Abstract
Background:
In general, the risk to develop Parkinson’s disease (PD) is higher in men compared to women. Besides male sex and genetics, research suggests diabetes mellitus (DM) is a risk factor for PD as well.
Objective:
In this population-level study, we aimed at investigating the sex-specific impact of DM on the risk of developing PD.
Methods:
Medical claims data were analyzed in a cross-sectional study in the Austrian population between 1997 and 2014. In the age group of 40–79 and 80+, 235,268 patients (46.6%females, 53.4%males) with DM were extracted and compared to 1,938,173 non-diabetic controls (51.9%females, 48.1%males) in terms of risk of developing PD.
Results:
Men with DM had a 1.46 times increased odds ratio (OR) to be diagnosed with PD compared to non-diabetic men (95%CI 1.38–1.54, p < 0.001). The association of DM with newly diagnosed PD was significantly greater in women (OR = 1.71, 95%CI 1.60–1.82, p < 0.001) resulting in a relative risk increase of 1.17 (95%CI 1.11–1.30) in the age group 40 to 79 years. In 80+-year-olds the relative risk increase is 1.09 (95%CI 1.01–1.18).
Conclusion:
Although men are more prone to develop PD, women see a higher risk increase in PD than men amongst DM patients.
INTRODUCTION
Previous studies have shown that the rates of Parkinson’s disease (PD) were significantly elevated following the diagnosis of type 2 diabetes mellitus (T2DM) [1, 2]. Diabetes mellitus (DM) is a chronic, non-communicable disease, associated with serious long-term micro-and macrovascular complications such as cardiovascular, peripheral vascular, ocular, neurologic, renal and reproductive abnormalities [3, 4]. The impact of T2DM on the risk to develop PD was increased in those with complicated T2DM and longer duration of T2DM [1]. Furthermore, T2DM is associated with a faster progression and a more severe phenotype of PD [5]. PD is a chronic, progressive disease of the nervous system due to the degeneration of nigrostriatal dopaminergic neurons and the second most common neurodegenerative disease in the world. Incidence rates in PD steadily increase with age in both females and males [6], leading to a higher worldwide prevalence of PD due to higher life expectancy and an aging global population [7]. With an average incidence rate of 61.21 per 100,000 person-years in men compared to 37.55 per 100,000 in women 40 years and older, there is a significant gender gap in PD. Contrastingly, the prevalence of DM is slightly higher in men than in women and increasing across all age groups [8, 9]. The connection between DM and the development of PD remains unclear. However, aberrant insulin signaling has been suggested as the culprit. In both DM and PD, mitochondrial dysfunction, neuroinflammation, increased endoplasmic reticulum stress, abnormal protein aggregation, and metabolic abnormalities are common, suggesting a pathophysiological link [10–12]. Anti-diabetic drugs such as glitazones, GLP-1 agonists, and metformin have been discussed as protective measures for PD risk and disease progression modification [13–15].
Women are often more negatively impacted by DM and its complications than men [16–18], which might apply to the risk of developing PD in patients with DM as well. Reflecting on these previous findings, we aimed at investigating the effect of DM on PD risk.
METHODS
This cross-sectional study is based on a medical claims database provided by the Austrian Federal Ministry for Health, covering approximately 45,000,000 hospital stays of about 9,000,000 individuals in Austria from 1997 until 2014. Each hospital visit is recorded and described with a unique patient identifier, patient’s age group, sex, primary diagnosis, secondary diagnoses, and hospital entry and exit dates. All diagnoses are given in the form of level-3 ICD-10 codes. Data on prescribed medication was derived from a six-year (2012–2017) medical claims data set on the entire Austrian diabetic population (n = 904,032 patients). This data set contains detailed information on prescribed medication (in form of Anatomical Therapeutic Chemical (ATC) codes) and general information on the patients’ date of birth and sex as well as data on hospital stays, including primary and secondary diagnoses (ICD-10 codes).
To ensure the comparability of the health status of our study population, we restricted the analysis to patients who are free of any relevant preexisting conditions before 2003. The patients with a diagnosis (range A00 to N99) during a hospital stay from 1997 to 2002 (in Austria) were excluded from the analysis. We extracted all DM patients, which are all patients with at least one diagnosis from the range E10 to E14 in the years 2003–2014 (main or side diagnosis), and age-matched controls in the ratio 1:3. We selected DM patients via the ICD 10 codes E10 to E14, which consists by large of T2DM but also includes, e.g., type 1 diabetes mellitus. However, as we only used patients which were diagnosed after the age of 40; almost all were T2DM patients except for a negligible number of patients with rare forms of DM, e.g., latent autoimmune diabetes in adults (LADA).
Patients with PD were identified with the ICD10 code G20 (main or side diagnosis). We then proceeded to calculate odds ratios for being diagnosed with PD in DM patients and the control group in the age group of 40 to 79 years and 80 + years. The gender gap was calculated as the ratio between these odds ratios for women and men.
A series of sensitivity analyses were conducted to assess the robustness and potential confounders of our results by excluding patients with diagnoses potentially promoting or preventing the development of PD (anxiety disorder (ICD-10 code F41) [19, 20], nicotine dependence (F17) [21], alcohol dependence (F10) [22], major depressive disorder (F32, F33) [19, 23], hypertension (I10) [19], hypercholesterolemia (E78) [24], and vitamin D deficiency (E55)) [25–27]. By excluding these patients, we were able to determine whether the observed relations between PD and DM are confounded by any of these diagnoses.
We additionally analyzed the patients’ prescribed medication using the second data set on medication for the years 2012 to 2014. We were especially interested in prescriptions of commonly used antidiabetic drugs like insulins (ATC codes starting A10A) or other blood glucose-lowering drugs (ATC codes starting A10B). We performed a sex-specific 1:3 case-control age-matching which, analogously as described above, is based on patients’ hospital diagnoses (primary and secondary) for DM (ICD-10 code E10–E14) and PD (ICD-10 code G20). The significance of sex-specific differences in proportions between cases and controls was assessed using Chi-squared tests.
To identify which disease is diagnosed first, we calculated the time difference between the first diagnosis of DM and the first diagnosis of PD for each patient over the period 2003–2014. Based on time differences between DM and PD, we grouped patients into four groups: (i) both diseases are diagnosed during the same hospital stay (time difference smaller than seven days) and the time difference is (ii) less than three months, (iii) greater than three months, and smaller than approximately one year (360 days) or, finally, (iv) greater than approximately one year. We proceeded to calculate the time order ratio TOR(DM⟶PD) = N(DM⟶PD)/N(PD⟶DM) and tested the null hypothesis that N(DM⟶PD) =N(PD⟶DM) to evaluate whether TOR(DM⟶PD) is significantly different from 1, assuming that both counts stem from a binomial distribution with equal success probability.
To analyze the time difference between the diagnosis of DM and the diagnosis of PD, we extracted only diabetic patients who got diagnosed with PD after DM. The time difference is defined as the difference between the time of the first diagnosis of DM and the time of the first diagnosis of PD. Kolmogorov-Smirnov test is performed on time difference for females and males to reject the null hypothesis that the time difference between females and males was drawn from the same continuous distribution.
RESULTS
Baseline characteristics
We identified a total of 235,268 diabetic patients (107,694 females, 127,574 males) and 1,938,173 controls (1,005,436 females, 932,737 males) in the age groups 40 to 79 and 80+ years in total. In the age group 40 to 79 years, as visible in Table 1, 1.71%of all patients with DM had been diagnosed with PD (1.8%of women, 1.65%of men). In the age-matched control group, 1.06% of all patients had received the diagnosis PD (1.03% women, 1.08% men). Patients suffering from DM had more hospital days, stays, a higher total number of diagnoses and were more likely to be diagnosed with risk factors for PD such as nicotine dependence, major depressive disorder, anxiety disorder, hypertension, vitamin D deficiency, and disorders of lipoprotein metabolism and other lipidemias when compared to the non-diabetic control group. Female patients with DM were older and had on average slightly more diagnoses and hospital days than their male counterparts.
Baseline characteristics and prevalence (in %) of Parkinson’s disease (PD) and possible confounding factors for PD amongst all patients between the age of 40 and 79 years with and without diabetes mellitus in Austria from 2003 to 2014
Impact of DM on the gender gap in PD
As illustrated in Fig. 1, male patients are more likely to be diagnosed with PD in all age groups. However, women displayed a higher relative risk increase for developing PD than men compared to the non-diabetic control group. Simply put, the gender gap in PD is smaller in the diabetes cohort than in non-diabetic controls (see Fig. 1). Here, female DM patients are at an increased risk to be diagnosed with PD compared to non-diabetic women (OR = 1.71, 95%CI 1.60–1.82, p < 0.001) whereas men with DM only have a 1.46 times higher risk when compared to non-diabetic men (95%CI 1.38–1.54, p < 0.001), which is significantly lower than in women. In other words, women with DM showed a gender gap with a 1.17-fold (95%CI 1.11–1.30) increased risk for the diagnosis of PD when compared to male patients with DM between the age of 40 and 79 years. Female DM patients older than 79 years old are at an increased risk to be diagnosed with PD compared to non-diabetic women (OR = 1.39 95%CI 1.33–1.46, p < 0.001), men with DM when compared to non-diabetic men have 1.28 times increased odds ratio (OR) to be diagnosed with PD (CI 1.20–1.37, p < 0.0001). The gender gap is GG = 1.09 (95%CI 1.01–1.18).
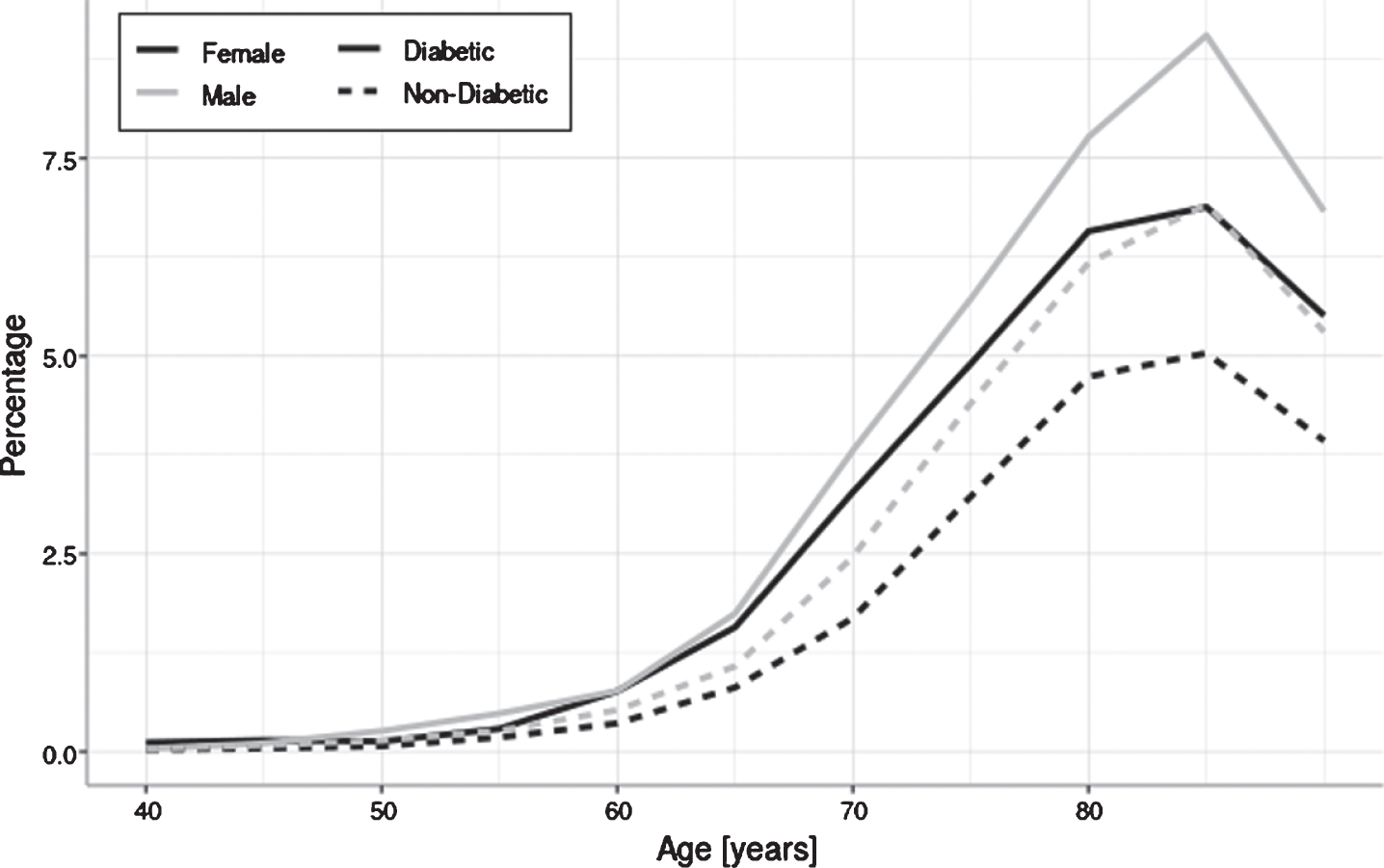
Prevalence of PD throughout life (in %). Percentage of DM patients diagnosed with PD compared to non-diabetic patients: the gender gap of female and male patients suffering from PD is larger in the DM cohort (female patients with DM: OR = 1.71, 95%CI 1.60–1.82, p < 0.001; male patients with DM: OR = 1.46, 95%CI 1.38–1.54, p < 0.001). In the population without DM, nondiabetic men are 1.3 times more likely to be diagnosed with PD than nondiabetic women in the age group 40–79.
To investigate possible confounding factors for the increased relative risk for PD amongst women with DM, we conducted a sensitivity test (Fig. 2) on diagnoses that potentially influence PD risk. Neither anxiety disorder (ICD-10 code F41), nicotine dependence (F17), alcohol dependence (F10), major depressive disorder (F32, F33), hypertension (I10), hypercholesterolemia (E78), nor vitamin D deficiency (E55) affected the increased relative risk for female DM patients to be diagnosed with PD.
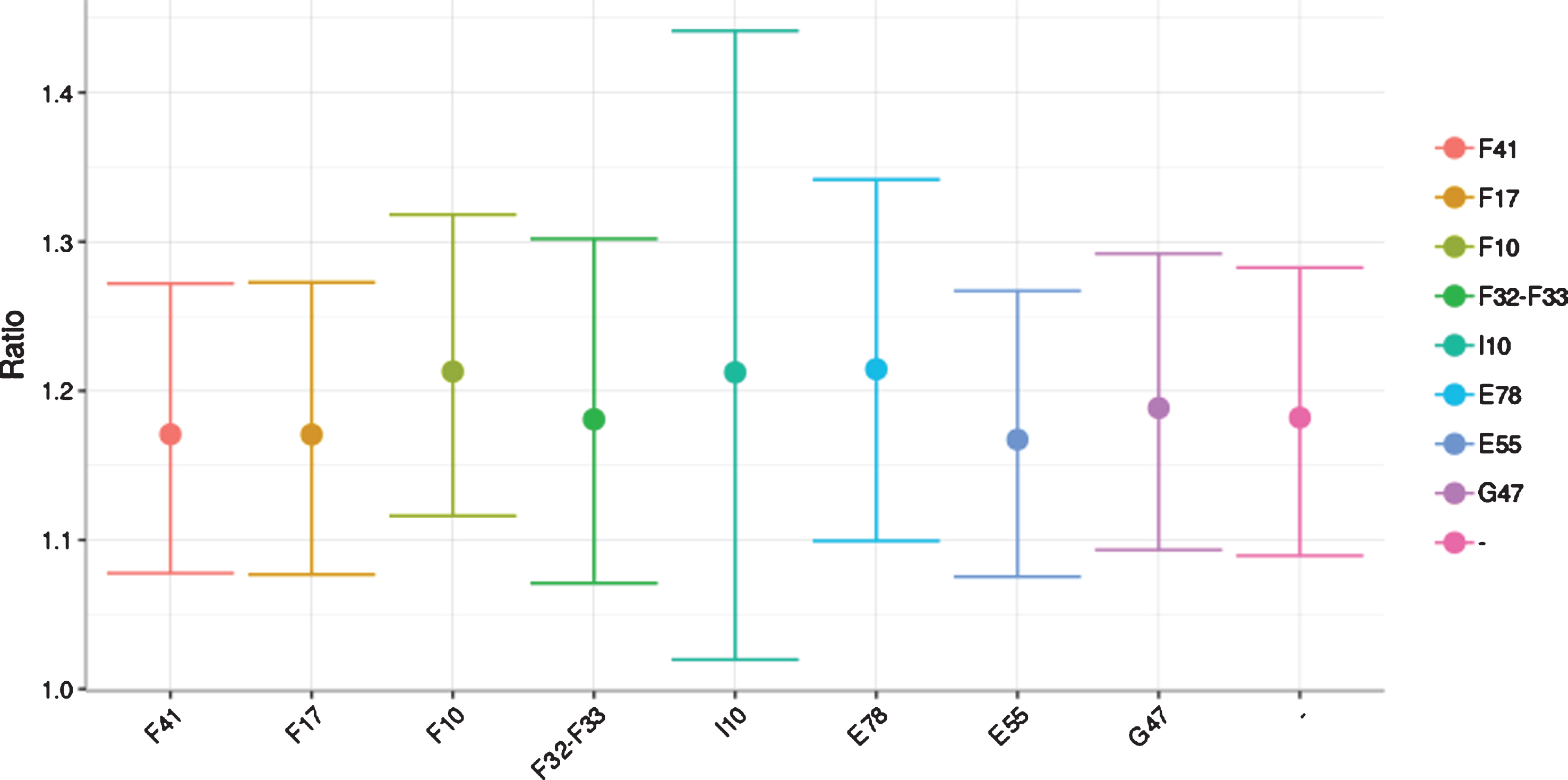
Sensitivity test investigating confounding factors for the increased relative risk for PD amongst female DM patients: anxiety disorder (F41), nicotine dependence (F17), alcohol dependence (F10), major depressive disorder (F32, F33), hypertension (I10), hypercholesterolemia (E78), vitamin D deficiency (E55), and G47 (Sleep disorders) did not affect the results. The pink line represents the results unadjusted for confounding factors.
To identify which disease is diagnosed first, we investigated the time directionality (see Methods and Fig. 3) for patients with a diagnosis of diabetes (E10–E14) and PD(G20). The larger the time difference between these two diagnoses, the stronger the dominance of patients first having a diabetes diagnosis (TOR > 1). DM is typically diagnosed before PD. Males and females showed no significant differences regarding their time-ordering of diagnoses (p = 0.29).
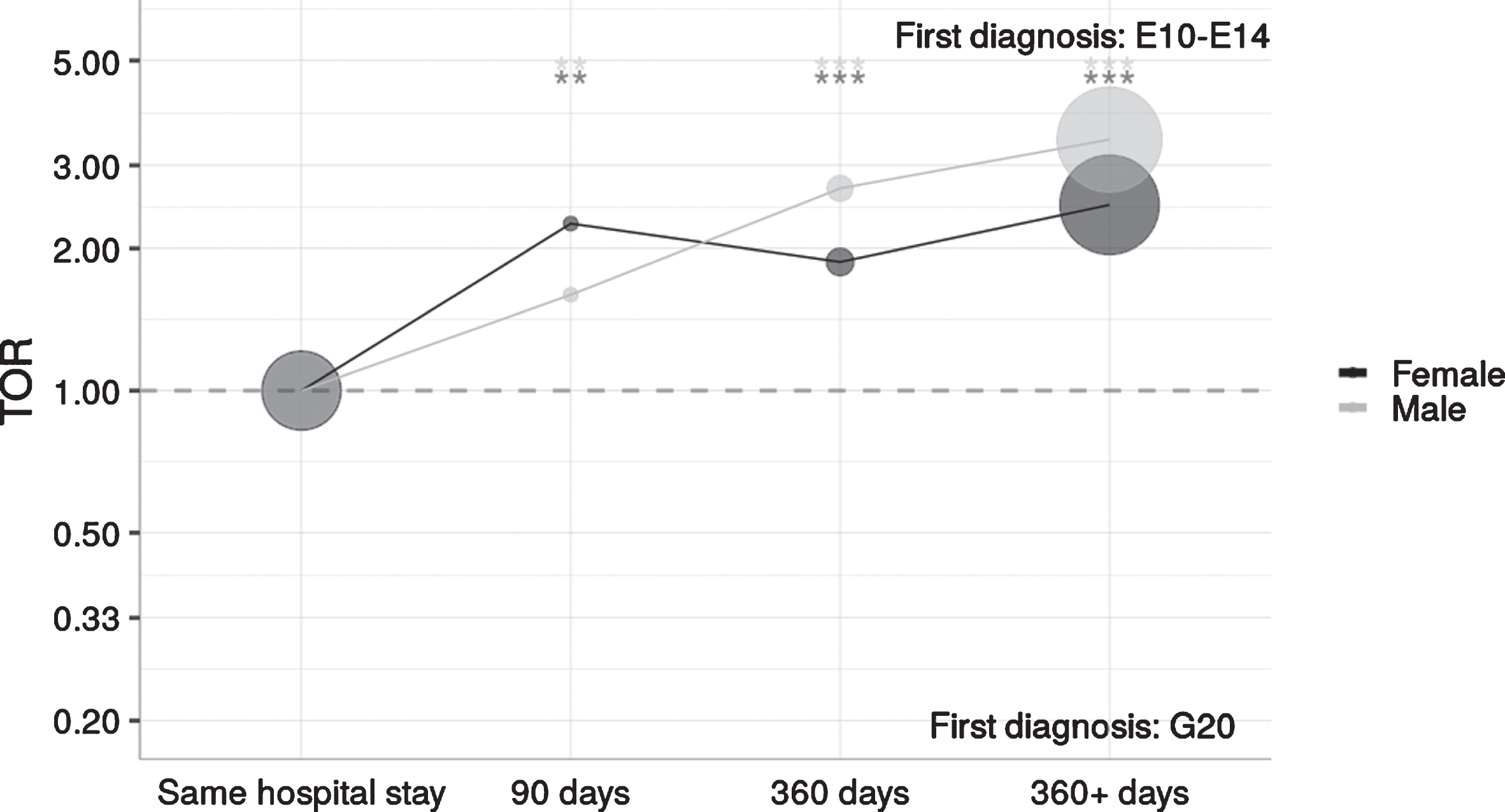
Time directionality for patients with DM (E10–E14) and PD (G20): DM is typically diagnosed before PD. The larger the time difference between these two diagnoses, the stronger the dominance of patients first being diagnosed with DM (TOR > 1). Significance levels of the TOR are indicated by asterisks (*p < 0.05, **p < 0.01, ***p < 0.0001).
A summary of case and control patients’ prescribed medications can be seen in Table 2. There were no significant differences between the PD group and the control group in the number of patients receiving insulin treatment (men: OR = 0.99, p = 0.90; women: OR = 0.92, p = 0.86) indicating no difference in DM disease severity between the two groups. Women with DM and PD were significantly more likely to take biguanides such as metformin than their controls without DM (OR = 1.27, p < 0.05). Sulfonylureas (OR = 0.92, p < 0.05), DPP4-inhibitors (OR = 0.73, p < 0.01), and combinations of oral blood glucose-lowering drugs, mostly biguanides with either sulfonylureas, glitazones or DPP4-inhibitors (OR = 0.66, p < 0.01) were associated with a significantly lower OR to develop PD in women. Glitazones were more often taken by men without PD.
Overview of prescribed medication in both case and control group for both sexes. The proportion (%) of patients with prescriptions of the specific ATC codes is given for each sex in both groups. P-values (p-val) from Chi-squared statistics indicate the significance of differences between groups. Odds ratios (OR) describe the ratio of the odds of being in the case group if exposed to some given medication to the odds of being in the control group if exposed to the same medication
DISCUSSION
We aimed at investigating the effect of DM on the gender gap in PD. This is the first study showing that DM is a stronger risk factor for PD in women than in men. Women with DM display an increased relative risk for PD.
Previous studies have shown increased rates of PD in patients with T2DM [1, 2]. Although men have a higher incidence rate in PD than women [6], women are often more negatively impacted by DM and its complications than men [16–18], which we were able to show for PD in this analysis as well. In general, the exact relationship of PD risk with DM remains unclear. In genome-wide association studies, more than 400 genes, which are linked to both diseases, have been identified using integrative network analysis [28], In older patients, the association of DM and PD might be provoked by disrupted insulin signaling in combination with lifestyle and environmental risk factors [29], Whether dysregulated insulin signaling is the cause or rather the consequence of neurodegeneration in PD remains elusive [12], Chronic levodopa treatment, though, has been shown to decrease glucose tolerance [30], Further, the restoration of brain insulin signaling via anti-diabetic medications might have neuroprotective effects [30], To investigate a potential beneficial effect of oral antidiabetics on PD risk in our database, we calculated the prevalence and ORs of common medications in the group of DM patients with versus without PD and found hints towards sex and gender differences. Female patients taking sulfonylureas, DPP-4 inhibitors, and combinations of oral blood glucose-lowering drugs (biguanides combined with either sulfonylureas/DPP-4 inhibitors/glitazones), but not glitazones, had a lower OR to develop PD, which is in concordance with previous studies showing a beneficial effect of DPP-4 inhibitors, GLP-1 agonists and the combination of metformin and sulfonylureas but not for glitazones [13, 31], Research on glitazones and PD risk is conflicted with some studies reporting a positive influence [32] whereas others dismiss any protective effect [14], In our study, male patients had a lower OR to develop PD when prescribed glitazones but not any other oral antidiabetic medication which is partly in conflict with previously published studies [13, 31], Biguanides alone were more often taken amongst female DM patients who developed PD in our study. Potential explanations include metformin being prescribed due to its known neuroprotective properties [33] or that patients who are only taking biguanides might have been inadequately treated for their DM and, thus, had an increased risk for PD. Complicated T2DM and a long DM disease duration increased the risk to develop PD in previous studies [29], In our dataset, we observed that female diabetes patients had more hospital diagnoses and hospital days than diabetic males. This could be due to women seeking medical help more often than men [34, 35] or it might indicate higher disease severity and could confound the results. Insulin dependency in T2DM is connected to longer disease duration and the insulin prescription rate was not different between the group who developed PD and those who did not. Therefore, DM disease duration is, most probably, not a confounding factor in our analysis.
To investigate other potential confounders for the smaller gender gap amongst DM patients, a sensitivity test on suspected covariates for PD was conducted [19], Neither a lack of smoking history [21], anxiety disorder [19, 20], alcohol dependence [22], major depressive disorder [19, 23], hypertension [19], hypercholesterolemia [24], nor vitamin D deficiency [25–27] had a significant impact on our results.
A limitation and strength of this study at the same time are the datasets themselves. We had access to hospital diagnoses of the whole Austrian population from 1997 to 2014, which is a large cohort with a high number of patients. However, due to the character of the dataset, outpatient visits were not recorded. Patients had to have been admitted to a hospital due to DM or any other disease at least once to be included in the analysis. We split the analysis into two age groups as we observed decreasing diagnose frequencies in patients > 80 years which makes the data less reliable. Furthermore, we were not able to correct the results for the potential confounder weight as this variable was not recorded in the database we analyzed. Moreover, we had no access to data on GLP-1 agonists and SGTL-2 inhibitors, which were registered from 2016 onwards, whereas our dataset only comprises the years 2003 until 2014. Due to the retrospective character of the dataset, we can only show tendencies and associations rather than causation; further prospective studies are needed to investigate the effects of antidiabetics on PD risk in greater depth. However, we were able to demonstrate via a time directionality analysis that DM was diagnosed before PD and, thus, was a risk factor for PD and not vice versa.
In conclusion, our results suggest that DM is a stronger risk factor for the diagnosis of PD in women than in men. Further prospective investigations are pivotal to assess the effect of DM on PD risk in more detail, especially including sex-specific prospective investigations on potential benefits of antidiabetic medication on PD risk. With this work, we aim at raising awareness of the complications of DM and their respective sex and gender-specific differences. This seems especially important in a condition such as PD, which predominantly affects men, but might be associated with a relative risk increase in female DM patients.
CONFLICT OF INTEREST
All authors have no relevant conflict of interest to disclose.
ETHICS
We made secondary use of a research database of medical claims records that are safeguarded and maintained by the Main Association of Austrian Social Security Institutions (HVB) and where it has been ensured that no individual is identifiable. This is a consolidated research database that is only available to selected partners under a strict data protection policy. Use of the data takes place in agreement and cooperation with HVB. The data contains no names of individual patients or health care providers. All unique identifiers including postal codes and date of birth have been removed. All team members working with the data have signed a confidentiality agreement and declaration of commitment ensuring that research will be undertaken in accordance with the applicable data protection regulations. Security clearance for this database signed by MUV’s legal department stating that individuals are identifiable can be provided upon request from the corresponding author.
Footnotes
ACKNOWLEDGMENTS
Funding was received from the WWTF –Vienna Science and Technology Fund (MA16–045).
