD cognition. Hypothetically, up regulation of NfL will be associated with a decline in overall cognitive ability. In de novo PD patients, a higher baseline serum NfL level was significantly associated with a higher increased rate of CSF T-tau (β= 0.014,
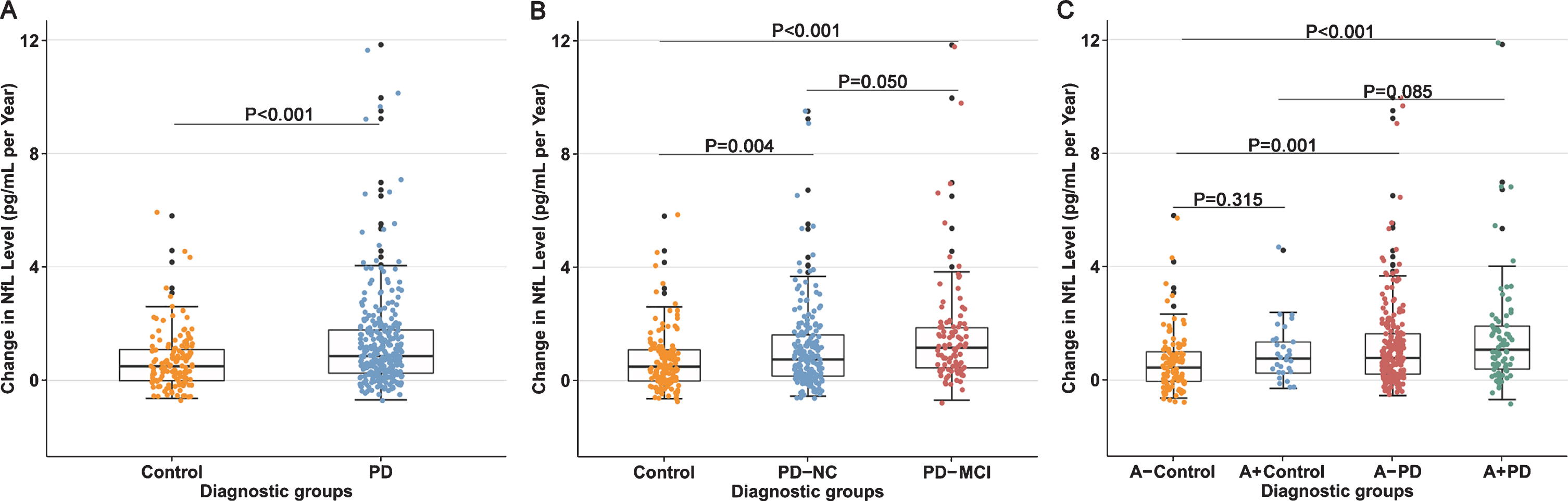
Longitudinal changes in serum NfL of controls and patients with
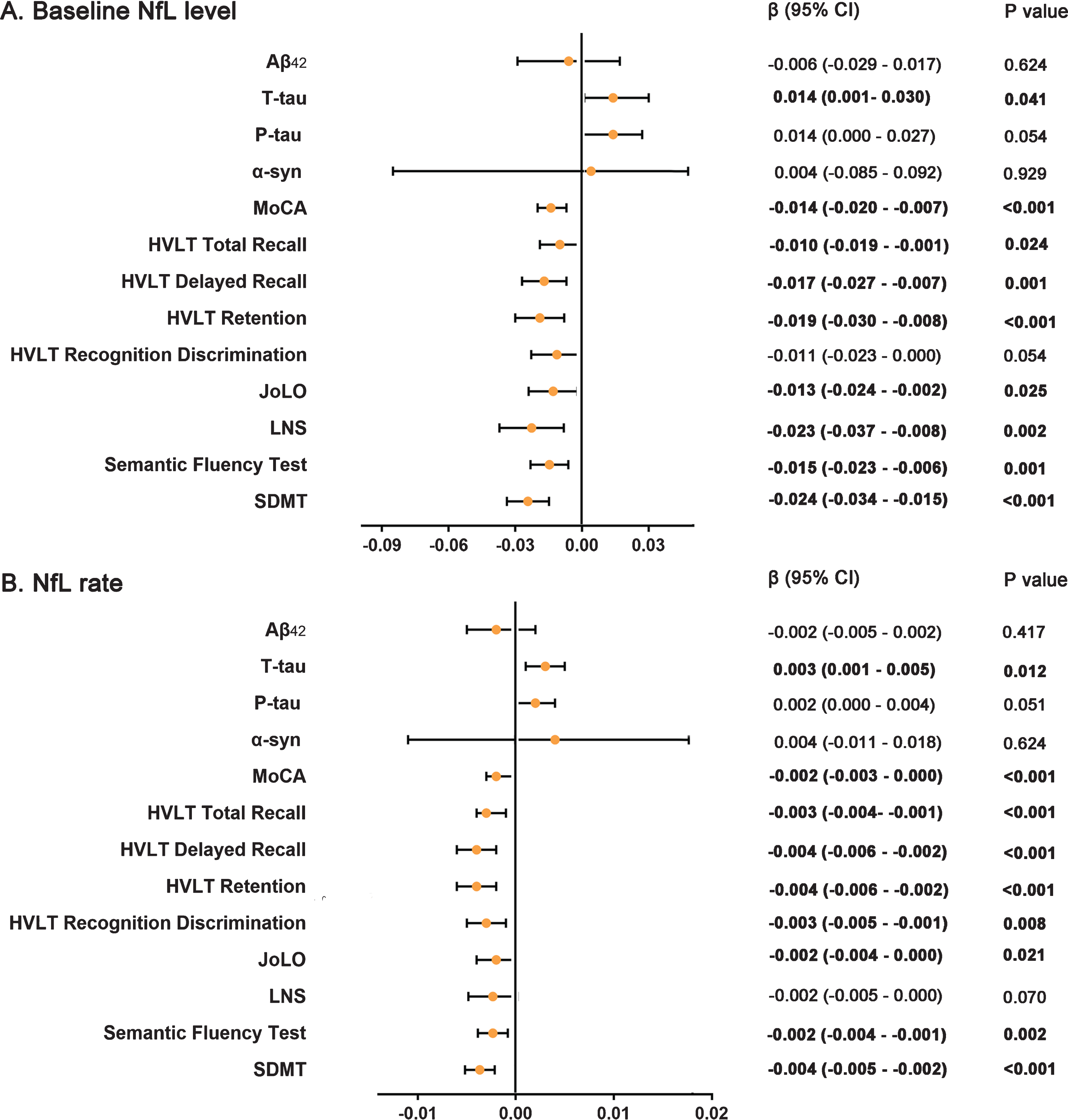
Effects of baseline NfL (A) and NfL accumulation rate (B) on biomarkers and cognition measurements in linear mixed-effects analysis among PD participants during follow-up. NfL, Neurofilament light; PD, Parkinson’s disease; Aβ42, Amyloid-β42; T-tau, Total tau; P-tau, Phosphorylated tau.
Figure 4A exhibits the results of a Kaplan-Meier analysis and the log-rank test. After a mean follow-up period of 6.37±1.84 years, 49 of 301 patients (16.28%) with PD displayed a sustained decline of MoCA score. Cox proportional-hazards models were developed to estimate the conversion risk from non-dementia to PD-D condition (Supplementary Table 4). PD individuals with the highest tertile add an increased risk of conversion to the PD-D compared with the lowest tertile (HR = 6.33, 95% CI: 2.62–15.29,
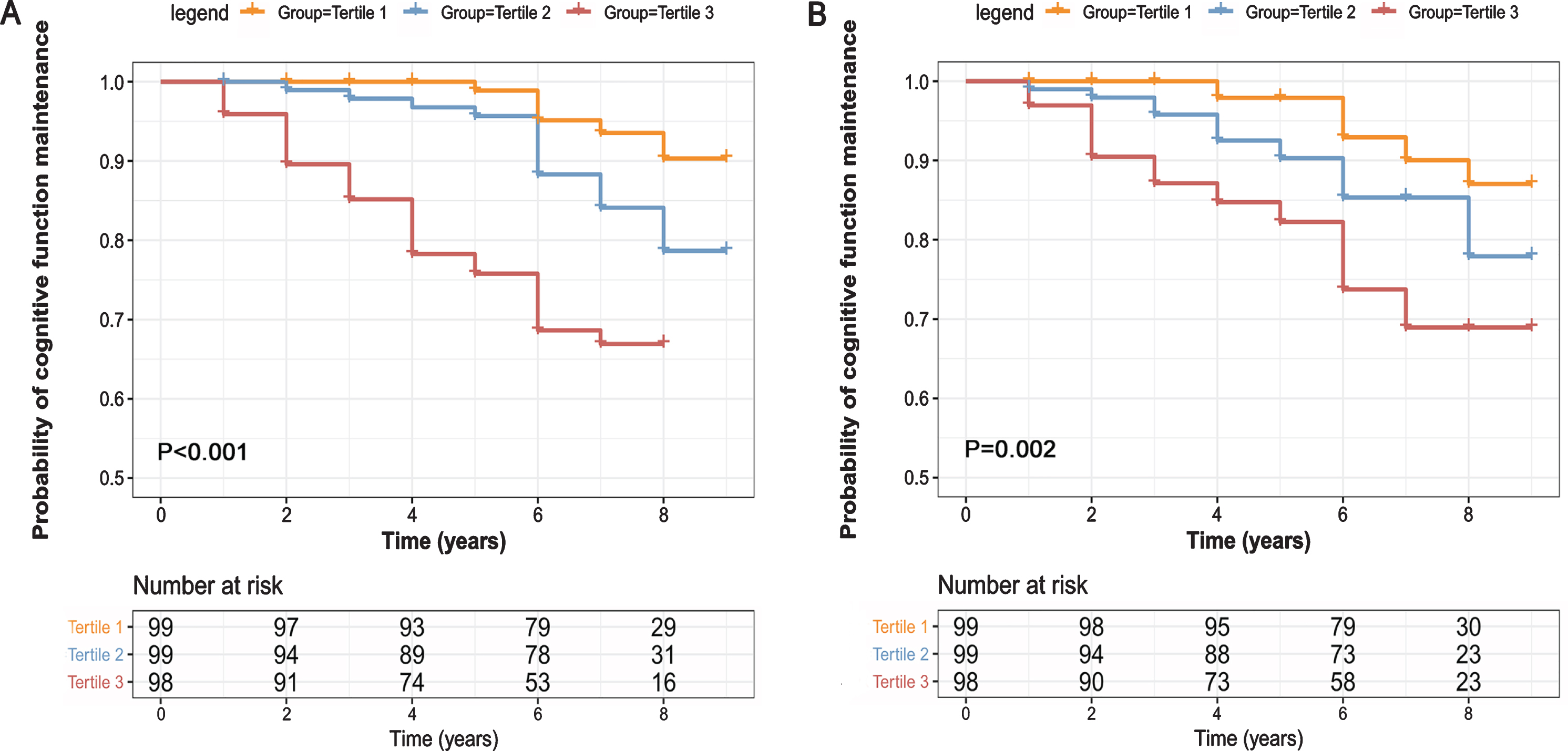
Kaplan-Meier curve for conversion from
Longitudinal change in serum NfL
To further investigate the profile of serum NfL longitudinally in PD and HC, we used LME models to test the average changes from baseline. The NfL rate remained slightly above zero. Serum NfL concentration increased significantly in all groups, with higher rates among de novo PD (1.42 pg/mL per year), PD-NC (1.27 pg/mL per year), and PD-MCI (1.71 pg/mL per year) patients than controls (0.69 pg/mL per year) (
Prediction of longitudinal changes of CSF biomarkers and cognitive decline using longitudinal serum NfL
Like the results shown in the analyses of baseline NfL, a longitudinal five-year increase in NfL levels was also significantly associated with a higher increased rate of CSF T-tau (β= 0.003,
Although we did not see a correlation between serum NfL and the risk of cognitive transformation in the results of Cox proportional-hazards models (Supplementary Table 4), the results of a Kaplan-Meier analysis and the log rank test suggest that there were still some differences between the different groups of the tertile (
Subgroup analyses
We then examined the relationship between serum NfL levels and various levels of cognitive ability among patients with PD by clinical diagnosis and Aβ status. We found that the predictive effect of baseline serum NfL levels and NfL rate on cognitive decline was reflected in global cognition, episodic memory, and processing speed/attention in the PD-NC population (Supplementary Table 5). In the PD-MCI population, this predictive effect seemed to be more obvious and reflected in more cognitive domains. More significant positive associations of NfL rates between CSF T-tau (β= 0.006,
DISCUSSION
The largest difference between our study and previous studies is that this longitudinal study allows us to observe the longitudinal changes of NfL through follow-up from the early stages of the disease. As expected, the PD-MCI group was apparently higher than the control group in terms of baseline level and longitudinal change rate [6, 8]. It is worth noting that although there was no difference in NfL levels between the control group and the PD-NC group at baseline, the rate of changes in serum NfL concentration in the PD-NC was significantly higher than that in the control group. This confirms that the detectable increase in NfL levels does not seem to be an early thing [27]. However, even without obvious cognitive decline or early motor symptoms, axon damage and neurodegenerative changes have begun to appear. Given that NfL is a well-established biomarker of neuroaxonal loss [6], we were able to provide evidence for the assumption that severe axon loss already occurs even in the early stage of PD [28, 29].
Importantly, a critical issue in clinical practice and clinical trials is the ability of predicting the time frame and time of disease progression. In this context we provided evidence whether biofluid biomarkers such as NfL might be helpful [27]. The results showed that over average 6.37 years follow-up, higher baseline NfL levels and higher increasing rate could predict more rapid individual cognitive progression among de novo PD patients. However, our study did not find a significant cross-sectional association of serum NfL with cognitive indicators. This result contradicted a previous study [8]. This may be due to the population in our study being at an earlier stage of the disease. Nevertheless, these data indicate that serum NfL profiles might have prognostic values for predicting the generally incipient cognitive decline in PD.
A large amount of biological heterogeneity is the basis of clinical manifestations and progression of PD [4]. In our distinctive analysis applying an reputable AD cut point for CSF Aβ42, we found around one-third of PD patients had pathologically-low CSF Aβ42 (A + PD), which is consistent with previous studies [24] and autopsy information in end-stage PD [30]. Moreover, these patients had, on average, higher NfL concentration and higher longitudinal growth rate of NfL compared with A- control group, suggesting that the distribution of serum NfL in PD may be consistent with aging and AD [31].
The etiology of cognitive decline in PD is complex and may involve multiple pathological mechanisms [32]. Longitudinal changes in serum NfL and other biomarkers in PD have not been systematically investigated in other cohorts. In a previous postmortem study, NfL protein was identified in senile plaque components in the hippocampus of patients with PD-D but not in patients with PD who had normal cognition [33]. That finding suggested that NfL might be involved in the pathological process of cognitive decline in PD. T-tau in the CSF is thought might correlating with cognitive decline in PD [34, 35]. Interestingly, our correlations found similar associations of CSF T-tau with majority of cognitive impairment, which further signifying that these biomarkers might in part share fundamental processes in PD. Another explanation for this is that NfL reflects neurodegenerative changes that occur independently of Aβ pathology. This result is similar to the results of brain damage caused by other neurodegenerative diseases [36, 37] and other causes [38, 39]. In general, blood based NfL concentrations seem to reflect the intensity of neuronal damage. Further research is necessary to clarify the involving biological mechanisms.
The main advantage of our study was that we used both cross-sectional and prospective follow-up study designs. Although the increase in NfL levels is not specific to PD, the findings in this study are relevant to understanding the progression of PD and emphasize their usefulness as a marker in clinical trials. Importantly, the average course of all patients at the time of study enrollment was 6.9 months, comparing to the average course of 2 to 9 years in other studies [8, 29]. In addition, validated serum or plasma biomarkers for pathophysiology in the central nervous system are rare, and most of the molecular biomarkers in neurodegenerative disease require CSF sampling, which are invasive and might not be achievable in all patients [40]. A few cognitive tests have previously been shown to predict conversion to dementia in PD. In future studies, NFL can be used as one of the indicators to establish a more reliable forecasting model. However, our study had some limitations. Firstly, there are some missing values in this longitudinal cohort, particularly at subsequent follow-up time points, which exerts a subtle influence on the results. Second, reasonable cut off values to classify CSF tau were lacking. For Aβ, we utilized CSF Aβ42 levels. It may also be useful to assess Aβ-PET imaging in future studies.
In summary, serum NfL level and rate of change in serum NfL were abnormally elevated in the de novo PD. Serum NfL predicted longitudinal cognitive progression of PD patients. And serum NfL was associated with CSF T-tau levels. The current findings suggest that the serum NfL might act as a very early cognitively prognostic marker in PD. A dynamic blood NfL measurements might assist clinicians and researchers in identifying patients with PD at risk of rapid cognitive decline and may have the potential for monitoring responses to therapy in future clinical trials.
Footnotes
ACKNOWLEDGMENTS
This study was supported by grants from the Shanghai Municipal Science and Technology Major Project (No.2018SHZDZX01) and ZJLab, Tianqiao and Chrissy Chen Institute, and the State Key Laboratory of Neurobiology and Frontiers Center for Brain Science of Ministry of Education, Fudan University.
Parkinson’s Progression Markers Initiative (a public–private partnership) is funded by the Michael J Fox Foundation for Parkinson’s Research and funding partners, including AbbVie, Allergan, Avid Radiopharmaceuticals, Biogen, BioLegend, Bristol-Myers Squibb, Celgene, Denali, GE Healthcare, Genentech, GlaxoSmithKline, Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi Genzyme, Servier, Takeda, Teva, UCB, Verily, Voyager Therapeutics, and Golub Capital. Data used in the preparation of this article were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (www.ppmi-info.org/data). For up-to-date information on the study, visit ![]() .
.
CONFLICT OF INTEREST
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. None of the authors of this article reported any financial disclosures.
