Abstract
Background:
It is known that music influences gait parameters in Parkinson’s disease (PD). However, it remains unclear whether this effect is merely due to temporal aspects of music (rhythm and tempo) or other musical parameters.
Objective:
To examine the influence of pleasant and unpleasant music on spatiotemporal gait parameters in PD, while controlling for rhythmic aspects of the musical signal.
Methods:
We measured spatiotemporal gait parameters of 18 patients suffering from mild PD (50%men, mean±SD age of 64±6 years; mean disease duration of 6±5 years; mean Unified PD Rating scale [UPDRS] motor score of 15±7) who listened to eight different pieces of music. Music pieces varied in harmonic consonance/dissonance to create the experience of pleasant/unpleasant feelings. To measure gait parameters, we used an established analysis of spatiotemporal gait, which consists of a walkway containing pressure-receptive sensors (GAITRite®). Repeated measures analyses of variance were used to evaluate effects of auditory stimuli. In addition, linear regression was used to evaluate effects of valence on gait.
Results:
Sensory dissonance modulated spatiotemporal and spatial gait parameters, namely velocity and stride length, while temporal gait parameters (cadence, swing duration) were not affected. In contrast, valence in music as perceived by patients was not associated with gait parameters. Motor and musical abilities did not relevantly influence the modulation of gait by auditory stimuli.
Conclusion:
Our observations suggest that dissonant music negatively affects particularly spatial gait parameters in PD by yet unknown mechanisms, but putatively through increased cognitive interference reducing attention in auditory cueing.
INTRODUCTION
In Parkinson’s disease (PD), auditory entrainment is more effective in positively influencing gait as compared to visual or tactile stimulation [1]. One app-lication of auditory entrainment is rhythmical auditory cueing, which seems to regulate internal timing that is deficient in PD [2–4]. Rhythmical auditory cueing has been suggested to work as an external time keeper [5]. Indeed, beneficial auditory cueing effects on gait parameters have been reported in several studies [6, 7]. Perceiving a metronome can modulate walking parameters such as velocity [8–10], stride length [9] and cadence [9, 11]. Moreover, a metron-ome embedded in music also positively affects spatiotemporal gait parameters such as velocity and stride length [5, 13] but also cadence [5, 12]. Al-though gait improvement via rhythmic stimulation in PD seems to be linked to rhythmic skills such as synchronization performance and low synchronization variability [14], it is still unclear whether only rhythm mediates positive effects of music on gait in PD or whether other features of music also play a role. The latter seems plausible given that music has been reported to support entrainment better than metro-nomes at medium and fast tempi in PD patients [15].
The aesthetic appreciation of a music, so called positive valence, underlies the motivation to listen to music. In music, perceived valence is commonly modulated by a variation of consonance/dissonance [16, 17]: Consonant harmony (i.e., high agreement of harmonic series) is generally perceived as pleasant whereas dissonant harmony (i.e., poor agreement of harmonics) is perceived as unpleasant. This is true even for participants unfamiliar with the type of music tested, for example ‘Western’ music [18], suggesting that some aspects of the valence of music may be hard-wired in the human auditory pathway. Most studies investigating change in gait parameters in response to music in PD patients used musical stimuli that matched individual patients’ preferences [5, 20]. It remains unclear if the perceived valence of musical stimuli has an influence on gait. Perceived pleasantness in music has been reported to correlate with endogenous dopamine release in the striatum [21]. Given that PD patients suffer from motor deficits that correspond to dopaminergic striatal depletion [22], it is plausible that perceived pleasantness in music, which seems to be positively correlated with dopamine release, has beneficial effects on motor performance and thus on gait kinematics in PD. However, a questionnaire study revealed that perception of motor symptoms in PD is only rarely improved by music [23].
Here we aimed here at examining the influence of valence in music on spatiotemporal gait parameters in PD patients, while controlling for rhythmic aspects of the musical signal. We hypothesized that (1) valence will modulate gait in PD, music perceived as pleasant will improve gait parameters when compared to unpleasant music, (2) music perceived as pleasant will improve spatiotemporal gait parameters compared to a beat condition, (3) music perceived to be unpleasant will deteriorate gait parameters compared to silence as it is assumed to function as an additional cognitive burden causing secondary task effects and (4) beat will improve gait parameters compared to the silence condition because of rhythmical auditory cueing effects. To dissect the various effects of the music/auditory stimuli on gait parameters, we herein differentiate harmony (consonant vs. dissonant music) from emotional valence (pleasantness/unpleasantness) as the individual perception of the presented music by study participants.
METHODS
Study participants
Participants of all ages were recruited at the Movement Disorder Outpatient Clinic at the Technische Universität Dresden between March and September 2013. The following eligibility criteria were used: idiopathic PD according to the United Kingdom PD Society Brain Bank Criteria [24], disease duration of at least one year and a disease stage of I–III (according to Hoehn & Yahr [25]): Patients thus had the ability to walk without an assistive device. Furthermore, the absence of atypical and secondary Parkinsonian syndromes, drug or alcohol abuse, and neuroleptic medication were required. Further exclusion criteria were: Severe hearing deficits, presence of dementia (Montreal Cognitive Assessment (MoCA) score of ≤26 [26] and depression (Beck depression Inventory (BDI-II) score ≥13 [27]). Anti-Parkinsonian medication was required to have been constant for at least four weeks prior to the start of the study and had to be maintained at the usual dose throughout the study. The study was approved by the local institutional review boards at the University of Leipzig and the Technische Universität Dresden. All participants provided written informed consent and the study was approved by institutional review boards at participating sites.
Clinical and neuropsychological assessments
Prior to the experiment, we assessed basic demographic and clinical data including Hoehn and Yahr stage [25], Unified PD Rating Scale [UPDRS] part III motor score [28, 29], depression (BDI-II) [27, 30], and cognitive impairment (MoCA) [26]. Furthermore, executive function (Trail Making Test Part A and B [31, 32]), and musical ability (Montreal Battery for the Evaluation of Amusia [MBEA], published cut-off for amusia is <20 [33]) were assessed to examine their influence on the modulation of gait parameters by perceived valence in music. Levodopa equivalent daily doses for each patient were calculated from the current PD medication according to Tomlinson and co-workers [34].
Music stimuli and experimental design
Three stimulus categories were generated based on eight musical excerpts used in our experiment: (1) a beat without melody, (2) the same beat embedded in consonant music, and (3) the same beat embedded in dissonant music, yielding 24 musical pieces in total. The consonant and dissonant music was identical with regard to tempo and dynamic. Patients received wireless headphones and listened to the first 15 seconds of each of the 24 musical pieces. They were then asked to rate the valence of the musical piece using a 12 cm long computer slider interface by indicating the subjective feeling of pleasantness/unpleasantness evoked by each stimulus. Patients pressed a button to confirm their evaluation and to start the next trial.
After this, patients were instructed to walk at a self-selected pace for 45 seconds, either in silence (baseline) or during musical stimulation, in a room specifically designed for an optimized experimental procedure (e.g., quiet condition, no visual cues). To measure the gait parameters, we used an established analysis system for spatiotemporal gait (GAITRite® [35]), which consisted of a walkway of approximately 10-meter length containing pressure-receptive sensors connected to a computer. Gait parameters were measured and defined as described in the GAITRite Electronic Walkway Technical Reference (WI-02-15) [36] (for details, see Supplementary Methods).
Musical stimulation was presented in four blocks comprising six musical stimuli, two of each category (consonance, dissonance, beat; see Fig. 1). The order of the musical pieces was determined by a constraint randomization, under the constraints that each block included two stimuli of each category, and that each half of the experiment was in itself balanced (such that four of the eight excerpts occurred in all category variations). The latter constraint was included to allow for partial data analysis, in the case that patients were unable to continue with the experiment after the second break. Each block was preceded and followed by a silence condition (Fig. 1).
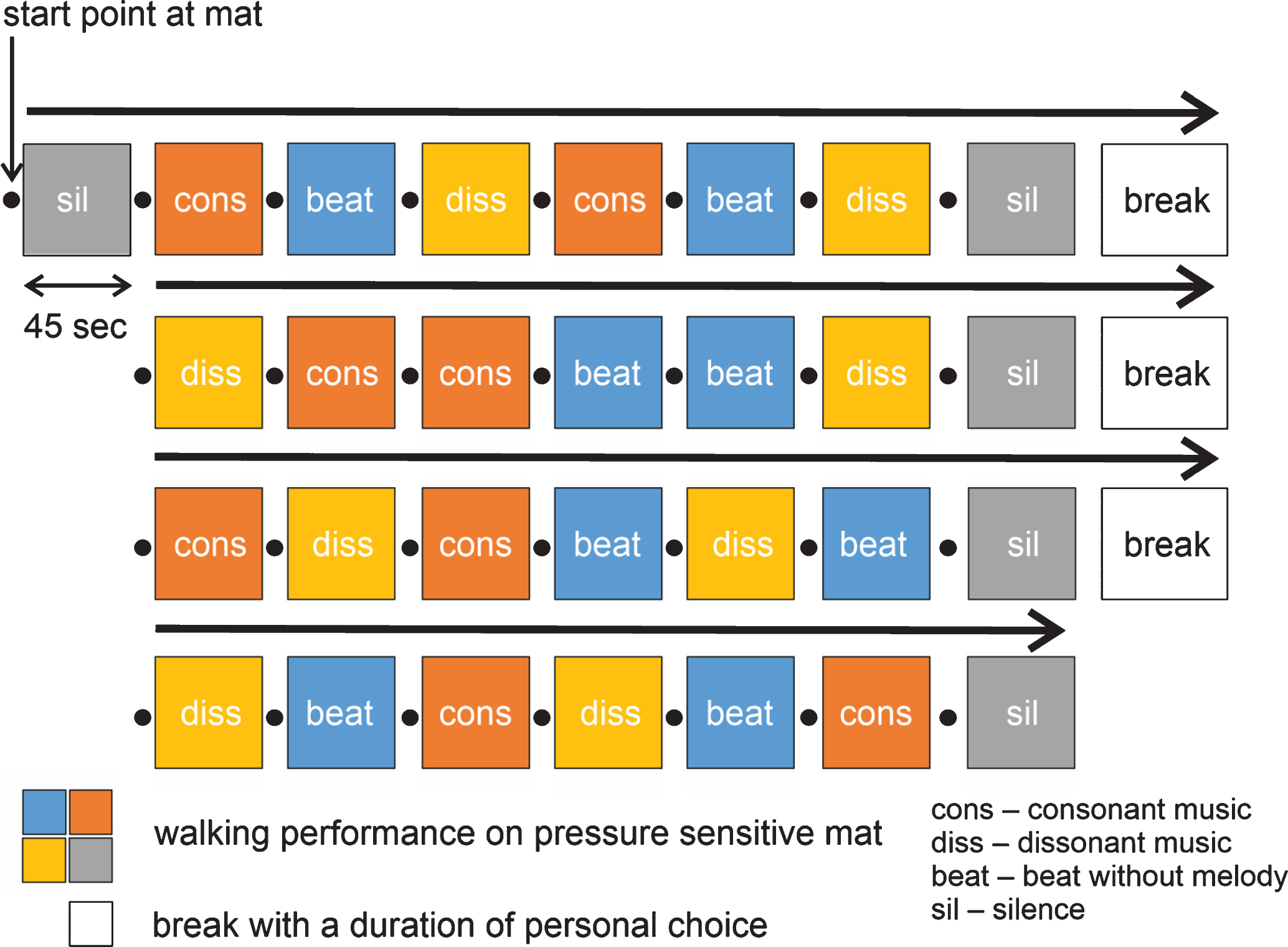
Experimental design.
In order to control for a deterioration of performance due to drop out effects, half of the participants performed the experimental procedure with a version of the stimulus sequence, where the musical pieces were presented in reverse order. The version of stimulus sequence was randomly assigned to each participant. The experimenter was blinded with respect to experimental condition, achieved by participants listening to the presented music over headphones. As this study is the first one to study the influence of musical valence on gait in PD patients, the sample size could not be calculated on the basis of effect sizes reported in previous studies. The determination of sample size was therefore based on studies with similar research questions and experimental designs which investigated gait performance using auditory stimulation by music (on 15–21 patients) [5, 20].
Statistical analysis
The study was designed as a within-participant paradigm. A repeated-measures analysis of variance (ANOVA) was carried out on gait parameters (stride length, cadence, swing time, and velocity) as dependent measures and music categories (beat, consonant, dissonant, silence) as independent variables. The test of the model assumptions showed a normal distribution as assessed by the Kolmogorov Smirnov test and sphericity as assessed by Mauchly’s test. In cases where sphericity was not given, we used the Greenhouse-Geisser correction. To investigate whether the overall effects of all music stimuli or the differential effects of the various music stimuli on gait parameters are modulated by motor or musical abilities, we computed a repeated measures analysis of covariance (ANCOVA) using the variables UPDRS and/or MBEA as continuous covariates. Post-hoc two-sided paired t-tests or repeated measures ANCOVAs were adjusted using the Bonferroni methods to avoid alpha error accumulation in multiple comparisons (in accordance with the four hypotheses tested in the experiment). Correlations of valence as rated by patients and gait parameters were assessed using Pearson’s correlation test and multivariate linear regression analyses as appropriate. Values are presented as means±standard deviation (SD) or median and interquartile range (IQR), as appropriate. p values <0.05 were considered statistically significant, and effect sizes Cohen’s d were additionally reported.
RESULTS
Study participants
In accordance with the eligibility criteria, a total of 18 PD patients (9 [50%] men) were enrolled into this study (for details on demographics and clinical data, see Table 1). Mean±SD age was 64.4±6.0 (IQR: 59–70) years with a mean disease duration of 6.1±5.0 (IQR: 2–15) years. The median Hoehn & Yahr stage was 2 (IQR: 1.5 to 2) and mean UPDRS part III motor score was 15.1±6.6 (IQR: 11–22). Most of the patients had a tremor-dominant phenotype, with the left side being slightly more affected than the right side. All patients were right-handed and wore glasses to correct vision if required; no patient required a hearing aid. Four patients had amusia as evaluated by the MBEA [33] and executive functions as measured by the Trail Making Test Part A and B were judged as not impaired [31, 32] (see Table 1 for details).
Demographic and clinical data of study cohort
Data are mean (SD), median (IQR), or number (%). Levodopa equivalent doses were calculated according to Tomlinson et al. (2010) [34]. BDI, Beck’s Depression Inventory; COMT, Catechol-O-methyltransferase; IQR, interquartile range); MAO-B, Monoaminoxidase B; MoCA, Montreal Cognitive Assessment; UPDRS, Unified PD Rating Scale. $Normative values of Trail Making Tests A and B generated from 287 normal adults covering the age range of our patient cohort as published by Giovagnoli and co-workers [32].
Valence of music stimuli
In order to test the effects of the music categories, a repeated measures ANOVA of the rated pleasantness was carried out. We found that patients perceived a significant difference between the three musical versions (F(2,30) = 118.6, p < 0.001). As shown in Fig. 2, the consonant versions were rated as the most pleasant, whereas dissonant versions were judged to be the least pleasant. The rhythmic beat was rated as neutral.
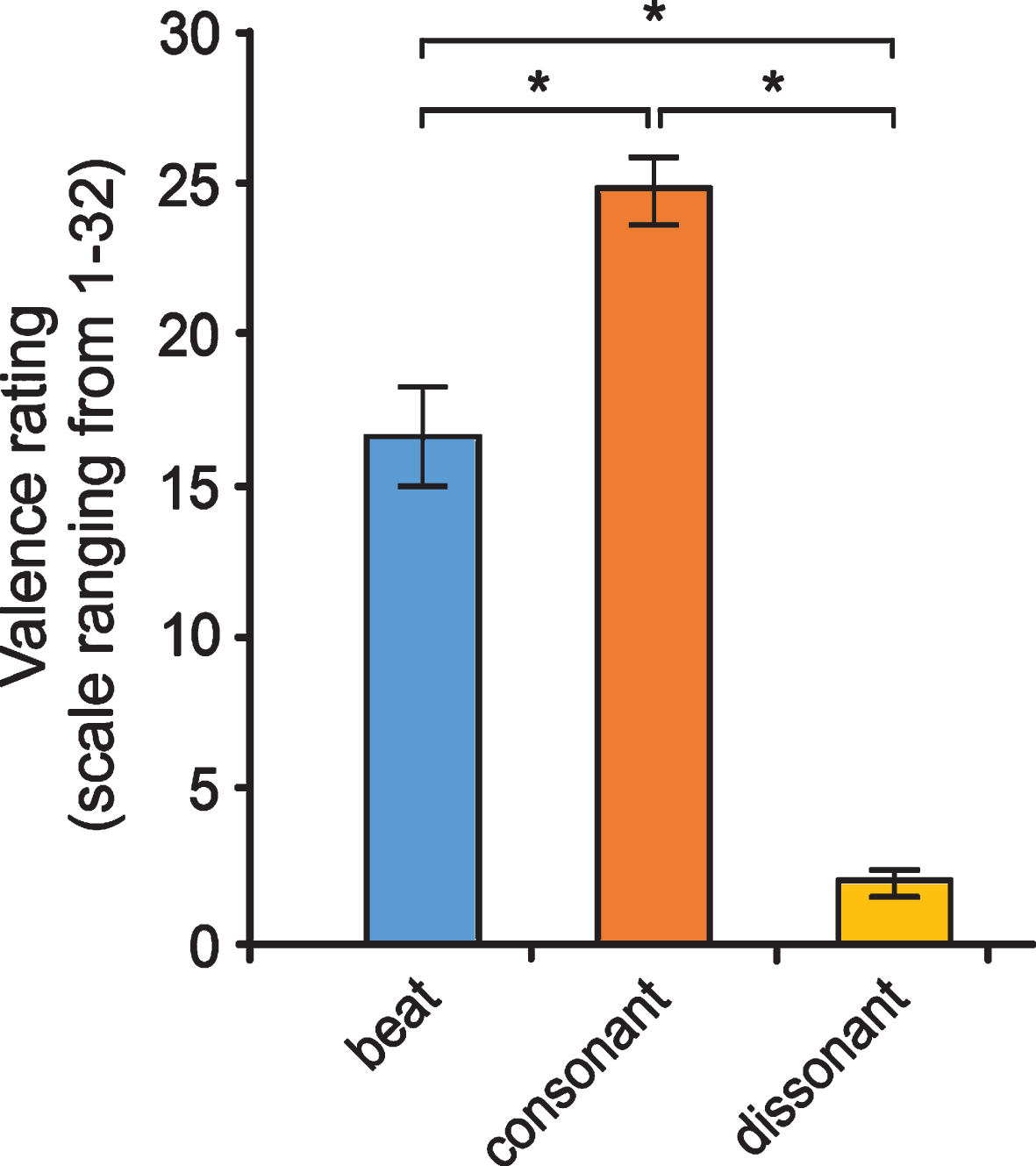
Valence ratings of the three music versions by Parkinson’s disease patients. Post-hoc tests using a Bonferroni correction showed that consonant music was perceived as significantly more pleasant than the beat (p = 0.001), while dissonant music was rated significantly more unpleasant as beat (p < 0.001) and consonant music (p < 0.001). Note: Ratings are based on a 32-point scale. Data are presented as mean values±SD.
Baseline gait parameters in silence
The first trial in silence was used to assess the baseline gait parameters in our cohort of PD patients. The average gait velocity was 104.9±14.9 cm/s, stride length was 114.4±13.6 cm, cadence was 110.8±10.2 steps/min and swing time 0.40±0.03 s. A comparison with normal values of healthy pro-bands [37] showed that gait performance of PD patients in our sample was significantly lower: tvelocity(17) = –4.52, p < 0.001; tsteplength (17) = –6.86, p < 0.001; tcadence(17) = –4.06, p = 0.001.
Overall effects of different music stimuli on gait parameters
We detected overall effects of the three music sti-muli and silence for the parameters velocity (Fvelocity (3,37) = 5.25, p = 0.006), and stride length (Fstridelength (3,37) = 7.49, p = 0.001), while the eff-ects on cadence (Fcadence (2,31) = 3.02, p = 0.067) and swing time (Fswingtime (3,51) = 0.73, p = 0.537) were not significant (Fig. 3). Repeated measures ANCOVAs to assess the influence of the participants’ motor abilities (as measured by UPDRS part III motor scoring) and/or musical abilities (MBEA scoring) on the overall effects of music stimuli revealed that both motor and musical abilities did not significantly affect how music stimuli modulated the spatiotemporal and spatial gait parameters velocity and stride length (Supplementary Table 1). In contrast, UPDRS part III score but not MBEA scores influenced the overall effects of music stimuli on the temporal gait parameters cadence and swing time.
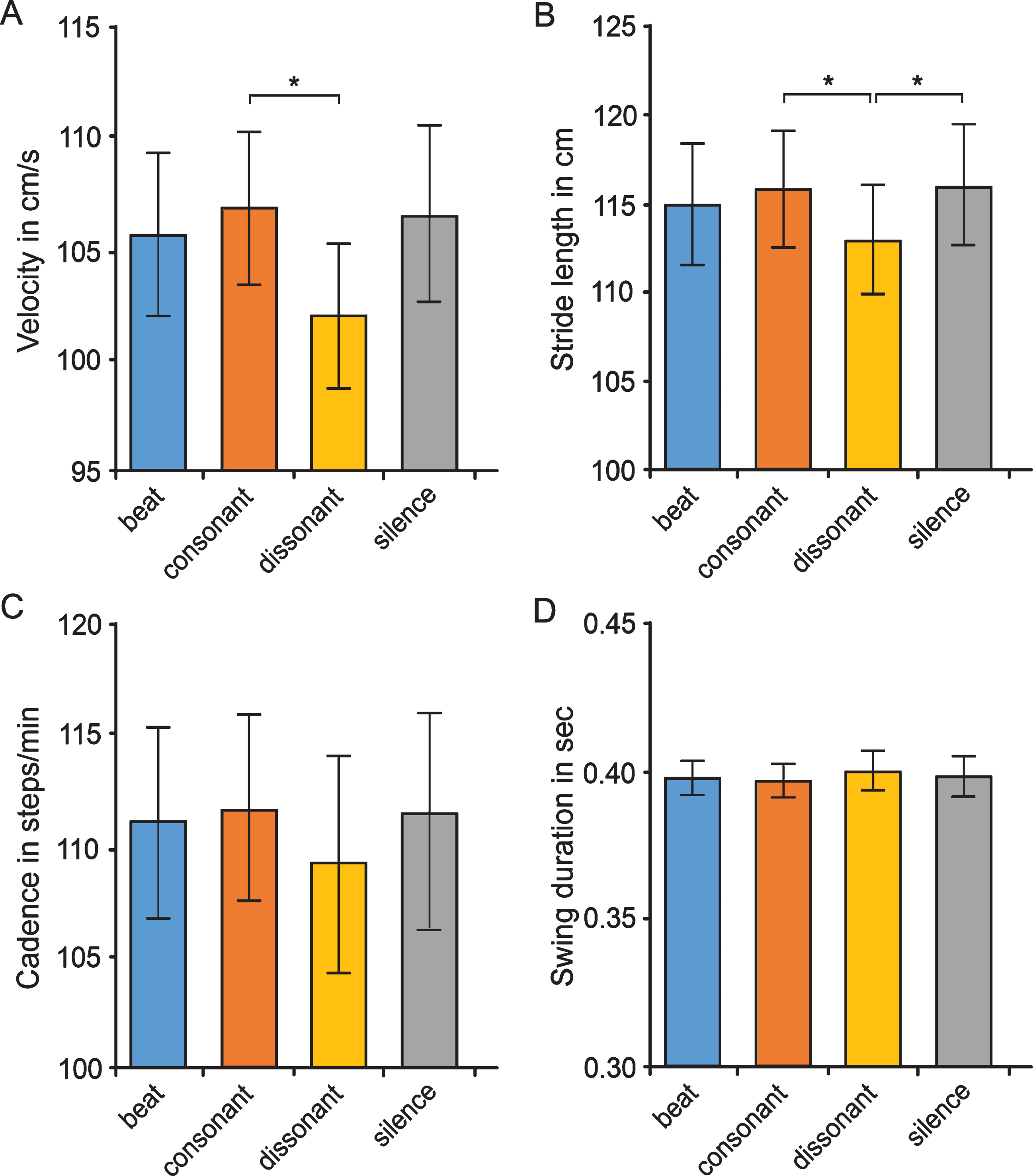
Effects of various auditory stimuli on gait parameters in Parkinson’s disease patients. Displayed are the effects of silence (baseline), beat, consonant and dissonant musical stimuli on spatiotemporal/spatial gait parameters (velocity [A], stride length [B]) and on temporal parameters (cadence [C], swing duration [D]). Post-hoc tests with Bonferroni correction revealed a significant (*) difference between consonant and dissonant music for velocity (p = 0.021; d = 0.3) and stride length (p = 0.026; d = 0.2). Dissonant music was also shown to significantly deteriorate stride length compared to silence (p = 0.020). Data are presented as mean values±SD.
Additional analyses to assess the overall association of valence and gait parameters however did not reveal any significant correlation of valence with the tested gait parameters (r = 0.105–0.237, p > 0.05; Pearson’s correlation test). Multivariate linear regression analyses with type of auditory stimulus, UPDRS and/or MBEA as additional independent variable confirmed Pearson’s correlation tests results (data not shown).
Differential effects of various music stimuli on gait parameters
As displayed in Fig. 3, we observed significant differences between consonant and dissonant music in spatiotemporal and spatial gait parameters with higher velocity (p = 0.021; d = 0.3) and larger stride length (p = 0.026; d = 0.2) with consonant as compared to dissonant music. No significant differences were detected between consonant and dissonant music in temporal gait parameters like cadence and swing time. When comparing the auditory stimulation of dissonant music with silence, we observed a significant difference in stride length with smaller stride length with dissonant music as compared to silence (p = 0.020, d = –0.2). In contrast, the comparisons of beat versus silence or consonant/dissonant music showed no significant differences in any gait parameter (Fig. 3). Repeated measures ANCOVAs with Bonferroni adjustment using the variables UPDRS and/or MBEA as continuous covariates to assess whether the differential effects of the various music stimuli on gait parameters are modulated by motor or musical abilities revealed that both motor and musical abilities did not significantly affect how the various music categories modulated gait parameters.
Further statistical analyses to assess the association of valence in music and gait parameters for the three auditory stimuli separately did not show significant correlations of valence with the tested gait parameters for all three music categories (p > 0.05; Pearson’s correlation test). Multivariate linear regression analyses with motor ability (UPDRS part III motor score) and/or musical ability (MBEA) as additional independent variables confirmed Pearson’s correlation tests results (data not shown).
DISCUSSION
The aim of this study was to investigate the influence of music on gait parameters of PD, apart from the known rhythmical effects of music. As valence (pleasantness/unpleasantness) in music has been previously reported to affect the dopamine system in healthy participants, we hypothesized that valence in music has an effect on gait parameters in PD patients, whose motor symptoms are the result of a dopamine deficiency in the basal ganglia. We thus studied the effects of various musical stimuli (silence as baseline, beat, consonant/dissonant music) on gait parameters in PD patients under highly standardized conditions. It is however important to note that an investigation of a relation of musical valence on dopamine release is not directly addressed in the current study. Moreover, dopamine release in response to music has not been studied and reported with PD patients yet.
Importance of valence for effects of music on gait in PD
As hypothesized in our first hypothesis, we observed that gait performance with respect to velocity (spatiotemporal gait parameter) and stride length (spatial parameter) is better when listening to consonant music than to dissonant counterparts of the same music with the same underlying beat. However, we did not detect a direct association of valence in music and gait. When we differentiate between dopamine sensitive and resistant parameters as suggested in previous studies [38, 39], our data show only effects for parameters that have previously been described as dopamine-sensitive, namely the spatiotemporal and spatial parameters velocity and stride length. Since gait velocity combines both spatial and temporal aspects of gait, it is thus likely that the effects of dissonant music on gait are particularly driven by actions on spatial gait parameters. Although statistical adjustment for motor abilities of the participants (UPDRS part III scores) revealed overall effects of musical stimuli also on dopamine-resistant gait parameters (the temporal parameters cadence, swing duration), these effects did not survive pairwise comparisons of the various auditory stimuli. Conclusively, if there is a modulation of temporal gait parameters by music harmony, these actions need to be considered as minor effects. These findings are in agreement with previous research that demonstrated that dopamine is released in healthy participants during experience of peak emotion to music [21]. However, contrary to our second hypothesis, where we suspected that consonant music would improve gait parameters compared to silence, gait parameters during stimulation with consonant music showed no significant improvement as compared to the silence condition. Although this may signify that valence as presented here by consonant music does not change gait in PD, this might be alternatively accounted for by the fact that the PD patients participating in the current study were in a rather early disease stage (median Hoehn & Yahr stage: 2 [IQR: 1.5–2]) and medicated leading to only mild motor symptoms (mean UPDRS motor score of only 15±7). This is reflected by the high velocity values measured during the baseline (silence) condition (mean velocity was 105 cm/s). A comparison with values of healthy participants [37] showed that gait performance of PD patients in our sample was significantly lower, but largely exceeded those reported in previous studies on PD, which ranged from 70 to 75 cm/s [5, 12]. It might thus be that in our patient cohort the PD motor deficit was relatively low, and, as a consequence, a stimulation by means of consonant music did not have any effects as compared to silence.
Negative effects of dissonant music generally perceived as unpleasant, as hypothesized in the third hypothesis, were observed in velocity and stride length and may correspond to previously described influences of musical timbre features on movement [40]. Our results with no correlations of valence as rated by the participants and gait parameters however point to the possibility that negative effects of dissonant music on spatial gait parameters is mediated by interference between listening to dissonance and motor behavior. It has been argued previously that listening to dissonant music may increase cognitive interference, resulting in a poorer performance in a Stroop interference task [41]. The current results may therefore indicate that similar interference takes place in the motor domain, an influence that may be more observable in PD patients whose motor system is more severely affected. Such large gait deterioration in PD patients as a consequence of interference has previously also been shown in a dual task condition [19]. The current results on pleasant/unpleasant auditory stimuli might therefore correspond to the role of one aspect of cognition, namely attention in auditory cueing, given that cueing strategies in PD have been reported to have large attentional effects [42].
Importance of beat for effects of music on gait in PD
Contrary to what we hypothesized (fourth hypothesis) and from what prior studies on auditory cueing have demonstrated [10, 11], our results show that the beat condition did not have any effects as compared to the silence condition on gait parameters. On the one hand, this can be explained by the fact that the PD patients participating in the current study were rather mildly affected by the disease (see above). Another interpretation of these results is that a metronome as used in the previous studies may have a more beneficial effect for PD patients than the complex rhythms applied in the current investigation. This interpretation is in line with previous assumptions that an easily discernible beat (such as a metronome) may have more beneficial effects in PD than complex rhythms [6], and is further supported by the findings that music with an embedded metronome could improve gait parameters [5, 13], whereas music without a metronome could not [19]. The absence of gait improvement in the beat condition could thus be explained by the fact that the beat consisted of a complex rhythm.
Surprisingly, while a difference in valence was observed between beat that was rated as neutral and consonance that was rated as pleasant, beat and consonant stimulation had similar effects on velocity and stride length (Fig. 3). Those data confirm the notion that valence is not directly related to gait parameters.
Limitations
Our study has several limitations that need to be addressed: Firstly, we did not run a control group consisting of matched healthy participants in parallel to the PD group to directly assess the specificity of the results for PD. Secondly, we investigated a relatively small cohort of rather mildly affected Parkinson’s disease patients with an intervention scheduled at one time point, which limits the possibility to draw definite conclusions for the general population of Parkinson’s disease patients, and is not sufficient to reveal whether or not advantageous effects of valence on gait parameters can be reproduced in an individual patient. Thirdly, the potential involvement of the dopamine system or cognitive interference in the effects of auditory stimuli had not been investigated in the present study. Future studies are thus warranted to dissect the factors mediating the negative effects of dissonant music on gait in PD.
CONCLUSIONS
In conclusion, we were able to demonstrate that effects of music on gait performance in PD patients are not only due to auditory cueing by rhythm as an external timekeeper [5]. The gait of patients walking to music with the same underlying beat correlated to the dissonance of the music, but was not associated with the perceived valence of the auditory stimuli. Together, dissonant auditory stimuli negatively affect particularly spatial gait parameters in PD putatively through increased cognitive interference reducing attention in auditory cueing. However, the mechanisms underlying the effects of musical stimuli on gait performance in PD beyond auditory cueing by easily discernible beats need to be elucidated in future studies.
CONFLICT OF INTEREST
GL has received a scholarship of the German Academic Scholarship Foundation and scholarship of the University of Leipzig. The authors have no conflict of interest to report related to this manuscript.
Footnotes
ACKNOWLEDGMENTS
The authors thank all patients for their participation and the study center personnel at the Department of Neurology at the Technische Universität Dresden. This study was financially supported by the Max Planck Society and the Department of Neurology, Technische Universität Dresden. The financial sponsors of the study had no role in the study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had the final responsibility for the decision to submit for publication.
