Abstract
Background:
The concept of motor reserve explains the individual differences in motor deficits despite similar degrees of nigrostriatal dopamine depletion in Parkinson’s disease (PD).
Objective:
To investigate glucocerebrosidase (GBA) variants as potential determinants of motor reserve for exploratory purposes.
Methods:
A total of 408 patients with drug-naïve PD were enrolled from the Parkinson’s Progression Markers Initiative cohort database. All patients underwent SPECT dopamine transporter (DAT) scans and had results for Sanger sequencing of
Results:
Fifty-four (13.2%) patients carried
Conclusion:
The results of this study demonstrated the detrimental effect of
Keywords
INTRODUCTION
The concept of reserve was introduced to explain the individual variability in clinical manifestations despite the similar degree of pathological brain changes in neurodegenerative disorders [1]. The most representative form of reserve is the cognitive reserve in Alzheimer’s disease (AD) [2], while the concept of motor reserve in Parkinson’s disease (PD) is also increasingly accepted to explain the mismatch between parkinsonian motor deficits and the degree of nigrostriatal dopamine depletion in individuals with PD [3]. Our previous works demonstrated that educational experience [4], premorbid exercise engagement [5], and dominant-side laterality [6] could enhance the motor reserve in patients with newly diagnosed PD, providing resilience in the face of substantial PD-related pathology [7]. We also identified the functional brain network associated with motor reserve, comprising the basal ganglia, hippocampus, amygdala, inferior frontal cortex, insula, and cerebellar vermis [8]. As the clinical heterogeneity of PD may reflect different genetic backgrounds [9], some genetic variants could determine the motor reserve in individuals with PD. While some reports have suggested genetic factors related to cognitive reserve or resilience to AD-related pathology [10, 11], few studies have investigated the link between genetic factors and motor reserve in PD populations.
Mutations in the glucocerebrosidase (GBA) gene are one of the most common genetic risk factors for PD. Although the exact mechanisms underlying the increased risk of PD in patients with
METHODS
Subjects
We used the database from the Parkinson’s Progression Markers Initiative (PPMI) cohort, an observational, international study cohort designed to identify clinical, imaging, genetic, and biospecimen PD progression markers to accelerate disease-modifying therapeutic trials [16]. We downloaded clinical, imaging, and genetic data of newly diagnosed drug-naïve patients with PD from the PPMI database in April 2021. A total of 408 drug-naïve and untreated patients with PD, who underwent dopamine transporter (DAT) imaging using 123I-Ioflupane single-photon emission computed tomography (SPECT) with results for Sanger sequencing of
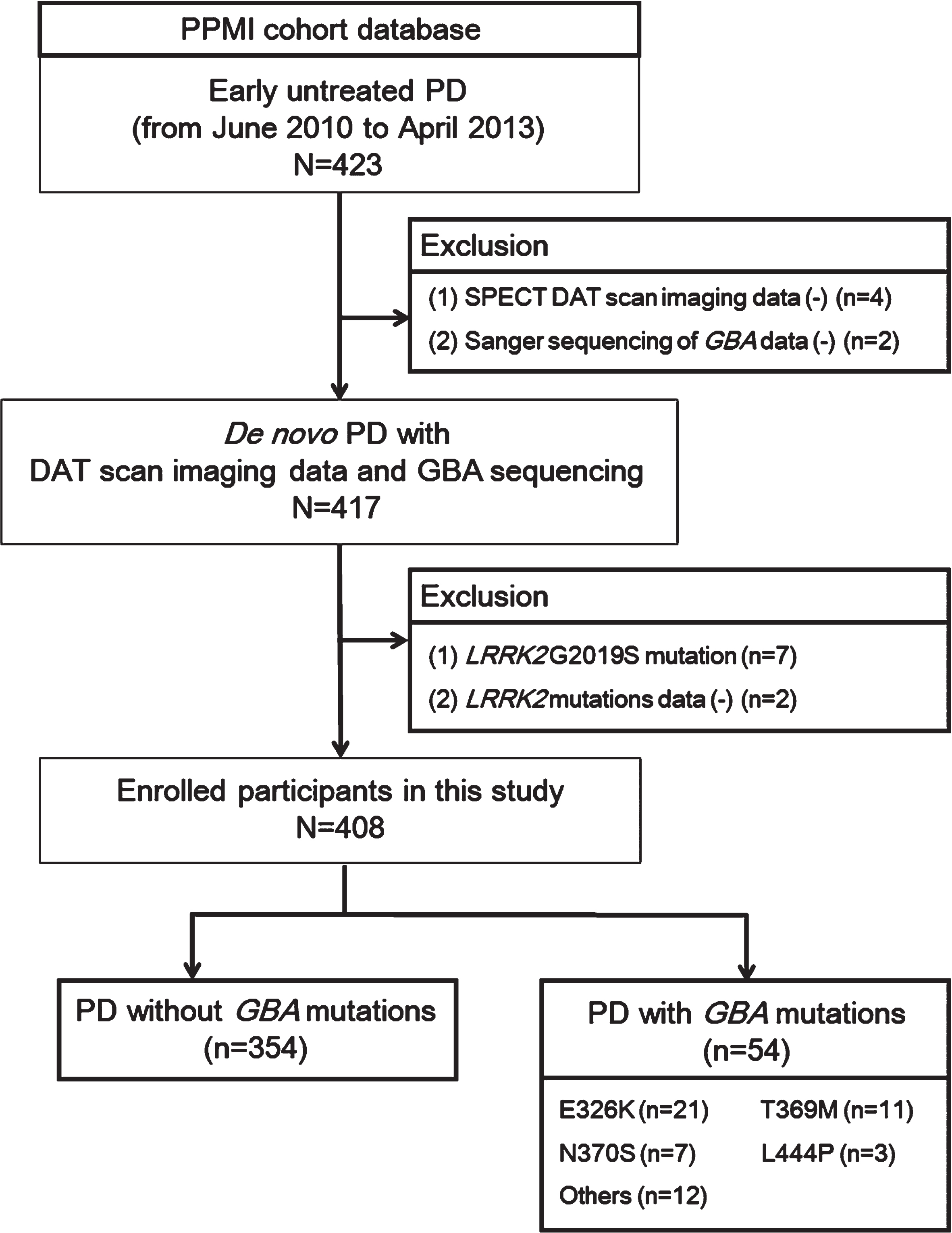
A flowchart of participants and enrollment.
Standard protocol approvals, registration, and consent
The study was approved by the Institutional Review Boards of each participating PPMI site. Written informed consent was obtained from all participants. The study is registered at http://www.clinicaltrials.gov (identifier: NCT01141023).
Quantitative analyses of SPECT DAT scan imaging data
SPECT DAT scan imaging data were acquired at PPMI imaging centers per the PPMI imaging protocol and sent to the Institute for Neurodegenerative Disorders for processing and calculation of striatal binding ratios (SBRs) [19]. The regions of interest were placed on the left and right caudate, left and right putamen, and occipital cortex (reference region). The SBRs for each of the four striatal regions were then calculated as (target region/reference region) –1. The mean of SBRs in the bilateral putamen was also calculated to estimate the motor reserve as DAT availability in this region is closely associated with the severity of PD motor symptoms [20].
GBA genetic testing in the PPMI
Sanger sequencing was performed on whole-blood extracted DNA samples collected according to the PPMI Research Biomarkers Laboratory Manual [18]. Exons 1–11 within
Statistical analyses
The baseline demographic characteristics and striatal DAT availability were compared between the PD groups according to the presence of
Additionally, the motor reserve of each patient was estimated using the residual-based approach, as described in our previous works [8, 21]. In detail, we used the general linear model to predict the MDS-UPDRS-III score by using age, sex, disease duration, and the natural logarithm of SBR in the putamen (Supplementary Table 1). Then, the residuals (i.e., differences between the actual and predicted MDS-UPDRS-III scores) in the general linear model were calculated and standardized. A greater standardized residual indicated that the subject had higher MDS-UPDRS-III score than predicted (i.e., lower motor reserve). We defined the “motor reserve estimate” of each patient as a negative value of standardized residual in the general linear model, with high values indicating high motor reserve. A similar approach has been applied in some previous studies [22, 23], where the residual model was used to define the cognitive reserve in AD populations. The proportions of patients with low motor reserve estimates and high motor reserve estimates (cut-off, 0) were compared between the PD groups using Pearson’s χ2 tests. The same analyses were applied to the MDS-UPDRS-III sub-scores of the more affected and less affected sides. IBM SPSS Statistics for Windows, version 25.0 (IBM Corp., Armonk, NY), and R software (version 4.0.3, http://www.r-project.org/) were used to perform all statistical analyses.
All data are available in the PPMI repository (http://www.ppmi-info.org).
RESULTS
Baseline demographic and clinical characteristics of patients with PD
Fifty-four (13.2%) of 408 patients carried
Demographic characteristics and striatal DAT availability of patients with PD
Values are expressed as the mean±standard deviation, estimated mean (standard error), or number (percentage). DAT, dopamine transporter; PD, Parkinosn’s disease; GBA(–), PD group without β-glucocerebrosidase (GBA) gene mutations; GBA(+), PD group with GBA gene mutations; MDS-UPDRS-III, Movement Disorders Society Unified Parkinson’s Disease Rating Scale Part III; MoCA, Montreal Cognitive Assessment; Benton’s JLO, Benton’s Judgement of Line Orientation; HVLT-R, Hopkins Verbal Learning Test-Revised. aMDS-UPDRS-III scores were compared between the PD groups using an analysis of covariance while adjusting for age, sex, disease duration, and the natural logarithm of DAT availability in the putamen as covariates. bOne patient for each variant including G115R/G193E, I489L, IVS2 + 1G > A, K(–27)R, N370S/N370S, R44C, R463C, and T369M/R120W.
Effects of glucocerebrosidase gene mutations on the MDS-UPDRS-III scores
MDS-UPDRS-III, Movement Disorders Society Unified Parkinson’s Disease Rating Scale Part III; DAT, dopamine transporter; GBA, β-glucocerebrosidase gene; SE, standard error. aSince the DAT availability in the putamen was not normally distributed, its natural logarithm was used in the general linear model. DAT availability in the more severely affected putamen and less severely affected putamen were used for predicting the MDS-UPDSR-III sub-scores of the more affected side and less affected side, respectively.
Motor reserve according to the presence of GBA mutations
The scatter plots of the natural logarithm of SBR in the putamen (mean, more affected, and less affected sides) and MDS-UPDRS-III scores (total, more affected, and less affected sides) are shown in Fig. 2. For the MDS-UPDRS-III total scores, the prevalence of patients with low motor reserve (motor reserve estimate < 0) or cases above the regression line in the general linear model did not differ between the PD groups according to the presence of
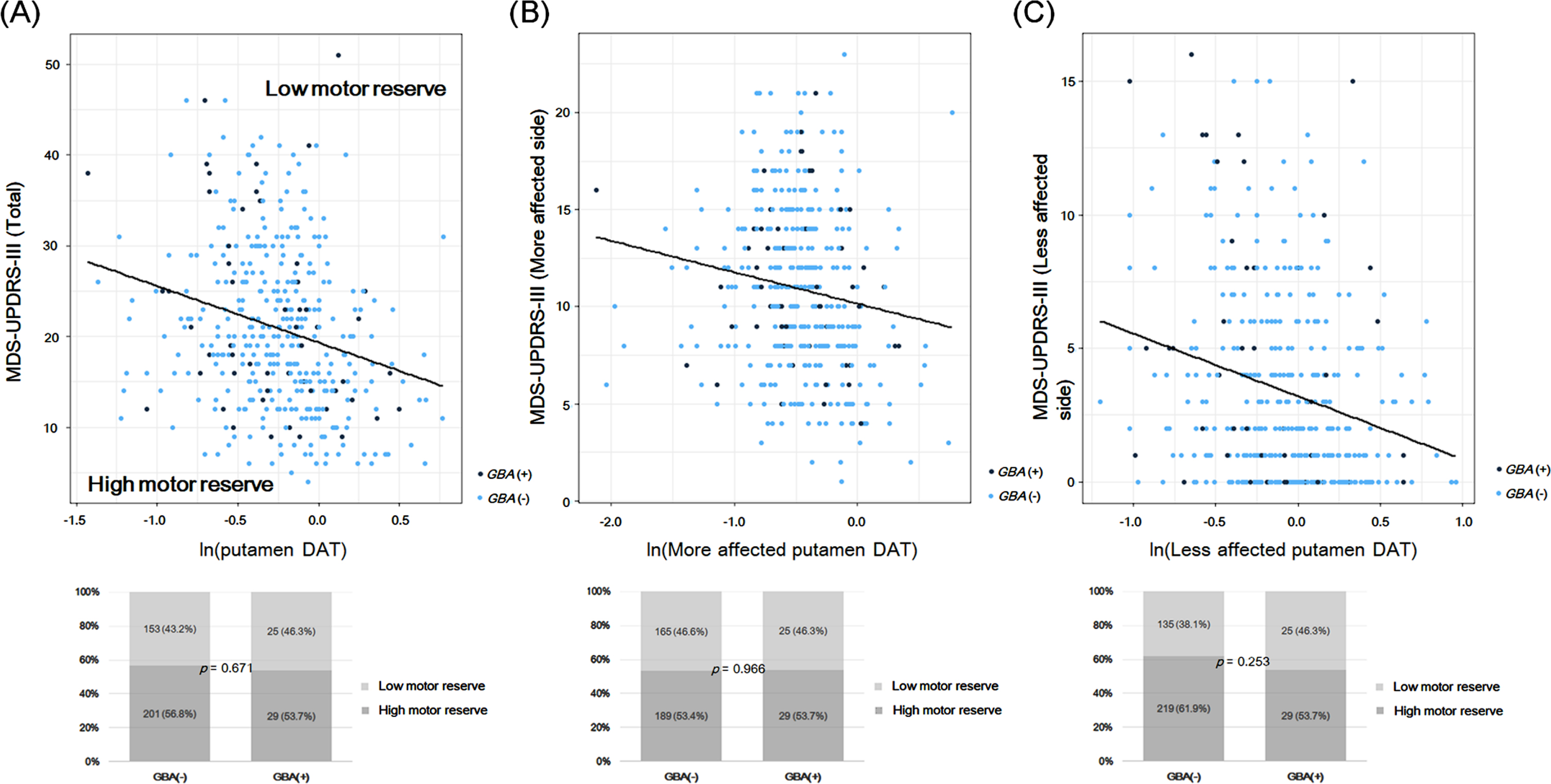
A) Scatter plots of the natural logarithm of SBR in the putamen (mean, more affected side, and less affected side) and MDS-UPDRS-III scores (total, more affected side, and less affected side). The general linear model was used to predict MDS-UPDRS-III scores by using age, sex, disease duration, and the natural logarithm of DAT availability in the putamen. The solid line (black) indicates the regression line of the general linear model. Cases above this line have a higher MDS-UPDRS-III score than the predicted score (i.e., low motor reserve or motor reserve estimate < 0), while cases below this line have a high motor reserve (motor reserve estimate > 0). B) The prevalence of patients with low motor reserve and those with high motor reserve in the PD groups according to
Additionally, we compared the clinical characteristics between the patients with high motor reserve (motor reserve estimate > 0,
DISCUSSION
The present study investigated whether
Motor symptoms in PD do not develop until the nigrostriatal dopamine is depleted by 60–80%[24], suggesting the presence of compensatory mechanisms in early-stage PD to delay the manifestation of motor deficits. This compensatory capacity appears to vary among patients with PD, and the concept of motor reserve has been proposed to explain the individual differences in the susceptibility to PD-related pathologies [3, 21]. We previously reported several factors that can enhance or reduce the motor reserve in patients with PD [3]. In particular, some modifiable factors including education attainment [4, 25–27], physical activity [5], body mass index [28], and white matter hyperintensity [29], could modulate an individual’s capacity to cope with the pathological changes related to PD. Moreover, although genetic factors associated with motor reserve have not yet been reported, the discovery of genetic factors may help in understanding the underlying pathophysiology and establishing therapeutic strategies to enhance the motor reserve.
The strong association between
In terms of motor laterality, the present study showed that patients with PD carrying
Our study has some limitations. First, we used SPECT DAT scan imaging data from the PPMI cohort, from which detailed segmentation of the striatum was difficult due to a low spatial resolution to differentiate the subdivisions of the putamen in to the anterior (i.e., associative striatum) and posterior parts (sensorimotor striatum), which have distinct anatomical and functional connections [47, 48]. Second, DAT availability in the putamen may not be the ideal measure for nigrostriatal dopaminergic degeneration [49]. Furthermore, estimation of motor reserve using the residual model could differ between studies, depending on the dataset. Third, although not statistically significant, PD patients with
In summary, the results of the present study demonstrated the detrimental effects of
Footnotes
ACKNOWLEDGMENTS
Data used in the preparation of this article were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (![]() ). For up-to-date information on the study, visit www.ppmi-info.org. PPMI (a public-private partnership) is funded by the Michal J. Fox Foundation for Parkinson’s Research and multiple funding partners, including AbbVie, Avid, Biogen, Bristol-Myers Squibb, Covance, GE Healthcare, Genetech, GlaxoSmithKline, Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Roche, Servier, Teva, and UCB. This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (grant number: NRF-2021R1I1A1A01059678).
). For up-to-date information on the study, visit www.ppmi-info.org. PPMI (a public-private partnership) is funded by the Michal J. Fox Foundation for Parkinson’s Research and multiple funding partners, including AbbVie, Avid, Biogen, Bristol-Myers Squibb, Covance, GE Healthcare, Genetech, GlaxoSmithKline, Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Roche, Servier, Teva, and UCB. This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (grant number: NRF-2021R1I1A1A01059678).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
