Abstract
Background:
In Parkinson’s disease (PD), sustained aerobic exercise is a promising therapy in delaying motor disability. Brisk walking is a moderate intensity aerobic training, which could be translated to community practice at low cost, but its effects on motor symptoms remains unclear.
Objective:
To determine the effectiveness of a six-month brisk walking and balance program in alleviating motor symptoms, and promoting functional, gait, and balance performance in people with PD.
Methods:
Seventy individuals with mild to moderate PD were randomly assigned to a brisk walking (BW) group or an active control (CON) group. BW group received ten 90-minute supervised brisk walking and balance exercise for six months (weeks 1–6: once/week, weeks 7–26: once/month). CON group received upper limb training. Both groups performed 2-3 self-practice sessions weekly. Primary outcome was Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) motor score. Secondary outcomes were fast gait speed (FGS), timed-up-and-go (TUG) time, six-minute walk distance (6MWD), and Mini-Balance Evaluation Systems Test (Mini-BEST) score.
Results:
Sixty-four participants (33 BW/31 CON) completed training. BW group showed greater significant decreases from baseline than CON group in MDS-UPDRS motor score after six weeks (–5.5 vs –1.6,
Conclusion:
The six-month brisk walking and balance program alleviates motor symptoms, promotes functional and gait performance, walking capacity, and dynamic balance in people with mild to moderate PD.
INTRODUCTION
Parkinson’s disease (PD) is a chronic progressive disorder affecting around seven million people in the year 2020 [1], characterized by motor symptoms including bradykinesia, rigidity, tremor, and postural instability [2]. As the disease progresses, these impairments further deteriorate causing marked functional and walking disability [3], and substantial economical and global burden [4, 5]. The ultimate therapeutic aim is to modify the disease and delay the progression of symptoms [6, 7]. While the pharmacological options for PD continue to advance, physical therapy has been found to exert positive short- and long-term effects on muscle strength, aerobic capacity, balance, and gait performance [8, 9].
Among different modes of intervention, sustained aerobic exercise is considered a promising potential therapeutic target in delaying motor disability [10], with higher-intensity exercise possibly more beneficial for symptomatic improvement [11]. Aerobic training using a treadmill attenuated motor symptoms in de-novo, non-medicated PD [12]. A home-based remotely supervised aerobic exercise program, using a stationary home-trainer with virtual reality, significantly decreased off-state (not on-state) motor symptoms in mild PD [13]. Nordic walking studies reported positive effects in alleviating motor symptoms and improving functions in mild to moderate PD [14, 15]. Most of these training studies lasted for six months, suggesting that an extended training period may delay motor disability. Despite their effectiveness, these interventions required large specialized equipment set-up [12, 13] or continual and intensive supervision of two to three sessions a week [14, 15]. Exercise adherence is very challenging for people with chronic illness such as PD [16], and it is highly possible that they could terminate practice when the equipment or supervision is not available, causing detraining effects [17].
Brisk walking is a moderate-intensity aerobic training requiring no exercise equipment, which can be practiced outdoors with a faster pace and larger arm swing at any convenient time. A pilot study indicates that six weeks of brisk walking and balance exercise significantly improves walking capacity and dynamic balance among individuals with mild to moderate PD [18]. In the present study, the training period was extended to six months. Along with accomplishment of the goal using the smartwatch tracker to attain an optimal exercise intensity with brisk walking, we hope to empower PD individuals to assume their responsibility for carrying out regular exercise. In addition, training with no exercise equipment, combined with a shift from under supervision to self-management, could enhance sustainability over a longer term [19]. We hypothesize that this novel combined aerobic and balance training protocol would be beneficial in reducing motor symptoms, as well as enhancing gait and functions, walking capacity, and dynamic balance in people with PD, as compared with an active control intervention. The present study aimed to investigate the effects of a six-month brisk walking and balance program on the motor symptoms of people with PD. The secondary objective was to investigate its effects on gait and mobility functions, walking capacity, and dynamic balance. We further explored the associations of the changes of MDS-UPDRS motor scores with the changes of other clinical outcomes in the BW group after training.
METHODS
Study design and participants
This was a prospective, two-arm, parallel single-blinded randomized controlled trial with group allocation (ratio 1:1) concealed and masked to the principal assessor. Participants were recruited from self-help groups from the Hong Kong PD Association. Inclusion criteria were aged 30 years or over, diagnosed with idiopathic PD [20], stable on anti-PD medications, and able to walk independently for 30 meters without aid. Individuals were excluded if they had significant cardiopulmonary, neurological (other than PD) or musculoskeletal conditions, had received neurosurgery, had cognitive impairment with a Montreal Cognitive Assessment (MoCA) score < 25 [21], or had joined a structured exercise program in past three months. The Ethics Committee of The Hong Kong Polytechnic University approved the use of human participants in this study. Written informed consent from each participant was obtained before the baseline assessment. The trial was registered on ClinicalTrials.gov (Identifiers: NCT04048291).
Sample size calculation
Based on the previous pilot study [19], the corresponding effect sizes obtained for the five outcomes (Movement Disorder Society Unified Parkinson’s Disease Rating Scale motor score, fast gait speed, timed-up-and-go time, six-minute walk distance, and Mini-Balance Evaluation System Test score), were 0.70, 0.19, 0.23, 0.49, and 0.64. Using the G*power 3.1.9.3 software [22], the sample sizes computed with 5%type I error and 90%power were 6, 52, 14, 6 and 8, respectively. To extend the generalizability and anticipating a 15%attrition rate, the required sample size should be at least 62.
After enrollment and screening for eligibility by the research personnel, participants were randomly allocated into either a six-month experimental brisk walking (BW) group or an active control group (CON), using the web-based computer software Research Randomizer Version 4.0 [23]. The randomization with assignment of participants to intervention was conducted before the baseline assessment by a team member not involved in this study. The group allocation was accessible to the training therapists, but not to the research staff conducting the assessment, data entry and data analysis. The participants were instructed not to disclose their group allocation to any staff involved. The two groups were treated at different venues to minimize any exercise contamination effect [24].
Outcome measures
The baseline measurements were age, gender, body mass index, disease duration, modified Hohen and Yahr stage [25], non-fallers to fallers ratio in the past six months, MoCA score [21], MDS-UPDRS motor score [26], number of comorbidities, Physical Activity Scale for the Elderly (PASE) score [27], daily levodopa equivalent dosage [28] (Table 1). Using a physical activity tracker (Model A370 from Polar Electro Oy, Finland) with photoplethysmography sensors [29], the average seven-day resting heart rate (HR) was collected before the participants started their daily activities (Table 1). We established the accuracy of the HR measurement, recorded by Polar A370, by comparing it with the gold standard telemetry electrocardiogram (NIH001-WEP-5208K, Nihon Kohden). Under sitting and fast walking conditions (at fast speed continuously for 6 minutes to achieve 40–60%HR reserve plus resting HR), excellent intraclass correlation coefficients (ICC3,1) of 0.974 and 0.961, respectively, were observed. HR reserve (HRR) was calculated by subtracting maximum predicted HR (220 minus age) by resting HR.
Comparison of demographic characteristics and clinical outcomes at baseline between two groups (N = 64)
Values are mean±standard deviations, or as otherwise indicated. §Chi-square tests; #Mann-Whitney U tests. MDS-UPDRS motor score (A higher score indicates more severe motor symptoms in past 7 days); Mini-BEST score (A higher score indicates better dynamic balance); MoCA score (A higher score represents less cognitive impairments); Modified Hohen & Yahr Stage (1–5, a higher stage indicates greater disease severity); PASE score # (A higher score indicates higher physical activity level in the past 7 days). 6MWD, six-minute walk distance; BW, brisk walking group; CON, control group; F, female; FGS, fast gait speed; HR, heart rate; M, male; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; Mini-BEST, Mini-Balance Evaluation Systems Test; MoCA, Montreal Cognitive Assessment; PASE, Physical Activity Scale for the Elderly; PD, Parkinson’s disease; Post6 m, after 6-month of training; Post6wk, after 6-week of training; Pre, 1-week before training; TUG, timed-up-and-go.
The primary outcome measure was the Movement Disorder Society Unified Parkinson’s Rating Scale (MDS-UPDRS) motor score to indicate motor performance [26]. The scale was found to be valid and reliable with excellent internal consistency > 0.9 [26, 30]. The secondary outcomes were fast gait speed (FGS), timed-up-and-go (TUG) time, six-minute walk distance (6MWD), and Mini Balance Evaluation Systems Test (Mini-BEST) score. FGS in cm/s was assessed by taking an average of two trials of a 10-meter walk test (as fast as one can safely walk). FGS has excellent test-retest reliability with an ICC of 0.97 in people with PD [31]. The TUG test is a valid tool to measure the time taken to stand up from a chair, walk three meters at a comfortable speed, turn around, walk back and sit down, with a shorter duration indicating better mobility [32]. The average duration of two TUG test trials was used to evaluate mobility function. Walking capacity was evaluated by the maximum 6MWD covered in a validated six-minute walk test [31]. The Mini-BEST is a validated test used to measure dynamic balance ability in four domains: anticipatory postural adjustment, postural responses, sensory integration, and gait stability [33, 34]. Mini-BEST scores range from 0 to 28 and higher scores indicate better dynamic balance. All participants were assessed in their on-medication phase at the University Balance and Gait Laboratory at three assessment intervals: one week before training (Pre), after six weeks (Post6wk), and after six months of training (Post6m). Other measures included exercise compliance and characteristics in terms of six-month class attendance, training HR expressed as percentage HRR plus resting HR in the BW group, and weekly exercise duration and frequency.
Interventions
Both the BW and CON groups performed three weekly exercise sessions for six months. During weeks 1–6 (the training phase), the BW participants received one weekly session (90 minutes) in groups of six-to-eight supervised by a physiotherapist and an assistant, and two self-practice sessions (60–90 minutes each) in the community. Training sessions were conducted at the university’s campus to ensure safety and protocol familiarization in the first two weeks. In next four weeks, training took place at the jogging track of an indoor sportsground. In each training session, the BW group received postural re-education, warm-up and cool-down exercises, balance training with musical cues [35], and progressive brisk walking (Supplementary Material). Brisk walking required the participants to ambulate at a moderate to fast speed, emphasizing long strides and large arm swings. Additional balance training enabled the participants to maintain good stability during ambulation under different environmental contexts in the community.
A smartwatch tracker was loaned to BW participants to monitor and record real-time HR changes (indicating actual data and under which HRR zones), total exercise duration and own exercise intensity (percentage of total time spent in each of the three preset personalized zones) during brisk walking. Exercise training below 40%of HRR plus resting HR was considered as low intensity, 40–60%of HRR plus resting HR as moderate intensity and above 60%plus resting HR as high intensity. The participants were instructed to walk at a fast pace gradually to reach a steady moderate exercise intensity level targeting at 40–60%of HRR plus resting HR in each training session [36]. Daily exercise records were accessible in the smartwatch displays to allow self-monitoring. The accumulative records were synchronized and downloaded from the official Polar Flow website at each supervisory session for the therapist’s reference. The rate of perceived exertion was self-monitored using modified 10-point Borg’s scale scores of 4–6 [37]. The weekly brisk walking duration was gradually increased from 60 minutes in week 1 to 150 minutes in week 6, aiming for “150 minutes of moderate-intensity exercise” per week as recommended by the American College of Sports Medicine guidelines [38]. At the end of week 6, participants would be able to perform brisk walking at the target moderate exercise duration and intensity. At weeks 7–26 (the empowerment phase), the supervisory sessions were tapered to once a month and took place at the outdoor park or open field. BW participants were trained to walk faster in order to reach the upper limits of their moderate exercise intensity while maintaining good balance (Supplementary Material). Participants continued to practice two to three sessions a week on their own in the local community such as park and outdoor sport ground, using smartwatch to monitor HR activity and to maintain the targeted exercise intensity and duration. Participants received feedback on their goal accomplishment at each monthly supervisory session. In addition, the therapists sent biweekly phone messages to motivate individual participants to complete the recommended dose, while the participants could also send their concerns and questions about self-practice to the therapists for advice and assistance.
The CON group provided placebo effects and maintained motivation without walking and balance training. They received six months of upper limb and hand dexterity training in groups of six-to-eight at community centers, including postural re-education, stretching, strengthening, and hand dexterity exercises. Their contact hours with physiotherapists, exercise progression and duration, level of supervision, home exercise dosage, and trainer feedback frequency and method were all equivalent to those in the BW group (Supplementary Material). The CON group recorded their exercise duration in monthly written diaries. Throughout the six-month training period, both subject groups were given handouts and exercise videos so that they could practice the taught exercises regularly. Exercise adherence in terms of six-month class attendance and the average weekly exercise duration (weeks 7–26) was evaluated and compared between the two groups. Exercise-related adverse events included falls with or without physical injury, other injuries, hypotension, shortness of breath, dizziness, joint pain, muscle soreness, fatigue and other discomfort affecting training [9]. A fall is defined as an event that results in a person coming to rest unintentionally on the ground or other lower level [39]. Both adverse effects and falls were recorded during supervised classes and self-practice sessions using fall diary.
Statistical analysis
All data were analyzed using SPPS Statistical Software 23.0, with a significance level of
RESULTS
Participants and baseline characteristics
Seventy participants were randomly assigned to either a BW (
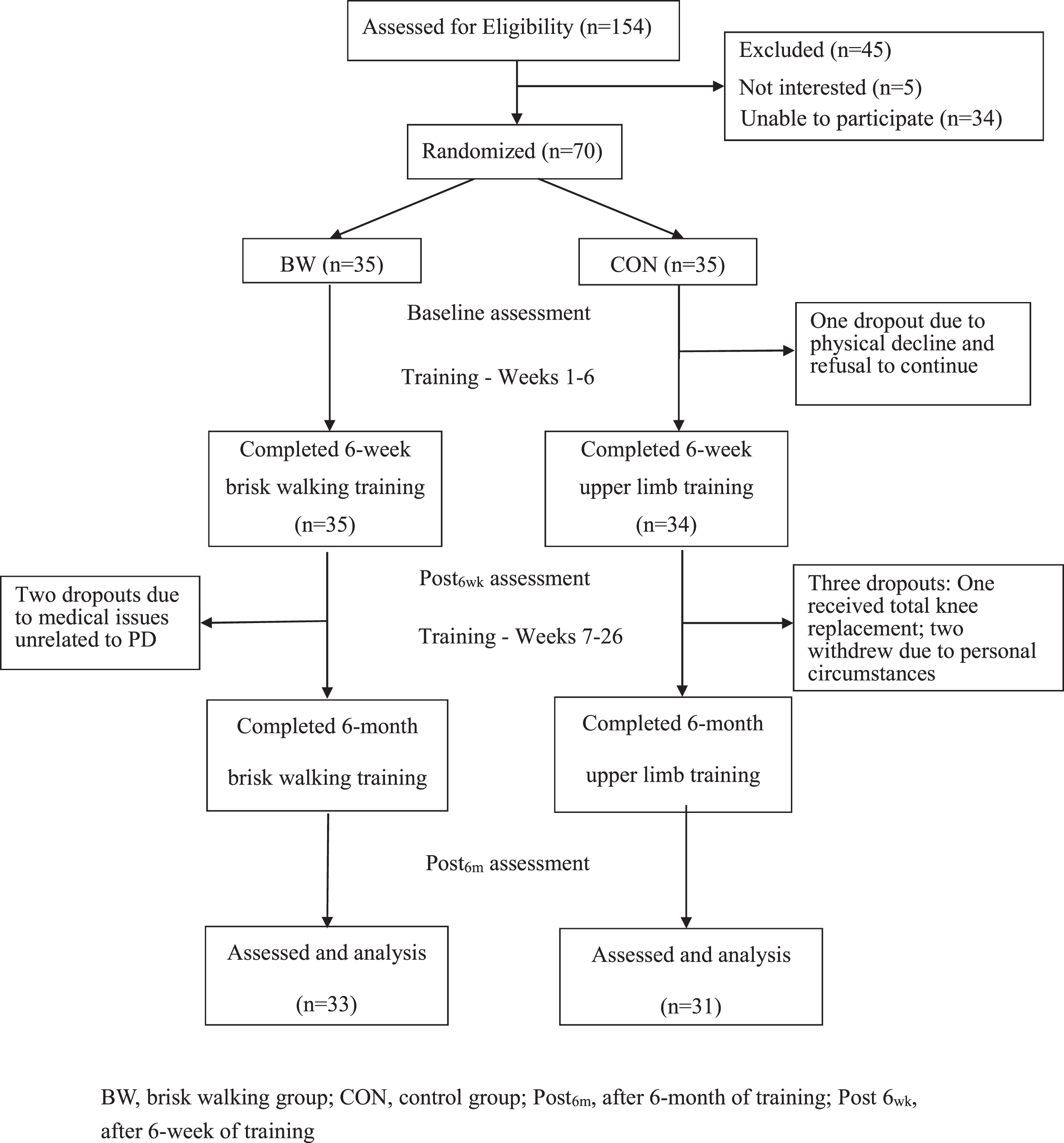
CONSORT flow diagram showing the course of study after recruitment.
Six-month exercise compliance, exercise characteristic and adverse effects (N = 64)
Values are mean±standard deviations, or as otherwise indicated. BW, brisk walking group; CON, control group; HRR, heart rate reserve.
Within the intervention period, both groups reported no falls or injuries during exercise training or self-practice in the community. There were also no serious adverse events. Three mild musculoskeletal adverse events related to exercise were reported in the BW group including knee pain (two episodes) and toe pain (one episode) during supervisory sessions. These painful conditions subsided quickly after the participants took a rest before continuing training. The therapist reiterated the importance of adopting a proper walking pattern and correct shoe wearing, and there was no recurrence of painful conditions. No other adverse effects were reported during self-practice sessions in both groups.
Treatment effects in primary and secondary outcomes
Tables 34 summarize the six-month training effects on the primary and secondary outcomes. When compared with the CON participants at Post6wk, the BW participants had significantly greater reductions from baselines in MDS-UPDRS motor scores (
Within-group comparisons of clinical outcomes (N = 64)
Values are mean±SD or as otherwise indicated. ∧
Between-group comparisons of changes from baselines for clinical outcomes (N = 64)
Values are mean±SD or as otherwise indicated. *
At Post6 m, the BW group showed greater significant decreases from baselines than the CON group in MDS-UPDRS motor scores (
Associations between changes in MDS-UPDRS motor scores and changes in other clinical outcomes
At Post6wk, there was a trend of association between reduced MDS-UPDRS motor score and better Mini-BEST score. At Post6 m, significant associations were shown between reduced MDS-UPDRS motor scores with improved Mini-BEST scores and less TUG time (
Associations between changes in MDS-UPDRS motor score and other clinical outcomes in the brisk walking group (
r, Pearson product-moment correlation coefficient. *
DISCUSSION
The present study reports that a six-week community-based brisk walking and balance program alleviates motor symptoms and promotes functions among people with mild to moderate PD, and that the benefit persists for six months after tapering of supervision frequency. The on-state MDS-UPDRS motor scores of BW individuals significantly decreased by 5.5 and 6.0 points at Post6wk and Post6 m, respectively. These positive changes were greater than the 3.25 points estimated for a meaningful clinically important difference [41]. Previous studies reported that aerobic training with a treadmill [12] or a stationary home-trainer [13], or Nordic walking requiring intense supervision [14, 15] were effective in modulating motor symptoms. In addition, aerobic training at high intensity, but not at moderate intensity, was effective in attenuating PD motor symptoms in an on-medicated state [13, 14]. Our study is the first to report that sustained aerobic training with no exercise equipment at moderate intensity combined with balance exercise, under a low level of supervision, alleviates motor symptoms in individuals with PD under optimal medication. In addition, this brisk walking and balance exercise regime improves walking capacity, gait functions and dynamic balance.
The adoption of coordinated aerobic walking in PD while practicing good postural skills could have lowered MDS-UPDRS motor scores and alleviated motor symptoms such as rigidity, bradykinesia, gait and posture impairments, and postural instability. PD is chronic and progressive; evidence has shown the long-term benefits of exercise training [9, 42], but it is very challenging for those with PD to develop sustained exercise habits and exercise adherence. The high attendance rate during the supervisory sessions reflects the good exercise compliance of the BW group. More importantly, as the participants mastered the brisk walking technique after six weeks of training, they sustained their training even with a tapering of weekly to monthly supervision. It is encouraging to observe that participants adhered well to the protocol by completing over 80%of the recommended weekly duration from Week 7 to 26. The sustained aerobic walking might facilitate training-induced neuroplasticity by increasing serum levels of brain-derived neurotrophic factor, leading to improved aerobic fitness and motor functions, as well as decreased symptom progression and in PD [9]. The use of a smartwatch tracker to monitor their exercise intensity and planned goal achievement, as well as the biweekly reminder from the therapists could have served as motivational purposes [43]. The ability to practice at moderate exertion with enjoyment could be another important motivator in increasing compliance [44]. PD individuals could be empowered to assume greater responsibility in carrying out regular exercise training with flexible options of time and location for practice.
In addition to alleviating motor symptoms, there was increase in walking capacity of a magnitude comparable to Nordic walking [14]. PD gait is usually affected by small steps, while cadence remains less affected [45]. To maximize treatment effects, BW participants were instructed to perform brisk walking with large steps at a faster pace to achieve moderate-intensity aerobic levels. These strategies could have improved their gait ability and cardiovascular function, leading to increased walking speed and walking capacity. In each BW session, the participants practiced balance exercises using musical cues to optimize their dynamic balance [35]. These crucial balance skills included shifting the body’s center of mass in different directions, changing from double- to single-leg stances, and incorporating large-amplitude arm and trunk movements in repetitive steps. After the acquisition of these useful postural control strategies, those in the BW group were able to optimize their gait and balance functions to negotiate different terrain during aerobic walking in the community. As a result, the BW participants outperformed the CON participants with large effect sizes in Mini-BEST scores, reaching a score of 23.3 and 24.2, at Post6wk and Post6 m, respectively. These scores were higher than the cut-off point (≥21.0) used to differentiate PD individuals with or without postural deficits [34]. The community-based BW and balance practice improved both balance and walking speed, thus contributing to the enhancement of sequential motor performance in TUG tests. Interestingly, the reduced TUG time and improved Mini-BEST score had significant associations with reduced MDS-UPDRS motor scores after training, implying that better balance performance and complex walking function might contribute to delaying motor disability related to PD. Further study is needed to confirm the causative effects between these clinical outcomes. Joint pain and muscle soreness are common adverse events in long-term physical interventions for PD [9], therefore programs should be safely adapted to the abilities of the participants [44]. Our findings of no injuries or serious adverse side effects during training indicate that it is a safe and feasible protocol for 6-month practice.
Study limitations
This study has several limitations. First, no smartwatch tracker was provided to the CON group during training and the CON treatment only served as a placebo effect. Thus, the measurement of exercise duration was based on self-report records for CON group and electronic records for BW group may limit accuracy of comparison. Second, therapists performed data synchronization of the smartwatch tracker during supervisory sessions using a laptop, and more regular data update could be possible if participants were taught to record the HR and exercise intensity data using mobile apps. Third, Borg scale scores were only reported during BW supervisory sessions. Fourth, this study has only addressed the effects after six months of training; adding a follow-up period without supervision would examine if the clinical benefits and exercise adherence during self-practice could be maintained. Further investigations with a six-month follow up have been planned to evaluate the carry-over effects. Finally, this brisk walking treatment protocol was applicable to people with mild to moderate PD who could walk outdoors independently, and its effects may not be generalized to those with more advanced disease and severe freezing of gait symptoms.
CONCLUSIONS
This community-based brisk walking and balance program alleviates motor symptoms and improves functions, walking capacity, and dynamic balance of people with mild to moderate PD after six months of training. Further investigations are required to determine the long-term carry-over effects of this mobile technology augmented exercise intervention for the PD population.
Footnotes
ACKNOWLEDGMENTS
We would like to express sincere thanks to the funding sources of support by the Shun Hing Education and Charity Fund (847P), and all the volunteers who had participated in the study.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
