Abstract
Background:
Glucagon-like peptide 2 (GLP-2) is a peptide hormone derived from the proglucagon gene expressed in the intestines, pancreas and brain. Some previous studies showed that GLP-2 improved aging and Alzheimer’s disease related memory impairments. Parkinson’s disease (PD) is a progressive neurodegenerative disorder, and to date, there is no particular medicine reversed PD symptoms effectively.
Objective:
The aim of this study was to evaluate neuroprotective effects of a GLP-2 analogue in the 1-Methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine (MPTP) PD mouse model.
Methods:
In the present study, the protease resistant Gly(2)-GLP-2 (50 nmol/kg ip.) analogue has been tested for 14 days by behavioral assessment, transmission electron microscope, immunofluorescence histochemistry, enzyme-linked immunosorbent assay and western blot in an acute PD mouse model induced by MPTP. For comparison, the incretin receptor dual agonist DA5-CH was tested in a separate group.
Results:
The GLP-2 analogue treatment improved the locomotor and exploratory activity of mice, and improved bradykinesia and movement imbalance of mice. Gly(2)-GLP-2 treatment also protected dopaminergic neurons and restored tyrosine hydroxylase expression levels in the substantia nigra. Gly(2)-GLP-2 furthermore reduced the inflammation response as seen in lower microglia activation, and decreased NLRP3 and interleukin-1β pro-inflammatory cytokine expression levels. In addition, the GLP-2 analogue improved MPTP-induced mitochondrial dysfunction in the substantia nigra. The protective effects were comparable to those of the dual agonist DA5-CH.
Conclusion:
The present results demonstrate that Gly(2)-GLP-2 can attenuate NLRP3 inflammasome-mediated inflammation and mitochondrial damage in the substantia nigra induced by MPTP, and Gly(2)-GLP-2 shows neuroprotective effects in this PD animal model.
INTRODUCTION
Parkinson’s disease (PD) is the second most common nervous system degenerative disease besides Alzheimer’s disease. Its typical clinical symptoms are resting tremors of the upper limbs, accompanied by bradykinesia, rigidity and gait difficulties [1]. The principal pathological feature of PD is the gradual decrease and loss of dopaminergic neurons in the substantia nigra [2–4], and the loss of dopamine transmitters in the striatum. The substantia nigra is a key brain area that controls movement. Tyrosine hydroxylase (TH) plays a critical role in regulating dopamine synthesis, and its protein expression level is the primary marker of dopaminergic neurons [5]. To date, there is no drug treatment that can arrest PD progression. Anticholinergics and dopaminergic receptor agonist as well as the dopamine precursor L-DOPA are mostly used in the treatment of PD [6, 7]. However, the efficacy is not good and side effects are apparent. Furthermore, long-term use of L-DOPA leads to serious side effects, and the effect on improving motor symptoms wanes due to the fact that the neurons that metabolize L-DOPA to dopamine continue to die [8]. Therefore, developing effective drugs that stop disease progression in PD is the focus of current research in drug discovery.
It has been reported that neuroinflammation, mito-chondrial dysfunction and oxidative stress play important roles on the pathogenesis of PD [9–11]. Neuroinflammation significantly promotes PD development by activating microglia and releasing pro-inflammatory factors, which can accelerate neuronal damage, reduction and death [12, 13]. On the other hand, damaged or dead neurons further promote the inflammatory response of microglia [14]. An imbalance between fission and fusion causes mitochondrial dysfunction. Abnormal protein expression connected to fission and fusion causes changes in mitochondrial morphology, number, and structure, further affecting its function in PD [15, 16].
Glucagon-like peptide 2 (GLP-2) is a 33 amino acid peptide that acts as a hormone and a growth factor [17, 18]. GLP-2 has various physiological functions, including reducing gastric emptying, promoting the absorption of nutrients, restoring and maintaining epithelial barrier function mucosal epithelium [19, 20], decreasing food intake [21, 22], regulating energy balance and glucose homeostasis [23, 24]. It works by binding to its specific GLP-2 receptor (GLP-2R) [25, 26] which is expressed in the central nervous system such as hippocampal neurons, brainstem, and hypothalamus [18, 27]. Activation of GLP-2Rs in the brain can have a neuroprotective effect. Studies have shown that GLP-2 improves memory impairment related to aging [26, 28] and vascular dementia [29], rescues lipopolysaccharide (LPS)-induced memory decline by activating receptors in the hippocampus, which is due to the anti-inflammatory properties of GLP-2 [26]. GLP-2 restores the learning and memory decline and neurogenesis induced by streptozotocin, and can reduce oxidative stress [28]. In addition, GLP-2 also alleviates the cognitive impairment caused by chronic cerebral hypoperfusion, which is related to restoration of the number and function of hippocampal neuron dendritic spines, and the improvement of synaptic plasticity [30]. Studies furthermore found that GLP-2 showed antidepressant and anti-anxiety effects, which may be caused by changes in 5-HT expression and the involvement of the hypothalamic-pituitary-adrenal axis regulation [26, 31].
GLP-1 and glucose-dependent insulin-promoting polypeptide (GIP) are incretin hormones that are members of the same family [32]. GLP-1 and GIP analogues have different degrees of neuroprotection in AD and PD animal models [33–37]. Dual-Agonist-5 (DA5-CH), a new long-lasting GLP-1/GIP dual agonist, can simultaneously activate GLP-1 and GIP receptors and cross the blood-brain barrier. DA5-CH significantly improves spatial memory impairment, reverses hippocampal long-term potentiation (LTP), and reduces hippocampal Aβ plaques and neurofibrillary tangles of the APP/PS1 transgenic mouse model [38]. Compared with other peptides, DA5-CH protects the brain from MPTP toxicity more efficiently and increases dopamine synthesis in PD mouse models [39]. Therefore, in this study, DA5-CH was used as a positive control and comparator. GLP-1 and GLP-2 are both glucagon-like peptides. We have previously shown that the sister hormone GLP-1 shows good neuroprotective effects in different models of PD [40]. However, the neuroprotective effects of GLP-2 analogues in PD animal models have not been tested. In this study, we used the 1-methyl-4-phenyl 1, 2, 3, 6-tetrahydropypridine (MPTP)-induced PD animal model to study the effects of a protease resistant GLP-2 analogue with a longer half-life in the blood stream [41] on behavior, neuroinflammation and mitochondrial dysfunction, and to explore the effects of Gly(2)-GLP-2 in the treatment of PD and its related mechanisms, which lays the foundation for the application of GLP-2 as a the treatment of neurodegenerative diseases.
MATERIALS AND METHODS
Drugs
The solutions used in this study were all prepared fresh before use. MPTP was purchased from Sigma-Aldrich (St. Louis, MO, USA). The GLP-2 analogue and DA5-CH were obtained from GL Biochem Ltd (Shanghai, China).
Amino acid sequences of the peptides:
YXEGTFTSDYSIYLDKQAAXEFVNWLLAGGPSSGAPPPSKRRQRRKKRGY-NH2, X is amino-isobutyric acid.
Gly(2)-GLP-2 [41]:
HGDGSFSDEMNTILDNLAARDFINWLIQTKITDR.
Animals and drug treatments
Two-month-old male C57BL/6 mice (20–25 g) were purchased from Animal Experiment Center of Henan University of Chinese Medicine (Zhengzhou, China). The mice maintained at 22±1°C under a cycle of 12 h of light and 12 h of darkness, being allowed free access to food and water. All animal procedures complied with the regulations of the Institutional Animal Care Committee of the Henan University of Chinese Medicine and following the National Institutes of Health (NIH) guidelines.
The 42-month-old male mice used in this study randomly divided into four groups, each with ten mice. (1) A control group treated with saline; (2) A group treated with MPTP; (3) A group treated with GLP-2 and MPTP; (4) and a positive control group treated with DA5 and MPTP. MPTP (20 mg/kg body weight), GLP-2 analogue (50 nmol/kg body weight), and DA5 (25 nmol/kg body weight) were dissolved in saline (0.9%) and injected intraperitoneally. MPTP was administered to the mice once a day for 7 days. GLP-2 analogue and DA5 were administered for 14 days. The time interval between two drug administration was 2 h.
Behavioral assessment
Open-field test
To evaluate the spontaneous locomotor and ex-ploratory ability of PD mice, it performed on the 6th day after MPTP treatment. Each mouse was subjected to adaptive training one day before the experiment. The device consists of two parts: a square arena (40×40 cm) and an opaque wall 25 cm high (Ruiwode, Shenzhen, China). According to some previous study [42, 43], at the beginning of the test, each mouse was placed in the center of the open area, allowing it to explore freely. The total distance and rearing times of each mouse were recorded within 5 minutes [44]. After each test, the arena was cleaned with 75% alcohol and dry it thoroughly to avoid affecting the experiment of the next mouse. Repeat the test twice for each animal and analyzed using Smart 3.0 software (Panlab SL, Barcelona, Spain). 10 mice per group were used in this test.
Rotarod test
To assess the effect of drugs on the motor coordination on mice, the rotarod (Ruiwode, Shenzhen, China) test was conducted from the 7th day to the 13th day of the experiment. The rotaord device consisted of two parts: a rotating spindle and five independent compartments capable of testing five mice simultaneously. The motor function of the mice was evaluated by measuring the latency to fall. The mouse was placed on the rod, which steadily accelerated from 5 rpm to 20 rpm in 3 min. The time for the mice to complete the task was 3 min [45]. After an adaptive training on the rotary instrument, the mice began to test for 7 consecutive days. When the mouse is completely dropped from the rod and the time spent on the rod is recorded, that is, the latency to fall off the rotating rod of each mouse was used for statistical analysis. The latency to fall was measured using Smart 3.0 software (Panlab SL, Barcelona, Spain). 10 mice per group were used in this test.
Pole test
To observe the bradykinesia and motor balance ability of mice, a pole test was performed on the 14th day after Gly(2)-GLP-2 treatment. We used the experimental designs as previously described [37, 46]. Briefly, the animal is placed face up near the top of a pole with a rough surface (1 cm in width and 55 cm in height). Record the time it takes for their head to turn down (T-turn) and the time it takes to reach the bottom of the pole (T-LA). After an adaptive training, each mouse tested three times, and the average of them was calculated and used for statistical analysis. 10 mice per group were used in this test.
Sample preparation
Mice were sacrificed on the 14th day after Gly(2)-GLP-2 treatment. The brain was removed after being anesthetized with 20% urethane, and bilateral substantia nigra were carefully dissected on ice. They were stored at –80°C for ELISA and western blot analysis respectively. After the substantia nigra was taken out, cut them into 1 mm3 tissue pieces, and put into 4% glutaraldehyde fixative for transmission electron microscope analysis. In addition, the right atrial appendage was cut and perfused with pre-chilled 0.9% saline and 4% paraformaldehyde. The whole brain was taken out and placed in a fixative for immunofluorescence histochemical analysis.
Transmission electron microscope (TCM)
TCM was used to observe mitochondria ultrastructure of substantia nigra neurons. The tissue block was fixed in 2.5% glutaraldehyde and phosphate buffer for 2 h, and in 1% osmium acid for 2-3 h. Different concentrations of ethanol and acetone mixtures were used to dehydrate and solidify at different temperatures in an oven. The tissue was sliced into 70 nm sections by an ultramicrotome and placed on the copper grid. 3% uranyl acetate-lead citrate was used for double staining and then observed mitochondria ultrastructure under a TCM (JEM-1230, JEOL Ltd., Tokyo, Japan). TCM services are supported by the Academy of Chinese Medicine Sciences of Henan University of Chinese Medicine. 3 mice per group were used in this test.
Immunofluorescence histochemistry
Immunofluorescence histochemistry was used to detect the expression of TH and Iba1 in the substantia nigra of midbrain. The frozen tissue was sliced into 20-μm coronal sections on a cryostat (CM1950, Leica Microsystems, Wetzlar, Germany). The sections were then permeabilized with 0.3% Triton X-100 for 30 min. Non-specific binding sites were blocked by incubation with goat serum (Solarbio Science & Technology Co., Beijing, China). Then the rabbit anti-TH (ab112, 1:200; Abcam, Cambridge, UK) and rabbit anti-Iba1 (ab178846, 1:500; Abcam, Cambridge, UK) were used at 4°C overnight. After washing with PBS three times, the slides were incubated with a secondary antibody, which was fluorescein isothiocyanate (FITC)-conjugated Donkey anti-rabbit IgG/Alex Fluor 594 (711–545–152, 1:500, Jackson Immuno Research, Baltimore, MD, USA), for 1.5 h at 37°C. DAPI (4′, 6-diamidino-2-phenylindole) (Solarbio Science & Technology Co., Beijing, China) was added to stain the nuclei for 5–10 min. The sections were observed and photographed under an optical microscope (BX61, Olympus Microsystems Imaging Solutions Ltd, Tokyo, Japan). Image-pro plus 6.0 software (Media Cybernetics, Bethesda, MD, USA) was used to determine the average optical density of dopaminergic neurons. N = 3 sections per brain were analyzed, with n = 3 per group.
Enzyme-linked immunosorbent assay
Then ELISA assay was used to detect the levels of IL-1β in the substantia nigra. The frozen substantia nigra from each group was homogenized by a tissue homogenizer (Bertin Technologies, Montigny-le-Bretonneux, France). After homogenization, the samples were centrifuged at 12000 rpm for 10 min at 4 (20R, Beckman Coulter Inc, Brea, CA, USA). An ELISA kit (Solarbio Science & Technology Co., Beijing, China) was used to measure IL-1β level following the manufacturer’s instructions. Firstly, the standard and sample were added to the microelisa stipulate respectively (100μl standard and test sample were added to the corresponding reaction well). Secondly, added 100ul biotinylated antibody to each well, covered with an adhesive strip, and incubated in a 37°C incubator for 60 min. Then, the chromogenic substrate was added to each well, and incubated for 15 min at 37° C in a dark incubator. Finally, the stop solution was added to each well, and the optical density (O.D.) was measured at 450 nm with a multifunction microplate reader (Thermo Fisher Scientific, MA, USA), and the concentrations of IL-1β was calculated from the standard curve. 4 mice per group were used in this test.
Western blot
The mouse’s frozen substantia nigra was homogenized in ice-cold RIPA lysis buffer containing phenyl methyl methane sulfonyl fluoride (PMSF) and protease inhibitor 1 (Solarbio Science & Technology Co., Beijing, China). After homogenization, the samples were centrifuged at 12000 rpm for 10 min at 4. The supernatant was then transferred to Eppendorf tubes. The bicinchoninic acid (BCA) protein assay kit (Beyotime Biotechnology, Shanghai, China) was used to detect protein concentration. Then, 30μg of protein from each group was separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The gel was run for 30 min at 80 V and for 1 h at 120 V, and proteins were transferred to a polyvinylidene difluoride (PVDF) membrane (Millipore, Boston, MA, USA) by using a semidry transfer apparatus (Bio-Rad, Hercules, CA, US). The proteins were subsequently blocked for 2 h with 5% BSA (Boster Biotechnology Co., Ltd., Wuhan, China) at room temperature to block remaining protein binding sites, and then incubated overnight at 4 with the primary antibody rabbit anti-mouse Mfn2 (ab124773, 1:1000; Abcam, Cambridge, UK), rabbit anti-mouse FIS1 (ab229969, 1:1000; Abcam, Cambridge, UK) added, rabbit anti-mouse IL-1β (ab9722, 1:400; Boster Biotechnology Co., Ltd., Wuhan, China), rabbit anti-mouse Caspase-1 (ab1872, 1:1000; Abcam, Cambridge, UK), rabbit anti-mouse NLRP3 (ab263899, 1:1000; Abcam, Cambridge, UK) and rabbit anti-mouse β-actin (ab8227, 1:1000; Abcam, Cambridge, UK). The membranes were then washed three times in TBST at room temperature and incubated with a secondary antibody goat anti-rabbit immunoglobulin (ab6721, 1:5000; Abcam, Cambridge, UK) for 1.5 h at room temperature. After adding ECL-enhanced chemiluminescence (Beyotime Biotechnology, Shanghai, China), the antibodies were visualized and observed western blot images in a chemiluminescence imaging system (Thermo Fisher Scientific, MA, USA). All bands were quantified using the imaging system of Image J v1.51 (National Institutes of Health, Bethesda, MD, USA). N = 4 per group.
Data and statistical analyses
Data were shown as the mean±standard error of the mean (SEM). The statistical significance of differences was evaluated using a parametric one-way analysis of variance (ANOVA) followed by followed by Bonferroni’s post hoc test. In all cases, significance was set at p < 0.05. Statistical analyses were performed using Graphpad Prism 6 (Graphpad Sofware, San Diego, CA, USA).
RESULTS
Gly(2)-GLP-2 ameliorated deficits in the spontaneous locomotor and exploratory ability caused by MPTP treatment
We examined MPTP-induced deficits and the effects of GLP-2 analogue using an open field test. Total distance and rearing numbers are shown in Fig. 1A, respectively. MPTP injection-induced deficits in the spontaneous locomotor and exploratory ability of mice, with less distance and the number of times, compared to the control group (p < 0.01, p < 0.001). However, treatment with Gly(2)-GLP-2 or DA5-CH effectively improve these effects induced by MPTP (p < 0.05, p < 0.001). It shows that Gly(2)-GLP-2 and DA5-CH can ameliorate deficits in the spontaneous locomotor and exploratory ability of mice caused by MPTP.
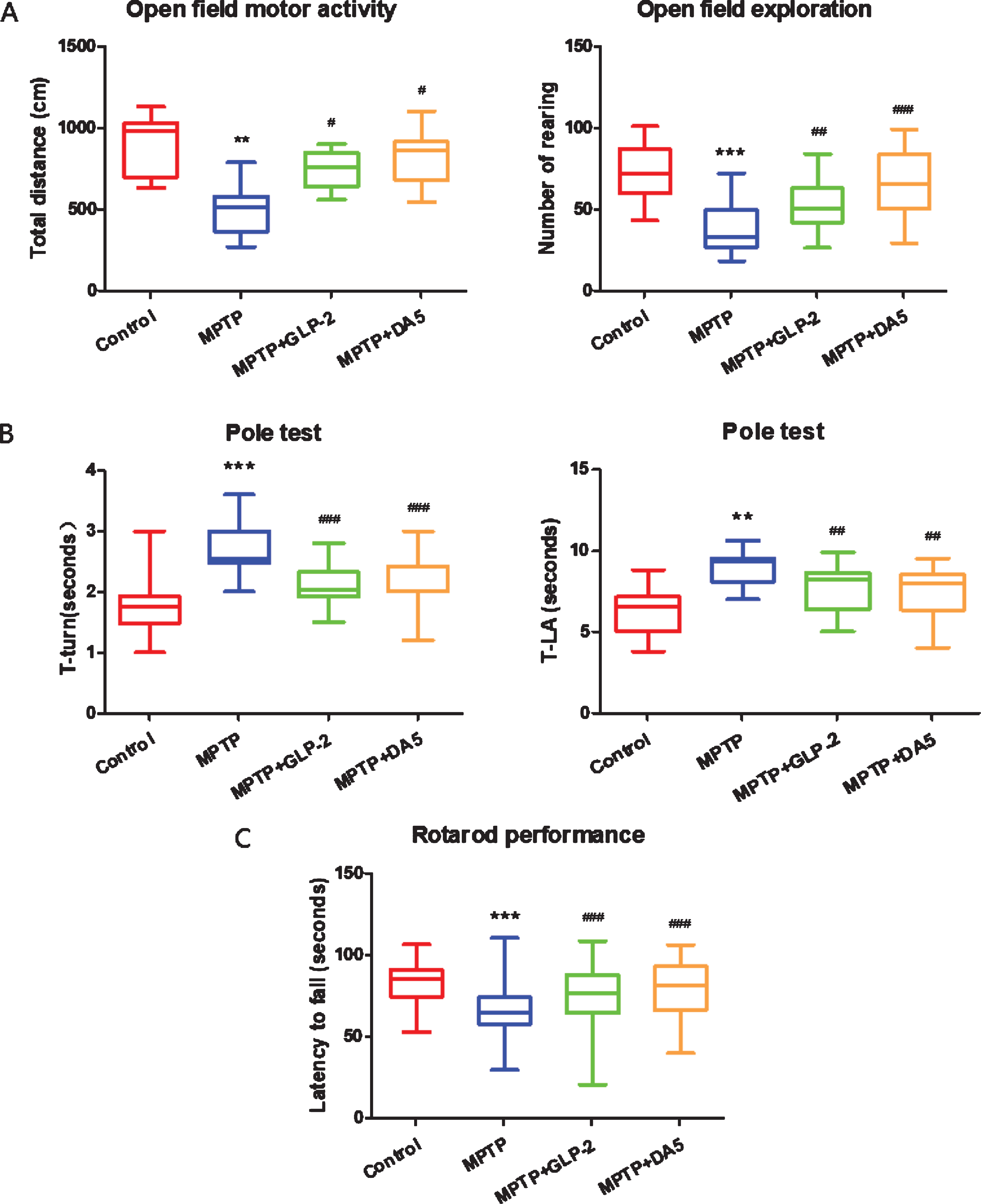
Effects of the administration of GLP-2 analogue on MPTP induced motor impairments. A) open field test, B) Pole test, C) Rotarod test. **p < 0.01 and ***p < 0.001 compared to the control group; #p < 0.05, # #p < 0.01, and # # #p < 0.001 compared to the MPTP group. n = 10 per group.
The GLP-2 analogue improved bradykinesia and movement imbalance caused by MPTP
We performed the pole test to investigate the effects of Gly(2)-GLP-2 on bradykinesia and movement imbalance responses after MPTP administration. The time to turn at the top (T-turn) and the time to climb down (T-LA) of pole were shown in Fig. 1B, respectively. MPTP injection induced the bradykinesia of mice, with more time to turn at the top (p < 0.001) compared to the control group. And MPTP intraperitoneal injection also caused movement imbalance of mice, with more time to climb down (p < 0.01) compared to the control group. However, treatment with Gly(2)-GLP-2 or DA5-CH significantly reversed these effects induced by MPTP (p < 0.001, p < 0.01). These results indicate that Gly(2)-GLP-2 and DA5-CH improved bradykinesia and movement imbalance in MPTP-treated mice.
Gly(2)-GLP-2 ameliorated dyskinesia caused by MPTP
After the last MPTP treatment, a rotarod performance was performed daily for 7 consecutive days. Compared with the control group, the staying time on the rotating rod of the mice in the MPTP group, that is, the latency to fall was significantly shorter (p < 0.001). After treatment with Gly(2)-GLP-2 or DA5, the latency to fall was significantly prolonged (p < 0.001), and there were significant differences. This indicates that Gly(2)-GLP-2 and DA5-CH can improve the dyskinesia caused by MPTP and increase their motor coordination ability (Fig. 1C).
The GLP-2 analogue protected against dopaminergic neuronal damage induced by MPTP
TH is commonly used as a marker of dopaminergic neurons in the substantia nigra [5]. To observe the position and expression of TH, we used immunohistofluorescence staining to detect it. As shown in the Fig. 2, compared with the control group, MPTP has obvious neurotoxicity to substantia nigra TH-positive cells, and the expression of TH is reduced. While both Gly(2)-GLP-2 and DA5-CH administration significantly increased the mean fluorescence intensity of TH positive cells, reduced cytotoxicity caused by MPTP. It suggests that Gly(2)-GLP-2 and DA5-CH might exhibit neuroprotective effects against dopaminergic neuronal damage in PD.
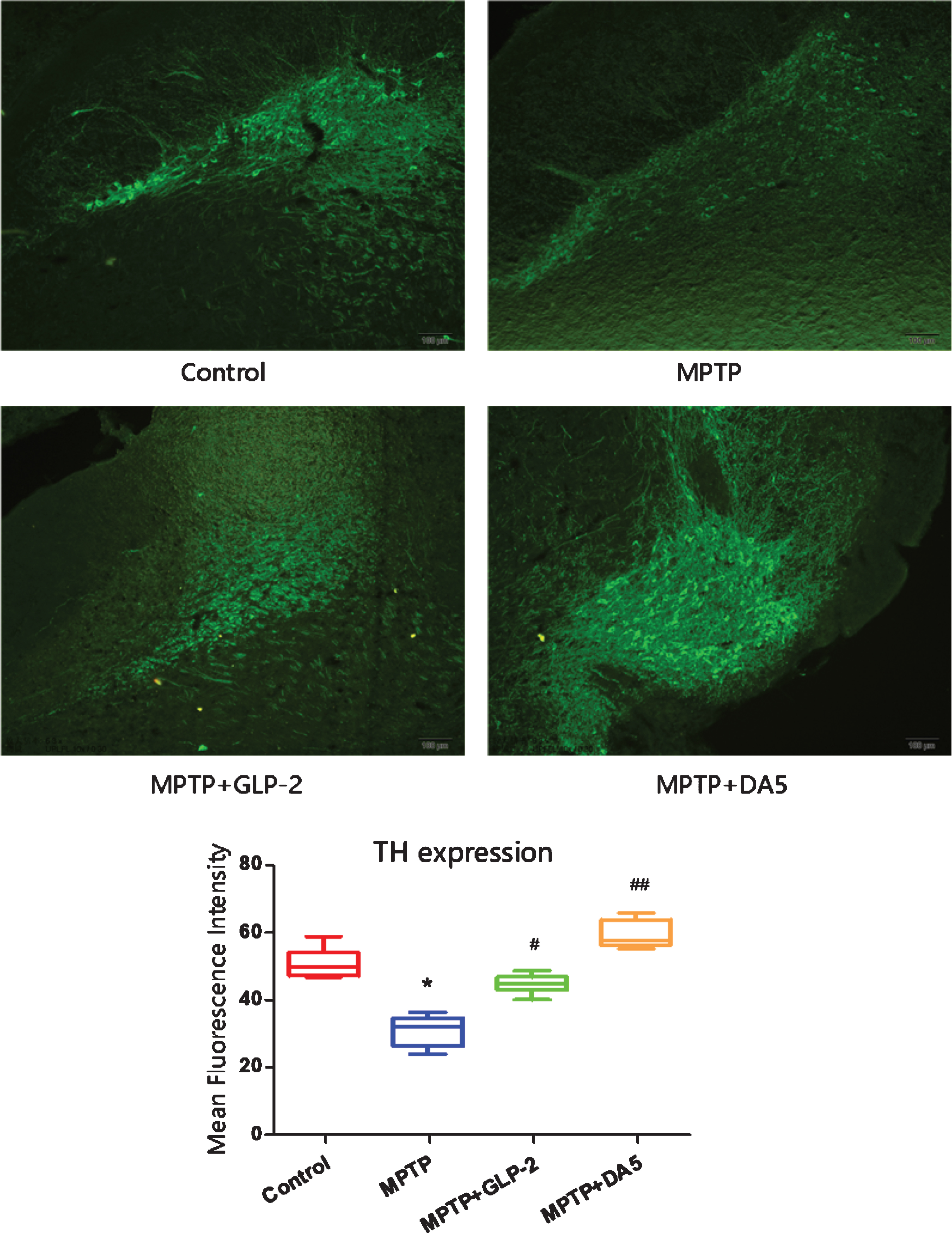
Immunofluorescence detection of TH positive expression in the substantia nigra. *p < 0.05 compared to the control group; #p < 0.05 and # #p < 0.01 compared to the MPTP group. Scale bar = 50μm. n = 3 per group.
(2)-GLP-2 decreased microgliosis induced by MPTP
Iba1 is a biomarker for microglia activation [47]. Iba1 is mainly distributed in the cytoplasm [48]. As shown in the Fig. 3, the mean fluorescence intensity increased in the MPTP group, which was significantly higher than that of the control group (p < 0.01). However, the mean fluorescence intensity of Iba-1 in the substantia nigra of the GLP-2 analogue and DA5 treatment groups was effectively lower than that of the MPTP group (p < 0.05, p < 0.01)
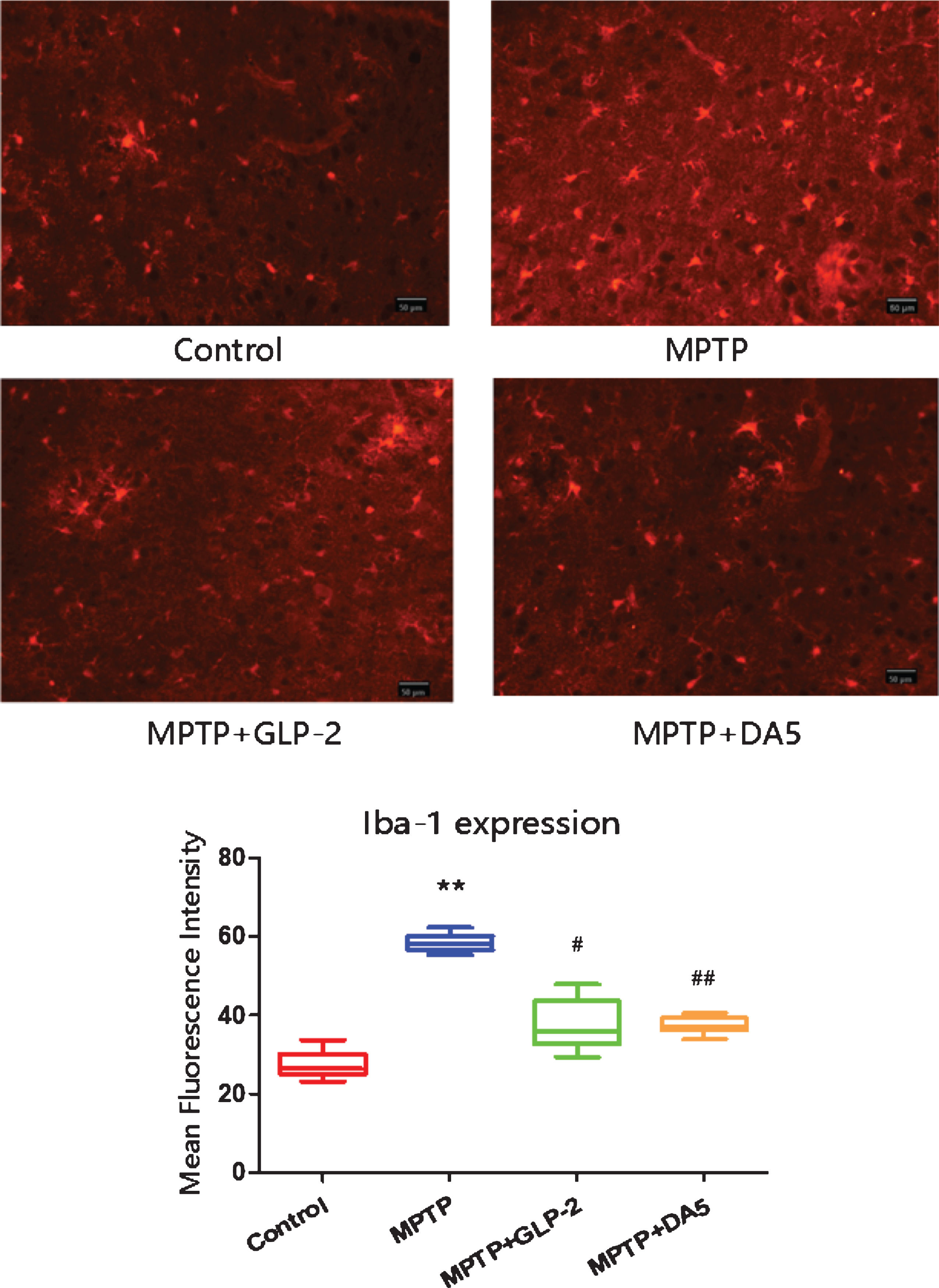
Activation of microglia in the substantia nigra. **p < 0.01 compared to the control group; #p < 0.05 and # #p < 0.01 compared to the MPTP group. Scale bar = 50μm. n = 3 per group.
Gly(2)-GLP-2 reduced the expression of IL-1β, caspase-1 and NLRP3 protein
The results showed that the levels of IL-1β, cas-pase-1 and NLRP3 in the substantia nigra of MPTP induced mice were significantly higher than those in the control group. However, the expression levels of IL-1β, caspase-1 and NLRP3 in the substantia nigra of Gly(2)-GLP-2 and DA5-CH treatment groups were decreased compare with MPTP group (Fig. 4). These results indicate that Gly(2)-GLP-2 reduced neuron inflammation in the mouse substantia nigra induced by MPTP.
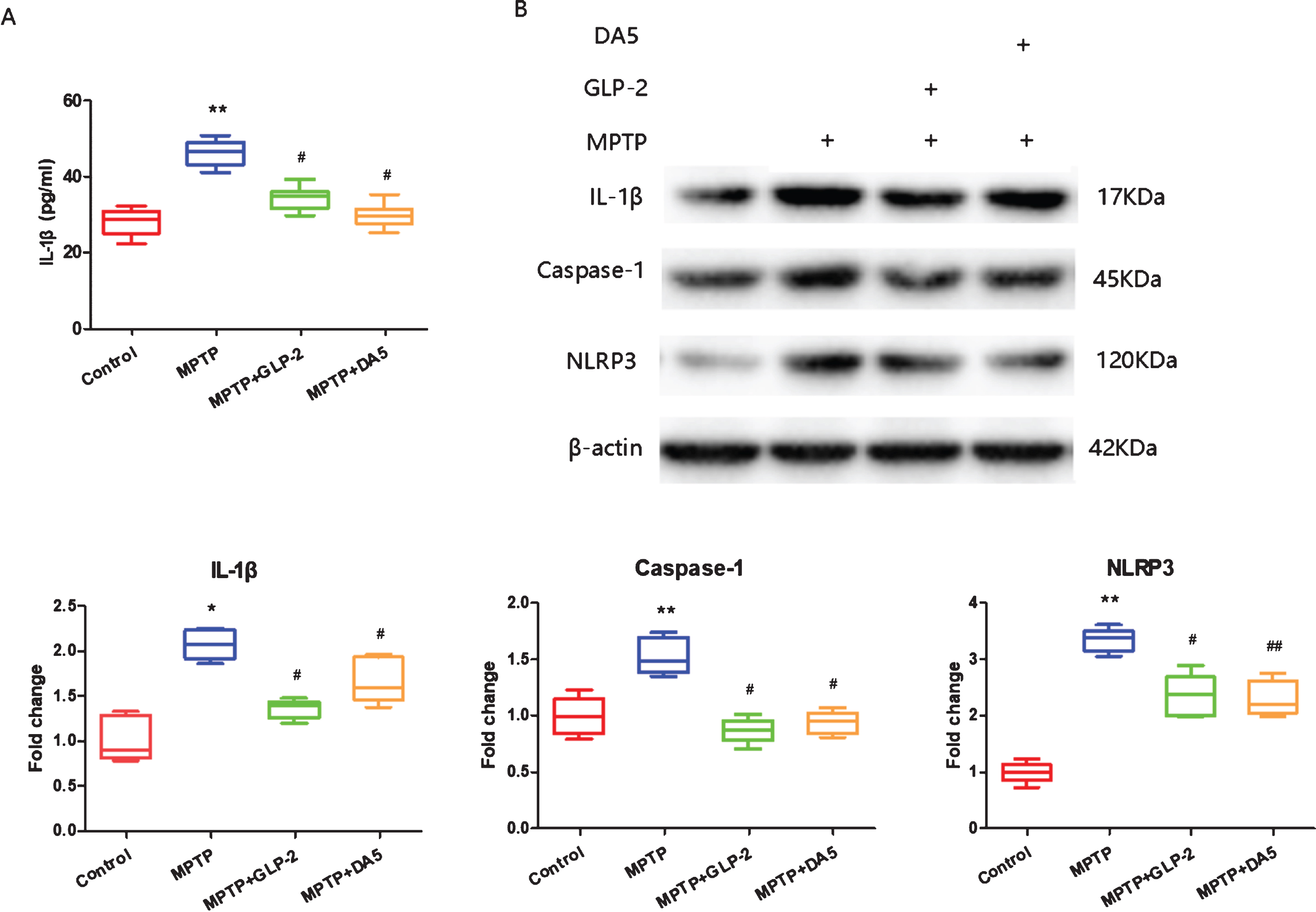
ELISA and western blot detection of IL-1β, caspase-1 and NLRP3 protein expression in the substantia nigra. A) ELISA of IL-1β, B) Western blot. Fold changes were normalized to the value in the control samples. *p < 0.05 and **p < 0.01 compared to the control group; #p < 0.05 and # #p < 0.01 compared to the MPTP group. n = 4 per group.
Gly(2)-GLP-2 improved mitochondrial damage induced by MPTP
The morphological structure of the mitochondria of the substantia nigra neurons was detected by transmission electron microscopy. The results showed that compared to the normal control group, the mitochondria of the substantia nigra neurons in the MPTP group were notably swollen, the vacuolarized mitochondria increased, cristae was fractured and the number was reduced. After Gly(2)-GLP-2 or DA5-CH treatment, the swelling of mitochondria was alleviated, vacuolized mitochondria were reduced, cristae was visible and the number increased (Fig. 5). It indicates that Gly(2)-GLP-2 can protect the mitochondria of substantia nigra neurons induced by MPTP.
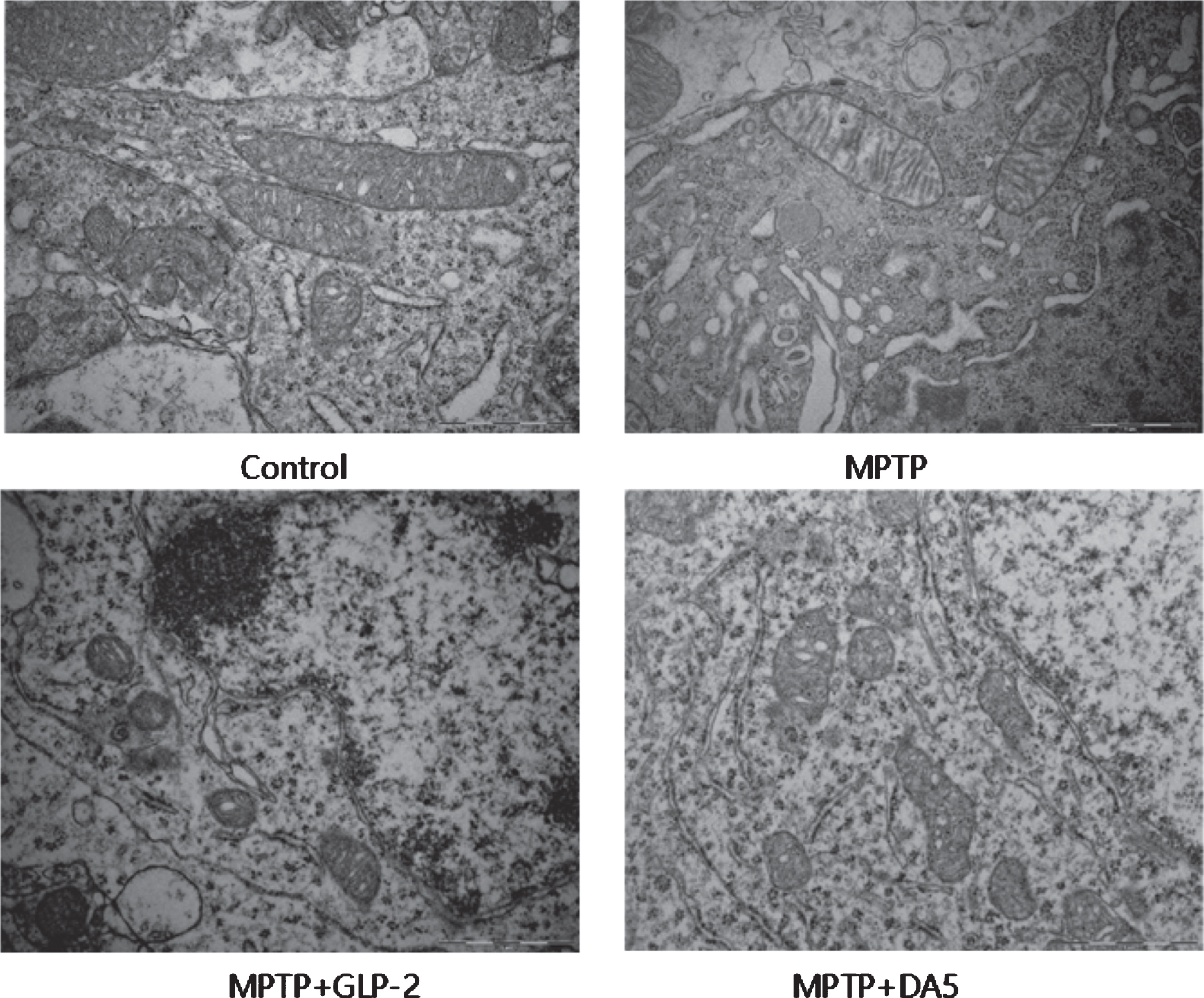
Transmission electron microscope detection of mitochondria in the substantia nigra. Scale bar = 1μm. n = 3 per group.
GLP-2 analogue reversed the expression of mitofusin2 (Mfn2) and mitochondrial fission protein 1(FIS1) levels induced by MPTP
Western blot was used to measure the expression levels of Mfn2 and FIS1 protein. Mfn2 is an indispensable dynamin-related protein that regulates mitochondrial morphology and function [49]. FIS1 is necessary for mitochondrial fission [50]. The results showed that compared to the control group, the levels of Mfn2 and FIS1 in the substantia nigra of PD mice decreased and increased respectively. After Gly(2)-GLP-2 or DA5-CH treatment, the expression levels of Mfn2 and FIS1 increased and reduced in the substantia nigra (Fig. 6). It indicates that Gly(2)-GLP-2 can reversed the expression of Mfn2 and FIS1 in the substantia nigra of mice induced by MPTP.
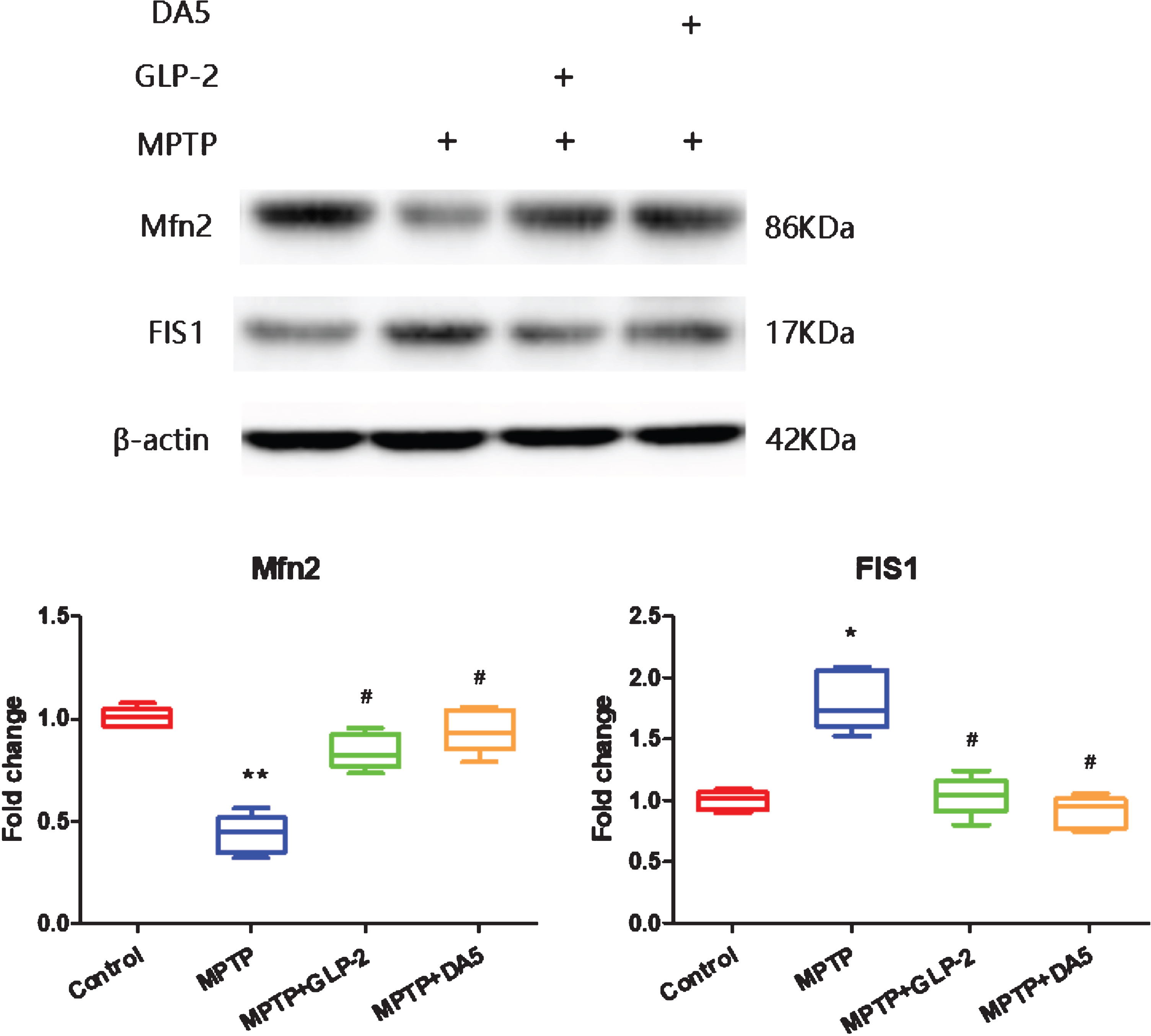
Western blot detection of expression of proteins involved in mitochondrial fission/fusion in the substantia nigra. Fold changes were normalized to the value in the control samples. *p < 0.05 and **p < 0.01 compared to the control group; #p < 0.05 compared to the MPTP group. n = 4 per group.
DISCUSSION
In this present study, the results indicate that the GLP-2 analogue Gly(2)-GLP-2 reduced motor impairment in an animal model of PD induced by the MPTP. Gly(2)-GLP-2 also protects dopaminergic neurons in the substantia nigra. Furthermore, it alleviates the neuroinflammation response, the NLRP3 inflammasome activation, and mitochondrial damage in the midbrain of PD mice induced by MPTP. These results suggest that Gly(2)-GLP-2 improves behavioral impairments by inhibiting the activation of NLRP3 inflammasome and reducing mitochondrial damage in PD mice induced by MPTP, demonstrating that GLP-2 analogue has neuroprotective properties in PD mice, and it may has potential in the prospective treatment of PD.
The MPTP rodent model is a standard model for PD which displays the main symptoms found in PD. MPTP can induce PD in humans [51]. It is lipophilic and can cross the blood–brain barrier [52]. In neurons, MPTP is metabolized into the toxic cation 1-methyl–4-phenylpyridinium (MPP+) by monoamine oxidase B [53–54]. MPP+has been shown to kill primarily dopamine-producing neurons in the substantia nigra [55]. MPP+blocks complex 1 in the mitochondrial electron transport chain, which leads to the production of free radicals and to cell death [56, 57]. The limiting factor of this model is the fact that animals can recover from the treatment and neuronal loss is limited.
MPTP reduces the protein synthesis of key en-zymes such as TH, which is necessary for dopamine synthesis. Gly(2)-GLP-2 and DA5-CH treatment were able to increase the level of TH positive neurons and protect dopaminergic neurons to some degree, with the dual agonist DA-CH5 being the most effective drug. GLP-1 and GIP receptors express in the brain and perform similar roles as GLP-2 [58–60]. Some previous studies have shown that dual GLP-1/GIP receptor agonists such as DA5-CH reduced neuron impairments induced by MPTP [61–63] and 6-OHDA [64]. DA5-CH containing a TAT sequence can cross the blood-brain barrier [40]. DA-CH5 increases the synthesis of TH, reduces neuroinflammation in the induced by MPTP [65], reduces the expression of phosphorylated tau protein, and normalized growth factor signaling in rats treated with streptozotocin to induce insulin desensitization [66]. This was another reason that we chose DA5-CH as the positive control.
In our study, Gly(2)-GLP-2 displayed anti-infla-mmatory properties. Although the pathogenesis of PD has not yet been elucidated, inflammation may be one of the critical factors [67, 68]. Previous studies have shown that LPS caused an increase in pro-inflammatory cytokines (IL-1β, IL-6 and TNF-α) in BV-2 cells [69]. Microglia are the most important effector cells for neuroinflammation. Over-activated microglia produce a variety of neurotoxic factors, such as pro-inflammatory cytokines and reactive oxygen species, which may lead to the damage, reduction or loss of dopaminergic neurons [70–72]. Therefore, the critical mechanism step for the treatment of PD is to inhibit the excessive activation of microglia. In the current study, Gly(2)-GLP-2 treatment was confirmed to reduce Iba-1 expression in the substantia nigra of PD mouse model, indicating that the GLP-2 analogue Gly(2)-GLP-2 reduces MPTP-induced activation of microglia.
NLRP3 inflammasome plays a significant role in the pathological mechanism of PD neuroinflammation [73–76]. Inflammasomes are high-molecular-weight complexes composed of a variety of proteins, including receptor protein (receptor), adaptor protein (adaptor) ASC and the downstream caspase-1 [77]. The assembly of NLRP3 inflammasome induces the cleavage of Caspase-1, which further promotes the maturation and secretion of pro-inflammatory cytokines such as interleukin-1 (IL-1β) and interleukin-18 (IL-18) [78]. Recent studies have found that an NLRP3 inflammasome inhibitor reduces the activation of inflammasomes. In addition, oral administration of an NLRP3 inflammasome inhibitor can inhibit inflammasome activation and effectively alleviate motor dysfunction and dopaminergic degeneration of the substantia nigra [67]. NLRP3 inflammasome may be a major driver of dopaminergic neuropathology. Thus, it is considered a potential target for the treatment of PD [68]. We hypothesized that Gly(2)-GLP-2 inhibited the neuroinflammation caused by the NLRP3 inflammasome. The data indicates that Gly(2)-GLP-2 reduces the protein levels of NLRP3, caspase-1 and IL-1β. Therefore, we demonstrate that Gly(2)-GLP-2 attenuates NLRP3 inflammasome-mediated neuroinflammation in the PD mice induced by MPTP.
In our study, Gly(2)-GLP-2 alleviated neuron mitochondrial dysfunction in the substantia nigra of PD mouse model. Mitochondrial dysfunction may be another critical factor in the loss of dopaminergic neurons [79–81]. Mitochondrial fission and fusion play critical roles in maintaining mitochondria function [82]. This study investigated whether Gly(2)-GLP-2 could improve MPTP-induced mitochondrial abnormal fission and fusion in the substantia nigra of PD mice. We found that Gly(2)-GLP-2 increased the expression of mitochondria fusion protein Mfn2 and reduced the level of mitochondria fission protein FIS1. The result indicates that Gly(2)-GLP-2 improves MPTP-induced mitochondrial abnormal fission and fusion. Therefore, we demonstrate that Gly(2)-GLP-2 alleviates MPTP-induced mitochondrial dysfunction in the PD mice.
This study found that GLP-2 ameliorated motor deficits in MPTP-treated mice by reducing the inflammation and mitochondrial dysfunction in the substantia nigra, very similar to the dual GLP-1/GIP receptor agonist DA5-CH. The combination of both GLP-1 and GIP receptors activation turned out to be superior to one of them [39, 40]. It may be that combinations of GLP-1, GLP-2 and GIP receptor agonists are even more potent. Further experiments that compare different doses and combinations of receptor agonists are required to answer the question if GLP-2 can add significantly to the neuroprotective effects of GLP-2 and GIP. Additionally, this study shows that Gly(2)-GLP-2 is only tested in acute PD mouse model. In order to study the role of GLP-2 in neuroprotection in more detail, further studies on the detection of Gly(2)-GLP-2 in the other PD animal models are required to investigate whether Gly(2)-GLP-2 has neuroprotective properties.
In conclusion, our study indicate that GLP-2 analogue Gly(2)-GLP-2 improves motor dysfunction by reducing NLRP3 inflammasome-mediated neuroinflammation and mitochondrial damage in the substantia nigra induced by MPTP. These results demonstrate that Gly(2)-GLP-2 may be a latent neuroprotective drug with good clinical application prospects in PD.
CONFLICT OF INTEREST
The authors declare no competing interest.
Footnotes
ACKNOWLEDGMENTS
The project was funded by a grant to CH as a Zhongyuan science and technology innovation leading talent (number 204200510022), Scientific and Technological Project of Henan Province (numbers 202102310078, 172102310286), The key Scientific Research Project of Universities in Henan Province (numbers 19A310012, 18B310004).
