Abstract
Parkinson’s disease (PD) is a progressive neurological motor control disorder. A key feature is the loss of midbrain dopaminergic neurons and the accumulation of aggregated alpha-synuclein (α-syn). No current treatment is on the market that slows or halts disease progression. Previous studies have shown that glucagon-like peptide-1 (GLP-1) receptor agonists have neuroprotective effects in animal models of PD. In addition, in a phase II clinical trial, the GLP-1 receptor agonist exendin-4 has shown good protective effects in PD patients. In the present study, we have investigated the neuroprotective effects of the GLP-1 analogues semaglutide (25 nmol/kg ip. once every two days for 30 days) and liraglutide (25 nmol/kg ip. once daily for 30 days) in the chronic MPTP mouse model of PD. Both drugs are currently on the market as a treatment for Type II diabetes. Our results show that both semaglutide and liraglutide improved MPTP-induced motor impairments. In addition, both drugs rescued the decrease of tyrosine hydroxylase (TH) levels, reduced the accumulation of α-syn, alleviated the chronic inflammation response in the brain, reduced lipid peroxidation, and inhibited the mitochondrial mitophagy signaling pathway, and furthermore increased expression of the key growth factor GDNF that protects dopaminergic neurons in the substantia nigra (SN) and striatum. Moreover, the long- acting GLP-1 analogue semaglutide was more potent compared with once daily liraglutide in most parameters measured in this study. Our results demonstrate that semaglutide may be a promising treatment for PD. A clinical trial testing semaglutide in PD patients will start shortly.
INTRODUCTION
Parkinson’s disease (PD) is the second most common degenerative disease characterized by progressive loss of dopaminergic neurons in the substantia nigra pars compacta, motor impairments, and deposition of intraneuronal inclusions known as Lewy bodies [1]. The main symptoms are resting tremors, muscular rigidity, bradykinesia, and postural and gait abnormalities [2]. Recently, studies have shown a link between PD and type 2 diabetes (T2DM), another common chronic neurodegenerative disorder characterized by progressive hyperglycemia, pancreatic β-cell dysfunction and insulin resistance (IR) in peripheral tissues [3]. Both PD and T2DM are age-related chronic diseases, and these also share several genetic susceptibilities, such as single nucleotide polymorphisms in the growth factor signaling kinase gene Akt, which can increase individual’s risk for developing PD and diabetes [4]. Insulin signaling was found to be impaired in the brains of PD patients, impairing energy utilization and cell repair [5–7]. Insulin is a key growth factor that protects neurons [8, 9].
Drugs that had been initially developed to treat type II diabetes have been re-purposed as treatments for PD [10, 11]. These drugs are long-lasting, protease resistant mimetics of the hormone and growth factor glucagon-like peptide –1 (GLP-1) [12–14]. GLP-1 is neuroprotective and can re-sensitize insulin signaling [15–17]. The GLP-1 mimetic exendin-4 (exenatide, Bydureon), which is on the market to treat T2DM, showed a therapeutic effect in different animal models of PD [18–21]. Exendin-4 was protective in a pilot clinical trial in PD patients (NCT01174810) [22, 23]. Importantly, a phase II clinical trial showed protective effects in PD patients even 3 months after treatment had been discontinued [24]. Liraglutide is a GLP-1 analogue [25, 26] that has an extended survival time in the blood stream and has a half-life of approximately 12 hours in humans [27, 28]. Therefore, it requires a once-daily dosing for treating diabetes [29]. Liraglutide also showed neuroprotective effects in animal models of PD and Alzheimer’s disease [30, 31]. Liraglutide is currently being tested in a phase II trial in PD patients (clinical trial identifier NCT02953665).
Semaglutide is a modification of liraglutide that is protease-resistant by changing the amino acid at position 8 and an extended spacer for the attached fatty acid [32], and is on the market as a new once- weekly drug to treat type II diabetes. It has been approved in the USA and Europe as a treatment for diabetes [33, 34]. A phase II clinical trial testing semaglutide in PD patients will start early 2019 (NCT03659682). Previously, we have investigated the neuroprotective effects of the once-weekly GLP-1 analogue semaglutide and compare it with liraglutide in the acute MPTP mouse model of PD [35]. However, the acute model is not considered to be a good representation of the pathology observed in PD, as the disease develops slowly over time. Therefore, we investigated these neuroprotective effective in the chronic MPTP treatment PD mouse model. Additionally, we analyzed the effects of the drugs on α-synuclein expression and on the impairments of autophagy that has been observed in the MPTP mouse model. If autophagy is impaired in the disease, proteins such as α-synuclein can accumulate and aggregate in the cell. As semaglutide will be tested in PD patients in a clinical trial, it is of vital importance to investigate the underlying molecular mechanisms of its actions.
MATERIALS AND METHODS
Chemicals and peptides
Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) was purchased from Sigma-Aldrich (St Louis, MO, USA). Other chemicals used were of the highest quality commercially available. Semaglutide and liraglutide (peptide purity: 95.77%) was purchased from Synpeptide Co. (Shanghai, China). The quality was tested using HPLC and MALDI-TOF analysis.
The amino acid sequence of liraglutide:
HAEGTFTSDVSSYLEGQAAK[(γE)-(Pal)]EFI AWLVRGRG-OH Pal = palmitoyl acid
The amino acid sequence of semaglutide [32]: HXEGTFTSDVSSYLEGQAAKN6-(N-(17-carboxy-1-oxoheptadecyl)-L-gamma-glutamyl-2-(2 -(2-aminoethoxy)ethoxy)acetyl-2-(2-(2-aminoethox y)ethoxy)acetyl)EFIAWLVRGRG-OH X = aminoisobutyric acid.
Animals and drug treatments
Male C57BL/6 mice 8 weeks old (20–25 g) were purchased from the Experimental Animal Center, Shanxi Medical University. The animals were maintained on 12-hour light/dark cycle and provided food and water ad libitum. Mice were randomly divided into six groups (N = 12 animals per group). A: control group treated with saline alone; B: liraglutide group treated with liraglutide (25 nmol/kg ip. once daily for 30 days); C: semaglutide group treated with semaglutide (25 nmol/kg ip. once every two days for 30 days), D: MPTP group treated with MPTP alone (once daily 20 mg/kg ip. for 30 days); E: MPTP (once daily 20 mg/kg ip. for 30 days) + liraglutide treated group (25 nmol/kg ip. once daily for 30 days). F: MPTP (20 mg/kg ip. once daily for 30 days) + semaglutide- treated group (25 nmol/kg ip. once every two days for 30 days). At the end of drug treatments, behavioral changes, neuronal damage, inflammatory markers, and other biomarkers were assessed.
All animal experiments were approved by the ethics committee of Shanxi Medical University.
Behavioral assessment
Open-field test
The open-field test is used to evaluate locomotor and exploratory activity of PD mice and was conducted on the 31th day after MPTP treatment. The open-field apparatus consisted of a circular arena (35 cm diameter floor and 40 cm high walls), and a computer tracking system (Etho Vision XT software, Noldus information technology, Wageningen, Netherlands). Each mouse was placed in the center of the apparatus, and tracking started immediately. After acclimatizing for 10 min, the distance travelled by the animal was recorded by the tracking system. The area was wiped with 75% alcohol and dried between each trial.
Rotarod performance
The rotarod test is to measure motor coordination in the mouse model of PD. The rotarod equipment (YLS-4C, Academy of medical sciences in Shandong, China) consisted of a rotating spindle and five individual compartments. All mice were pre-trained for 3 days prior to drug administration. The test consisted of three consecutive runs with a gradual increase in rpm up to a maximum 30 rpm for up to 180 seconds. The length of time was recorded as the latency period to fall. The experiment was repeated three times for each animal at 10 min rest intervals.
Footprint gait test
The footprint test was described previously [35]. Briefly, the animal forelimbs were dipped in blue ink and the hind limbs in red ink to record footprints as they walked through a dark tunnel (10×10×50 cm). The footprints were recorded on a clean sheet of white paper placed on the floor of the tunnel. The two initial steps were excluded from the measurements, and only steps performed in a straight line were recorded. To avoid differences in the stride length as a result of velocity variations, the footprints were only recorded when the mice walked along the tunnel with a regular velocity, and excluding the mice that performed the test with perceptible velocity alterations. Stride lengths were determined by measuring the distance between each step on the same side of the body. The length of the shortest stride was subtracted from the length of the longest stride to determine the stride variability.
Grip strength test
Grip strength was measured by the digital grip strength meter 47200 (Ugo Basile, Italy). Grip strength testing is commonly used as an objective measure of muscle strength in the front legs. All mice were pre-trained for 3 days prior to test. Each mouse was placed in the platform apparatus, and to grasp a lever that can transmit the force value by its forelimbs, then they were pulled at the tail until release of grip to measure the muscle strength of their forelimbs. Each group was tested 3 times and measured in Newtons (N). If the third value was highest, the subject was tested until the value stopped increasing. The maximum muscle strength of each mouse was taken for statistical analysis.
Brain tissue preparation
All animals were killed on the 31th day of MPTP injection. After ethyl carbamate anesthetization, the brains of 6 mice per group were selected and the substantia nigra and striatum were dissected and immediately frozen at –80°C for immunoblot analysis. Another 6 mice per group were intracardially perfused with 20 ml saline and then fixed with 20 ml of cold 4% paraformaldehyde (PFA). Brains were immediately removed and post-fixed in 4% PFA 24 hours for immunohistochemistry analysis.
Immunohistochemistry
The fixed brain tissue samples were embedded in paraffin, and sections were cut at 4μm with a semiautomatic microtome (Leica, Wetzlar, Germany). The sections encompassing the substantia nigra pars compacta (SNpc) and the striatum were placed on glass slides, then the paraffin was removed from the tissue sections with xylene, and the sections were rehydrated in descending concentrations of ethanol solutions. Then the sections were put into H2O2 (3%) for 10 min to block the activity of endogenous peroxidase. Antigen retrieval was performed by heating in 10 mmol/L citrate buffer for 10 min. After blocking with 5% BSA, sections were incubated with the primary antibody for tyrosine hydroxylase (TH) (rabbit anti-TH; 1:200; cat. No. ab75875, Abcam, Cambridge, UK), GFAP (rabbit anti-GFAP; 1:200; cat. No. PB0046, Boster Biotechnology Co., Ltd. Wuhan, China) and IBA1 (goat anti-IBA1; 1:200; cat. No. PB0517, Boster Biotechnology Co., Ltd. Wuhan, China), 4-HNE (rabbit anti-4-HNE; 1:400; cat. No. ab46545, Biosynthesis Biotechnology Co., Ltd. Beijing, China) at 37°C for 1 hour. Then they were rinsed in PBS and incubated a secondary peroxidase-conjugated antibody kit (Boster, Wuhan, China) at 37°C for 0.5 h. Stained sections were viewed under a Zeiss light microscope, and images were captured by a digital camera (Motic BA410; Motic, Xiamen, China). Quantitative analysis of DA neurons in SNpc was carried out in the region spanning from –2.92 mm to –3.40 mm relative to bregma. The region corresponding to the SNpc was clearly delineated, according to the mouse brain atlas of Paxinos and Franklin [36]. The magnification was kept the same for all measurements. Each mouse had one section analyzed with n = 6 per group. Numbers of GFAP, IBA1, 4-HNE positive cells in SNpc were determined using Image-pro plus 6.0 software. All data were expressed as a percentage of control values. Abbreviations: 4-Hydroxynonenal (4-HNE); Glial fibrillary Acid Protein (GFAP); ionized calcium-binding adapter molecule 1 (IBA-1); B-cell lymphoma 2 (Bcl-2); Bcl-2 associated X protein (BAX); dopamine (DA).
Western blots
Brain tissue with substantia nigra was stored at –80°C for western blot analysis. The tissue was weighed and cut into pieces in cold radio immune precipitation (RIPA) buffer (Beyotime Institute of Biotechnology, Shanghai, China). Two hours later, tissue lysates were added to phenylmethanesulfonyl fluoride (PMSF) and put on ice for 30 min. Tissue lysates were obtained by centrifugation at 12,000 rpm for 20 min at 4°C. Protein concentration was measured by the bicinchoninic acid protein assay (Boster Biotechnology Co., Ltd. Wuhan, China). Samples mixed with loading buffer to the same concentration were boiled for 5 min. Samples with equivalent amounts of protein were run on 8%, 10% or 12% SDS-polyacrylamide gel and transferred protein band onto polyvinylidene difluoride (PVDF) membranes. Then, the membranes were blocked with 5% bovine serum albumin for two hours. The membranes were probed overnight at 4°C with primary antibodies that specifically detect Bcl-2 (1:1000; #BS70205, Bioworld Technology, lnc. MN, USA), Bax (1:500; #BS2538, Bioworld Technology, lnc. MN, USA), and β-Actin (1:5000; #ab8227, Abcam, Cambridge, UK), α-Syn (1:1000; #2642, Cell Signaling Technology, lnc.), ATC7 (1:000; #BS6046, Bioworld Technology, lnc.MN, USA), LC3 (1:1000; #L7543, Sigma-Aldrich, lnc. USA)], Beclin 1 (1:000; #AP0768, Bioworld Technology, lnc. MN, USA), SQSTM1 (1:000; #AP6006, Bioworld Technology, lnc. MN, USA), or GDNF (1:500; #ab18956, Abcam, Cambridge, UK), followed by labeling with secondary antibodies (goat-anti-rabbit -IgG-horseradish peroxidase, HRP), 1:5000; (Abcam, Cambridge, UK) shake for 2 h. The relative immunoreactive bands were captured by a chemiluminescence imaging system (Sagecreation, Beijing, China), and visualized by using ECL-enhanced chemilluminescence (Boster Biotechnology Co., Ltd. Wuhan, China), and digitalized by the image analysis system of Quantity One 4.31 (Bio-Rad, Hercules, CA, USA). As an indicator for mitochondrial apoptosis, the ratio of BAX/Bcl-2 levels was computed and graphed. Abbreviations: autophagy chaperone mediator 7 (ATC7); sequestosome 1 (SQSTM1); 1A/1B-light chain 3 (LC3); LC3 binding protein 62 (p62).
Statistical analysis
All data were expressed as means±S.E.M. All analysis was conducted using GraphPad Prism (Graph-Pad software Inc., San Diego, CA, USA). Statistical significance was performed by one-way ANOVA for multiple comparisons followed by Tukey’s Multiple Comparison Test. Statistical significance was set at p < 0.05.
RESULTS
Semaglutide and liraglutide normalized motor impairments induced by MPTP
In the open field test, semaglutide alleviated the locomotor impairments induced by MPTP. A one-way ANOVA found an overall difference of distance travelled (F = 13.19, p < 0.0001) between all groups. There was no difference between control group and ns + liraglutide group and ns + semaglutide group. However a difference was found between the control groups and MPTP group (p < 0.001). Furthermore, a difference was found between MPTP + liraglutide and MPTP group (p < 0.01), and MPTP + semaglutide and the MPTP group (p < 0.001). This shows that liraglutide and semaglutide were able to normalize the MPTP-induced impairments in locomotor and exploratory activity of mice. There was no significant difference between MPTP + semaglutide group and MPTP + liraglutide groups (p > 0.05). N = 12 per group, see (Fig. 1a).
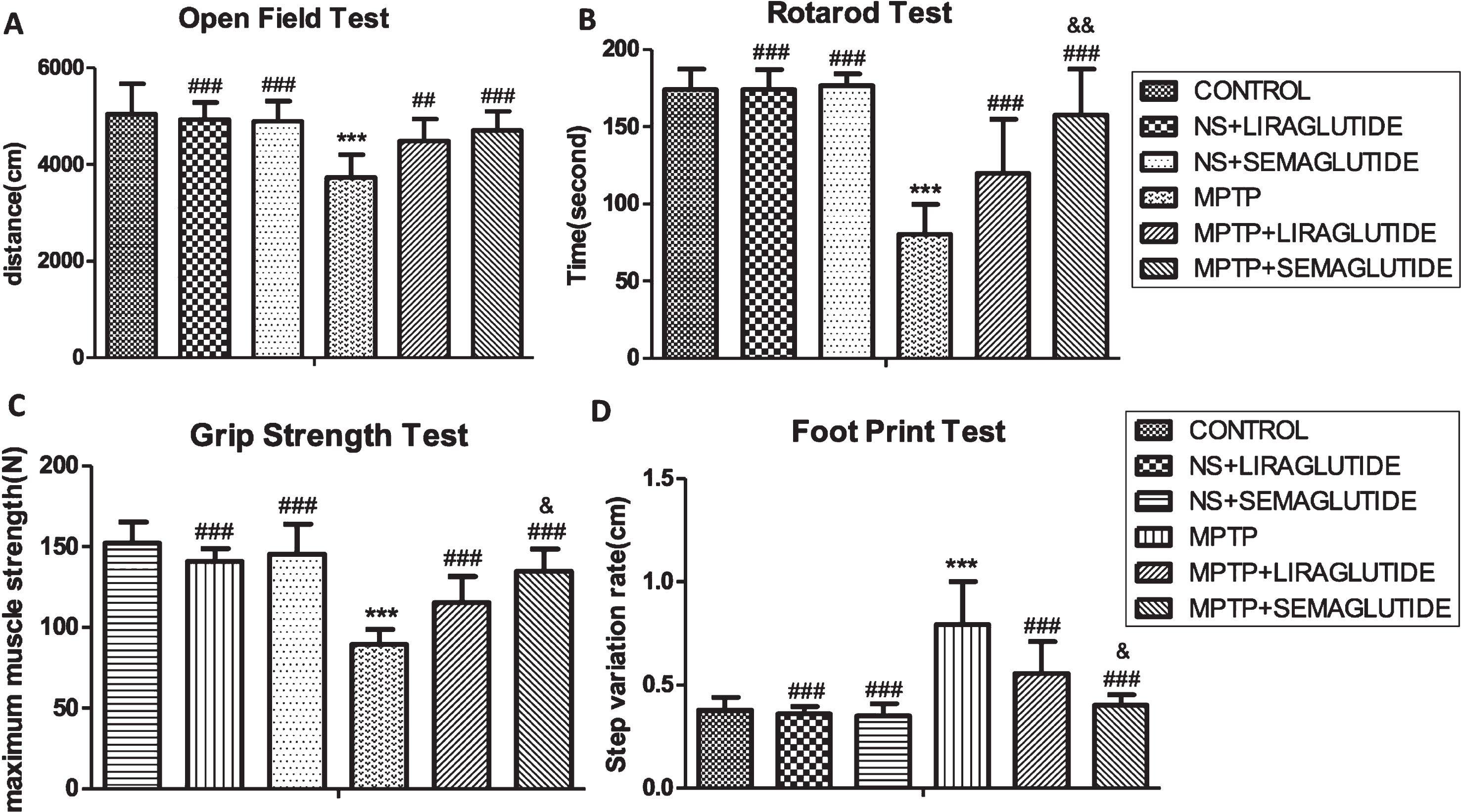
A) GLP-1 analogues protect from the MPTP-induced impairments in motor activity of mice. A difference was found between the control group and MPTP group. Furthermore, a difference was found between MPTP + liraglutide and MPTP + semaglutide and the MPTP group. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # #p < 0.01, # # #p < 0.001 compared with the MPTP group. n = 12. B) GLP-1 analogues improve the bradykinesia and imbalance of mice induced by MPTP. A difference was found between the control group and MPTP group. Furthermore, a difference was found between MPTP + liraglutide and MPTP + semaglutide and MPTP group. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # # #p < 0.001 compared with the MPTP group. &&p < 0.01 compared with MPTP + liraglutide group. n = 12. C) GLP-1 analogues improve the muscle strength weakening of mice induced by MPTP. A difference was found between the control group and MPTP group. Furthermore, a difference was found between MPTP + liraglutide and MPTP + semaglutide and MPTP group. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # # #p < 0.001 compared with the MPTP group. &p < 0.05 compared with MPTP + liraglutide group. n = 12. D) GLP-1 analogues improve the abnormal posture and gait of mice induced by MPTP. A difference was found between the control group and MPTP group. Furthermore, a difference was found between MPTP + liraglutide and MPTP + semaglutide and MPTP group. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # # #p < 0.001 compared with the MPTP group. &p < 0.05 compared with MPTP + liraglutide group. n = 12. NS, normal saline.
In the rotarod test, a one-way ANOVA found an overall difference of the time spent on the rotating rod (F = 37.87, p < 0.0001) between all groups. There was no difference between control group and ns + liraglutide group and ns + semaglutide group. However a difference was found between the control group and MPTP group (p < 0.001). Furthermore, a difference was found between MPTP + liraglutide and MPTP + semaglutide and the MPTP group (p < 0.001). That is to say the two drugs were able to improve the bradykinesia and imbalance of mice induced by MPTP. Semaglutide was more effective than liraglutide (p < 0.01). N = 12 per group, see (Fig. 1b).
In the Grip Strength Test, a one-way ANOVA found an overall difference of maximum muscle strength (F = 35.86, p < 0.0001) between all groups. There was no difference between control group and ns + liraglutide group and ns + semaglutide group, However a difference was found between the control groups and MPTP group (p < 0.001). Furthermore, a difference was found between MPTP + liraglutide and MPTP + semaglutide and the MPTP group (p < 0.001). That is to say the two drugs were able to improve the muscle strength of mice that was impaired by MPTP. Semaglutide was more effective than liraglutide (p < 0.05). N = 12 per group, see (Fig. 1c).
In the foot print test, a one-way ANOVA found an overall difference on the Step variation rate (F = 27.61, p < 0.0001) between all groups. There was no difference between control group and ns + liraglutide group and ns + semaglutide group. However a difference was found between the control groups and MPTP group (p < 0.001). Furthermore, a difference was found between MPTP + liraglutide and MPTP + semaglutide and the MPTP group (p < 0.001). That is to say the two drugs were able to improve the abnormal posture and gait of mice induced by MPTP. Semaglutide was more effective than liraglutide (p < 0.05). N = 12 per group, see (Fig. 1d).
Semaglutide and liraglutide attenuated dopaminergic neuronal loss in the SN induced by MPTP
In the histological analysis of the number of cells positive for the dopamine biomarker tyrosine hydroxylase (TH) in the substantia nigra, MPTP reduced the number of neurons significantly. In a one-way ANOVA with Tukey’s multiple comparison test (F = 42.12, p < 0.0001), MPTP groups showed fewer TH positive neurons in the SN than saline-treated mice (p < 0.001). There was a difference between MPTP + liraglutide and MPTP group (p < 0.01), and a difference between MPTP + semaglutide and MPTP group (p < 0.001). Semaglutide was more effective than liraglutide (p < 0.05), see (Fig. 2).
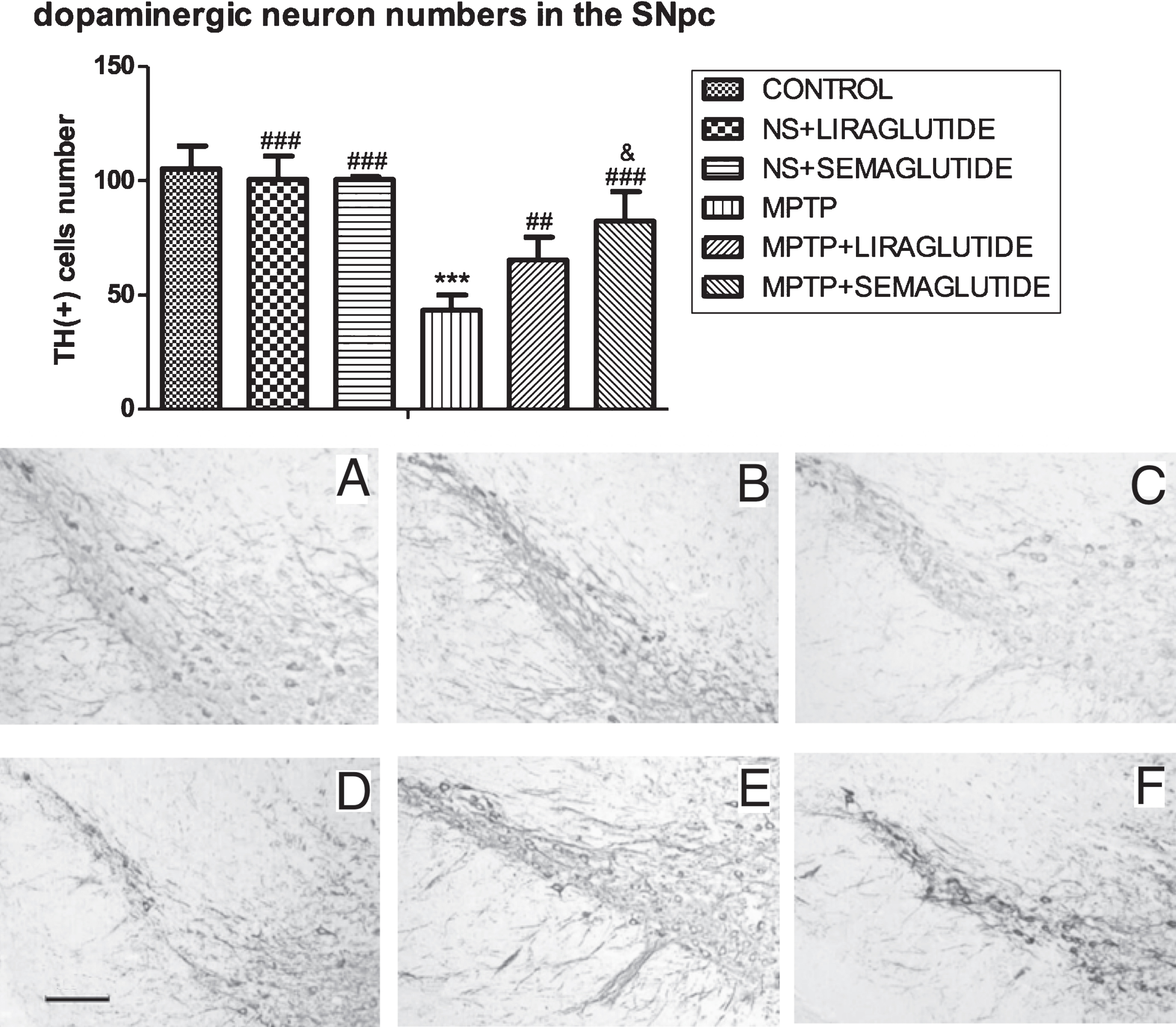
GLP-1 analogues restored tyrosine hydroxylase (TH) positive dopaminergic neuron numbers in the substantia nigra. NS = normal saline. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # #p < 0.01, # # #p < 0.001 compared with the MPTP group. &p < 0.05 compared with MPTP + liraglutide group. n = 6. Examples of micrographs are given. A: Control; B: NS + Liraglutide; C: NS + Semaglutide; D: MPTP; E: MPTP + Liraglutide; F: MPTP + Semaglutide. Scale bar in image D: 100μm.
Semaglutide and liraglutide alleviated astrocyte and microglia activation in the striatum
When analysing GFAP (astrogliosis) levels in the striatum: In a one-way ANOVA with Tukey’s multiple comparison test (F = 432.2, p < 0.0001). There was no difference between the control group and the ns + liraglutide group and the ns + semaglutide group. GFAP levels in the MPTP group were found to be far higher in the striatum compared to the control group (p < 0.001), a difference was found between the MPTP + liraglutide, MPTP + semaglutide and the MPTP group (p < 0.001), and semaglutide was more effective than liraglutide (p < 0.05); N = 6 per group, see (Fig. 3a).
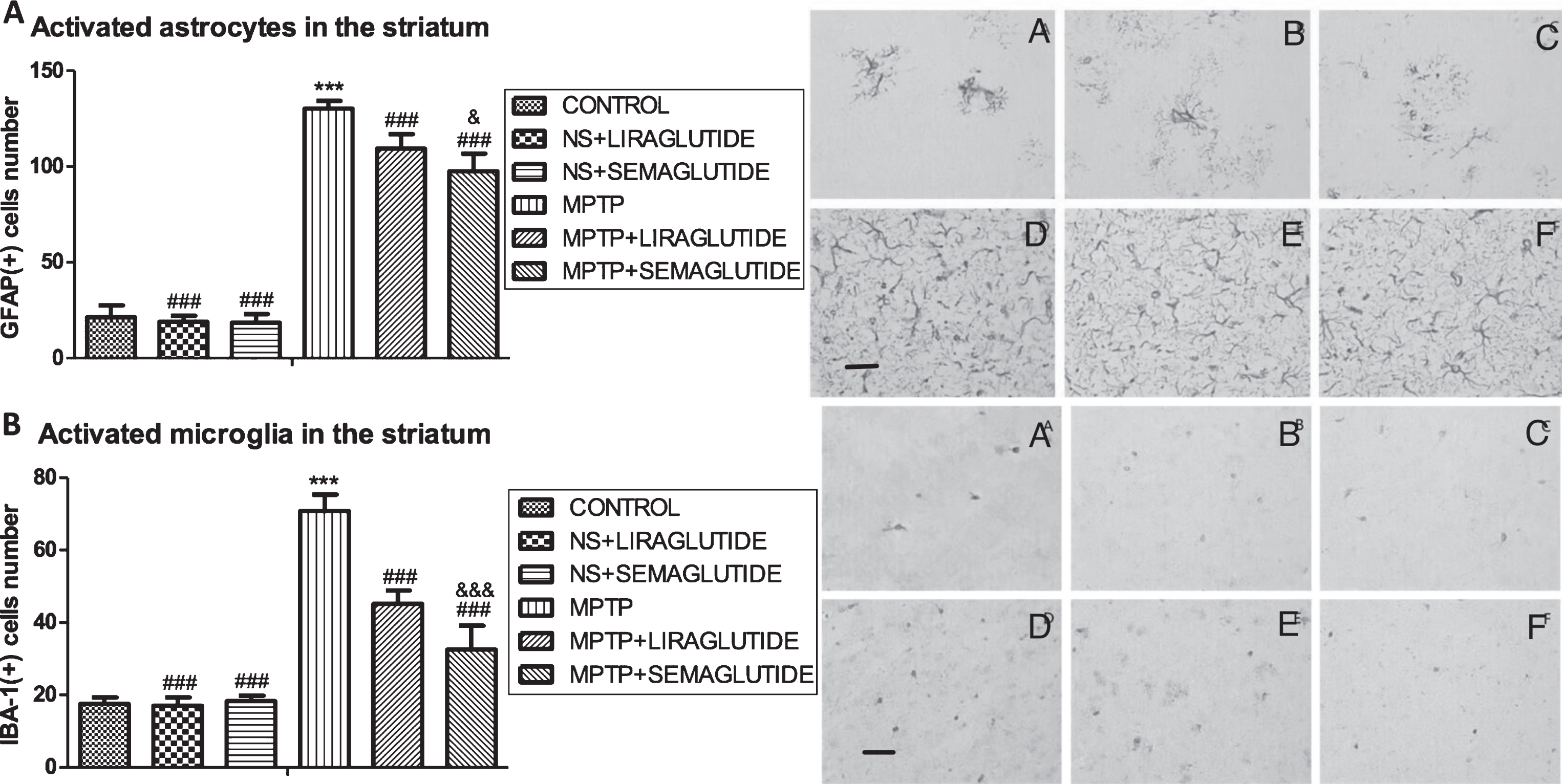
A) GLP-1 analogues reduced the astrocyte activation in the striatum of mice induced by MPTP. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # # #p < 0.001 compared with the MPTP group. &&&p < 0.001 compared with MPTP + liraglutide group. n = 6. Examples of micrographs are given. A: Control; B: NS + Liraglutide; C: NS + Semaglutide; D: MPTP; E: MPTP + Liraglutide; F: MPTP + Semaglutide. Scale bar in image D: 25μm. B) GLP-1 analogues reduced the microglia activation in the striatum of mice induced by MPTP. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # # #p < 0.001 compared with the MPTP group. &&&p < 0.001 compared with MPTP + liraglutide group. n = 6. Examples of micrographs are given. A: Control; B: NS + Liraglutide; C: NS + Semaglutide; D: MPTP; E: MPTP + Liraglutide; F: MPTP + Semaglutide. Scale bar in image D: 25μm. NS, normal saline.
When assessing IBA-1 (microgliosis) levels in the striatum: In a one-way ANOVA with Tukey’s multiple comparison tests (F = 187.2, p < 0.0001), there was no difference between the control group and ns + liraglutide group and the ns + semaglutide group. In the MPTP group, IBA-1 levels were found to be higher than the control group (p < 0.001). A difference was found between the MPTP+liraglutide, MPTP + semaglutide and the MPTP group (p < 0.001), and the MPTP + semaglutide group differed from the MPTP + liraglutide group (p < 0.001), demonstrating that liraglutide and semaglutide can reduced microgliosis. Semaglutide was the more potent drug. N = 6 per group. See (Fig. 3b).
Semaglutide and liraglutide reduced lipid peroxidation in the striatum induced by MPTP
In the immunohistochemical analysis, 4-HNE was monitored as an indicator of lipid peroxidation. A one-way ANOVA found an overall difference of 4-HNE expression in the SN (F = 157.8, p < 0.001); There was no difference between the control group and ns + liraglutide group and the ns + semaglutide group. In the MPTP group, 4-HNE levels were found to be higher than in the control group (p < 0.001), but identical to the MPTP + liraglutide group and MPTP + semaglutide group. This shows that the liraglutide and semaglutide drugs reduced 4-HNE levels (p < 0.001). The MPTP+semaglutide group differed from the MPTP + liraglutide group (p < 0.001), demonstrating that both liraglutide and semaglutide reduced 4-HNE levels. Semaglutide was the more potent drug. N = 6 per group. See (Fig. 4).
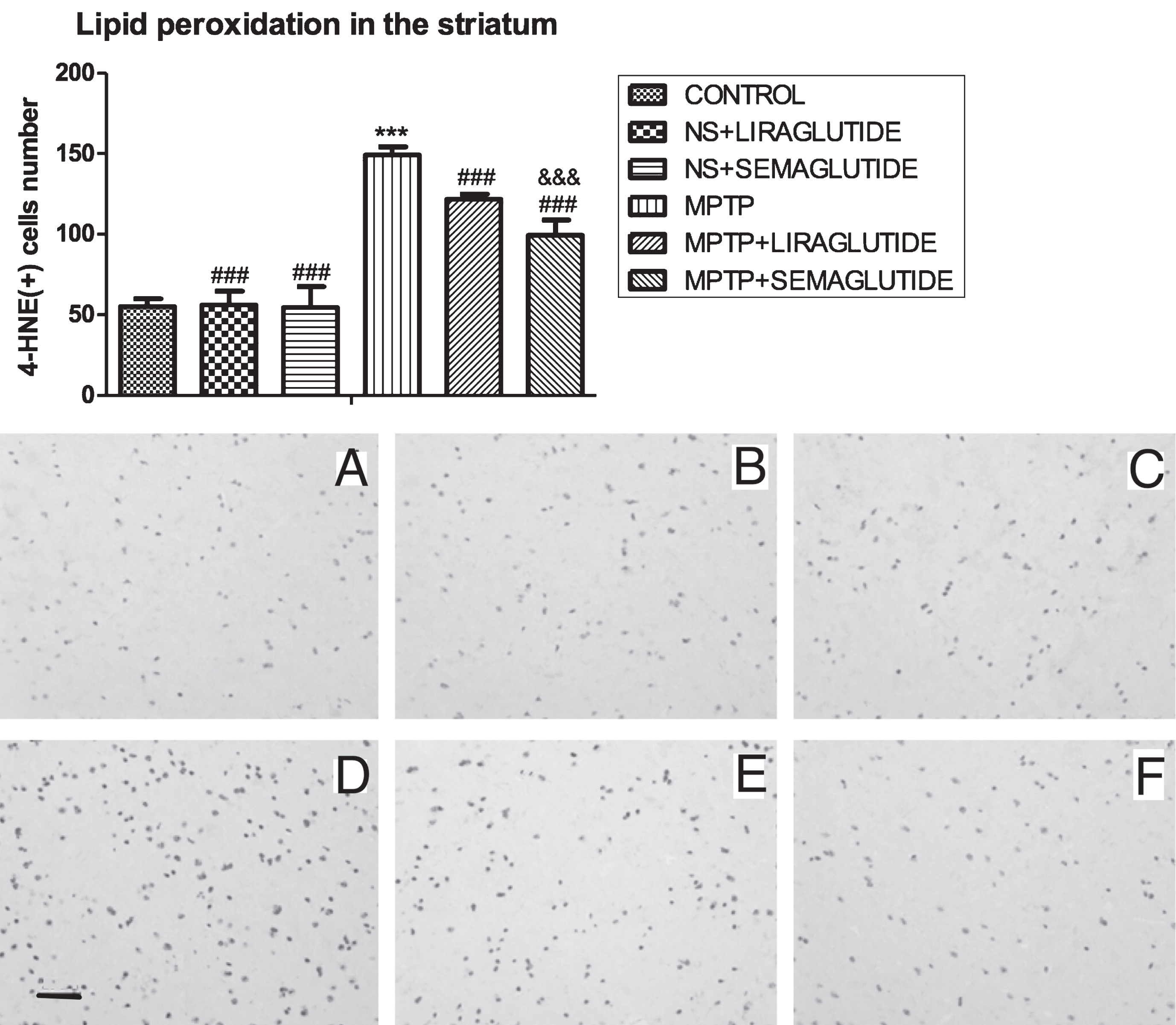
GLP-1 analogues reduced the oxidative stress 4-Hydroxynonenal expression in the striatum of mice induced by MPTP. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # # #p < 0.001 compared with the MPTP group. &&&p < 0.001 compared with MPTP + liraglutide group. n = 6. Examples of micrographs are given. A: Control; B: NS + liraglutide; C: NS + Semaglutide; D: MPTP; E: MPTP + Liraglutide; F: MPTP + Semaglutide. Scale bar in image D: 25μm. NS, normal saline.
Semaglutide and liraglutide reduced the levels of α-syn in the SN enhanced by MPTP treatment
In the western blot analysis, we investigated the levels of a-syn in the SN. A one-way ANOVA showed an overall difference (F = 139.7, p < 0.0001). In Tukey’s multiple comparison tests, there was no difference between control group and ns + liraglutide group and ns + semaglutide group. In the MPTP group, α-syn levels were found to be higher than in the control group (p < 0.001), the MPTP + liraglutide group and MPTP + semaglutide group (p < 0.001). The MPTP + liraglutide group was different from the MPTP + semaglutide group (p < 0.01). This shows that both liraglutide and semaglutide drug reduced α-syn levels, and that semaglutide was more effective. N = 4 per group. See (Fig. 5).
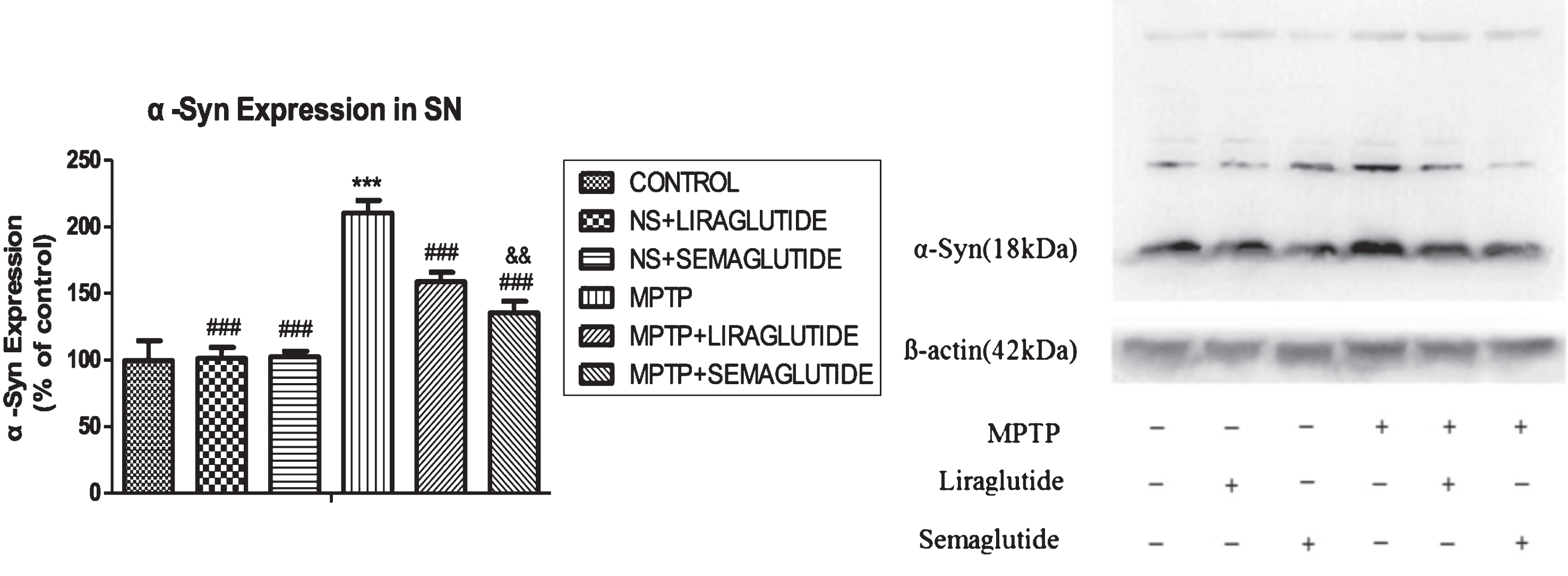
GLP-1 analogues reduced the accumulation of α-Syn in the substantia nigra of mice induced by MPTP. NS = normal saline. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # # #p < 0.001 compared with the MPTP group. &&p < 0.01 compared with the MPTP + LIRAGLUTIDE group; n = 4 per group.
Semaglutide and liraglutide normalized the Bcl-2/BAX ratio in the SN impaired by MPTP
The increase of Bax/Bcl-2 levels in the substantia nigra of mice induced by MPTP was reversed by the two drugs. A one-way ANOVA showed an overall difference (F = 324.8, p < 0.0001). In Tukey’s multiple comparison tests, there was no difference between control group and ns + liraglutide group and ns + semaglutide group. The overall levels of the anti-apoptotic signaling molecule Bcl-2 in SN was reduced by MPTP treatment, levels of the pro-apoptotic signaling molecule Bax in SNpc was increased by MPTP treatment, and the ratio of Bax/Bcl-2 was increased (p < 0.001), compared with control group. Liraglutide and semaglutide partly decreased the ratio of Bax/Bcl-2 by enhancing Bcl-2 levels and decrease Bax levels (p < 0.001). Semaglutide was the more potent drug (p < 0.001). N = 4 per group, see (Fig. 6).
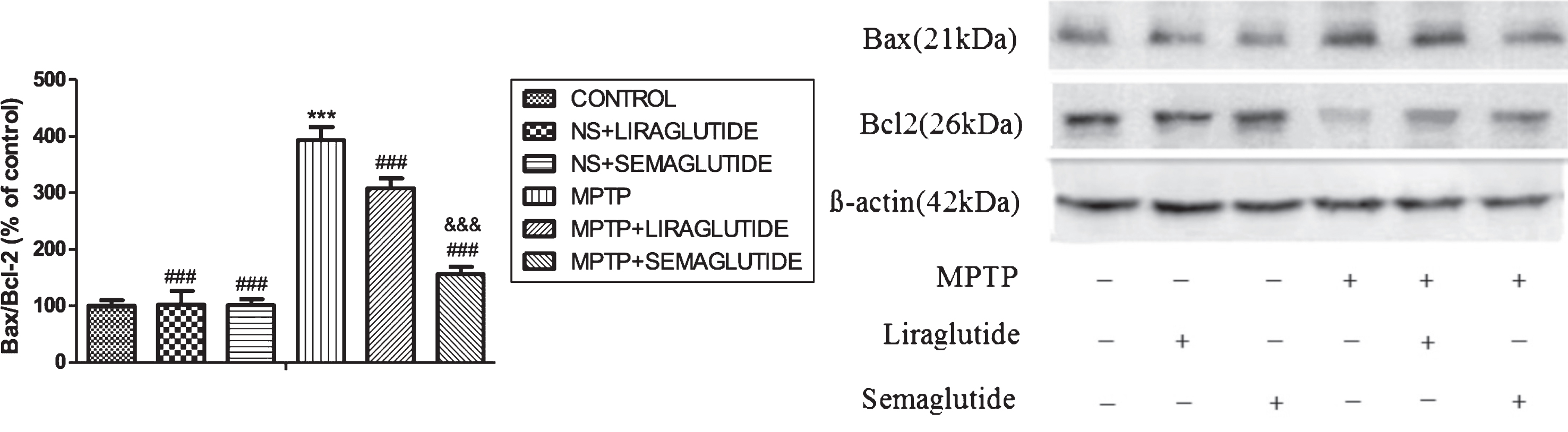
GLP-1 analogues reversed the increase of ratio of mitophagy markers Bax/Bcl-2 in the substantia nigra of mice induced by MPTP. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # # #p < 0.001 compared with the MPTP group. &&&p < 0.001 compared with the MPTP + Liraglutide group; n = 4 per group. NS, normal saline.
Semaglutide and liraglutide increased autophagy-related proteins expression in the SN reduced by MPTP
In the substantia nigra, we investigated the expression of a set of autophagy-related (Atg) proteins. Protein expression of Beclin1, Atg7, LC3 and P62 significantly differs among groups as evident by one-way ANOVA analysis (p < 0.0001). Tukey’s multiple comparison tests demonstrated that insurmountable ER stress induced by MPTP significantly suppresses Beclin1, Atg7, LC3, and P62 expression. Semaglutide and liraglutide treatment significantly enhanced levels of Beclin1, Atg7, L3 and P62. Additionally, semaglutide was more effective than liraglutide. N = 4 per group, see (Fig. 7).
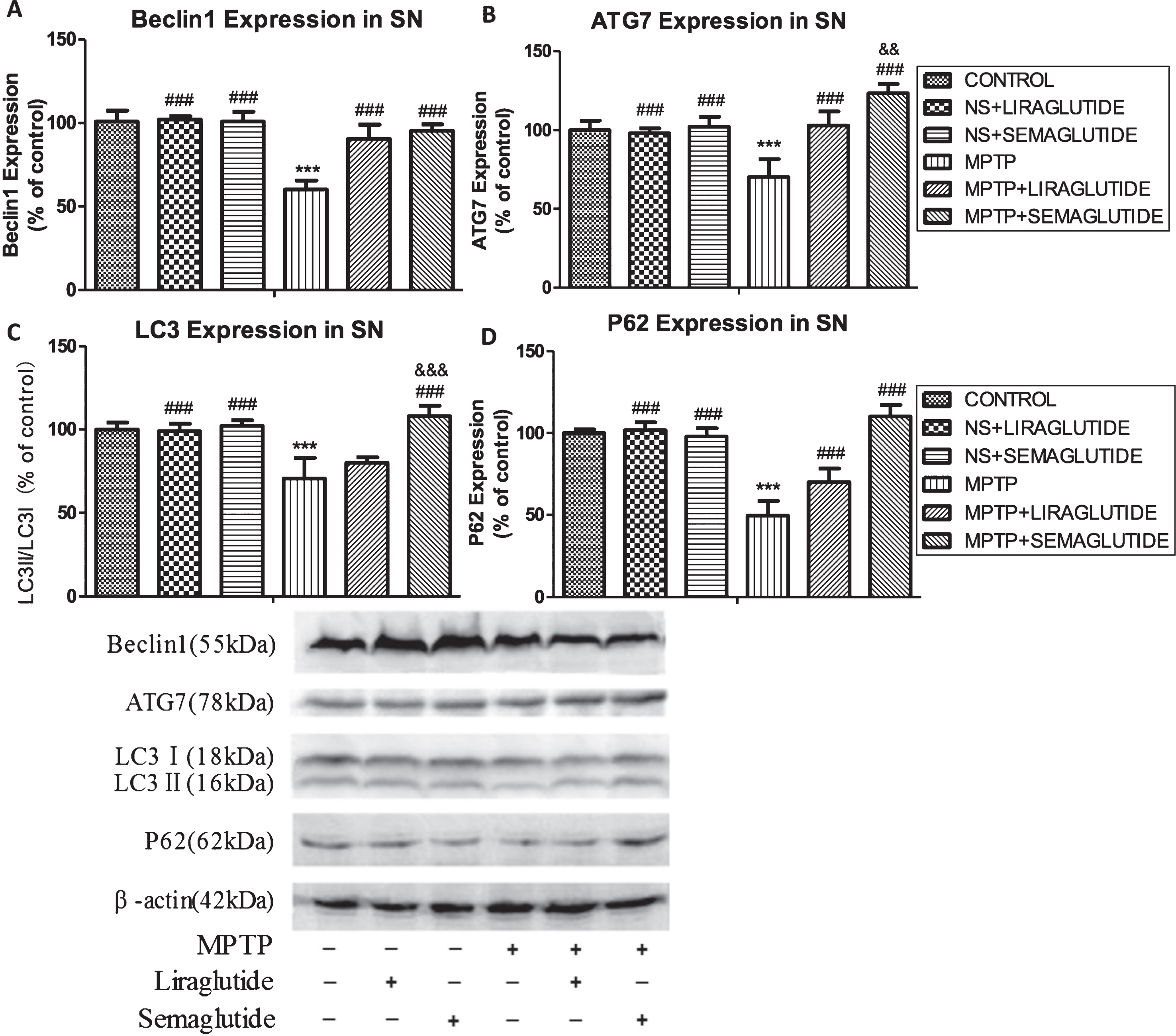
GLP-1 analogues reverse the decrease of autophagy-associated markers. A) Beclin1; B) ATG7; C) LC3; D) P62 expression and even upregulated autophagy in the substantia nigra of mice reduced by MPTP; E) Sample western blot scans are shown. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # # #p < 0.001 compared with the MPTP group. &&p < 0.01 and &&&p < 0.001 compared with the MPTP + Liraglutide group; n = 4 per group. NS, normal saline.
Semaglutide and liraglutide increased GDNF expression in the SN reduced by MPTP
In the western blot analysis, we investigated the expression of GDNF in the SN. A one-way ANOVA showed an overall difference (F = 67.88, p < 0.0001). In Tukey’s multiple comparison tests, there was no difference between the control group and ns + liraglutide group and ns + semaglutide group. In the MPTP group, GDNF levels were found to be lower than in the control group (p < 0.001), but identical to the MPTP + liraglutide group and MPTP + semaglutide group. This shows that the liraglutide (p < 0.01) and semaglutide (p < 0.001) partly increased GDNF levels. Semaglutide was the more potent drug (p < 0.001) compared to liraglutide. N = 4 per group. See (Fig. 8).

GLP-1 analogues reverse the decrease of GDNF expression in the substantia nigra of mice reduced by MPTP. The values represent the means±S.E.M. ***p < 0.001 compared with the control group. # #p < 0.01 and # # #p < 0.001 compared with the MPTP group. &&&p < 0.001 compared with the MPTP + Liraglutide group; n = 4 per group. NS, normal saline.
DISCUSSION
PD is characterized by the progressive functional loss of dopaminergic neurons in the SN. One hypothesis is that the progressive deterioration of SN dopaminergic neurons may be caused by misfolding and aggregation of the protein alpha synuclein, disruption of the autophagy system, and mitochondrial dysfunction [37]. 1-methyl-4-phenyl-1,2,3,6-tetra- hydropyridine (MPTP) is widely used to induce a Parkinson-like state in rodents [38, 39], which can cross the blood brain barrier. Then, MPTP is metabolized into the toxic cation1-methyl-4-phenylpyridinium (MPP+) by monoamine oxidase B [40]. MPP + can kill primarily dopamine-producing neurons in SN [41, 42]. MPP + interferes with complex1of the mitochondrial electron transport chain, which leads to the production of free radicals and ultimately to neuronal death in the SN [43, 44]. The MPTP animal model is a commonly used model of PD as this chemical can induce a PD-like phenotype in humans [45].
Our study demonstrates that both the GLP-1 analogue semaglutide and liraglutide effectively normalized locomotor and exploratory activity, improved bradykinesia, movement coordination and balance of mice, restored a weakening of muscle strength, and improved postural and gait abnormalities of MPTP-treated mice. These results are in agreement with our previous studies in the acute MPTP model [35]. Importantly, we have previously shown that GLP-1 mimetics do not affect food intake or insulin plasma levels in non-diabetic and non-obese animals [46]. We and others furthermore demonstrated that these drugs can enter the brain and are activate receptors on neurons [47–49].
Tyrosine hydroxylase (TH) is a key enzyme in the synthesis of the catecholamine neurotransmitters [50] and is the principal regulator of dopamine synthesis in the CNS [51]. In order to investigate whether GLP-1could directly protect against MPTP-induced nigrostriatal degeneration, we measured the number of TH positive cells. Our results demonstrate that both drugs could increase the number in the SN, and show that the once-weekly semaglutide was more effective than the once-daily liraglutide. Chronic inflammation is playing a central role in PD pathogenesis because the release of cytokines promotes disease progression [52, 53]. Damaged dopaminergic neurons and activated microglial cells can stimulate astrocytes into immune-active status [54]. Moreover, the status of astrocyte activation and the release of pro-inflammatory cytokines is associated with impairment of the nigrostriatal system of MPTP treated mice [41, 55]. Recently a study reported that the presence of activated microglia in the SN and putamen of patients with a PD diagnosis [56]. Both central and peripheral inflammation responses are responsible for sustained progression of PD [57]. Our results demonstrate that both GLP-1 analogues can inhibit the inflammatory response. Importantly, semaglutide was more effective compared with liraglutide in our study where both drugs were tested at the same concentration.
Oxidative stress is a key feature of PD and of chronic inflammation that drives disease progression [58]. One study found that lipid peroxidation and the level of 4-Hydroxynonenal (4-HNE) in SNpc are increased in PD [59]. 4-HNE is one of the markers of membrane lipid peroxidation induced by the excessive generation of reactive oxygen species (ROS) [60, 61]. The generation of ROS by MPTP administration is partly due to the inhibition of the mitochondrial complex I activity [62]. 4-HNE furthermore activates BAD, a sensor for mitochondrial dysfunction, and accelerates mitochondrial mitophagy [63]. Our study demonstrates that both drugs can reduce 4-HNE levels in the midbrain of the mouse induced by MPTP treatment, protecting cells of oxidative stress. Again, semaglutide was more potent compared with liraglutide in this experiment.
The mechanisms underlying accumulation and aggregation of α-syn are considered to be based on over-expression and failure to clear α-syn by proteolysis and autophagy pathways [64, 65]. In addition, aberrant forms of α-syn, including oligomers and fibrils, are seen to interfere with normal cellular processes, promoting further aggregation of protein, leading to the spread of these toxic forms of α-syn from neuron to neuron, and ultimately to neuronal death [66–68]. Oligomeric α-syn is proposed to play a central role in spreading protein aggregation in the brain with associated cellular toxicity, contributing to a progressive neurological decline. One study demonstrated that the cerebrospinal fluid (CSF) of patients with PD contained increased levels of α-syn oligomers when compared to controls [69]. We therefore decided to measure α-syn expression in the brain. We have previously shown that in the MPTP mouse model, α-syn expression is very much increased [63]. Our study demonstrates that the MPTP-induced increase of α-syn expression in the brain is reduced back to almost control levels by the drug, semaglutide again being more potent than liraglutide.
Apoptosis (the most common form of programmed cell death) is closely related to mitochondrial function, because the intrinsic apoptosis pathway is linked to mitochondrial depolarization [70]. B cell leukemia/lymphoma 2 (Bcl-2) -family proteins regulate the intrinsic apoptosis pathway by controlling mitochondrial outer membrane permeability [71]. The anti-apoptotic protein Bcl-2 can bind to the pro-apoptotic protein BAX (Bcl-2-associated X protein) to form heterodimers that modulate apoptosis [72]. Therefore, we measured the ratio of Bax/Bcl-2 levels. Our result show that MPTP treatment led to a decline of Bcl-2 levels and an increase of BAX in the SN, and GLP-1 analogues partly reversed this process. Our results show that the rate of mitophagy and eventually apoptosis is reduced in the brain after drug treatment. In addition, semaglutide showed an advantage compared with liraglutide.
Autophagy removes misfolded proteins and damaged mitochondria to prevent apoptosis caused by mitochondrial dysfunction [73–75]. Some studies show that the autophagy-lysosome system is impaired in PD animal models and evidence for this was also found in the analysis of postmortem PD brain tissue [76, 77]. In physiological conditions, apoptosis is blocked and autophagy maintains intracellular homeostasis; this balance is perturbed in PD [78, 79]. The activation of Beclin-1 leads to autophagosome formation and initiation of autophagy, and a reduced expression of this protein will impair this process. We also measured the conversion of microtubule-associated protein 1 light chain 3 beta-I/LC3B-I to LC3B-II which is important for the sequestration of the phagosome in autophagy. The conversion of LC3B-I to LC3B-II increased after GLP-1 treatment compare with the MPTP group, and GLP-1 analogues upregulated beclin1 expression, indicating that GLP-1 signalling alleviates the inhibition of autophagy induced by MPTP. Autophagy dysfunction along with persistent ER stress can further trigger the excess accumulation of the autophagy adaptor protein p62, which contains a KEAP1 binding motif similar to the promotor of oxidative stress-reducing genes Nrf275 [80]. Accumulation of p62 leads to KEAP1 sequestration and inactivation, which, in turn, blocks nuclear Nrf2 localization and transcription of Nrf2 target genes [81]. This will lead to an impaired response to enhanced oxidative stress. These results are in line with our previous studies of liraglutide effects on autophagy [82].
Glial cell line-derived neurotrophic factor (GDNF) is one of the most potent trophic factors that have been identified for midbrain dopamine (DA) neurons, and plays an important role in the postnatal survival of mesencephalic dopamine neurons [83, 84]. In 1993 GDNF was first been shown to protect embryonic dopaminergic neurons in vitro [85]. The therapeutic benefit of GDNF and NRTN has been demonstrated in phenotypic, toxin-induced (MPTP) rodent and nonhuman primate models of PD [86–89]. Our study demonstrates that the MPTP-induced loss of GDNF in the brain was reversed by both drugs, demonstrating that GLP-1 signalling can rescued the decrease of GDNF levels induced by MPTP. This is in line with previous studies [90, 91]. In addition, semaglutide showed an advantage compared with liraglutide.
In summary, our result showed that semaglutide and liraglutide normalized impaired motor activity, increased the number of TH positive neurons in the SN, reduced the expression of α-syn, and decreased inflammation, oxidative damage and mitophagy while increasing autophagy, and furthermore increased GDNF expression. These conclusion confirm our previous findings that GLP-1 receptor agonists have neuroprotective effects in PD mouse models [15]. In this mouse model of PD, semaglutide appears to be more effective than liraglutide under the conditions chosen in this study. As both liraglutide and semaglutide are in clinical trials in PD patients, we will be able to see if this outcome translates into the clinic.
CONFLICT OF INTEREST
CH is a named inventor on patent applications that cover the use of GLP-1 receptor agonists as a treatment for Parkinson’s disease. The patent is owned by Ulster university, UK.
Footnotes
ACKNOWLEDGMENTS
The research project was supported by a Shanxi Scholarship Council of China (2017- importance 4), and by the Fund for Shanxi “1331 Project” Key Subjects Construction.
