Abstract
Background:
Instrumented measures of balance and gait measure more specific balance and gait impairments than clinical rating scales. No prior studies have used objective balance/gait measures to examine associations with ventricular and brain volumes in people with Parkinson’s disease (PD).
Objective:
To test the hypothesis that larger ventricular and smaller cortical and subcortical volumes are associated with impaired balance and gait in people with PD.
Methods:
Regional volumes from structural brain images were included from 96 PD and 50 control subjects. Wearable inertial sensors quantified gait, anticipatory postural adjustments prior to step initiation (APAs), postural responses to a manual push, and standing postural sway on a foam surface. Multiple linear regression models assessed the relationship between brain volumes and balance/gait and their interactions in PD and controls, controlling for sex, age and corrected for multiple comparisons.
Results:
Smaller brainstem and subcortical gray matter volumes were associated with larger sway area in people with PD, but not healthy controls. In contrast, larger ventricle volume was associated with smaller APAs in healthy controls, but not in people with PD. A sub-analysis in PD showed significant interactions between freezers and non-freezers, in several subcortical areas with stride time variability, gait speed and step initiation.
Conclusion:
Our models indicate that smaller subcortical and brainstem volumes may be indicators of standing balance dysfunction in people with PD whereas enlarged ventricles may be related to step initiation difficulties in healthy aging. Also, multiple subcortical region atrophy may be associated with freezing of gait in PD.
INTRODUCTION
Parkinson’s disease (PD) is a neurodegenerative disorder characterized by a multitude of balance and gait disturbances [1]. In fact, gait in PD is characterized by short, shuffling steps, freezing of gait, and difficulty initiating gait. Standing postural sway is also affected by PD, even early in the disease [2, 3]. These disturbances, together with impaired postural responses, lead to impaired balance and gait, falls and poor quality of life [4, 5]. Patterns and severity of balance and gait disfunction vary among patients with PD, as well as among the older adults without neurological disease.
In older adults without overt neurologic disease, slow gait has been associated with ventricular dilation, generalized brain atrophy, and white matter disease [6]. Although ventricular enlargement has not been associated with PD in general, ventricular dilatation has been associated with dementia in people with PD [7] and increased motor impairments in PD as measured by the MDS-UPDRS Part-III scores [8, 9]. However, the relationship between structural changes in the brain and balance and gait dysfunction in PD remains poorly characterized. To our knowledge, no prior studies have used instrumented measures of balance and gait to examine specific aspects of mobility that relate to ventricular dilation or global and regional cortical and subcortical volume atrophy in people with PD.
Balance and gait control depend on both cortical and subcortical structures [10]. However, balance and gait are likely independent functions that may be controlled by different brain regions [11, 12]. Functional MRI studies have shown activations in cortical motor regions, brainstem and cerebellum while performing mental imagery of locomotion both in healthy adults and in people with PD [13, 14]. For balance control, a PET study has shown decreased thalamic cholinergic innervation associated with increased sway measured on a balance platform in people with PD [15]. Freezing of gait in PD has been related to atrophy of subcortical structures, cortical motor areas as well as cortical non-motor areas, include the inferior parietal lobe [16]. Despite the fact that neuroimaging studies show that subcortical and cortical regions are both impacted in PD, either by volumetric changes, loss of neurotransmitters or change in functional connectivity, the relationship between loss of cortical and subcortical gray matter volumes and specific types of balance and gait impairments is unknown. Since gray matter loss in many cortical and subcortical regions is associated with freezing of gait [17], the relationship between brain volumes and balance and gait impairments may differ in freezers and non-freezers. There has been no previous comparison of brain structural changes related to specific aspects of balance and gait in freezers versus non-freezers.
Here, we hypothesize that larger ventricular volume and reduced cortical and subcortical volumes are associated with impaired balance and gait measures in people with PD. Specifically, we predict that impairments of different domains of balance and gait in PD will be associated with atrophy in different brain areas. We also hypothesize that atrophy in different cortical and subcortical regions are associated with different domains of balance and gait in freezing of gait. To objectively characterize multiple domains of balance and gait we used wearable inertial sensors [18] and according to a common clinical test, characterize four domains of balance and gait: gait, anticipatory postural adjustments (APA) prior to gait initiation, automatic postural responses (APR) to external perturbations and postural sway in stance [19].
This study examines the relationship between ventricular size, global and regional brain volumes and objective measures of four domains of balance and gait.
MATERIALS AND METHODS
Study sample
Data were collected from 100 people with PD and 58 healthy control subjects. Subjects with PD had idiopathic PD clinically diagnosed by a movement disorders specialist using UK Brain Bank Criteria [20]. Inclusion criteria for subjects were: 1) between 50 and 90 years old, 2) no major musculoskeletal or peripheral disorders (other than PD) that could significantly affect their balance and gait, and 3) ability to stand, walk unassisted for 20 feet and to follow instructions. Healthy, elderly adults within the same age range were recruited from the community. Exclusion criteria for all groups were: metal implants or any other contraindications for brain imaging. In addition, people with PD and severe tremor were excluded to ensure good quality MRI. All subjects gave written informed consent in accordance with the Declaration of Helsinki. The protocol was approved by the Oregon Health & Science University, OHSU (#4131) and the OHSU/VAPORHCS joint IRB (#8979). These participants were a subset of subjects in a larger interventional study (Clinical Trials NCT02231073 and NCT02236286) and these MRI scans were acquired on average 6 (±13) days before or after the baseline balance and gait testing (median: 2 days, range: 0–105 days).
Out of 158 subjects (healthy controls and people with PD), 12 subjects (4 PD and 8 control subjects) were removed from the imaging data due to issues with registration of native brain to the atlas resulting in poor segmentation of white and gray matter caused by motion artifacts. Out of these 12 subjects, 3 PD and 2 control subjects had larger ventricles, characterized as 1.5 standard deviation from the control mean. Our final sample sizes were 96 people with PD and 50 control subjects (see Table 1 for more details). Of the 96 with PD, 40 had freezing of gait as indicated by the New Freezing of Gait Questionnaire [21].
Demographics and clinical characteristics
*median and inter quartile range was computed instead of mean/SD; PD, Parkinson’s disease; MDS-UPDRS, Movement Disorders Society Unified Parkinson’s Disease Rating Scale; PIGD, 4-items of Postural instability and gait dysfunctions; LEDD, L-dopa equivalent daily dose; H&Y, Hoehn and Yahr; MoCA, Montreal Cognitive Assessment; MiniBESTest, Mini Balance Evaluation Systems Test; ABC, Activity Specific Balance Confidence scale. For group comparisons, t-test/ Kolmogorov–Smirnov/chi-square were performed to test significant differences between groups.
MRI acquisition and processing
T1 weighted images were acquired using MPRAGE sequence using a 3 Tesla Siemens Trio scanner with a 12-channel head coil. T1-weighted image protocol is as follows: TR = 2300 ms, TE =3.58 ms, voxel size = 1 mm×1 mm×1.1 mm, sli-ces = 160, FoV = 256 mm, flip angle = 10 degrees. T2-weighted images were acquired using SPC sequence using the following parameters: TR =3200 ms, TE = 497 ms, resolution = 1 mm isotropic, slices = 160, FoV = 256 mm. Due to an upgrade of MRI scanner during the phase of data collection, part of subjects were scanned with a 3 Tesla Siemens Prisma scanner with a 32-channel head coil. T1-weighted image acquisition protocol: TR = 2500 ms, TE = 2.88 ms, voxel size = 1 mm×1 mm×1 mm, slices = 176, FoV = 256 mm, flip angle = 8 degrees. T2-weighted images were acquired using SPC sequence using the following parameters: TR =3200 ms, TE = 565 ms, resolution = 1 mm isotropic, slices = 176, FoV = 256 mm.
Data was processed using an adapted version of the workflow pipelines from the Human Connectome Project, as reported previously [22–25]. Briefly, denoised T1-weighted and T2-weighted volumes were first aligned to the MNI atlas’ AC-PC axis and then non-linearly registered to the MNI atlas [26–28]. Then, optimally aligned T1-weighted images were segmented using recon-all from FreeSurfer. Segmentations were improved by using the enhanced white matter-pial surface contrast of the T2-weighted image. All volume measures were divided by intracranial volume (ICV) to control for differences in head size. Total ventricular volume was computed as the sum of the lateral (including the inferior lateral), third and fourth ventricular volumes from the FreeSurfer outputs. Subjects were classified as normal or large size ventricle based on mean ventricle volume of healthy controls to select subjects to check for evidence of normal pressure hydrocephalus.
The following brain volumes were extracted from FreeSurfer: Total cortical, subcortical and cerebellar volumes. Right and left cerebellar gray matter volumes from the FreeSurfer outputs were combined to get the total cerebellar gray matter volume. Total subcortical volumes outputs from FreeSurfer consisted of the following regions: sum of right and left putamen, pallidum, caudate, thalamus, hippocampus, accumbens, and amygdala. Bilateral cortical parcellations (Desikan-Killiany) were mapped into four lobes: frontal, parietal, temporal and occipital lobes using the FreeSurfer lobe mapping [29].
Balance and gait assessment outcome measures
Participants underwent instrumented assessment of balance and gait in the laboratory. All data in people with PD were collected in the practical “off” levodopa state, withholding medication for at least 12 h. People with PD were clinically rated by a trained examiner on the Motor Section (III) of the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS). Postural instability and gait dysfunction components were estimated using the sum of 4 posture and gait items (sit-to-stand, standing posture, walking and pull test) of the MDS-UPDRS Part-III. Each participant was outfitted with 8 inertial sensors (APDM wearable technologies, Portland, OR, USA), worn on the sternum, lumbar spine, bilaterally on the wrists, shins, and feet. Each inertial sensor recorded tri-axial accelerations, tri-axial angular velocities, and magnetic field at 128 Hz. Subjects from both cohorts were sometimes unable to complete the balance or gait tasks due to various reasons, including postural instability, fatigue or limited time, or data quality was compromised. Those data were marked as missing.
We chose the balance and gait metrics for the analysis based on our previous study identifying the most reliable, discriminative, nonredundant measures most sensitive to PD compared to age matched control subjects [30]. Best performing metrics from each of the four different domains of balance and gait were chosen: 1) Anticipatory postural adjustments (APA) prior to gait initiation: first step Range of Motion of the leg (N = 127), 2) Automatic postural responses (APR) to a backward external perturbation: step height in vertical direction (N = 114), 3) Postural Sway during quiet stance: root mean square of acceleration time series in anteroposterior direction (Sway RMS AP standing on foam) when standing 30 seconds with eyes open on a foam surface (N = 122), and 4) Dynamic balance during walking (Spatiotemporal gait measures; N = 134). We previously showed that standing on foam with eyes open was the most sensitive and specific metric to PD compared to standing on firm surface or with eyes closed on firm/foam surface [30]. Since gait is considered to have multiple sub-domains of its own [6], the following gait metrics were chosen for pace and turning, rhythm, upper body stability and variability: gait speed, turn velocity, stance time, arm range of motion and gait cycle duration variability (SD), respectively. This study was performed on a subset of subjects from our previous publications (N = 236; [30, 31]) who had high quality brain MR images at baseline (N = 146). For detailed definitions of the metrics and how the metrics were selected, refer to Hasegawa et al. (2019) [30].
Statistical analysis
Using a general linear model, analysis of covariance (ANCOVA) was used to assess the relationship between PD status (PD/Control) and brain volumes (ventricular volumes, global and regional subcortical and cortical brain volumes) with balance and gait metrics, while controlling for covariates. The ANCOVA model was run with group and normalized brain volumes as main effects, in addition to the interaction between group and brain volumes as independent variables. The reported main effects use healthy controls as the reference group and PD as the indicator group. Balance and gait metrics were dependent variables. We controlled for the covariates age and sex since both are known to affect brain volumes [32, 33] and age is known to affect balance and gait metrics [34]. ICV was not included as an additional covariate in the model because the brain volumes were used as a proportion of ICV. A sub-analysis was performed on people with PD. ANCOVA was used to assess whether the relationships between brain volumes with balance and gait metrics differed for freezers versus non-freezers (controlling for age, sex, and disease duration). The reported main effects use non-freezers as the reference group and freezers as the indicator group. Regression analysis between balance and gait metrics and clinical scores (MDS-UPDRS Part-III and PIGD components) were performed for the PD group, controlling for sex and age. Multiple comparisons correction was performed at the model level for all the outcome metrics and clinical scores, as well as brain volumes, with a False Discovery Rate (FDR) threshold of 0.05. The reported model p-values are FDR corrected. Seven subjects had Cook’s distance more than 4/n while checking for potential outliers but removing these 7 subjects did not change the results, so they were kept in the cohort. All continuous variables were standardized to Z-scores in the regression analysis.
We assessed the group differences on following variables: T-tests were performed to check for PD versus control group differences for age, Montreal Cognitive Assessment, Mini Balance Evaluation Systems Test score, Activity Specific Balance Confidence Scale, and brain volumes for normally distributed data or Kolmogorov-Smirnov-test otherwise. A chi-square test was performed to test differences between sex in PD and control groups. We used Pearson’s correlation to assess the relationship between the following variables: 1) Brain volumes were correlated with age and 2) Subcortical brain volumes were correlated with ventricle volume.
All the data analyses were performed using R, version 3.4.3.
RESULTS
Sample characteristics
Average ages did not differ between the healthy controls and people with PD. 71 people with PD in the cohort were Hoehn and Yahr (H&Y) stage 2, 7 subjects were stage 3 and 8 subjects were stage 4. The demographic and clinical features of the participants are in Table 1.
Brain volumes and balance and gait metrics in PD and control groups
Group average ventricular volumes were similar between people with PD and healthy controls. Mean ventricular volume for people with PD was 1.9%of ICV (SD: 0.9%of ICV) and for the healthy controls was 2.1%of ICV (SD: 1%of ICV) (Table 2). When controlled for age and sex, bilateral cortical gray matter volume, occipital, parietal and temporal lobe volumes and putamen volumes were smaller in the PD group (Table 2). When classified as larger ventricle size based on the control mean plus 1.5 SD threshold, 8 healthy control subjects and in 15 PD subjects showed larger ventricles. A neurologist (A.G.) reviewed the brain images and videos of the MDS-UPDRS of subjects with larger ventricles and did not see evidence of normal pressure hydrocephalus, as accounted by either callosal angle or wide-based gait. The callosal angle was greater than 90 degrees in all subjects with enlarged ventricles showing absence of normal pressure hydrocephalus.
Mean (SD) of the brain volumes and balance and gait metrics in Control and PD groups with group difference test significance values
*median and inter quartile range was computed instead of mean/SD and KS test significant value computed instead of t-test; var, variability; SD, standard deviation; deg, degree; s, seconds; cm, centimeter; m, meter; t-values and p-values have been corrected for age and sex.
As observed in our larger, parent study (n = 223) [30], the balance and gait metrics in our sub-cohort (n = 146) were worse in people with PD compared to healthy controls (Table 2). However, this study is a subset (N = 146/223) of those previously reported results, limited to those with high quality brain imaging data.
Brain volumes related to balance and gait
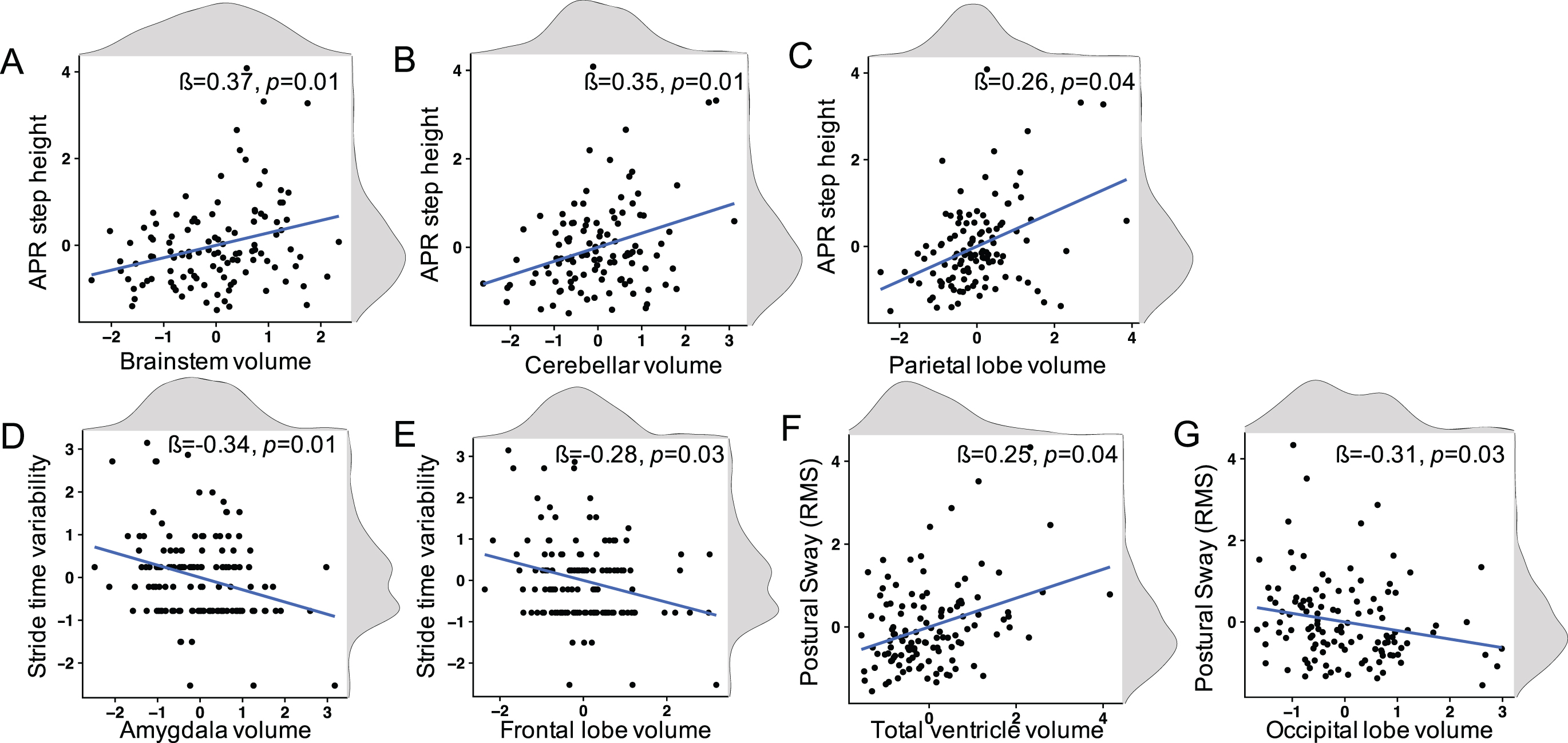
Using a general linear model, ANCOVA was performed to determine how normalized brain volume was associated with balance/gait metrics in PD and healthy control groups (controlling for age and sex). The main effect for group status was statistically significant for all balance and gait metrics, with the exception of stride time variability. The group with PD showed worse balance and gait. The models that also had statistically significant brain volume main effects or interaction effects are summarized in Table 3. The following main effects relating brain volumes with balance or gait metrics were found to be significant in healthy controls (reference group): 1) smaller brainstem volumes, smaller cerebellar volumes, and smaller parietal lobe volumes were associated with smaller (APR) reactive step height (Fig. 1A-C), 2) smaller amygdala volumes and smaller frontal lobe volumes were associated with larger stride time variability (Fig. 1D, E), and 3) larger ventricular volumes and smaller occipital lobe volumes were associated with larger postural sway (root mean square in anteroposterior direction (RMS AP)) standing on foam (Fig. 1F, G).
Significant standardized beta coefficients with p-values from ANCOVA for main and interaction effects between brain volumes and balance/gait metrics in people with PD and healthy controls
†Main effects coefficients are reported for healthy controls (reference group). *To get the beta coefficient of the PD group sum the interaction effects coefficients and the brain volume main effects in HC. Model p < 0.05 FDR corrected, uncorrected p < 0.05 for each predictor. HC, healthy controls; RMS, root mean square; AP, anteroposterior; APA, anticipatory postural adjustments; RoM, range of motion; GM, gray matter; β, standardized beta coefficient.
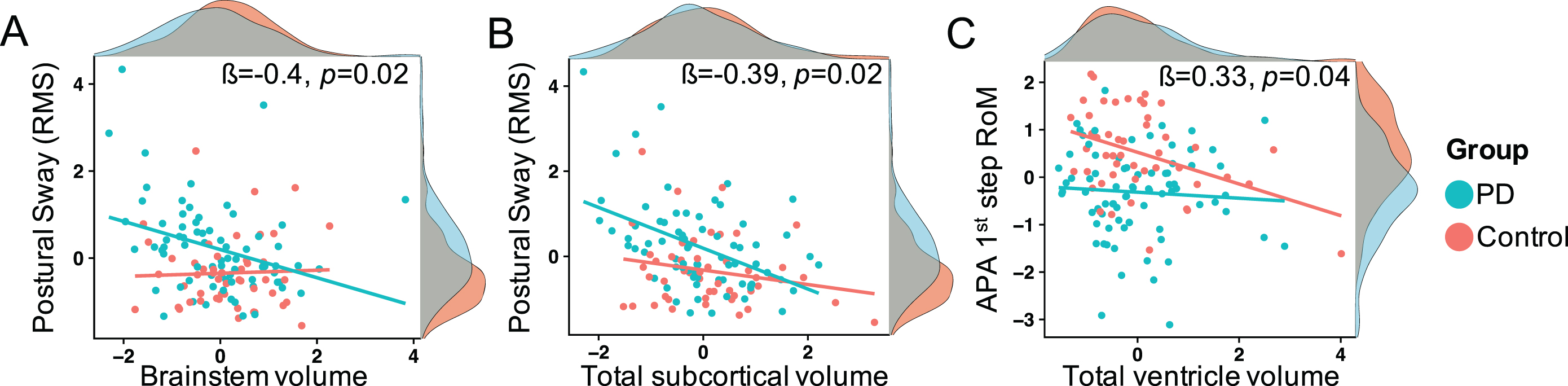
The significant interactions reflecting how the groups differed in their relationships between brain volumes and balance/gait metrics were: 1) people with PD, but not healthy controls, with smaller brainstem volumes and smaller total subcortical volumes showed larger (more abnormal) postural sway standing on foam (RMS AP; Fig. 2A, B) and 2) healthy controls, but not people with PD, with larger ventricular volume was associated with smaller (APA) first step of gait initiation (Fig. 2C).
Sub-analysis for freezers and non-freezers in PD: ANCOVA was used to determine how normalized brain volume was associated with balance/gait metrics in freezers versus non-freezers (controlling for age, sex and disease duration). The main effect for group (freezer/non-freezer) was statistically significant for (APA) first step of gait initiation and for gait speed with worse balance and gait in freezers. The main effect for brain volume for non-freezers (reference group) was significant for subcortical areas associated with sway standing on foam and for several cortical areas associated with (APA) first step of gait initiation and (APR) reactive step height (See Supplementary Table 1 for all results). We found several significant interactions between group (freezers/non-freezers) and how brain volumes associated with balance/gait metrics. Freezers with larger ventricle volume (β= 0.47, p = 0.02) and smaller cerebellum (β= –0.49, p = 0.02), smaller brainstem (β= –0.49, p = 0.01), and hippocampal (β= –0.45, p = 0.03) volumes showed larger stride time variability. Freezers with smaller cerebellar volumes (β= 0.41, p = 0.04) showed slower gait speed. Non-freezers with smaller cerebellar volumes (β= 0.44, p = 0.04) showed shorter first step of gait initiation (APAs) while freezers showed larger APAs (For main and interaction effects plots see Supplementary Figures 1 and 2).
Brain volumes related to clinical scores
Significant associations between brain volumes and gait/posture were found only with the 4-item, PIGD component of the MDS-UPDRS Part-III and not the total score. Smaller brain volumes were associated with larger PIGD. Smaller total subcortical volumes (β= –0.24, p = 0.04), smaller putamen volumes (β= –0.25, p = 0.017), smaller accumbens volumes (β= –0.26, p = 0.03), smaller hippocampus volumes (β= –0.33, p < 0.01), smaller amygdala volumes (β= –0.25, p = 0.02), and smaller temporal lobe volumes (β= –0.39, p < 0.01) were associated with larger PIGD.
Relationship between the brain volumes and age
Correlation of brain volumes with age showed that thalamus (r = –0.42, p < 0.01), brainstem (r = –0.26, p = 0.03), and occipital lobe (r = –0.25, p = 0.03) were correlated with age in the PD group alone, but not in the control group. The rest of the brain volumes were correlated with age in both the control and PD groups.
The normalized total ventricle volume correlated with volumes of normalized total subcortical volume (r = –0.58, p < 0.01), putamen (r = –0.36, p < 0.01), thalamus (r = –0.63, p < 0.01), accumbens (r = –0.52, p < 0.01), hippocampus (r = –0.68, p < 0.01), amygdala (r = –0.57, p < 0.01), and brainstem (r = –0.47, p < 0.01). However, the volumes of normalized pallidum and caudate had no significant correlation with ventricle volume. (for all significant correlation values, see Supplementary Table 2).
DISCUSSION
Summary of results
This study demonstrated that brain ventricular and gray matter volumes are significantly associated with instrumented measures of balance and gait, albeit with different patterns, in people with PD and age-matched healthy controls. In the primary analysis that consisted of both healthy controls and people with PD, the following associations were significant in healthy controls: enlarged ventricle and smaller occipital lobe volumes were associated with larger postural sway; smaller brainstem, cerebellar and parietal lobe volumes were associated with smaller reactive step height; and smaller amygdala and frontal lobe volumes were associated with larger stride time variability. Smaller brainstem and subcortical volumes were predictive of increased postural sway (while standing on foam causing disrupted surface somatosensory inputs) in people with PD, but not the healthy controls. Larger ventricular volume in healthy controls, but not in people with PD, were associated with impaired gait initiation. A sub-analysis in people with PD, alone, showed that subcortical volumes associated with postural sway whereas cortical areas associated with APAs and APRs in non-freezers. Freezers, but not non-freezers showed associations between atrophy in several brain areas with larger stride time variability and showed the opposite relationship between cerebellar volume and APA size than non-freezers. People with PD with smaller total subcortical, putamen, accumbens, hippocampus, amygdala and temporal lobe volumes also showed worse PIGD (4 items from the MDS-UPDRS Part-III). As shown previously, putamen volume was significantly smaller in PD than in the control group when controlled for age and sex [35]. Although ventricular volume was not larger in people with PD, total cortical GM volume and occipital, parietal and temporal lobe volumes were smaller in people with PD than in healthy controls, consistent with the literature [36].
Brain volumes related to postural sway
We found that ventricular enlargement and occipital lobe volumes were related to postural sway standing on foam with eyes open in healthy controls. Postural sway, especially when standing on foam, is known to be larger than normal in people with PD, older people without PD, and is related to fall risk [30, 37]. Ventricular volume is an indirect measure of brain atrophy and could be used as a surrogate marker of neuronal loss. Our results are consistent with a previous study showing that larger volumes of supratentorial cerebrospinal fluid and the ventricular system are associated with increased postural sway in healthy adults [38]. Since there is a correlation between many subcortical volumes and ventricular volume, loss of gray matter in the subcortical volumes maybe an important contributor to ventricular enlargement and increased standing postural sway. The occipital lobe may be involved in visual feedback for control of postural sway, especially when surface somatosensory inputs are disrupted and in older people, who rely on vision for postural control more than younger people [39]. Subjects with smaller occipital lobe volumes due to right-sided stroke showed greater sway eyes open, consistent with a role of the occipital lobe in visual control of postural sway [40].
People with PD show an association between brainstem volume and total subcortical volumes with postural sway when standing on foam with eyes open. We previously showed that standing on foam with eyes open, which compromises use of somatosensory information for balance, was the most sensitive and specific standing condition for PD [30]. The brainstem is known to be one of the critical regions involved in both static [13] and dynamic [41] posture control. Brainstem, along with other subcortical regions like basal ganglia, thalamus and cerebellum play a key role in human balance control and are most affected in people with balance disorders like PD [42, 43]. Also, functional imaging studies suggest that postural sway is primarily controlled by subcortical regions including the thalamus, globus pallidus and subthalamic nucleus [44].
Brain volumes related to anticipatory postural adjustments
We found that ventricle volume is associated with gait initiation, specifically the APA measure, first step range of motion, in healthy control subjects, but not in people with PD. While we did not find significant associations between the APA measures and the subcortical or cortical volumes in the primary analysis, we did see parietal, frontal and total cortical gray matter related to APAs in the PD-only cohort (see Supplementary Table 1). The parietal and frontal cortex have been implicated in control of APAs for step initiation since transcutaneous magnetic stimulation over the posterior parietal cortex or over the SMA disrupts APAs in healthy subjects, and even more in subjects with PD [45–47] and APAs can be absent in humans with lesions/tumors in the SMA [48].
Surprisingly, we found the opposite relationship between cerebellum volume and APA size in freezers versus non-freezers. That is, the smaller the cerebellum, the larger the APAs in freezers but the smaller the APAs in non-freezers. This is surprising because small APAs and steps are a hallmark of PD progression. It is also surprising because smaller APAs and smaller first steps are found in freezers than non-freezers. However, APAs are only small in freezers when they are not actively freezing when they become much larger than normal, suggesting that small APAs are a compensatory mechanism to avoid actual freezing events [49]. Thus, APAs may become smaller and smaller as the disease progresses (as a cerebellar compensation strategy) until the compensation fails and smaller cerebellar volume is associated with larger and larger APAs associated with actual freezing events. The cerebellum is considered one of the locomotor hubs and implicated in freezing of gait [50].
Brain volumes related to gait
We found that amygdala and frontal lobe volumes were related to stride time variability in healthy controls. Greater variability in gait is often associated with fall risk and future mobility disabilities [51]. Although impairments in amygdala are typically related to anxiety and cognitive decline, smaller amygdala, along with smaller frontal regions like medial prefrontal and inferior frontal gyrus have been associated with slow gait in the elderly [52, 53]. In general, structural imaging studies in people with PD have demonstrated neuroanatomical correlates of gait related to both cortical (frontal, dorsolateral and medial frontal cortical thickness) and subcortical (basal ganglia volume reduction and increased diffusivity in the substantia nigra and pedunculopontine nucleus) regions [16]. While we did not find any brain regions significantly associated with gait metrics for people with PD, we did find stride time variability and gait speed associated with cortical volumes in freezers, more than non-freezers with PD (Supplementary Table 1). Stride time variability has been related to severity of freezing, even when measured during gait without any freezing episodes [54]. Perhaps, combining freezers and non-freezers as one group reduced associations between brain volumes and gait in the PD cohort.
Brain volumes related to postural stepping responses
We found brainstem, cerebellum, and parietal lobe volumes to be associated with (APR) reactive step height in response to a backward perturbation in healthy controls. Although the brainstem and cerebellum play a substantial role in control of automatic postural responses, the cortex is also involved, especially for stepping responses [55]. The cortex is activated at 50 ms after a postural perturbation with leg muscle responses occurring a latency longer than 100 ms [56, 57]. Stepping response latencies are much longer and more variable and more dependent on instruction/intention/environmental conditions than feet-in-place postural responses, consistent with involvement of parietal, motor planning areas [58–60].
Brain volumes related to clinical scores
The clinical judgement about standing balance, walking, sit to stand and postural responses (pull test) reflected in the (4-items) MDS-UPDRS PIGD component was related to total subcortical, putamen, accumbens, hippocampus, amygdala, and temporal lobe gray matter volumes. Thus, subcortical volumes were associated with both the clinical and objective measures of postural control. Putamen volume was related to postural sway and temporal lobe volumes were associated with APRs (similar to the pull test) in the PD sub-analysis.
Putamen atrophy is known to be responsible for poor motor function and we found reduced putamen volume and cortical regions in our PD group, compared to the control group [36, 61]. However, the ventricular and other subcortical GM volumes were not significantly different between the 96 people with PD and the 50 healthy control subjects, although we cannot rule out age-related atrophy in both groups. While some brain regions like thalamus and brainstem show atrophy due to PD, most regions show atrophy in both PD and control groups related to aging effects.
Limitations
One limitation of our study is inherent in examining brain volume measures. Structural thickness or surface area changes may be missed with total volume measurements. It is not possible to capture all the complex neuronal changes with volume measures, alone. It is also important to note that segmentation of subcortical structures is tricky and is prone to partial volume effects. In order to minimize such issues, manual quality checks were performed to ensure good segmentation. Additionally, our study is cross-sectional and does not examine longitudinal volumetric changes, so, we cannot infer causality. Another limitation is that subjects with the worst balance and gait could not be included if they could not stand and walk independently [30]. We did not perform lateralized brain region analysis, since it would increase the number of regression models and reduce the statistical power. Finally, the PD group consisted of both freezers and non-freezers, who we showed had different relationships between brain volumes and gait/balance. Nevertheless, this is the first study to examine the associations between instrumented balance and gait metrics and brain volumes.
CONCLUSION
In conclusion, we found several significant models relate brain volumes and balance and gait in people with and without PD. Smaller subcortical and brainstem volumes may indicate standing balance dysfunction in people with PD. Smaller total ventricular volume may indicate smaller step initiation lateral movements (APAs) in healthy adults. Finally, atrophy of multiple subcortical regions seems to be associated with freezing of gait in PD. Thus, clinicians may expect to see balance dysfunction and freezing in people with PD who have reduced subcortical volumes.
Footnotes
ACKNOWLEDGMENTS
This work was funded by the grants: NIA grant # AG006457, VA Merit Award # RX001075. We thank Graham Harker and Patty Carlson-Kuhta for their help with data collection and study organization. We also thank Moosa Ahamed, Anders Perrone and Eric Earl for their support with setting up the pipeline for processing the MRI data.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
