Abstract
Background:
Urinary dysfunction and constipation, manifestations of pelvic floor dysfunction are common sources of disability and impaired quality of life in women with Parkinson’s disease (PD).
Objective:
We sought to evaluate the pelvic floor health amongst women with PD and their reporting of bladder and bowel symptoms.
Methods:
We surveyed women with PD and age-matched controls about pelvic floor health using validated questionnaires. All participants completed the Pelvic Floor Disability Index (PFDI-20), the Pelvic Floor Impact Questionnaire (PFIQ-7) and the Patient-Reported Outcomes Measurement Information System (PROMIS) short form version 2.0 Cognitive Function 8a. Additionally, PD patients underwent the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) scale and the Montreal Cognition Assessment (MoCA).
Results:
Women with PD (n = 59; age, 70.4±8.6 years, PROMIS cognitive score, 52.0±7.8) self-reported urinary symptoms to a greater extent than controls (n = 59; age, 70.2±8.7 years, PROMIS cognitive score, 51.0±10) (68% vs 43%, p < 0.01). The difference was mirrored by higher (worse) scores on both PFDI-20 (35.4 vs 15.6; p = 0.01) and PFIQ-7 (4.8 vs 0; p < 0.01) for PD women compared to controls. Only 63% of all participants with self-reported pelvic floor symptoms had previously reported these symptoms to a health care provider. There was no difference in utilization of specialty care between the two groups (30% vs 46%, p = 0.2).
Conclusion:
Pelvic floor dysfunction, more common amongst women with PD, is underreported and undertreated. Our study identifies a key gap in care of women with PD.
Keywords
INTRODUCTION
Parkinson’s disease (PD) affects 1% of the population older than 60 years [1] with 6.2 million individuals affected globally. PD is the fastest growing neurodegenerative disorder in the world with a projected 12 million individuals affected by 2040 [2]. Urinary dysfunction is a common non-motor manifestation in PD with a high prevalence of urinary urgency and frequency, also known as overactive bladder (OAB) [3–7] and is associated with increasing age, worse quality of life, dysautonomia, cognitive impairment and falls [8–11]. Slow colonic transit, secondary to bowel dysfunction and dysautonomia, can lead to constipation which is also a common pelvic floor symptom in this patient population [12].
National estimates of pelvic floor disorders (PFDs) including urinary incontinence, fecal incontinence or pelvic organ prolapse, have found that almost 50% of non-institutionalized women in the United States (US) over the age of 80 report at least one symptom [13]. Specific to lower urinary tract symptoms (LUTS), there are differential bother and health seeking behaviors between men and women [14]. While LUTS was more bothersome in women, men were more likely to seek treatment with increasing age and severity of symptoms. Treatment-seeking in women was driven only by increasing bother, regardless of age or symptom severity [14]. Epidemiologic studies in PD typically include both men and women and report a wide range of prevalence of LUTS between 24–96% [15]. Despite the increasing prevalence of both PD and PFDs amongst all women as they grow older [13, 16], the literature describing PFDs in only women with PD is scarce, compromising our ability to provide adequate care for these patients. Earlier diagnosis and referral to appropriate specialists can enable patients to receive optimal care specific to their PFD and improve their quality of life, including but not limited to behavioral therapies and botulinum toxin injections [15].
To address this gap in knowledge, we sought to assess pelvic floor health in women with PD as compared to age- and sex matched healthy controls. We hypothesized that women with PD would be more affected by PFDs than age-matched controls.
MATERIALS AND METHODS
Experimental design and patient population
This was a survey-based, cross-sectional study of consecutive, female participants presenting to the movement disorders clinic at University of Louisville (November 2018 to September 2019) and University of Cincinnati (November 2019 to May 2020) with a diagnosis of PD (cases) or presenting to primary care clinics without a diagnosis of PD (controls). Inclusion criteria for cases were 1) females with PD diagnosis fulfilling the United Kingdom Brain Bank criteria [17], 2) age between 40 to 85 years, 3) able to understand oral and written English, and 4) able to consent or presented with a caregiver who could provide consent. The exclusion criteria for cases were 1) patients with atypical Parkinsonism and/or 2) those unwilling to consent. Age-matched controls were selected in a 1:1 ratio to cases from amongst women who presented for primary care and annual appointments with Family Medicine, Geriatric Medicine and Gynecology at University of Louisville. Controls were included if 1) their age was within±1 year of cases, 2) they had presented for an annual exam within the last 6 months, and 3) they did not have a diagnosis of PD. To minimize confounding, controls were excluded if they presented to the primary care clinics with primary complaint related to PFDs. This study was independently approved by the institutional review boards at both the University of Louisville and the University of Cincinnati. Informed consent was received from all subjects prior to participation in the study. All human studies were approved by the appropriate committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Data collection
Eligible cases were identified during their routine clinic visit for PD. After verbal and written consent, participants completed the following validated scales: Pelvic Floor Disability Index (PFDI-20) [18], Pelvic Floor Impact Questionnaire (PFIQ-7) [18], and the Patient-Reported Outcomes Measurement Information System (PROMIS) short form version 2.0 Cognitive Function 8a [19]. Data on the following scales were collected from the electronic clinical charts for case patients: Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) [20], modified Hoehn Yahr Scale [21] and the Montreal Cognition Assessment (MoCA) score [22]. These scales were repeated at the time of the visit if available scores were older than 6 months. Controls were asked to complete the same surveys as their PD counterparts except for MDS-UPDRS, modified Hoehn Yahr Scale and MoCA. Lastly, participants completed a non-validated semi-structured questionnaire with questions about their general health, parity, Charlson comorbidity index [23], symptoms of vaginal bulge, urinary symptoms and whether they received subspecialty care or treatment for their pelvic floor symptoms (Supplementary Material). Due to social distancing guidelines, implemented during recruitment for this study after the onset of the COVID-19 pandemic, eligible participants could complete this survey online (through a confidential email link) or via fax, per their preference. All responses were recorded in REDCap [24].
Description of selected data collection tools
The PFDI-20 questionnaire is a scale validated in women which focuses on symptom distress [25]. It is comprised of 3 subscales, the Urinary Distress Inventory (UDI-6), the Pelvic Organ Prolapse Distress Inventory (POPDI-6), and the Colorectal-Anal Distress Inventory (CRADI-8), recording urinary, prolapse and bowel (constipation, anal incontinence, fecal urgency and rectal prolapse) symptoms, respectively. Each subscale is scored from 0–100 and the sum of the three scores is reported as the overall score, ranging from 0–300. The PFIQ-7 focuses on the impact of symptoms on everyday activities, has 3 similar subscales: the Urinary Impact Question-naire (UIQ), the Pelvic Organ Prolapse Impact Ques-tionnaire (POPIQ) and the Colorectal-Anal Impact Questionnaire (CRAIQ), and provides a score between 0–300 [25]. Higher scores in both the PFDI-20 and the PFIQ-7 correspond with a higher perceived impact or bother of pelvic floor symptoms, regardless of the cause of the pelvic floor disorder [26]. The PROMIS Short Form version 2.0 - Cognitive Function 8a is an 8-item questionnaire which assesses the extent of patient-perceived cognitive deficits and has been validated in older patients at risk of cognitive decline [27]. Scores are standardized such that a score of 50±10 represent the mean ± standard deviation for the general United States (US) population [19]. Higher scores represent better cognitive functions.
Outcomes and statistical analysis
The primary outcome of this study was to compare prevalence of participant-reported symptoms of PFD to age-matched controls based on a positive response to the questions “Do you experience bulging or something falling out you can see or feel in the vaginal area?” or “Do you have complaints of urgency of urination or leaking of urine?” [13]. We sought to further characterize pelvic floor symptoms by comparison of scores on the three PFDI-20 and PFIQ-7 subscales. Additional outcomes included participant reporting of symptoms to health care providers and barriers to subspecialty referrals amongst PD participants, as compared to controls.
Using data from prior studies specific to urinary dysfunction, we estimated a 40% prevalence of urinary incontinence in the control population [28] compared to 65% in the PD population [29]. To achieve 80% power with 95% confidence, we required a sample size of 59 participants in each group. Categorical variables were analyzed using χ2 and Fisher’s exact tests while continuous variables were analyzed using 2-sample t-tests. For non-parametric data, Mann-Whitney test was conducted. Missing responses (usually confined to one or two per group) were analyzed in an all available cases fashion. Significance level was two-sided and set at 0.05 for all tests. Data were analyzed using R 3.6.2.
RESULTS
Demographic characteristics
Between November 2018 to May 2020, we recruited 59 women with PD and 59 age- and sex-matched controls at University of Cincinnati and University of Louisville (cases, 70.4±8.6 years; controls, 70.2±8.7 years). Apart from a higher proportion of Caucasian race (93% in cases vs 78% in controls, p = 0.03), there were no significant differences in demographic data between the two groups (Table 1). Cases had similar cognitive function to controls when measured using PROMIS Cognitive function (52.0±7.8 vs 51.0±10, p = 0.54). For cases, the median time after diagnosis of PD was 4 years [Interquartile Range (IQR) 3–10], the median modified Hoehn Yahr Scale was 3 [IQR 3–5] and MoCA was 26 [IQR 24–28]. The median MDS-UPDRS scores were Part 1:11 [IQR 5.5–14.5], Part 2:9 [IQR 4–13.5] Part 3:24 [IQR 16–35], and Part 4:3 [IQR 1–6].
Demographic characteristics
All data are reported as Mean±Standard Deviation or Median [25th percentile- 75th percentile] or n (%) unless otherwise specified. BMI, body mass index; CRADI, Colorectal-Anal Distress Inventory; CRAIQ, Colorectal-Anal Impact Questionnaire; MDS-UPDRS, Movement Disorders Society Unified Parkinson’s Disease Rating Scale; MOCA, Montreal Cognition Assessment; PD, Parkinson’s disease; PFDI, Pelvic Floor Disability Index; PFIQ, Pelvic Floor Impact Questionnaire; POPDI, Pelvic Organ Prolapse Distress Inventory; POPIQ, Pelvic Organ Prolapse Impact Questionnaire; PROMIS, Patient-Reported Outcomes Measurement Information System; UDI, Urinary Distress Inventory; UIQ, Urinary Impact Questionnaire. *Missing 1 respondent. **Missing 2 respondents. ***Missing 4 respondents.
Primary outcome
There was significantly higher self-reported urinary urgency or incontinence in cases when compared with controls (68% vs 43%, p < 0.01) but no statistically significant evidence of a difference in vaginal bulge symptoms between the two groups (2% cases vs 11% controls, p = 0.06). Cases had higher overall scores for both the PFDI-20 [35.4 (IQR 13.4–57.8) vs 15.6 (IQR 4.2–37.5), p = 0.01] and the PFIQ-7 [4.8 (IQR 0–28.6) vs 0 (IQR 0–7.1), p < 0.01] compared to controls. On sub-score analysis, cases had significantly worse CRADI-8 scores [12.5 (3.1–18.3) vs 0 (0–9.4) controls, p = 0.01] and UIQ scores [4.8 (0–19) vs 0 (0–4.8), p < 0.01] when compared with controls (Table 1). Stratification by age revealed that PFIQ-7 scores were significantly worse in cases younger than 66 years when compared to controls of similar age (Fig. 1). There was no significant difference between PFDI-20 and PROMIS 8a scores when stratified by age (Fig. 1).
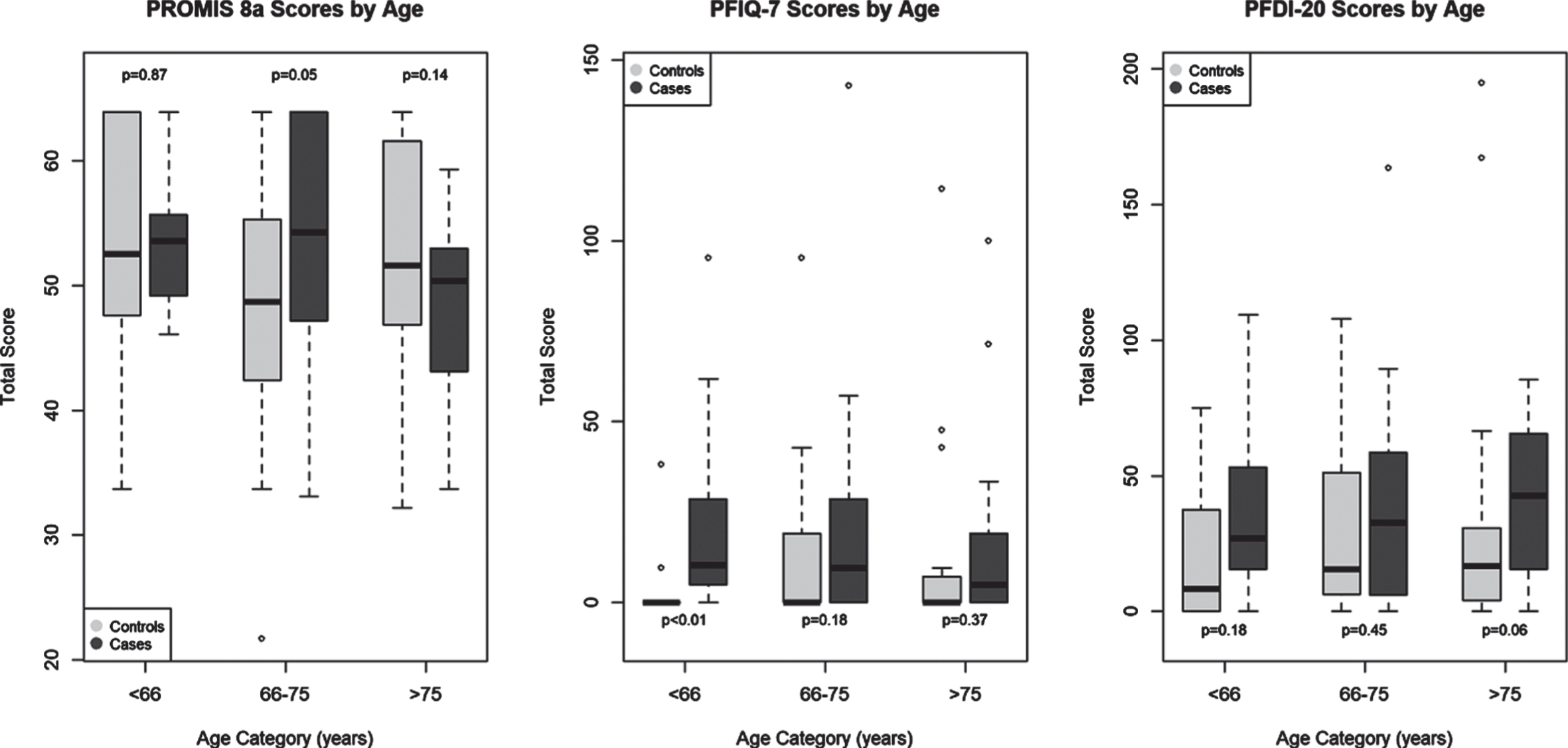
Age stratification of questionnaires. Data reported as box plot: median with interquartile range, whiskers: minimum and maximum, outliers. The groups are reported in tertiles of age: < 66 (n = 14 cases and controls), 66–75 years (n = 24 cases; 26 controls), and > 75 years (n = 21 cases; 19 controls). PROMIS 8a, Patient-Reported Outcomes Measurement Information System short form version 2.0 Cognitive Function 8a; PFIQ-7, Pelvic Floor Impact Questionnaire; PFDI-20, Pelvic Floor Disability Index.
Health care utilization
Twelve percent of all participants had received previous treatment for prolapse or urinary dysfunction, similar between cases and controls (7/59 or 12% in both groups). Of the 66 patients who reported urinary or bulge symptoms, only 63% of participants had reported their symptoms to a health care provider, and there was no difference in symptom reporting between cases and controls (63% vs 62%). The most common type of provider to which patients in this study reported PFDs was their primary care provider (Table 2). Only 34% of participants with either prolapse or OAB symptoms had seen a specialist, and this was similar between cases and controls (30% vs 46%; p = 0.2). Most participants who had not seen a specialist reported that their symptoms were not bad enough to seek specialty treatment, although 31% (5/13) of cases with symptoms had not been offered a specialty referral.
Physician reporting and specialty referrals in participants who reported bulge or urinary incontinence
All data are reported as n (%) unless otherwise specified. CRS, colon & rectal surgery; PCP, primary care provider. *Missing 1 respondent.
DISCUSSION
Our study found that women with PD had worse pelvic floor health, as seen by worse PFD associated symptoms, compared to age-matched controls per self-report and scores on validated scales. By controlling for potential confounding by age, we demonstrate that PFDs are an important, inadequately addressed problem in women with PD regardless of age.
Literature reports that more than half of PD patients report bowel dysfunction, which may be confounded by age-related constipation [7]. In this study, we found that women with PD had significantly greater bother from bowel dysfunction when compared to controls (as demonstrated by a higher CRADI-8 score) but the greatest impact on their life was due to their urinary symptoms (significantly worse UIQ scores). Overall, symptom bother and impact was much worse (higher PFDI-20 and PFIQ-7 scores) in women with PD when compared with controls. Such pelvic floor symptom burden should trigger a clinically appropriate work up for etiology and directed management, in addition to symptomatic management. Stratification by age revealed that the impact of PFDs was significantly greater in PD patients younger than 66 years of age, suggesting that PFDs may either present earlier or be more deleterious to the quality of life of younger PD patients.
Despite worse pelvic floor health, women with PD were as likely to report symptoms as controls and perhaps less likely to see a specialist due to poor access and/or education about management options amongst reported reasons. Only a small fraction of our PD sample reported these symptoms to their movement disorders neurologist. In addition to possible discomfort with discussing pelvic floor symptoms, the most common reason for declining specialty evaluations was that these symptoms “were not bad enough to seek treatment”, even in patients who initially reported their complaints to a physician. It is possible that patients get accustomed to living with associated discomfort, downplay their symptoms to their physician, do not realize the association of symptoms with PD and/ or are not counseled appropriately by the treating physician. Apathy, commonly seen with PD [30], may also be a contributory factor which could delay seeking care. Although cognition may impact reporting of urinary symptoms, it is unlikely that cognitive impairment discordantly influenced our results given the similar PROMIS scores between the two groups. Finally, 30% of participating women with PD who had not seen a specialist reported that they had not been offered a referral from their physician. Our data highlight the need for greater awareness and appreciation of these symptoms and available treatment modalities amongst physicians and patients. Such understanding of specific symptoms and appropriate referral could lead to different approaches based on the presenting complaint, as seen in management of types of urinary incontinence.
The strengths of this study lie in the use of validated questionnaires to address questions regarding female pelvic health. Our study evaluated patients with PD who were cognitively unimpaired, and this was confirmed by similar PROMIS cognition scores in both patients and controls. Both groups were also demographically similar and had similar general health and health-seeking behaviors. This study also age-matched controls to provide valuable data on differences in PFD symptoms between PD and non-PD women without the confounding effect of age. Lastly, we included controls from a similar patient population, but excluded those who presented to clinic with PFD related symptoms to minimize confounding.
Our study is not without limitations. The study sample was selected from tertiary referral centers and may have a higher rate of reporting symptoms and seeking specialty care when compared to community-dwelling women. This study was also not powered to detect granular differences in subscale scores of quality of life questionnaires. The results and conclusions of our study are based on validated pelvic floor questionnaires for women. These are not generalizable to men with PD who have distinct pelvic floor pathology which may present as different pelvic floor symptoms. While we attempted to age-match controls, we did not match for race or parity, and there were disproportionately more Caucasian women with PD when compared to controls, which is consistent with prior PD literature [31]. Historic studies have suggested a difference in PFD symptoms based on race but more recent, population-based studies have reported similar PFD prevalence in women of different race and ethnicities [13]. We did not individually match for parity but had similar childbearing across the two groups. Lastly, while this study was entirely computer based, the implementation of social distancing guidelines prompted the completion of surveys electronically via email by more controls compared to PD participants, most of whom completed the questionnaires on a computer or tablet in person at the clinic. The impact of this difference on patient reported outcomes is unknown.
In conclusion, our study shows that pelvic floor disorder-associated symptoms, more prevalent and impactful in women in PD, are underreported and undertreated likely contributing to impaired quality of life. Physicians taking care of women with PD should inquire about these symptoms and have a low threshold for referral for subspecialty care. Better education and greater outreach is imperative to fill this gap in care for our patients.
CONFLICTS OF INTEREST
AG reports no relevant financial disclosures or conflicts of interest.
KL has received consulting fees from Acorda and honoraria from the American Academy of Neurology and the International Movement Disorder Society.
KVM receives travel support as a Board Member of the Society of Gynecologic Surgeons (Research Committee Chairman), receives book royalties from Elsevier publishing, and is a consultant for RBI Medical.
AJE has received grant support from the NIH and the Michael J Fox Foundation; personal compensation as a consultant/scientific advisory board member for Abbvie, Neuroderm, Neurocrine, Am-neal, Adamas, Acadia, Acorda, InTrance, Sunovion, Lundbeck, and USWorldMeds; publishing royalties from Lippincott Williams & Wilkins, Cambridge University Press, and Springer; and honoraria from USWorldMeds, Acadia, and Sunovion.
AM has received funding from Dystonia Medical Research Foundation, Sunflower Parkinson’s disease foundation and Parkinson’s Foundation outside the scope of this work. He reports no conflicts of interest.
The remaining authors report no relevant financial disclosures or conflicts of interest.
Footnotes
ACKNOWLEDGMENTS
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Availability of data and material (data transparency)
Dr. Mahajan had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Ethics approval (include appropriate approvals or waivers)
This study was independently approved by the institutional review boards at both the University of Louisville and the University of Cincinnati. Informed consent was received from all subjects prior to participation in the study. All human studies were approved by the appropriate committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
