Abstract
Background:
Parkinson’s disease (PD) is a progressive neurodegenerative disease for which there is no cure. In a clinical trial, the glucagon-like peptide-1 (GLP-1) receptor agonist exendin-4 has shown good protective effects in PD patients. The hormone glucose-dependent insulinotropic polypeptide (GIP) has also shown protective effects in animal models of PD.
Objective:
We tested DA-CH5, a novel dual GLP-1/GIP receptor agonist.
Methods:
DA-CH5 activity was tested on cells expressing GLP-1, GLP-2, GIP or glucagon receptors. The ability to cross the blood-brain barrier (BBB) of DA-CH5, exendin-4, liraglutide or other dual receptor agonists was tested with fluorescein-labelled peptides. DA-CH5, exendin-4 and liraglutide were tested in the MPTP mouse model of PD.
Results:
Analysing the receptor activating properties showed a balanced activation of GLP-1 and GIP receptors while not activating GLP-2 or glucagon receptors. DA-CH5 crossed the BBB better than other single or other dual receptor agonists. In a dose-response comparison, DA-CH5 was more effective than the GLP-1 receptor agonist exendin-4. When comparing the neuroprotective effect of DA-CH5 with Liraglutide, a GLP-1 analogue, both DA-CH5 and Liraglutide improved MPTP-induced motor impairments. In addition, the drugs reversed the decrease of the number of neurons expressing tyrosine hydroxylase (TH) in the SN, alleviated chronic inflammation, reduced lipid peroxidation, inhibited the apoptosis pathway (TUNEL assay) and increased autophagy -related proteins expression in the substantia nigra (SN) and striatum. Importantly, we found DA-CH5 was superior to Liraglutide in reducing microglia and astrocyte activation, improving mitochondrial activity by reducing the Bax/Bcl-2 ratio and normalising autophagy as found in abnormal expression of LC3 and p62.
Conclusion:
The results demonstrate that the DA-CH5 is superior to liraglutide and could be a therapeutic treatment for PD.
INTRODUCTION
Parkinson’s disease (PD), the second most common neurodegenerative disease, is characterised by the loss of dopaminergic neurons in the substantia nigra (SN) [1] which results in impaired motor functions, displayed as compromised postural reflexes, muscular rigidity and bradykinesia [2]. PD affects millions of people worldwide [3]. The main treatment currently is levodopa that can temporarily reverse the disability and improve quality of life [4, 5]. However, levodopa does not affect disease progression, and the declining effectiveness in long-term therapy is a serious limitation [6]. Drugs that initially have been developed to treat type II diabetes have been re-purposed as treatment for Parkinson’s disease [7, 8]. These drugs are long-lasting, protease resistant mimetics of the hormone and growth factor glucagon-like peptide 1 (GLP-1) [9–11]. GLP-1 and GLP-1 receptors are expressed in the brain and have shown good protective effects [12]. Recently, a phase II clinical trial showed protective effects of the GLP-1 receptor agonist exendin-4 (Exenatide, Bydureon) in PD patients. The progressive degeneration was stopped in the drug group but not in the placebo group, and the effect that was still present 3 months after treatment had stopped [13]. When analysing neuronal exosomes in the blood, it was found that drug treatment normalised insulin signalling as shown in Akt and mTOR activation [14], which mirrors our findings in animal studies [12]. This proof of principle demonstrates that activation of the GLP-1 growth factor receptor can arrest disease progression not only in animals but in PD patients, too. The underlying mechanism appears to be the re-activation of insulin signalling, energy utilisation, improvement of mitochondrial functions, and the normalisation of gene expression of key proteins such as tyrosine hydroxylase, an enzyme involved in the synthesis of dopamine [12]. In addition, GLP-1 has anti-inflammatory properties [15, 16]. We and others have previously shown that GLP-1 receptor agonists can reverse the impairments observed in animal models of PD [17–19]. We also showed that analogues of the growth factor glucose-dependent insulinotropic polypeptide (GIP) also showed good neuroprotective effects in these models [20, 21]. Both growth factors normalised the expression of tyrosine hydroxylase in dopaminergic neurons, reversed motor impairments, reduced mitochondrial dysfunction and apoptosis, normalised expression of key growth factors such as GDNF and BDNF, and reduced the chronic inflammation response in the brain in animal models [12]. The underlying mechanism appears to be a normalisation of growth factor signalling. In patients with PD, it was found that insulin, IGF-1 and other growth factors have lost their efficacy [22–25]. This de-sensitisation is most likely caused by the chronic inflammation response in the brain that is inherent with PD, and the actions of pro-inflammatory cytokines that are released by activated microglia [26, 27]. Recently, novel dual GLP-1/GIP receptor agonists have been developed [28]. We previously tested two of these (DA-JC1 and DA3-CH), and they showed good effects in animal models of PD [29–31]. Based on these encouraging effects, we developed new dual GLP-1/GIP receptor agonists that showed improved effects compared to the older ones. In a previous comparative study, the novel dual agonists DA-JC4 and DA-CH5 showed improved neuroprotective effects in the MPTP mouse model of PD compared to the older dual agonist DA1-JC [32]. In that study, all drug doses were equal for direct comparison. Here, we report the receptor binding specificity to GLP-1 and GIP receptors, the ability of single and double receptor agonists to cross the BBB, a dose-response relationship in comparison with the effects of the first GLP-1 receptor agonist to be brought to the market as a diabetes treatment, exendin-4 (exenatide), and furthermore compared the protective effects of DA-CH5 and liraglutide in the MPTP mouse model on motor activity, numbers of dopaminergic neurons, inflammation, oxidative stress, apoptosis, and autophagy in substantia nigra (SN) and striatum. Due to the lower bio-availability of liraglutide in the blood stream [33], we used a lower dose of DA-CH5 to compare both drugs at more equal terms.
MATERIALS AND METHODS
Reagents and peptides
Reagents used were Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP, Sigma-Aldrich, USA), RIPA lysis buffer (Beyotime Biotechnology, China), BCA protein assay kit (Boster Biotechnology, China), paraformaldehyde perfusion (PFA, Boster Biotechnology, China), ECL-enhanced chemoluminescence (Boster Biotechnology, China), Goat IgG SABC immunohistochemical Kit (Boster Biotechnology, China), TUNEL Apoptosis Detection Kit (BOSTER, China), rabbit anti-tyrosine hydroxylase (TH) antibody (Abcam, UK), rabbit anti-BCl-2 antibody (Bioworld Techology, USA), rabbit anti-Bax antibody (Bioworld Techology, USA), rabbit anti-LC3 antibody (Bioworld Techology, USA), rabbit anti-Atg7 antibody (Bioworld Technology, USA), rabbit anti-β-actin antibody (Bioworld Technology, USA), rabbit anti-GFAP antibody (Boster Biotechnology, China), rabbit anti-IBA-1 antibody (Boster Biotechnology, China), rabbit anti-4-HNE antibody (Bioss Biotechnology, China), HRP-labelled goat anti-rabbit immunoglobulin secondary Antibody (Boster Biotechnology, China).
Incretin peptides were synthetized by Chinapeptides and GL Biochem (Shanghai, China). The purity of each peptide was analysed by reversed-phase HPLC and characterized using matrix-assisted laser desorption/ionization time of flight (MALDI-TOF) mass spectrometry, with a purity >95%. Peptides were reconstituted in ultrapure water (Milli-Q) to a concentration of 1 mg/mL and aliquots prepared and stored at –20°C.
Peptides tested and AA sequences:
Exendin-4
HGEGTFTSDLSKQMEEEAVRLFIEWLKNGGPSSGAPPPS-NH2
Liraglutide
HAEGTFTSDVSSYLEGQAAK[(γE)-(Pal)] EFIAWLVRGRG
Pal = palmitoyl acid;
Dual agonist 3 (DA3)
Y
Dual agonist 5 (DA-CH5)
Y
cAMP measurements
Transfection and cell culture
COS-7 cells were cultured at 10% CO2 and 37°C in Dulbecco’s modified Eagles medium 1885 supplemented with 10% FBS, 2 mmol/L glutamine, 180 units/mL penicillin, and 45 g/mL streptomycin. Transient transfection was performed using the calcium phosphate precipitation method [34] with all receptors: GIP, GLP1, GLP2 and the glucagon receptor.
Receptor cAMP study
Transiently transfected COS-7 cells were seeded in 96-well plates one day after transfection at 35.000 cells/well in white plates and the experiments carried out the following day. At the assay day, the cells were washed twice with Hepes-buffered saline (HBS) buffer and incubated with HBS and 1 mmol/L 3-isobutyl-1-methylxanthine (IBMX) for 30 min at 37°C [35]. The agonists were added and incubated for 30 min at 37°C. The HitHunterTM cAMP XS assay (DiscoveRx, Herlev, Denmark) was carried out according to the manufacturer’s instructions. In vitro pharmacological analysis with determination of EC50 and Emax were carried out using GraphPad Prism. Sigmoid curves were fitted logistically with a Hillslope of 1.0.
BBB penetration study
We tested the peptides exendin-4, liraglutide, DA1 (acetylated), DA2 (pegylated with a 40 kDa pegylation), DA3 (the peptide without any modifications), and DA5 (with the TAT sequence). 3-month-old C57BL6 mice were injected with fluorescein-labelled peptide at 50 nmol/kg ip. Two h min later, animals were anaesthetised with Dolethal pentobarbital (BAYER) at 50 mg/kg and were transcardially perfused with approximately 20 ml phosphate buffered saline (PBS, pH7.4), followed by approximately 20 ml 4% paraformaldehyde (PFA, pH7.4). N = 6 per group. Brains were post-fixed in PFA for 24 h in 20% sucrose solution and sectioned on a Leica cryostat at 40 μm, and fluorescence was quantified on a Zeiss Axio Scope A1 microscope under 488 nm illumination. Images were taken with a Sony ICX 267 digital camera at 525 nm. One image per brain was taken at a random location in the cortex. The image size was 250×250 μm. Fluorescence was quantified using Image-Pro Plus 6.0 software (Media Cybernetics, Inc., USA)
Animals and MPTP treatment
Adult (8 weeks old, weighing approximately 20∼22 g), male C57BL6/J mice were purchased from the Experimental Animal Center, ShanXi Medical University. The animals were maintained under standard temperature and humidity conditions with a 12 h light/dark cycle and fed a standard pellet diet and water ad libitum. For the dose-response study, mice were randomly divided into eight groups and were treated intraperitoneally (i.p.) injections with saline or MPTP (25 mg kg–1) in the presence of incretin analogues at different doses (0.1 mg kg–1, 1.0 mg kg–1, 10 mg kg–1) for seven consecutive days, including Group A: Saline; Group B: MPTP - only; Group C: 10 mg kg–1 DA-CH5 + MPTP; Group D: 1.0 mg kg–1 DA-CH5 + MPTP; Group D: 0.1 mg kg–1 DA-CH5 + MPTP; Group E: 10 mg kg–1 Exendin-4 + MPTP; Group F: 1.0 mg kg–1 Exendin-4 + MPTP; Group H: 0.1 mg kg–1 Exendin-4 + MPTP. The sample number of each group is 8. For the comparison between DA-CH5 and liraglutide, separate groups were set up. After 1 week of acclimatization, mice were randomly divided into six groups and were treated with i.p. injections with saline or MPTP (25 mg kg–1) in the presence or absence of incretin analogues (Liraglutide 25 mg/kg or DA-CH5 10 mg/kg), respectively. Group A: Saline; Group B: Liraglutide - only; Group C: DA-CH5 - only; Group D: MPTP - only; Group E: Liragultide + MPTP; Group F: DA-CH5 + MPTP. The sample number of each group is 12. Mice were rendered parkinsonian by the daily i.p. administration of MPTP for 1 week and were treated with saline and incretin analogues once a day for seven consecutive days. A timeline of the experiment is shown in Fig. 3.
Behavioural assessment
Open field test
In the present study, the apparatus was composed of a round arena (35 cm in diameter; 40 cm high wall) and used a computer tracking system (Etho Vision XT software, Noldus information technology, Wageningen, Netherlands) to record the animal’s movements. Twenty-four hours after habituation to the test room, each mouse was placed in the center of the open-field, and its locomotor activity during the subsequent 10 min was videotaped and analysed by the computer tracking system. The area was wiped with 75% ethanol after each mouse trial to prevent build- up of olfactory cues. The open-field test was performed on day 8, and conducted by an examiner who was blinded to the treatment groups.
Rotarod test
The rotarod apparatus (YLS-4C, Academy of medical sciences in Shandong, China) comprised a horizontal rod, 3 cm in diameter, separated by opaque plastic dividers in order to accommodate up to five mice per trial. In the rotarod task, all mice were pre-trained for consecutive 3 days prior to drug administration. On the assessment, the latency to fall from the rod was automatically recorded. The speed of rod accelerated from 0 to 30 rpm. The maximum duration allowed per trial was 3 min.
Footprint test
The gait analysis started by placing the animal in a handmade dark wooden tunnel (40 cm long, 10 cm wide, and 8 cm tall), the hind limbs were dipped in blue ink. During training and testing, the mouse was placed at the starting zone facing the dark tunnel. When the animal walked through the tunnel, the footprints were record in the white paper. To avoid velocity variations affecting stride length, the two initial steps for the measurements and the footprints recording when the mice walked through the tunnel with obvious velocity changes were discarded. Stride lengths were determined by measuring the distance between each step on the same side of the body. The shortest stride was subtracted from the longest stride to decide the stride variability.
Tissue preparation
After behavioural testing had been completed, mice were anesthetised and sacrificed. For immunohistochemistry, mice were perfused with approximately 20 ml phosphate buffered saline (PBS, pH7.4), followed by approximately 20 ml 4% paraformaldehyde (PFA, pH7.4). The fixed brains were removed, post-fixed overnight in the PFA at 4°C. Later, fixed brains were gradiently dehydrated with ethanol and xylene. Finally, the brains were embeded with paraffins. For western blot, brains were removed quickly and were dissected. Later, brains were rinsed with ice-cold PBS and stored at –80°C until western blot analysis was performed.
Immunohistochemistry
Coronal sections (thickness 4 μm) were cut on a semiautomatic Microtome (Leica, Wetzlar, Germany), and heat-mediated antigen retrieval was performed before the IHC staining protocol was begun. Brain sections were blocked in 10% fetal bovine serum (FBS) for 1 h and then incubated overnight at 4°C with the primary antibody and followed by incubation in room temperature with the secondary antibodies. Finally, the slices were mounted and captured using a microscope (Motic BA410; Motic, China). Quantification of stained cells in the substantia nigra pars compact (SNpc) and in the striatum was carried out in the region spanning from –2.92 mm to –3.40 mm and from 1.54 mm to 0.10 mm relative to Bregma respectively. The total number of TH cells was estimated from every 6–8 section covering the entire of these regions and the average cell count per animal was used for statistical analysis. To show the changes in a clear manner, controls were defined as 100% (the numerical average of all control animals) and drug groups were normalised according to that. Counting was manually performed using the Image-Pro Plus 6.0 software after digitalization of the images (Media Cybernetics, Inc., USA). For analysis of the inflammation response, we randomly took eight 40X magnification images in the region of striatum and SN per sections and the average cell count per animal was used for statistical analysis. Five brain slices per animal were quantified. Counting was manually performed using the Image-Pro Plus 6.0 software after digitalization of the images. Rabbit anti-TH (1:400; abcam, UK), rabbit anti-GFAP (1:200; Boster, China), rabbit anti-IBA-1 (1:200; Boster, China), rabbit anti-4-HNE (1:200; Bioss, China), rabbit anti-Bcl-2 (1:200; Bioworld, USA) and rabbit anti-Bax (1:200; Bioworld, USA) and a secondary peroxidase-conjugated antibody (Boster, China) were used for staining in this study.
TUNEL assay
Terminal deoxynucleotidyl Transferase Biotin-dUTP Nick End Labeling (TUNEL)-positive apoptotic nuclei were detected in brain paraffin sections using an TUNEL Apoptosis Detection Kit (BOSTER, China) according to manufacturer’s instructions. The number of apoptotic cells was counted by using the Image-Pro Plus 6.0 software in the striatum to calculate the ratio of apoptotic cells per brain area.
Western blot analysis
Western blot assay was performed using a standard protocol. Briefly, substantia nigra (SN) tissues were homogenized in the RIPA lysis buffer containing 1:100 phenyl-methylsulfonyl fluoride (PMSF). The lysates were centrifuged at 12,000 rpm for 20 min at 4°C and total protein concentration was evaluated with BCA Protein Assay Kit (Boster, Wuhan, China). Samples were separated on SDS page by using a 10% Bis-Tris gel and then transferred onto polyethylene difluoride (PVDF) membranes blocked with 5% BSA (Boster, Wuhan, China) for 1 h at room temperature and incubated with primary antibodies overnight at 4°C. The membranes were incubated with secondary antibodies (1:5000; Boster, Wuhan, China) subsequently at room temperature for 1 h and were then imaged with the ECL-enhanced chemilluminescence (Boster, China). Western blot images were made visible using a chemiluminescent imaging system (Sagecreation, China) and band intensities were quantified by optical densitometry using Image-Pro Plus 6.0 (Media Cybernetics, USA). Rabbit anti-Bcl-2 (1:1000; Bioworld, USA), rabbit anti-Bax (1:1000; Bioworld, USA), rabbit anti-ATG7 (1:1000; Bioworld, USA) and rabbit anti-LC3 were used for staining in this study. Rabbit anti- β-Actin (1:2000; Bioworld, USA) always served as an internal loading control.
Statistical analysis
Data were analysed using GraphPad Prism 5.0 software and the data are expressed as mean±standard errors of the mean (S.E.M.). The significance level of treatment effects was determined using a one way analysis of variance (ANOVA) followed by Tukey or Newman-Keuls multiple comparison test between groups. A probability of <5% (p < 0.05) was considered to be statistically significant.
RESULTS
Receptor binding cAMP study
COS-7 cells were successfully transfected with the hGCG-R, hGIP-R, hGLP2-R and hGLP-1R. The native peptide hormones glucagon, GIP, GLP2 and GLP1 activated their respective receptors with potencies similar to previously described [36] or GLP-1, we observed a potency (LogEC50±SEM) of –10,1±–0,11, for GIP:–10,9±0,09, for glucagon: –9,3±0,12 and for GLP2: –8,2±0,03 (Fig. 1A–D). The ability of DA-CH5 to activate the receptors was determined on all four receptors. We observed that DA-CH5 acted as a high potency agonist of the GLP-1R and the GIP-R with the same efficacies (Emax) as the endogenous ligands, thus acting as a full agonist on both receptors. Moreover, it acted with similar potencies on the two receptors with logEC50±SEM of –8,3±0,16 for the GLP-1R and –8,6±0,15 for the GIP-R (Fig. 1A, B). Compared to the respective endogenous agonists, DA-CH5 thus acted with a decreased potency of 63-fold at the GLP-1 receptor and 199-fold at the GIP receptor. In contrast, no activation by DA-CH5 was observed at the GCG-R and the GLP2-R, (Fig. 1C, D).
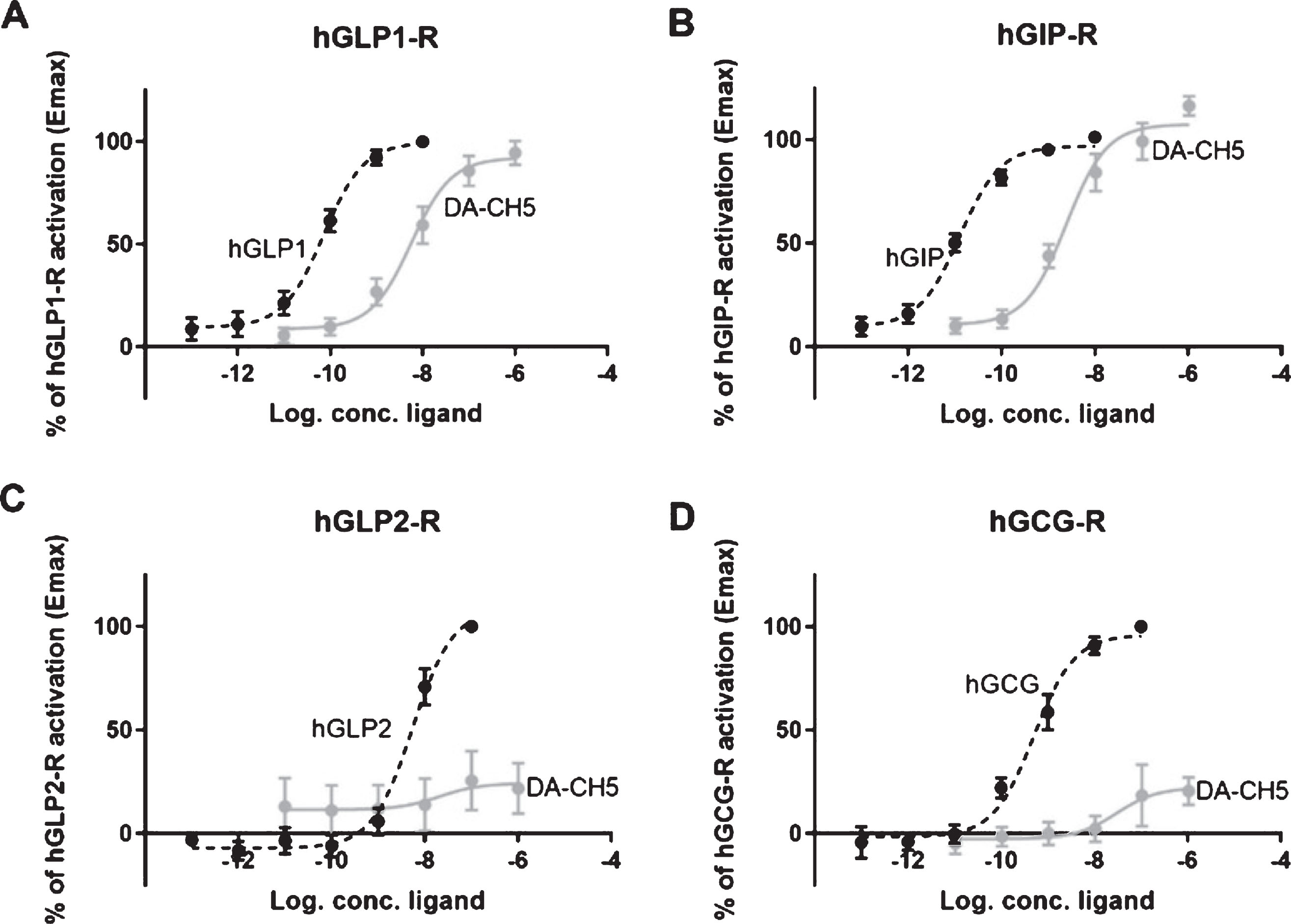
Activation of hGLP1-R, hGIP-R, hGLP2-R, and hGCG-R by endogenous agonist and DA-CH5 cAMP accumulation in COS-7 cells transfected with (A) hGLP-1 receptor, treated with increasing concentrations of hGLP-1 and DA-CH5 (B) hGIP-R treated with increasing concentrations of hGIP and DA-CH5 (C) hGLP2-R treated with increasing concentrations of hGLP-2 and DA-CH5 (D) hGCG-R treated with increasing concentrations of hGCG and DA-CH5.
BBB penetration study
In a one-way ANOVA, a difference was found between all groups (p < 0.0001). Post-hoc Tukey’s multiple comparison test found significant differences between groups. The pegylated version of the dual agonist did not readily cross the brain. The acetylated peptides liraglutide and DA1 crossed the BBB at a lower rate than unmodified peptides but at a higher rate than the modified peptides. Exendin-4 and DA3 crossed the BBB better than the modified peptides but not as well as DA5-CH which had the TAT sequence addition. See Fig. 2.
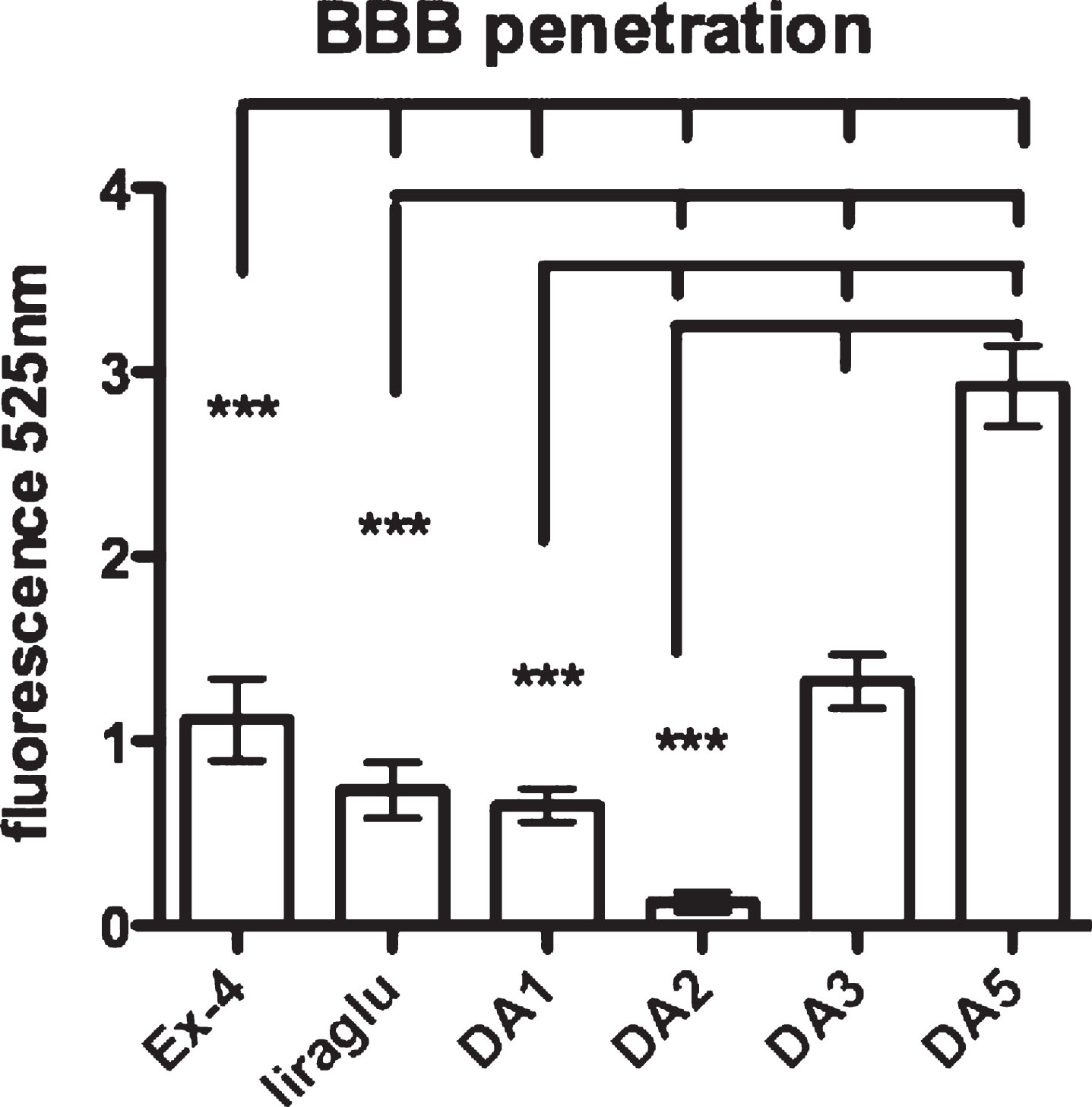
BBB penetration by Fluorescein-labelled peptides in brain tissue. One-way ANOVA (p < 0.001) and post-hoc tests ***p < 0.001. N = 6 per group. DA, dual agonist. DA1 and liraglutide were acetylated with a C16 fatty acid, while DA2 was pegylated (40 kDa).
Dose-response curve
Rotarod assessment
Comparison between groups in the Rotarod test. A one-way ANOVA found a difference between all groups (p < 0.0001). Post-hoc Tukey’s multiple comparison test found significant differences between groups. DA5-CH improved motor performance at 10 (p < 0.01) and 1 nmol/kg (p < 0.05) doses when compared to MPTP saline, while Exendin-4 improved motor performance at 10 nmol/kg only (p < 0.05). The saline control group was not different to the DA5-CH 1 nmol/kg group, but different from all other groups (p < 0.01; p < 0.001). See Fig. 4A.
Schematic presentation of the study design.
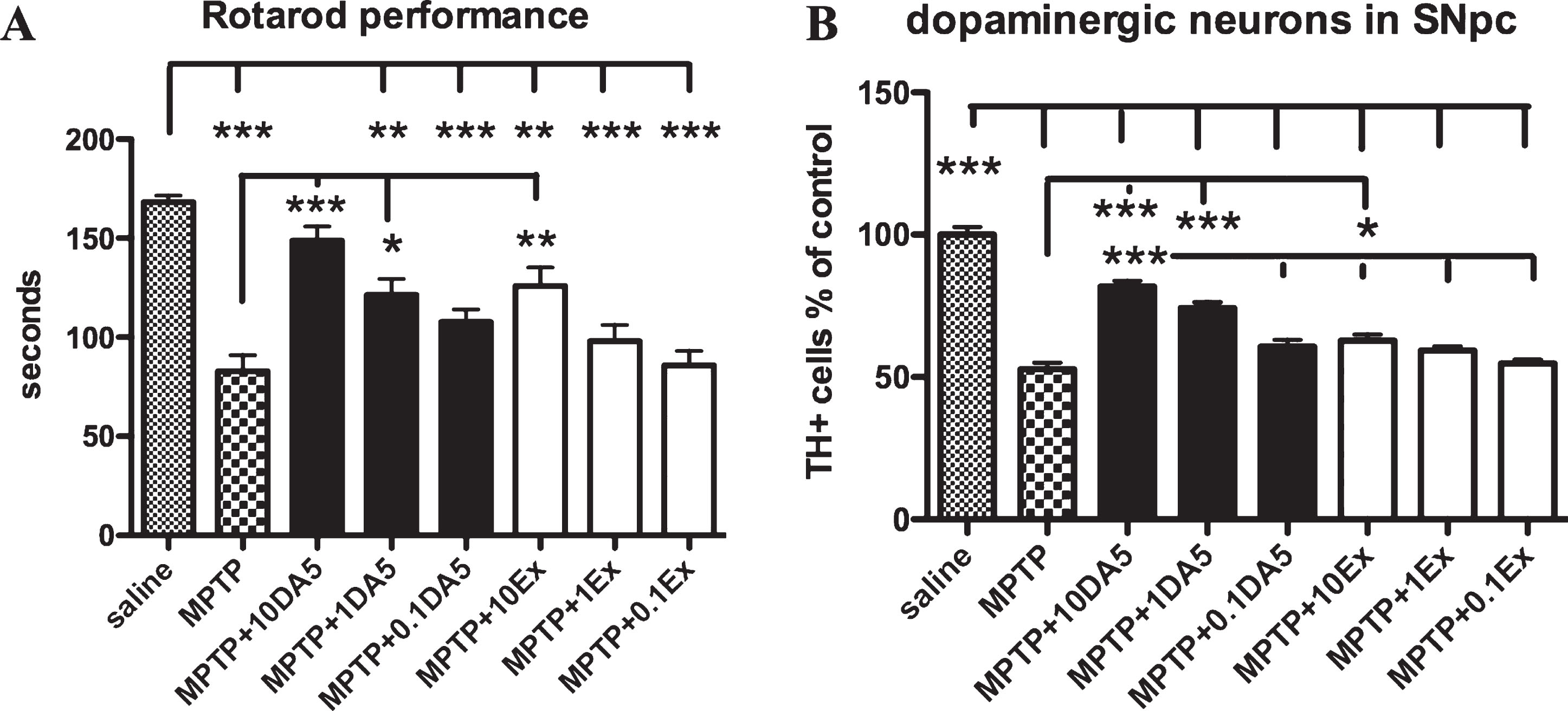
A) Comparison between groups in the Rotarod test. A one-way ANOVA found a difference between all groups (p < 0.0001). Post-hoc Tukey’s multiple comparison test found significant differences between groups. *p < 0.05; ***p < 0.001. DA5-CH improved motor performance at 10 and 1 nmol/kg doses, while exendin-4 improved motor performance at 10 nmol/kg only. B) Quantification of tyrosine-hydroxylase positive neurons (TH+) in the substantia nigra. A one-way ANOVA found a difference between all groups (p < 0.0001). Post-hoc Tukey’s multiple comparison test found significant differences between groups. *p < 0.05; ***p < 0.001.
Quantification of TH positive cells in the substantia nigra pars compacta
Quantification of tyrosine-hydroxylase positive neurons (TH+) in the substantia nigra showed clear differences between drug groups. A one-way ANOVA found a difference between all groups (p < 0.0001). Post-hoc Tukey’s multiple comparison test found significant differences between groups.
The dual agonist DA5-CH is more effective at protecting dopaminergic neurons than exendin-4. Only at the highest dose, exendin-4 showed a significant protection compared to the MPTP group (p < 0.05). DA5-CH showed a significant protection at the 1 (p < 0.001) and 10 nmol/kg (p < 0.001) ip. dose. DA5-CH at the 10 nmol/kg ip dose is significantly more effective than exendin-4 at the 10 nmol/kg ip dose. All groups were different from the saline only group (p < 0.001). Importantly, DA5-CH at the 10 nmol/kg ip dose is significantly different to all exendin-4 groups (p < 0.001). See Fig. 4B.
DA-CH5 and Liraglutide normalised motor impairments induced by MPTP
In the open field test, DA-CH5 alleviated the locomotor impairments induced by MPTP treatment. A one-way ANOVA found an overall difference in the distance travelled (p < 0.0001; Fig. 5A) between all groups. There was not any difference between the control, Liraglutide only injection, DA-CH5 only injection, MPTP + Liraglutide and MPTP + DA-CH5 groups (p > 0.05; Fig. 5A). Results showed that the distance travelled by MPTP group mice exhibited a significant reduction to 71.3% (p < 0.001) versus the control group mice. Liraglutide and DA-CH5 treatment normalised the distance reduction caused by MPTP to 103.7% (p < 0.001) and 111.2% (p < 0.001) compared with MPTP group. There was no significant difference between MPTP + DACH5 group and MPTP + Liraglutide groups (p > 0.05; Fig. 5A). n = 12 mice in each group.
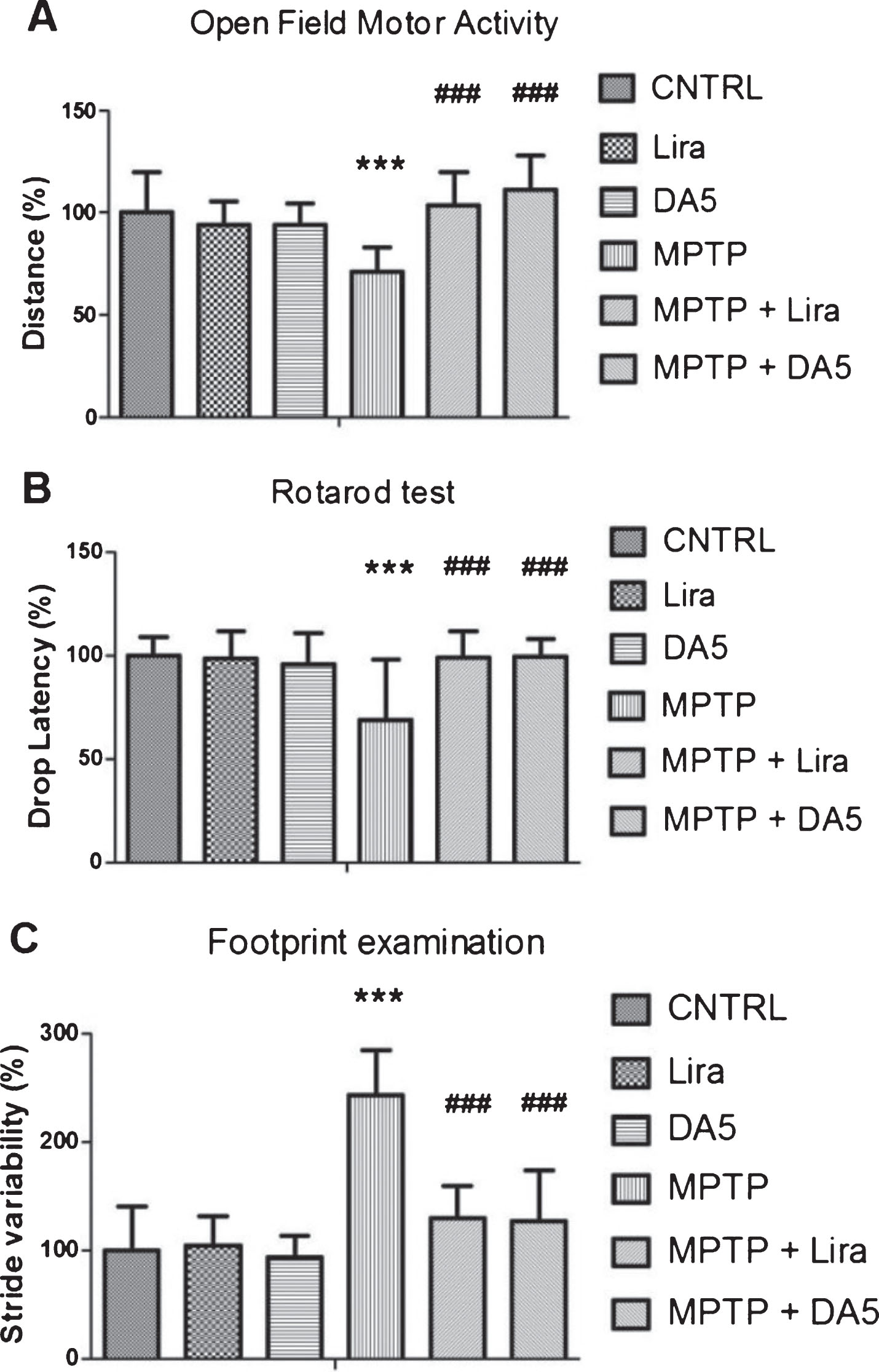
DA-CH5 and liraglutide protects the MPTP-induced impairment in motor activity of mice. A) Open field test. MPTP reduced the spontaneous locomotion of mice (***p < 0.001 vs. the control group), while DA-Ch5 and liraglutide reversed this impairment. B) Rotarod test. MPTP reduced drop latency (***p < 0.001 vs. the control group), while DA5 and liraglutide improved muscular strength and hypokinesia. C) Quantitative data for footprint examination. MPTP increased stride variability (***p < 0.001 vs. the control group), while DA-CH5 and liraglutide improved the motor coordination. The values represent the means±S.E.M.; n = 12 per group
In the rotarod test, a one-way ANOVA found an overall difference between groups (p < 0.0001). There was no difference between control, Liraglutide injection, DA-CH5, MPTP + Liraglutide and MPTP + DA-CH5 groups (p > 0.05). Results showed that MPTP injection impaired muscular strength and movement balance of mice, with less time to 68.9% (p < 0.001) staying on the rotating rod compared with the control group. However, treatment with Liraglutide and DA-CH5 significantly reversed the drop latency reduction induced by MPTP to 98.8% (p < 0.001) and 99.2% (p < 0.001), respectively. The drop latency between MPTP + DA-CH5 and the MPTP + Liraglutide group mice were not different (p > 0.05; Fig. 5B). n = 12 mice in each group.
In the footprint examination, a one-way ANOVA found an overall difference between groups (p < 0.0001; Fig. 5C). There was no difference between control, Liraglutide, DA-CH5, MPTP + Liraglutide and MPTP + DA-CH5 groups (p > 0.05). The results showed that MPTP-treated mice exhibited higher variability in their stride length to 243.1% (p < 0.001) indicating a deficiency in gait coordination with respect to the controls. Interestingly, this motor deficit was significantly ameliorated by Liraglutide treatment to 129.9% (p < 0.001) and DA-CH5 treatment to 127.3% (p < 0.001) respectively. Stride variation between MPTP + DA-CH5 and MPTP + Liraglutide group mice was not different (p > 0.05; Fig. 5C). n = 12 mice in each group.
DA-CH5 and Liraglutide attenuated dopaminergic neuronal loss in the SN and striatum induced by MPTP
To determine the neuroprotective effect of the drugs tested, we analysed the levels of tyrosine hydroxylase (TH), a key enzyme expressed in dopaminergic neurons. A one-way ANOVA found an overall difference in the SN (p < 0.001; Fig. 6A) and the striatum (p < 0.001; Fig. 6C). After 7 days of MPTP injection, the number of TH positive cells in the SN was decreased to 64.42% (p < 0.01; Fig. 6A, B), and the average optical density of TH staining in the striatum was decreased to 57.01% (p < 0.001; Fig. 6C, D) compared with the control group. There was no difference between the control group, Liraglutide group and DA-CH5 group both in the SN (p > 0.05) and striatum (p > 0.05). However, DA-CH5 and Liraglutide treatment restored numbers of dopaminergic neurons to 89.3% and 83.6% in SN (p < 0.05) in MPTP-treated mice. Besides that, DA-CH5 and Liraglutide treatment elevated overall levels of TH in the striatum to 70.3% (p < 0.01) and 71.3% (p < 0.01) respectively compared with the MPTP group. However, there is not any difference between the MPTP + DA-CH5 and MPTP + Liraglutide groups both in the SN (p > 0.05; Fig. 6A, B) and striatum (p > 0.05; Fig. 6C, D). n = 6 per group.
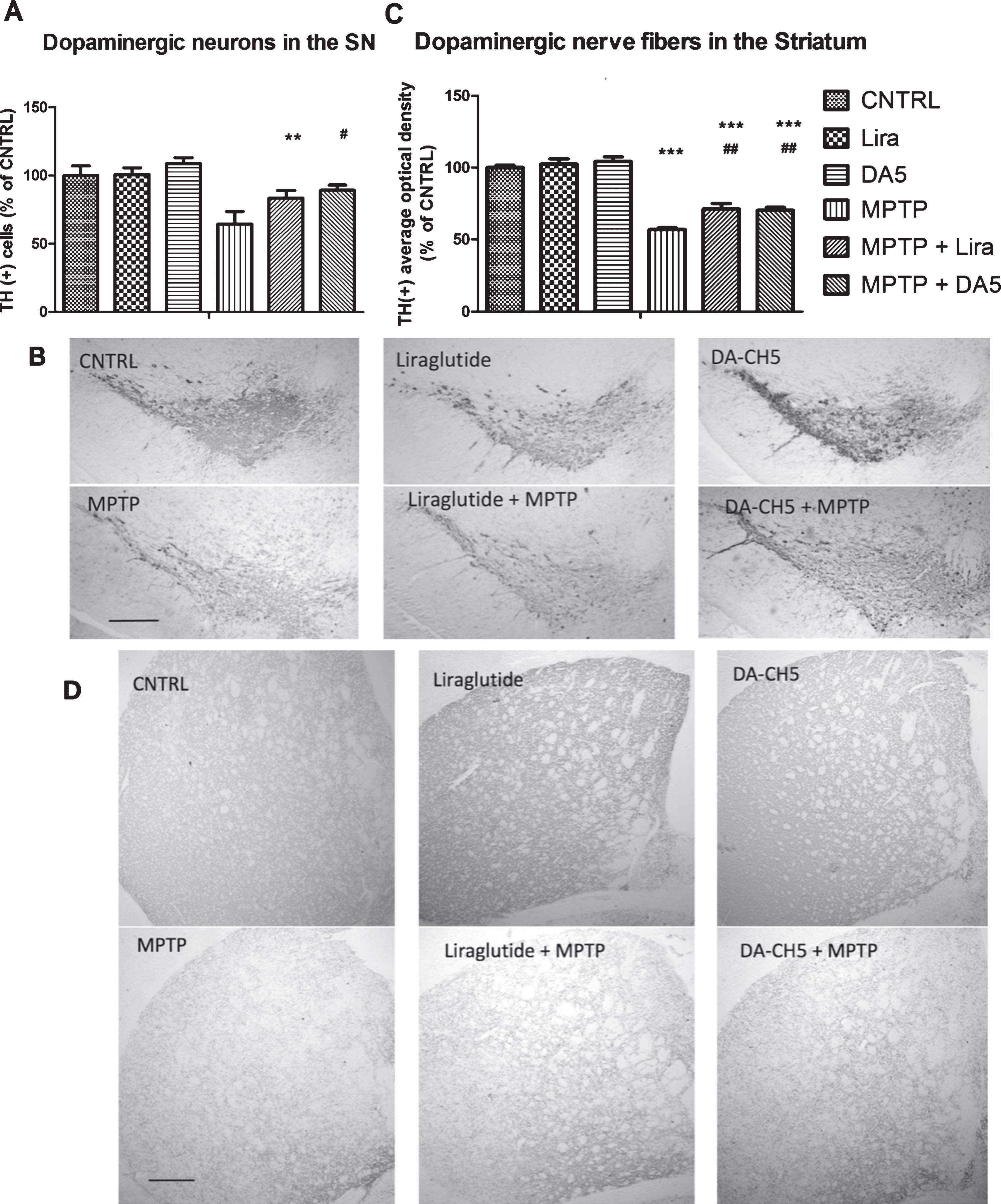
DA-CH5 and liraglutide normalised the number of TH positive neurons in the SN and gradient of TH expression in the striatum of MPTP-induced PD mice. A) Number of TH+ dopaminergic neurons in the SN. The number of TH+ neurons was decreased in the MPTP group (**p < 0.01 vs. the Control group). DA-CH5 and liraglutide restored TH positive dopaminergic neuron numbers (#p < 0.05 vs. the MPTP group). The values represent the means±S.E.M. n = 6 per group. B) Sample images of TH staining in the SN. Scale bar is 200 μm. C) Average optical density of TH staining in the striatum. The optical density was decreased in the MPTP group, liraglutide + MPTP group and DA-CH5 + MPTP group (***p < 0.001 vs. the Control group). DA-CH5 and liraglutide ameliorated the decreased of average optical density of TH staining induced by MPTP (# #p < 0.01 vs. the MPTP group). The values represent the means±S.E.M. n = 6 per group. D) Sample images of TH staining in the striatum. Scale bar is 200 μm.
After MPTP treatment, DA-CH5 and Liraglutide alleviated astrocyte and microglia activation in the SN and striatum
GFAP was used as a biomarker for astrocyte activation. A one-way ANOVA found an overall difference in the SN (p < 0.001; Fig. 7A) and the striatum (p < 0.001; Fig. 7C) of mice. There was no difference between control, DA-CH5 only and Liraglutide only groups both in the SN (p > 0.05; Fig. 7A, B) and striatum (p > 0.05; Fig. 7 C, D). GFAP-positive astrocytes increased to 203.8% in the SN (p < 0.001) and 384.3% in the striatum (p < 0.001) of MPTP-treated mice, which was significantly higher than in control mice. Liraglutide treatment of MPTP-treated mice reduced GFAP-positive astrocytes numbers to 171.8% in the SN (p < 0.001) and 281.6% in the striatum (p < 0.001), which was significantly less than in the MPTP group (384%). DA-CH5 treatment could also ameliorate the astrocyte activation induced by MPTP, the GFAP-positive level decreased to 141.3% in the SN (p < 0.001) and 244.7% in the striatum (p < 0.001) compared with MPTP group (384%). MPTP + DA-CH5 group differed from the MPTP + Liraglutide group in both the SN (p < 0.001; Fig. 7A, B) and striatum (p < 0.05; Fig. 7C, D). n = 6 per group.
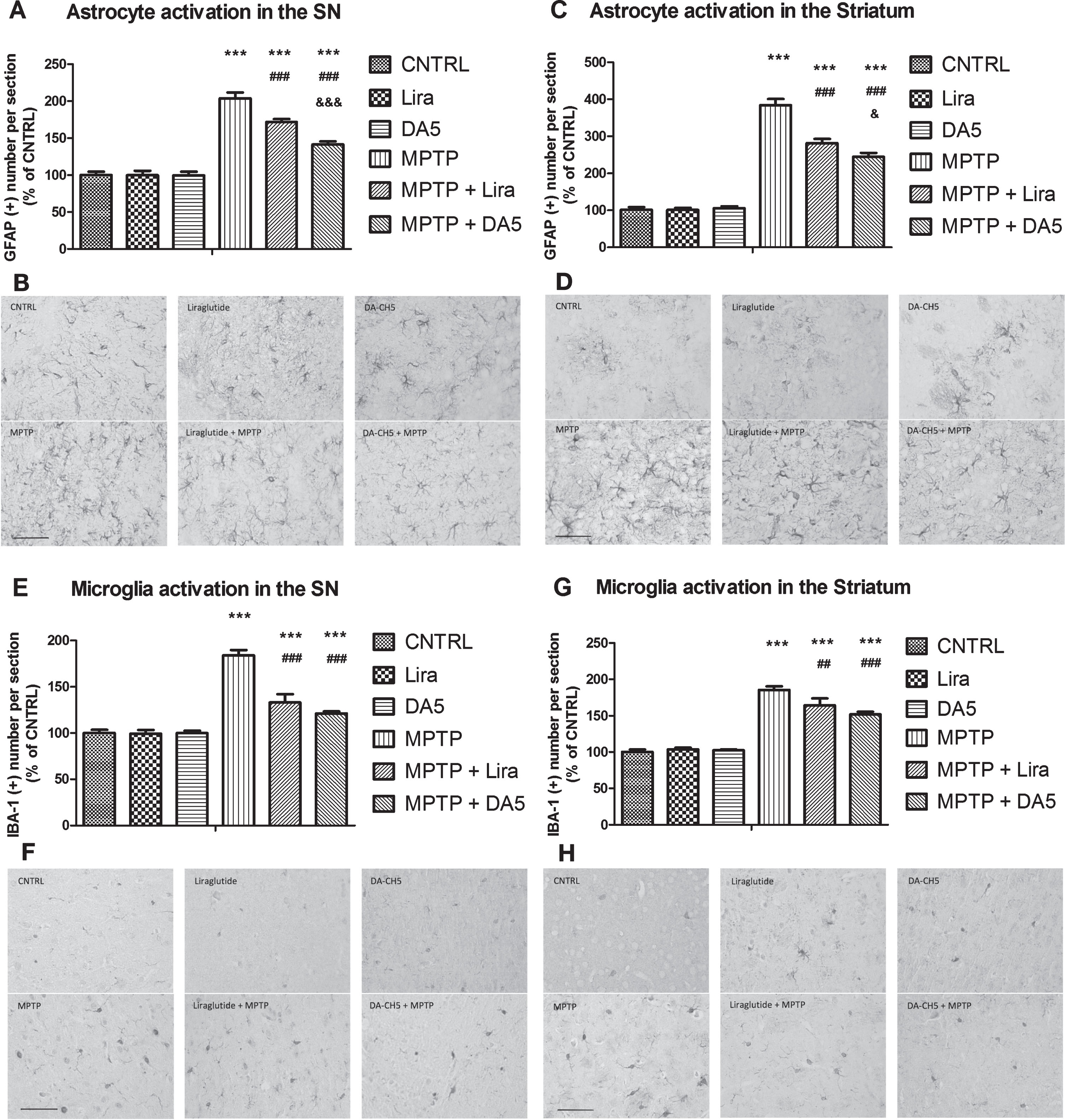
DA-CH5 and liraglutide reduced chronic inflammation in the SN and striatum induced by MPTP. A) The number of activated astrocytes in the SN. Individual star-shaped cells are clearly visible in the micrographs. The number was increased in the MPTP group, liraglutide + MPTP group and DA-CH5 + MPTP group (***p < 0.001 vs. the Control group). DA-CH5 and liraglutide reduced activated astrocyte numbers (###p < 0.001 vs. the MPTP group). The number of activated astrocytes in the DA-CH5 + MPTP group was less than in the liraglutide + MPTP group (&&&p < 0.001 vs. the liraglutide + MPTP group). The values represent the means±S.E.M. n = 6 per group. B) GFAP levels in the SN. Scale bar is 50 μm. C) The number of activated astrocytes was increased in the MPTP group, liraglutide + MPTP group and DA-CH5 + MPTP group (***p < 0.001 vs. the Control group). Individual cells are clearly visible in the micrographs. DA-CH5 and liraglutide reduced activated astrocyte numbers (###p < 0.001 vs. the MPTP group). The number in the DA-CH5 + MPTP group was lower than in the liraglutide + MPTP group (&p < 0.05 vs. the liraglutide + MPTP group). D) IHC staining of activated astrocyte marker GFAP in the striatum. Scale bar is 50 μm. E) The number of activated microglia was increased in the MPTP group, liraglutide + MPTP group and DA-CH5 + MPTP group (***p < 0.001 vs. the Control group). DA-CH5 and liraglutide reduced activated microglia numbers (###p < 0.001 vs. the MPTP group). F) IHC staining of activated microglia marker IBA-1 in the SN. Scale bar is 50 μm. G) The number of activated microglia was increased in the MPTP group, liraglutide + MPTP group and DA-CH5 + MPTP group (***p < 0.001 vs. the Control group). DA-CH5 and liraglutide restored activated microglia numbers (# #p < 0.01 and ###p < 0.001 vs. the MPTP group). The values represent the means±S.E.M. n = 6 per group. H) IHC staining of activated microglia marker IBA-1 in the striatum. Scale bar is 50 μm.
Anti-IBA1 was used as a biomarker for microglia activation. A one-way ANOVA found an overall difference in the SN (p < 0.001; Fig. 7E) and the striatum (p < 0.001; Fig. 7F) of mice. According to IBA1-positive microglia numbers, there was no difference between control and alone DA-CH5 injection group and alone of Liraglutide injection group both in the SN (p > 0.05) and striatum (p > 0.05). IBA-1 was rising to 184.0% in the SN (p < 0.001) and 185.6% in the striatum (p < 0.001) by MPTP procedure, which was significantly higher than control mice. Liraglutide treatment of MPTP-treated mice made IBA-1 positive microglia decrease to 133.1% in the SN (p < 0.001) and 164.3% in the striatum (p < 0.001), which was significantly less than MPTP group. While DA-CH5 treatment also ameliorated the microglia activation induced by MPTP, the IBA-1 positive level reduced to 121.1% in the SN (p < 0.001) and 151.8% in the striatum (p < 0.001) compared with the MPTP group. However, the MPTP + DA-CH5 group did not differ from the MPTP + Liraglutide group in both of the SN (p > 0.05; Fig. 7E, F) and striatum (p > 0.05; Fig. 7G, H). n = 6 per group.
DA-CH5 and Liraglutide reduced lipid peroxidation in the SN and striatum induced by MPTP
In the immunohistochemical analysis, 4-HNE was monitored as an indicator of lipid peroxidation. A one-way ANOVA found an overall difference of 4-HNE expression both in the SN (p < 0.001; Fig. 8A) and striatum (p < 0.001; Fig. 8B) of mice between groups. There was no difference between control, DA-CH5 only injection and Liraglutide only injection groups both in the SN (p > 0.05; Fig. 8A, B) and striatum (p > 0.05; Fig. 8C, D). 4-HNE positive cells were significantly increased by MPTP injection to 237.3% in the SN (p < 0.001) and 204.4% in the striatum (p < 0.001), respectively, compared with the control group. Although Liraglutide treatment decreased the 4-HNE levels induced by MPTP to 138.4% in the SN (p < 0.001) and 141.2% in the striatum (p < 0.001) compared with the MPTP group, the expression of 4-HNE in SN (p < 0.01) and striatum (p < 0.001) in the MPTP + Liraglutide group were still significantly higher than in the control group. DA-CH5 treatment also reduced the 4-HNE levels increased by MPTP to 124.1% in the SN (p < 0.001) and 130.4% in the striatum (p < 0.001) respectively compared with the MPTP group. Interestingly, the expression of 4-HNE in the SN of MPTP + DA-CH5 group mice was not significantly difference (p > 0.05) compared with control group mice. However, the expression of 4-HNE in the striatum of MPTP + DA-CH5 group mice was different compared with control group mice (p < 0.001). There was no significant difference between the MPTP + DA-CH5 group and the MPTP + Liraglutide group in both the SN (p > 0.05; Fig. 8A, B) and striatum (p > 0.05; Fig. 8C, D). n = 6 mice in each group.
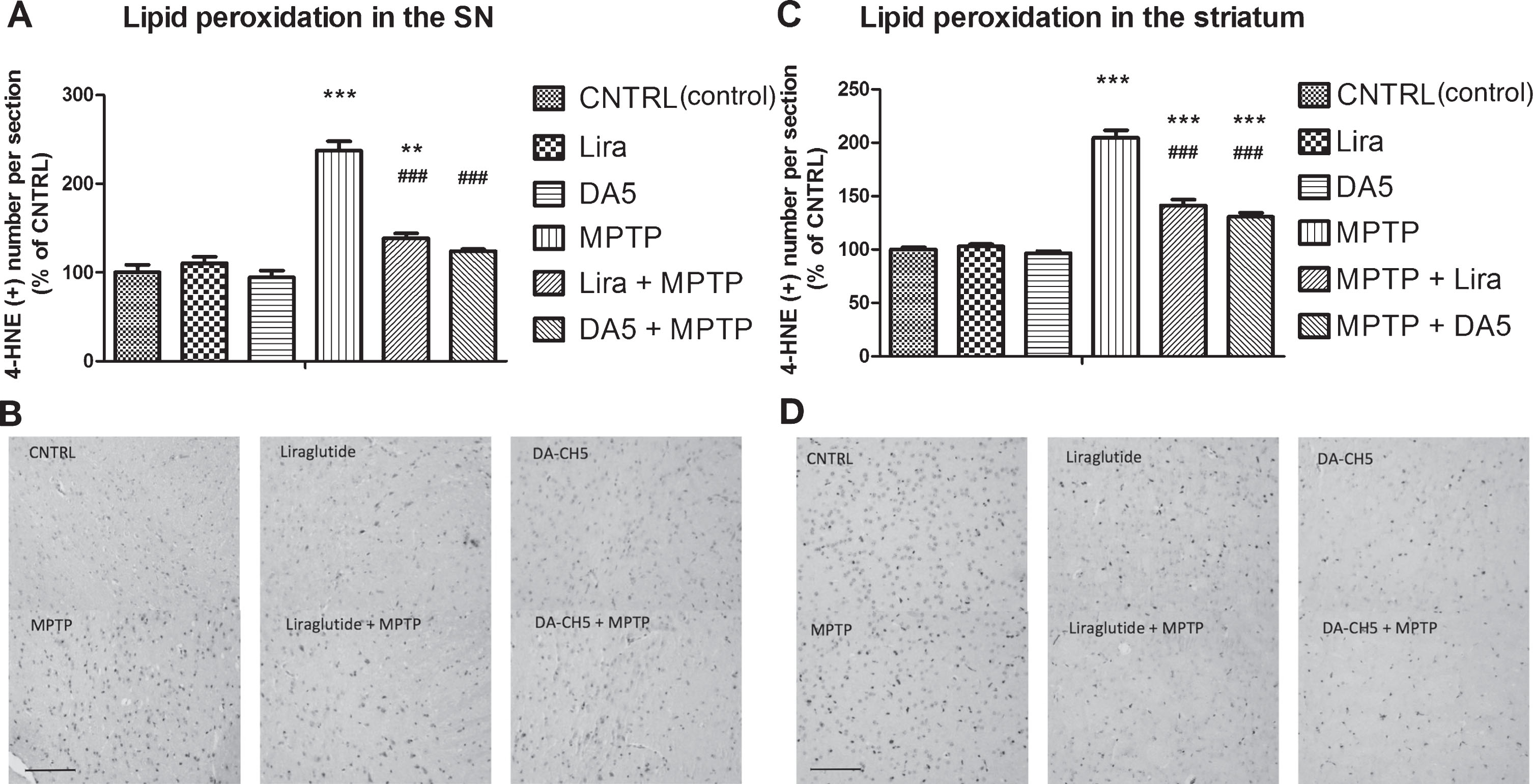
DA-CH5 and liraglutide reduced lipid peroxidation in the SN and Striatum of MPTP-induced PD mice. A) Levels of 4-HNE, a biomarker of lipid peroxidation, increased in the MPTP group and liraglutide + MPTP group (**p < 0.01 and ***p < 0.001 vs. the Control group). DA-CH5 and liraglutide lowered 4-HNE levels (###p < 0.001 vs. the MPTP group). The values represent the means±S.E.M. n = 6 per group. B) IHC staining of 4-HNE in the SN. Scale bar is 100 μm. Individual, stained cells are clearly visible in the micrographs. C) Levels in the striatum. 4-HNE increased in the MPTP group, liraglutide + MPTP group and DA-CH5 + MPTP group (***p < 0.001 vs. the Control group). DA-CH5 and liraglutide downregulated 4-HNE expression (###p < 0.001 vs. the MPTP group). The values represent the means±S.E.M. n = 6 per group. D) IHC staining of 4-HNE in the striatum. Scale bar is 100 μm. Individual, stained cells are clearly visible in the micrographs.
DA-CH5 and Liraglutide reduced apoptotic signalling in the SN and striatum induced by MPTP
To identify and quantify apoptotic cell death, TUNEL assay was used. This assay identifies DNA breaks in cells that undergo apoptosis. We found that TUNEL-positive cell numbers in the striatum of MPTP mice (p < 0.001; Fig. 9A) were significantly higher (136.88% of. control) in comparison with CNTRL animals, providing evidence that apoptotic cell death occurred in MPTP mice. The Liraglutide treatment (p < 0.001; Fig. 8A) and DA-CH5 treatment (p < 0.001; Fig. 9A) significantly reduced the presence of neurons with fragmented DNA to 111.24% and 107.20% respectively, suggesting decrease of apoptotic cellular death in treated animals. There was no significant difference between the MPTP + Liraglutide group and the MPTP + DA-CH5 group (p > 0.05; Fig. 9A). n = 6 mice in each group.
Bcl-2, as an anti-apoptotic member of the Bcl-2 family, can be bind to Bax to form Bcl-2:Bax heterodimers, thereby attenuating the pro-apoptotic effect of Bax. In the present study, we detected Bcl-2 and Bax protein by immunohistochemical analysis and western blotting respectively.
In the immunohistochemical analysis, a one-way ANOVA found an overall difference on Bcl-2 expression in the SN (p < 0.001; Fig. 9C) and the striatum (p < 0.001; Fig. 9E) between groups. There was not any difference between control group, Liraglutide only injection and DA-CH5 only injection groups, both in the SN (p > 0.05; Fig. 9C, D) and striatum (p > 0.05; Fig. 9E, F). The Bcl-2 expression was significantly reduced by MPTP injection to 58.7% in the SN (p < 0.001; Fig. 9C, D) and 59.9% in the striatum (p < 0.001; Fig. 9E, F) respectively, compared with the control group. Liraglutide treatment promoted the Bcl-2 expression to 101.2% in the SN (p < 0.001; Fig. 9C, D) and 89.6% in the striatum (p < 0.001; Fig. 9E, F) in MPTP-treated mice. The Bcl-2 positive cell numbers in the SN in the MPTP + Liraglutide group were no different from the control group (p > 0.05; Fig. 9C, D), but lower than the control group in the striatum (p < 0.01; Fig. 9E, F). DA-CH5 treatment promoted Bcl-2 expression to 109.9% in the SN (p < 0.001; Fig. 9C, D) and 94.5% in the striatum (p < 0.001; Fig. 9E, F) in MPTP-treated mice compared with MPTP group. The Bcl-2 positive cells in SN in MPTP + DA-CH5 group mice were no differences with the control group in the SN (p > 0.05; Fig. 9C, D) and in the striatum (p > 0.05; Fig. 9E, F). There was no significant difference between the MPTP + DA-CH5 group and the MPTP + Liraglutide group in both the SN (p > 0.05; Fig. 9C, D) and striatum (p > 0.05; Fig. 9E, F). n = 6 mice in each group.
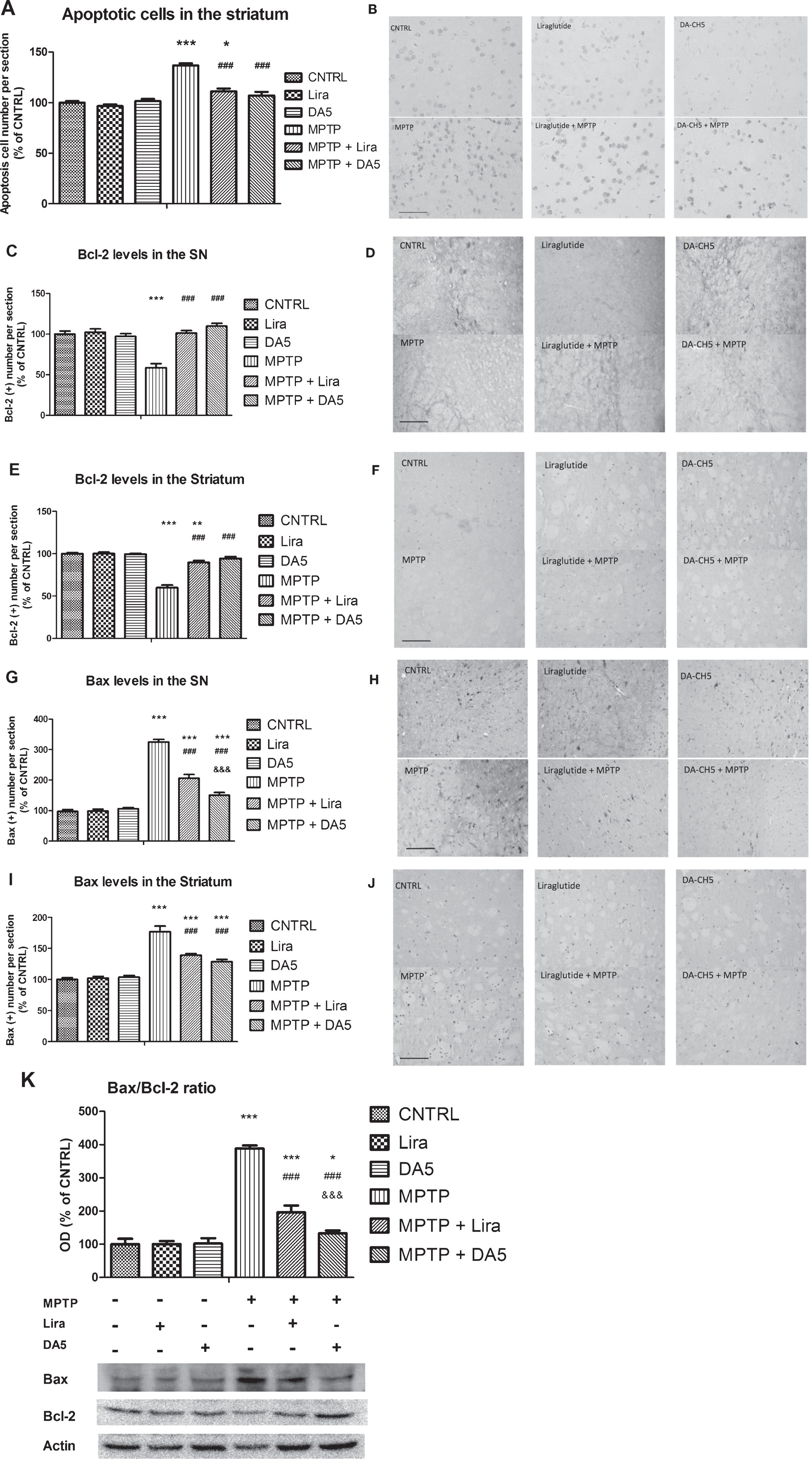
DA-CH5 and liraglutide reduces neuronal apoptosis in the SN and striatum MPTP-induced PD mice. A) The number of Tunel positive nuclei in MPTP group was higher than CNTR group (***p < 0.001 vs. the Control group). Liraglutide treatment and DA-CH5 treatment significantly reduced the presence of apoptotic neuron in striatum (###p < 0.001 vs. the MPTP group). The values represent the means±S.E.M. n = 6 per group. B) Apoptotic neuron numbers in the striatum. Scale bar is 100 μm. Individually stained cells are clearly visible in the micrographs. C) Bcl-2 positive cell numbers in the SN. Bcl-2 decreased in the MPTP group (***p < 0.001 vs. the Control group). DA-CH5 and liraglutide normalized Bcl-2 expression (###p < 0.001 vs. the MPTP group). The values represent the means±S.E.M. n = 6 per group. D) Bcl-2 positive cell numbers in the SN. Scale bar is 100 μm. Stained cells are visible as grey dots. E) Bcl-2 positive cell numbers decreased in the MPTP group and liraglutide + MPTP group (**p < 0.01 and ***p < 0.001 vs. the Control group). DA-CH5 and liraglutide normalised Bcl-2 expression (###p < 0.001 vs. the MPTP group). The values represent the means±S.E.M. n = 6 per group. F) Numbers of Bcl-2 positive cells in the striatum. Scale bar is 100 μm. Stained cells are visible as grey dots. G) Bax increased in the MPTP group, liraglutide + MPTP group and DA-CH5 + MPTP group (***p < 0.001 vs. the Control group). DA-CH5 and liraglutide improved Bax expression (###p < 0.001 vs. the MPTP group). Bax expression in the DA-CH5 + MPTP group was lower than in the liraglutide + MPTP group (&&&p < 0.001 vs. the liraglutide + MPTP group). The values represent the means±S.E.M. n = 6 per group. H) Bax positive cell numbers in the SN. Scale bar is 100 μm. Stained cells are visible as grey dots. I) Bax positive cell numbers increased in the MPTP group, liraglutide + MPTP group and DA-CH5 + MPTP group (***p < 0.001 vs. the Control group). DA-CH5 and liraglutide improved Bax expression (###p < 0.001 vs. the MPTP group). The values represent the means±S.E.M. n = 6 per group. J) Bax positive cell numbers in the striatum. Scale bar is 100 μm. Stained cells are visible as grey dots. K) Western blot analysis of Bcl-2 and Bax expression in the SN. The Bax/Bcl-2 ratio was increased in the MPTP group, Liraglutide + MPTP group and DA-CH5 + MPTP group (*p < 0.05 and ***p < 0.001 vs. the Control group). DA-CH5 and liraglutide improved the Bcl-2/Bax ratio (###p < 0.001 vs. the MPTP group). Bax/Bcl-2 ratio in the DA-CH5 + MPTP group was lower than in the liraglutide + MPTP group (&&&p < 0.001 vs. the liraglutide + MPTP group). The values represent the means±S.E.M. n = 4 mice for each group.
A one-way ANOVA found an overall difference on Bax expression in the SN (p < 0.001; Fig. 9G, H) and the striatum (p < 0.001; Fig. 9I, J) of mice between groups. There was not any difference between the control, DA-CH5 only injection and Liraglutide only injection groups both in the SN (p > 0.05; Fig. 9G, H) and striatum (p > 0.05; Fig. 9I, J). The Bax expression was significantly increased by MPTP injection to 325.0% in the SN (p < 0.001) and 176.8% in the striatum (p < 0.001) respectively, compared with the control group. Liraglutide treatment reduced the Bax expression to 206.3% in the SN (p < 0.001) and 139.3% in the striatum (p < 0.001) in MPTP-treated mice. Bax positive cell numbers in SN in MPTP + Liraglutide group mice were significantly different compared with the control group both in the SN (p < 0.001) and in the striatum (p < 0.001; p < 0.001). DA-CH5 treatment reduced Bax expression to 150.0% in the SN (p < 0.001) and 128.5% in the striatum (p < 0.001) in MPTP-treated mice. Bax positive cell numbers in the MPTP + Liraglutide group mice were significantly different compared with the control group both in the SN (p < 0.001) and in the striatum (p < 0.001). The MPTP + DA-CH5 group significantly differed from The MPTP + Liraglutide group in the SN (p < 0.001; Fig. 9G, H), but there was not any difference in the striatum (p > 0.05; Fig. 9I, J). n = 6 mice in each group.
The Bax/Bcl-2 ratio provides an indication of the activation of apoptosis signaling. A one-way ANOVA found an overall difference in Bax/Bcl-2 ratio in the SN (p < 0.001; Fig. 9K). There was no difference between the control, DA-CH5 injection and Liraglutide injection groups (p > 0.05; Fig. 9K). MPTP-induced toxicity resulted in a dramatic up-regulation of the Bax/Bcl-2 ratio to 388.5% compared with the control group (p < 0.001). The DA-CH5 (p < 0.001) and Liraglutide group (p < 0.001) reversed the increased of Bcl-2/Bax ratio caused by MPTP to 133.1% and 196.4% respectively compared with MPTP group. The Bax/Bcl-2 ratio in the MPTP + Liraglutide group mice was significantly different from the control group (p < 0.001), while there was moderately up-regulated in the MPTP + DA-CH5 group (p < 0.05; Fig. 9K). The Bax/Bcl-2 ratio in the MPTP + DA-CH5 group was significantly differed from the MPTP + Liraglutide group in the SN (p < 0.001). n = 4 mice in each group.
DA-CH5 and Liraglutide increased autophagy-related proteins expression in the SN induced by MPTP
Beclin 1, microtubule associated protein 1 light-chain 3 (LC3) and p62, are critical regulators of autophagy [37].
A one-way ANOVA found an overall difference in Beclin 1 expression in the SN (p < 0.001; Fig. 10B). There was no difference between control, Liraglutide only injection, DA-CH5 only injection, MPTP + Liraglutide, and MPTP + DA-CH5 groups (p > 0.05). MPTP-induced toxicity resulted in a down-regulation of Beclin 1 to 67.3% compared with the control group (p < 0.01). However, Liraglutide (p < 0.01) and DA-CH5 (p < 0.001) normalised the low expression of Beclin 1 caused by MPTP to 90.6% and 114.8% respectively compared with the MPTP group. Beclin 1 expression in MPTP + DA-CH5 group significantly differed from the MPTP + Liraglutide group (p < 0.05). See Fig. 10A and B). n = 4 mice in each group.
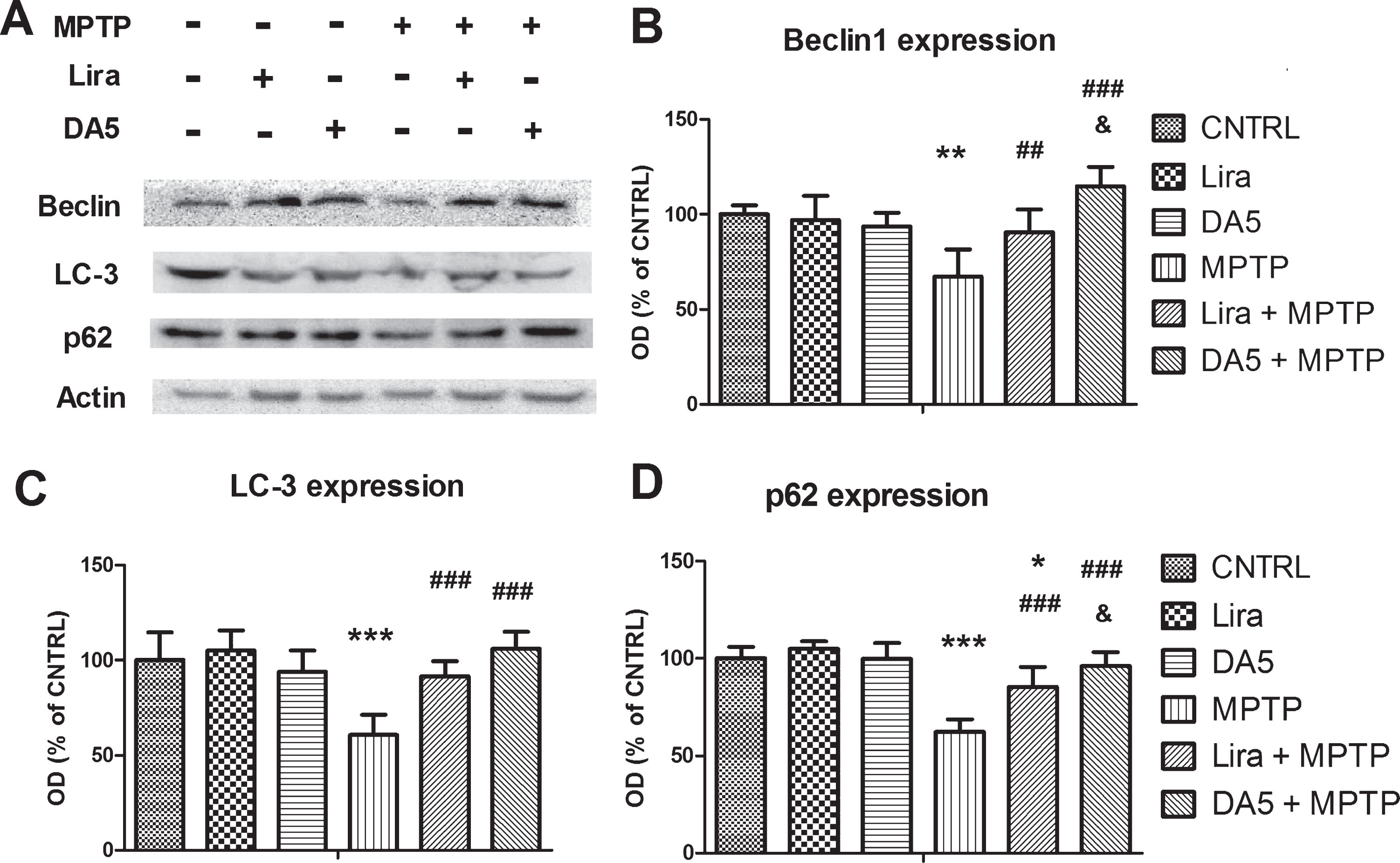
DA-CH5 and Liraglutide normalized the expression of autophagy related proteins in the SN. A) Western blots of Beclin-1, LC3 and p62 expression. B) Beclin-1 expression was downregulated in the MPTP group (**p < 0.01 vs. the Control group). DA-CH5 and Liraglutide normalized Beclin-1 expression (# #p < 0.01 and ###p < 0.001 vs. the MPTP group). Beclin-1expression in the DA-CH5 + MPTP group was higher than in the Liraglutide + MPTP group (&p < 0.05 vs. the Liraglutide + MPTP group). The values represent the means±S.E.M. n = 4 mice for each group. C) Levels of LC3 in the MPTP group (***p < 0.01 vs. the Control group). DA-CH5 and Liraglutide normalized LC3 expression (###p < 0.001 vs. the MPTP group). N = 4 mice for each group. D) The p62 expression was downregulated in the MPTP group and Liraglutide + MPTP group (***p < 0.001 and *p < 0.05 vs. the Control group). DA-CH5 and Liraglutide normalized p62 expression (###p < 0.001 vs. the MPTP group). P62 expression in the DA-CH5 + MPTP group was higher than in the Liraglutide + MPTP group (&p < 0.05 vs. the Liraglutide + MPTP group). The values represent the means±S.E.M. n = 4 mice for each group.
A one-way ANOVA found an overall difference in LC3 expression in the SN (p < 0.001; Fig. 10C). There was not any difference between control, Liraglutide only injection, DA-CH5 only injection group, MPTP + Liraglutide and MPTP + DA-CH5 groups (p > 0.05; Fig. 10A, C). MPTP-induced toxicity resulted in a dramatic down-regulation of LC3 expression to 60.7% compared with the control group (p < 0.001; Fig. 10A, C). However, Liraglutide (p < 0.001) and DA-CH5 (p < 0.001) normalised the low LC3 expression caused by MPTP to 91.6% and 106.1% respectively, compared with the MPTP group. But there was not any difference between MPTP + Liraglutide group and MPTP + DA-CH5 group (p > 0.05). n = 4 mice in each group.
A one-way ANOVA found an overall difference in p62 expression in the SN (p < 0.001; Fig. 10D). There was not any difference between control, Liraglutide only injection, DA-CH5 only injection group, and MPTP + DA-CH5 groups (p > 0.05; Fig. 10A, D). MPTP-induced toxicity resulted in a dramatic down-regulation of p62 expression to 62.3% compared with the control group (p < 0.001). p62 expression of MPTP + Liraglutide was down-regulated to 85.3% compared with control group (p < 0.05), but much higher than in the MPTP group (p < 0.001). Also, DA-CH5 (p < 0.001; Fig. 10A, D) normalised the low LC3 expression caused by MPTP to 96.1% compared with MPTP group. The LC3 expression in MPTP + DA-CH5 group significantly differed from MPTP + Liraglutide group (p < 0.05; Fig. 10A, D). n = 4 mice in each group.
DISCUSSION
PD is a complex neurodegenerative disorder in which the etiology remains largely unknown. Several studies have reported that the progressive deterioration of vulnerable SN dopaminergic neurons may be caused by misfolding and aggregation of the synaptic protein alpha synuclein, disruption of the autophagy system, and mitochondrial dysfunction [38]. The MPTP mouse model is widely used for mimicking nigrostriatal damage in PD [39]. After MPTP transit to the brain through the Blood Brain Barrier, it is converted to the toxic metabolite 1-methyl-4-phenylpyridinium (MPP+) in glia cells by monoamine oxidase-B (MAO-B) [40, 41]. When MPP+ is released from the astrocytes into the extracellular space, it is taken up by dopaminergic neurons via dopamine transporters where it shows toxic effects [42]. Once MPP+ accumulates in dopaminergic neurons, it induces neurotoxicity primarily by inhibiting complex I of the mitochondrial electron transport chain [43]. As a consequence, energy utilisation, mitochondrial function, and synaptic transmission is impaired, as well as gene expression and dopamine synthesis. A further consequence is the induction of a chronic inflammation response in the brain [44–47]. Our previous studies demonstrated that GLP-1 or GIP analogues could provide neuroprotective effects by normalizing apoptosis and autophagy pathways, inhibit the chronic inflammation response and the resulting oxidative stress in the brain, improving dopamine levels and the expression of the tyrosine hydroxylase as well as key growth factors such as BDNF and GDNF [18, 49].
Our results show that DA-CH5 is a selective and well-balanced agonist at both the GLP-1R and the GIPR, while showing little activity at the GLP-2R and the GcgR. This result is a conformation of the study that tested dual agonist peptides with the same receptor binding sites [28]. Furthermore, we observed that peptides with a C16 fatty acid attached or with a pegylation addition do not cross the BBB as easily as peptides without these functional groups. The TAT sequence modification of DA-CH5 enhanced penetration of the BBB significantly compared to the dual agonist DA-CH3 that does not have this modification. This sequence is well recognised as a ligand to cell membrane receptor binding sites that enhance cell uptake and enhances BBB penetration [50]. In the MPTP animal studies, DA-CH5 was more effective in protecting the brain than the older GLP-1 receptor agonist exendin-4 in a direct comparison in a dose-response assessment. In addition, we found that DA-CH5 and liraglutide effectively normalized locomotor moving distance, increased the duration time on rotating-stick and improved motor coordination of MPTP-treated mice. These results confirm previous work [12, 32] and suggested that both DA-CH5 and liraglutide might be potential agents for PD due to their improvement of motor function. In our previous study, we tested liraglutide and DA-CH5 at equal doses (25 nmol/kg once daily) to compare equal doses. However, as liraglutide is acetylated and associates with blood proteins much more than DA-CH5, its bio-availability to activate GLP-1 receptor is lower [33]. In clinical practice, liraglutide has to be dosed at 0.6–1.8 mg per person per day [51], while liisenatide (Lyxumia), which is not acetylated is dosed at 20 μg daily [52]. We therefore dosed DA-CH5 at 10 nmol/kg ip. once-daily to compare the neurophysiological effects of both drugs at more equal conditions.
Tyrosine Hydroxylase (TH) is the initial and rate-limiting enzyme in the synthesis of the catecholamine neurotransmitters [53] and is the principal regulator of dopamine synthesis in the CNS [54]. Therefore, in order to investigate whether DA-CH5 and liraglutide could directly protect against MPTP-induced nigrostriatal degeneration, immunohistochemistry (IHC) analysis for TH protein was conducted. Our results showed that both drugs could increase the number of TH positive cells in the SN and TH levels in the axons projecting to the striatum. Neuroinflammatory processes mediated by astrocytes and microglia cells are extensively involved in the progression of PD [27, 55]. The damaged dopaminergic neurons and activated microglial cells can stimulate astrocytes into immune-active status [56]. Activated microglia release pro-inflammatory cytokines which in turn impair neuronal function and inhibit growth factor signalling and energy utilisation [57, 58]. In addition, astrocyte activation is associated with the impairment of the nigrostriatal system of MPTP mouse models [39, 42]. Our results demonstrate that both peptides can inhibit the inflammatory response. Importantly, DA-CH5 showed superiority in inhibiting astrocyte activation compared with Liraglutide. These results confirm our previous findings that showed that GLP-1 or dual agonists can reduce the inflammation response in the brain [15, 60].
MPTP administration can inhibit mitochondrial complex I activity leading to the excessive generation of reactive oxygen species (ROS) which enter the cytosol causing oxidative damages [61]. This can enhance the levels of lipid peroxidation (LPO) products [62]. LPO causes cell membrane damage that leads to the production of conjugated dienic hydroperoxides that decompose into various aldehydes, such as 4-hydroxynonenal (4-HNE) [63]. In addition, 4-HNE accumulation has been observed in neurodegenerative diseases, including Alzheimer’s disease, PD and amyotrophic lateral sclerosis [64]. Importantly, 4-HNE activates the oxidative damage sensor and apoptosis trigger BAD and can initiate apoptosis of mitochondria [21, 65]. Our results demonstrate that MPTP treatment increased 4-HNE levels while DA-CH5 and Liraglutide treatment significantly reduced 4-HNE levels in MPTP-treated mice.
Mitochondrial impairment is always connected to apoptosis as inhibition of complex I will affect proton pumps and enhance mitochondrial membrane permeability for cytochrome c, which in turn triggers activation of caspase families and finally induces apoptosis [66].
One important feature of the apoptosis pathway is its regulation by the levels of signalling peptides. Bcl-2, which is anti-apoptotic, delays programmed cell death and is shown as an anti-oxidant, especially by preventing LPO [67]. Bax, a pro-apoptotic signalling peptide, is activated during oxidative stress and then oligomerizes and translocates to mitochondria from the cytosol. This in turn triggers cytochrome c release from mitochondria to the cytosol [68]. In the present study, we found that both DA-CH5 treatment and Liraglutide could inhibit the apoptosis process. MPTP treatment decreased Bcl-2 and increased the total Bax protein levels. In contrast, DA-CH5 and Liraglutide treatment significantly increased the total Bcl-2 expression and significantly reduced the Bax protein levels in MPTP-treated mice. Furthermore, the number of cells that undergo apoptosis and DNA fragmentation as shown in the TUNEL assay was much reduced by the drug.
Accumulation of misfolded proteins is one of the important features of neurodegenerative diseases. That is why autophagy, which is a major intracellular degradation and recycling system of the eukaryotic cell, plays a role in the development of PD. Autophagy can clear cellular components including misfolded macromolecules and damaged organelles using double-membraned autophagosomes and deliver them to lysosomes for degradation. LC3, the key regulator of autophagy, initiates autophagosome biogenesis after becoming membrane-conjugated through interactions with Beclin 1, which is important for localisation of autophagic proteins to a pre-autophagosomal structure [69] and other autophagic effectors [70]. P62, which plays a critical role in recognition and selective autophagy, can bind to LC3 family and transport ubiquitinated misfolded protein into autophagosomes [71]. Recent research has reported that impairment of autophagy is considered particularly important in the development of PD [72]. In our study, we find that both Liraglutide and DA-CH5 upregulated Beclin 1, LC3 and p62 expression, which indicated that these two drugs can improve autophagy in dopaminergic neurons. However, cell culture studies are required to actually monitor the autophagy flux, which is not possible in static histology images. In addition, we find DA-CH5 is more effective than Liraglutide. These results confirm our previous results testing the effects of liraglutide in rotenone-stressed SH-SY5Y cells [37]. Our results also support our previous findings, comparing the dual agonists DA1-JC, DA4-JC and DA-CH5 in the MPTP mouse model of PD. In that study, we showed that DA-CH5 was the most effective drug in protecting mice from MPTP-induced impairments in motor activity, TH expression, GDNF expression, and the expression of the synapse marker synaptophysin [32]. We repeated the direct comparison with a lower dose of DA-CH5 due to the lower bio-availability of liraglutide and the lower BBB penetration and extend the range of protective effects in the present study by showing that mitochondrial apoptosis markers and apoptotic cell numbers are reduced, survival factor expression is enhanced, lipid peroxidation and astrocyte activation is reduced, and the expression of key biomarkers of autophagy is normalised by DA-CH5. These findings add important information to our knowledge of the molecular processes that underlie the neuroprotective effects of this dual agonist. Activating the second messenger growth factor cascade by activating the GLP-1 and GIP receptors counteracted apoptotic signalling and the inflammation response which reduced oxidative stress and thereby reduced apoptotic signalling by reducing lipid oxidation and 4-HNE formation. Importantly, we also showed that a dual GLP-1/GIP receptor agonist is more effective than a single GLP-1 receptor agonist. The addition of the GIP receptor binding appears to enhance the protective properties. A recent clinical trial comparing a dual GLP-1/GIP receptor agonist with a single GLP-1 receptor agonist in patients with type II diabetes also demonstrated superiority to the dual receptor agonist [73].
In summary, our study demonstrates that DA-CH5 and liraglutide achieve neuroprotective effects by normalizing impaired motor activities, inhibiting the reduction of TH positive cells in the SN, decreasing inflammation, oxidative stress, apoptosis and increasing autophagy to prevent the loss of dopaminergic transmission. DA-CH5 was more effective than liraglutide even when given at a lower dose. Liraglutide is currently being tested in a phase II trial in PD patients (clinical trial identifier NCT02953665). On the basis of our findings, DA-CH5 might be more effective than liraglutide in treating PD.
CONFLICT OF INTEREST
CH is a named inventor on a patent application by Lancaster University which covers the use of dual GLP-1/GIP receptor agonists as a treatment for PD. The other authors declare no conflict of interest.
Footnotes
ACKNOWLEDGMENTS
The work was supported by a fellowship to CH of the funding programme of the State Administration of Foreign Experts Affairs in China, the Shanxi ‘1331 Project’ Key Subjects Construction fund.
