Abstract
Background:
Although abnormalities in gut microbiota are hypothesized to influence the pathogenesis and clinical phenotype of Parkinson’s disease (PD), prospective studies on de novo patients are lacking.
Objective:
To preliminarily investigate whether gut microbiota in early untreated PD may predict motor and non-motor features progression over a 3-year period.
Methods:
16S ribosomal RNA gene amplicons were sequenced on fecal samples of 39 de novo PD patients. Multiple confounders were taken into account, including dietary habits. Motor and non-motor symptoms were assessed using validated scales at baseline and followed-up yearly for 3 years. At last follow-up, a detailed neuropsychological assessment was additionally performed. A general linear model for repeated measurements— adjusted by dopaminergic therapy at follow-up— was used to investigate the relationship between bacterial taxa abundance at baseline (stratified by the median of distribution at baseline) and outcome variables.
Results:
Twenty-five patients were included (11 refused, 2 lost at follow-up, 1 died). Lower abundance of Roseburia (Firmicutes phylum) at baseline was associated with worse evolution of motor, non-motor and cognitive functions at 3-year follow-up. Similarly, lower abundance of Ruminococcaceae and Actinobacteria at baseline was associated with faster worsening of global cognitive functions. At follow-up, frontal lobe functions were the features most robustly associated with baseline microbial abnormalities.
Conclusion:
In the present exploratory study on de novo PD, we found an association between abnormal distribution of specific bacterial taxa and the progression of motor and non-motor features over a 3-year period. This proof-of-principle study supports the design of a larger observational study aiming to determine whether these differences survive multiple-comparison correction and define microbiota-specific subgroups suitable for therapeutic targeting.
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disease worldwide. It is associated with significant morbidity and a huge burden to the caregivers and families of those suffering from PD. Yet, there are no available interventions designed to prevent developing the disease or to modify its course [1]. Thus, there is a major unmet need to identify novel safer therapies for PD that could modify the course of the disease and even prevent PD in high-risk individuals, which can be met only by better understanding its pathogenesis. Aggregated alpha-synuclein found as Lewy bodies and Lewy neurites in pigmented neurons of the brainstem is a consequence of misfolding of native α-syn protein, which is likely the consequence of increased oxidative stress and neuroinflammation [2]. The gastrointestinal tract has been suggested to be the site where environmental factors trigger neuroinflammation and activate microglia and astrocytes [3], because it is the largest surface area between the host and the environment (with over 300 square meters of surface area) and it harbors the most complex microbial community capable of producing proinflammatory factors [4]).
Several studies have evidenced the presence of abnormalities in gut microbial community structure and composition (“dysbiosis”) in patients with PD as compared to healthy controls [4]. Nevertheless, results of case-control studies published so far are heterogeneous with discrepancies likely due to methodological differences (e.g., different inclusion/exclusion criteria, lack of adjustment for confounders that greatly influence gut microbial composition [4]). In a recent cross-sectional study [5], we compared gut microbial composition between 193 PD patients at different disease stages (including 39 early drug-naïve cases) and 113 healthy control subjects, adjusting for multiple confounders (including dietary and lifestyle habits, constipation, PD medications), aiming to investigate whether microbial composition could act as a modulator of disease severity and clinical phenotype. Interestingly, we found that reduced levels of Lachnospiraceae and increased Lactobacillaceae were associated with intellectual impairment and worse motor symptoms, including ‘nondopaminergic’ features, such as gait disturbances and postural instability.
In this scenario, longitudinal studies are needed to establish the causal link between dysbiotic microbiota and progression of PD clinical features. Considering that changes in clinical features might additionally be influenced by microbiota-induced variability in pharmacokinetics of PD medications [4], it is essential to minimise this bias by investigating PD patients without the confounding effect of pharmacological therapy. The aim of the present 3-year prospective exploratory study was to investigate whether gut microbial composition in early, untreated (de novo) PD patients may predict differences in the progression of motor and non-motor symptoms over time.
METHODS
Study design and population
This prospective observational study was performed at the tertiary care out-patient clinic (Parkinson Institute, ASST Gaetano Pini-CTO, Milan, Italy) and included the cohort of 39 patients diagnosed with PD [6] and never exposed to any antiparkinsonian therapy at baseline (de novo), out of the cohort of 193 cases recruited in the previous study [5]. At the baseline assessment, general characteristics were collected, including type of delivery and lactation, eating habits and lifestyle, such as consumption of coffee and cigarette smoking, and clinical features including past and current diseases, surgical interventions in the past and any ongoing pharmacological therapies. Exclusion criteria were: ongoing artificial nutrition; chronic inflammatory bowel disease; any type of autoimmune disease; acute inflammatory disease (e.g., viral/bacterial infections); history of important gastrointestinal surgery; use of antibiotics, probiotics, corticosteroids or other immunosuppressive drugs in the past month; radio-chemotherapy; severe cognitive impairments that precluded the execution of the assessments; advanced stage therapies (deep brain stimulation, continuous apomorphine or duodenal infusion of levodopa). Brain magnetic resonance imaging was performed in all cases.
As we aimed to investigate the influence of baseline gut microbial composition on the progression of PD motor and non-motor features, we included all patients who had been visited yearly for 3 years starting from the baseline assessment.
Clinical assessment
Clinical workup included the rating of motor and non-motor symptoms and disease staging at baseline and then every 12 months for 3 years, using the Unified Parkinson Disease Rating Scale (UPDRS) from part I to part IV [7] and the Hoehn and Yahr (HY) stage [8], respectively. We calculated a distinct score for dopaminergic and nondopaminergic motor signs using UPDRS-III data, according to the criteria proposed by Levy and colleagues [9], We assessed non-motor symptoms using the Non-Motor Symptoms Questionnaire (NMSQ) [10, 11], In addition, orthostatic hypotension was directly assessed, measuring blood pressure supine and after 3 minutes of standing. Information on pharmacological therapy inception during follow-up was also recorded. Specifically, dopaminergic drug therapy was calculated in levodopa equivalent daily doses (LEDD) [12],
Global cognitive functions were assessed at baseline and at 3-year follow-up using the Mini-Mental State Evaluation (MMSE) [13]. At the 3-year follow-up assessment, the following extensive neuropsychological testing battery was additionally performed: Montreal Cognitive Assessment (Mo CA), Trail Making Test (TMT scoring the parts A, B, and B-A) [14], and the copy of the figure of Rey-Osterrieth [15]. All scores were adjusted for age and education and considered pathological according to normative data in the Italian population [14, 16–18].
Microbiota analysis
At baseline, stool samples were collected from all patients and stored in a sterile container at –20°C. The procedures of DNA extraction, processing and sequencing have been described in detail elsewhere [5]. Briefly, the V3–V4 region of 16S rRNA gene amplicons were sequenced on Illumina MiSeq platform, using a 2×250 bp paired end protocol (Illumina, San Diego, CA). Raw reads were analyzed using Pandaseq, processed through the QIIME pipeline (release 1.8.0), clustered into Operational Taxonomic Unit (OTUs) at 97% identity level and taxonomically assigned via RDP classifier against the Greengenes database (release 13_8 http://greengenes.secondgenome.com). Bacterial genera were selected considering only those present at >0.5% of abundance in at least 30% of the samples, in order to exclude minor and transient contributors of the gut microbiota.
Statistical analysis
Continuous variables have been described as mean and standard deviation, while categorical variables have been reported with counts and percentages. The comparison of continuous and dichotomous variables between two groups was conducted using the Student’s t test and the Fisher’s exact test, respectively. The prognostic impact of the abundance of bacterial taxa (stratified on the basis of the median distribution) on the outcome variables of disease progression (HY stage, UPDRS-III score, UPDRS-III dopaminergic score, score UPDRS-III non-dopaminergic, UPDRS total score (Parts I + II + III), NMSQ score and MMSE score) was investigated using a general linear model for repeated measurements adjusted for follow-up dopaminergic therapy (expressed in LEDD mg/day); test for interaction between taxa and time was computed. Finally, taxa found to be associated with the progression of global cognitive functions (MMSE) were additionally investigated for their association with neuropsychological tests performed at follow-up. Given the exploratory nature of the study and the small sample size, uncorrected P-values were initially computed. Then, correction for multiple comparisons to address the false discovery rate (FDR) was performed using the Benjamini-Hochberg procedure. Based on the sample size we set the FDR cutoff at 0.2. All analyses were performed using the software Stata 16 (StataCorp, College Station, TX, USA). The level of significance was set at the two-tailed p value < 0.05.
Ethical standards
This study was conducted in accordance with the principles of the Helsinki Declaration and approved by the local ethics committee. All the subjects recruited provided their informed consent to participation.
RESULTS
Study population
Among the 39 patients of the original cohort, 25 patients were included in the final analysis. Reasons of exclusion were: refusal to attend the 3-year follow-up assessment (N = 11), lost at follow-up (N = 2) and death (N = 1). General and clinical characteristics of the study population are summarized by dropout status in Table 1. Considering that about one third of patients (14 out of 39) assessed at baseline could not be included in the present follow-up study, we compared their features with those of the study population and found no difference (Table 1). We found no significant change in clinical and dietary habits of the study population between baseline and follow-up. All patients initiated dopaminergic therapy, either levodopa, dopamine-agonist or MAO-B inhibitor (Table 1). The results of the analysis addressing the association (repeated-measure general linear model) between disease progression end-points and taxa are summarized in Table 2.
General characteristics of the study population and related sub-groups
UPDRS, Unified Parkinson’s Disease Rating Scale; NMSQuest, Nonmotor Symptoms Questionnaire; DA, dopamine agonists; iCOMT, catechol-O-methyltransferase inhibitors; iMAO-B, Monoamine Oxidase type B inhibitors; LEDD, levodopa equivalent daily dose. aSum of items 19-to-22, 24-to-26 (in medication-on condition) [9]. bSum of items 18, 27-to-30 (in medication-on condition) [9].
Association (repeated-measure general linear model) between disease progression end-points variables and taxa
UPDRS, Unified Parkinson’s Disease Rating Scale; NMS-Quest, Non-motor Symptoms Questionnaire; MMSE, Mini-Mental State Examination. aTaxa are listed as follows:
Motor symptoms
After stratifying patients according to the median distribution of taxa abundance at baseline, we observed a significantly faster worsening of the HY stage in patients characterized by lower abundance of Roseburia (Lachnospiraceae family; p = 0.038; Fig. 1A) and higher abundance of Lactobacillaceae (p = 0.047; Fig. 1B). Notably, the average HY stage at baseline was similar between the two subgroups and diverged during the follow-up. The opposite pattern was found for the Christensenellaceae. Patients with higher relative abundance for this family presented at baseline worse HY stage, which remained substantially stable over time. However, patients with lower concentrations showed a significant progression to a comparable disease severity at follow-up (for taxa distribution, p = 0.047; for interaction, p = 0.024).
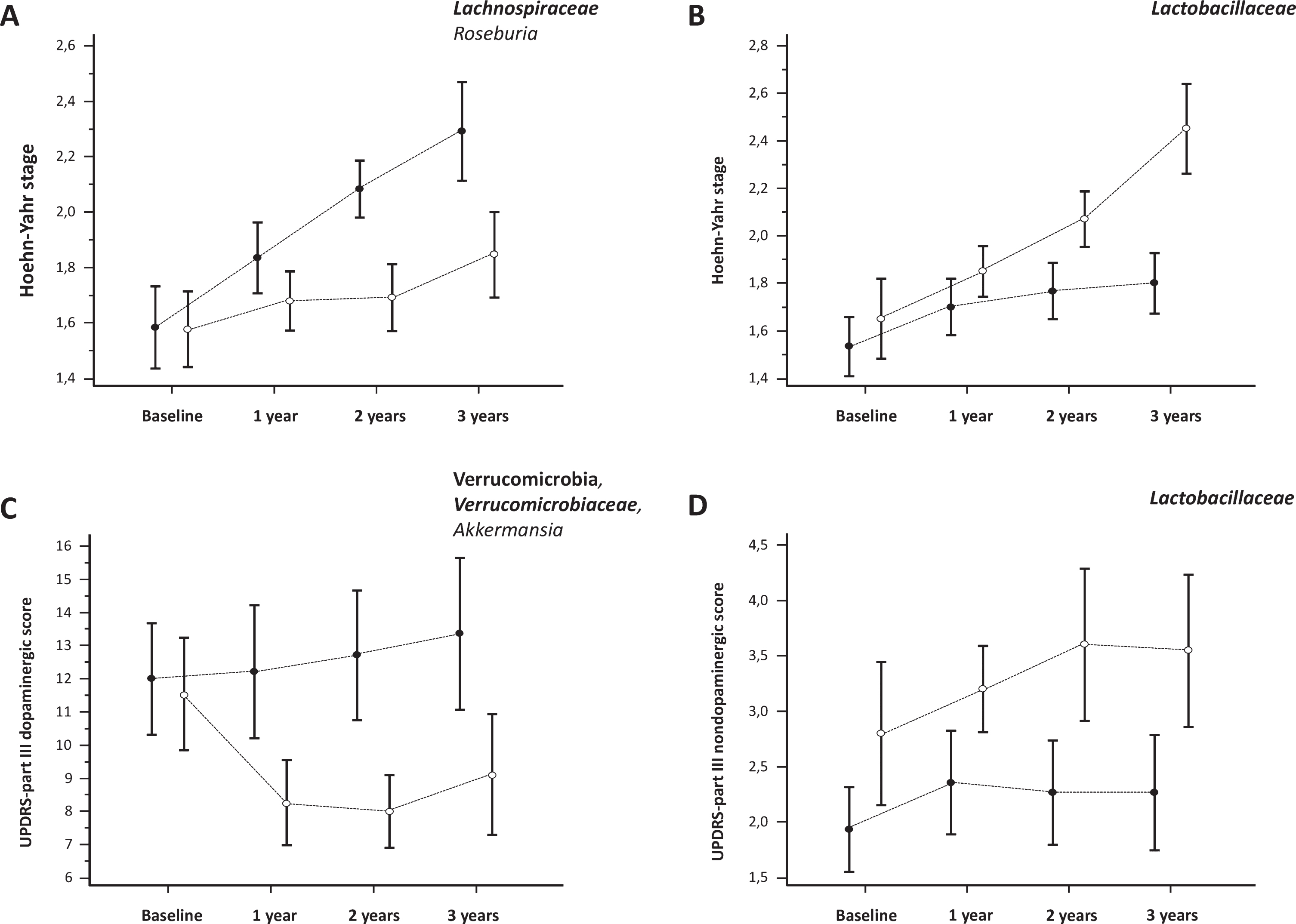
Progression (mean and standard error) of motor symptoms (UPDRS motor score) and disease severity (Hoehn and Yahr stage) according to the relative abundance of bacterial taxa. Empty (∘) and Full (•) dots represent relative abundance respectively above and below the median of the population.
We did not find significant associations between gut microbiota and the worsening of the UPDRS-III score (Table 2). Nonetheless, when we analyzed dopaminergic vs. nondopaminergic motor features, we found that patients with a higher abundance of Verrucomicrobia phylum, Verrucomicrobiacee family and Akkermansia genus showed a better response to dopaminergic therapy as showed by the significant improvement over time in the UPDRS-III ‘dopaminergic score’ (p = 0.045 for all; Fig. 1C). On the other hand, higher abundance of the Lactobacillaceae family was associated with greater scores of non-levodopa-responsive symptoms (such as postural instability and gait difficulties) at baseline, that persisted at follow-up (p = 0.048; Fig. 1D).
Non-motor symptoms
Patients with lower abundance of Roseburia and Unclassified genera of the Lachnospiraceae family developed a greater load of non-motor symptoms over the 3-year follow-up (Table 2, Fig. 2A,B). Similarly, lower abundance of Faecalibacterium (Ruminococcaceae family) at baseline resulted in worse NMSQ scores at follow-up (Table 2, Fig. 2C).
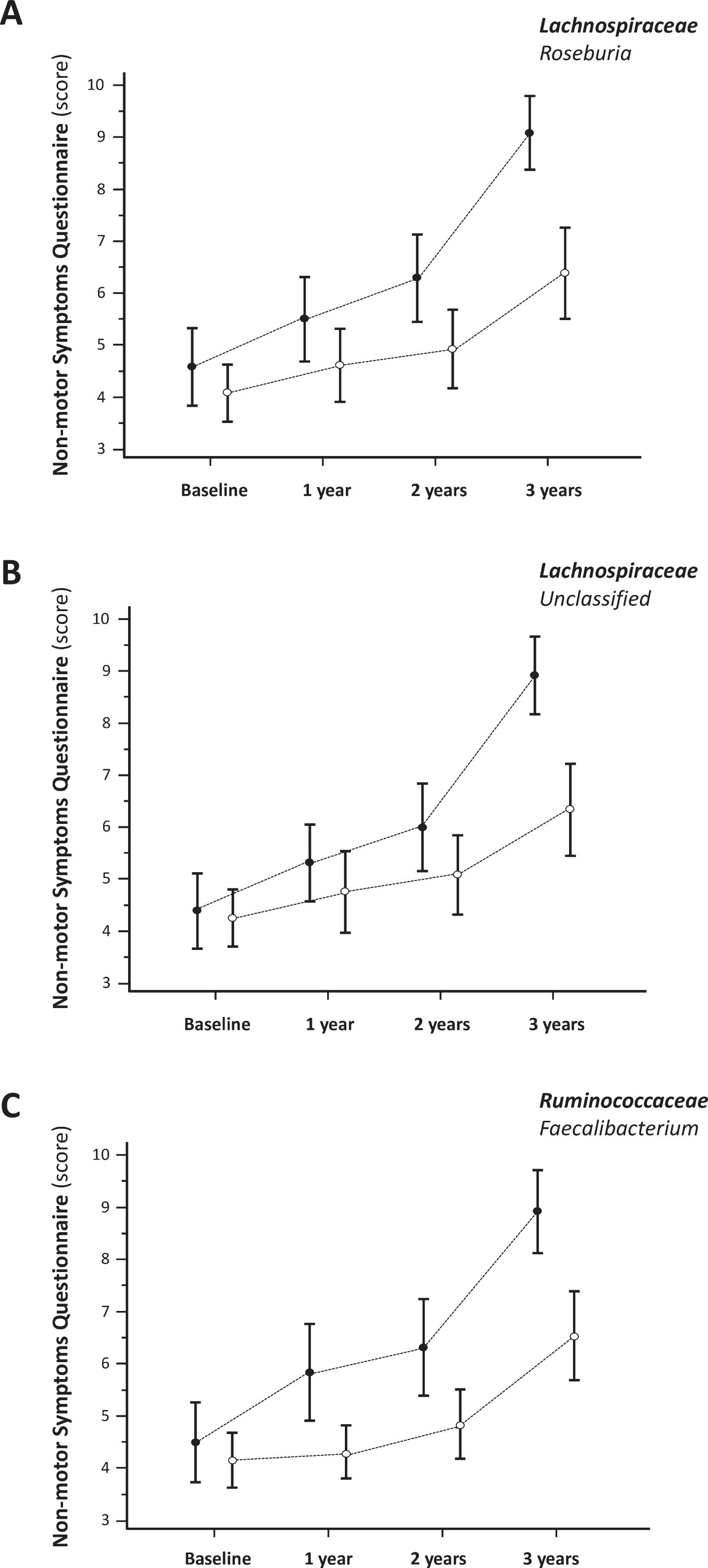
Progression (mean and standard error) of overall non-motor symptoms according to the relative abundance of bacterial taxa. Empty (∘) and Full (•) dots represent relative abundance respectively above and below the median of the population.
Regarding the nine NMSQ domains, patients with lower abundance of the Lachnospiraceae family were characterized by higher impairment in gastrointestinal (p = 0.030) and behavioral (p = 0.018) disturbances, with more specific association with the genera Roseburia (p = 0.041) and Unclassified (p = 0.023), respectively. Notably, lower abundance of Roseburia was associated with significantly worse mnesic-attention disorders at follow-up (p < 0.001).
Cognitive functions
Lower relative abundance of the genera Roseburia and Faecalibacterium at baseline was associated with faster decline in the global cognitive functions (MMSE) after 3 years of follow-up (p = 0.036 and p = 0.047, respectively; Fig. 3A,B). On the other hand, more rapid worsening of MMSE scores was observed in patients with higher relative abundance of the phylum Actinobacteria and the genus Oscillospira (Ruminococcaceae family, Firmicutes phylum) at baseline (p = 0.026 and p = 0.041 respectively; Fig. 3C,D).
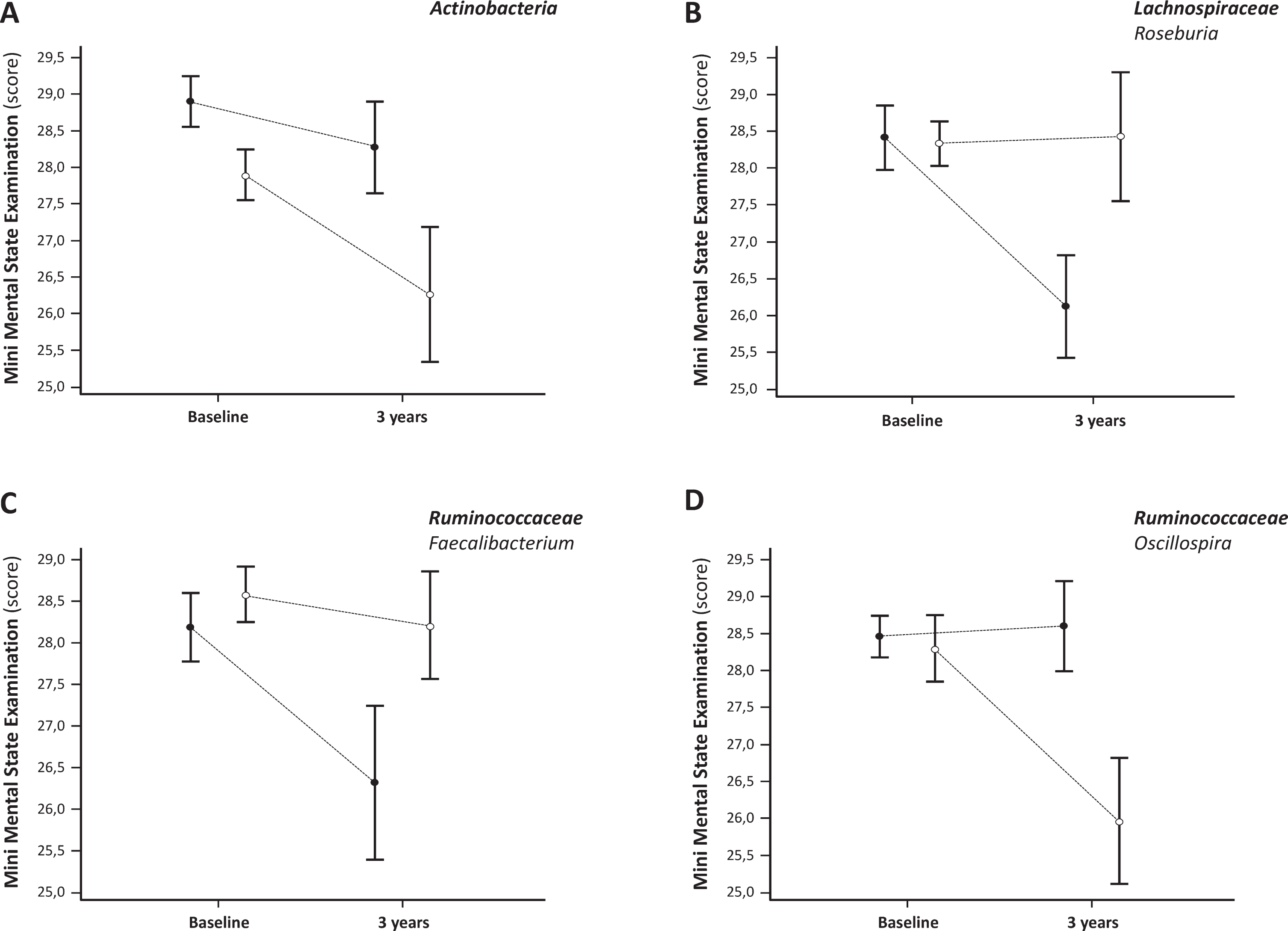
Progression (mean and standard error) of cognitive dysfunction according to the relative abundance of bacterial taxa. Empty (∘) and Full (•) dots represent relative abundance respectively above and below the median of the population.
Consistently with this finding, focusing on additional neuropsychological testing performed at follow-up and the bacterial species whose abundance was associated with differential progression of global cognitive functions, we found that the higher abundance of Oscillospira was associated with worse performance not only at the MMSE but also at the MOCA and in the Trail Making A test (p = 0.001 and p = 0.035, respectively).
Post-hoc correction for multiple testing
As many outcomes were investigated, we recalculated probabilities using a FDR cutoff of 0.2 (Benjamini-Hochberg procedure). After correcting for multiple comparisons, no significant differences were observed in all clinical endpoints.
DISCUSSION
In the present prospective exploratory study of patients with de novo PD, we have provided preliminary evidence supporting the hypothesis that the composition of gut microbiota at the onset of motor symptoms may influence the forthcoming clinical progression. We found that the progression of motor and non-motor symptoms over a 3-year period was consistently associated with the relatively lower abundance of a number of bacterial genera belonging to the Firmicutes phylum, which is the main constituent of the human luminal microbiota under physiological conditions (along with Bacteroidetes) and a major producer of butyrate, which is the preferred energy substrate for colic epithelial cells [4]. Since low levels of butyrate are associated with disruption of intestinal barrier integrity, the reduced abundance of Firmicutes at the earliest stages of PD might allow bacterial inflammatory products like lipopolysaccharide (LPS) to leak into the mucosa and systemic circulation triggering mucosal, systemic and even a neuroinflammatory state [3, 4]. Particularly, we found that reduced relative abundance in the genus Roseburia at baseline was associated with overall worse evolution of motor (Fig. 1A), non-motor (Fig. 2A) and cognitive features (Fig. 3B) at 3-year follow-up.
Motor disability
More rapid progression of motor disability was associated with reduced relative abundance of the genus Roseburia at PD onset. The relationship between Lachnospiraceae and PD motor disability is consistent with previous cross-sectional studies [5, 19]. Heintz-Buschart and colleagues (2018) found a significant correlation between motor symptoms severity and the relative abundance of Lachnospiraceae [19]. Lower abundance of Lachnospiraceae was associated with more severe clinical phenotype and negatively correlated with gait disturbances in our previous case-control study on 193 consecutive PD patients regardless of disease stage, a robust finding that remained significant after adjusting for multiple confounders, such as age, sex, constipation, duration of motor symptoms, dopaminergic medications, dietary and lifestyle habits [5]. Similarly, greater relative abundance of Lactobacillaceae appeared to be associated with faster progression of disease severity (according to the Hoehn and Yahr staging system) over a 3-year period, including a greater burden of non-dopaminergic motor symptoms, such as postural instability and gait impairment. This is in line with the significant relationship we previously found between increased Lactobacillaceae and UPDRS part III score as a whole and at a separate analysis of nondopaminergic features and postural instability [5].
Two longitudinal studies on the prognostic role of gut microbiota in PD have been published so far [20, 21]. Minato and colleagues (2017) subdivided 36 PD patients into two groups according to stability vs progression of clinical features over a 2-year period and found that the deteriorated group had lower genus Bifidobacterium counts than the stable group at baseline, suggesting that lower counts of Bifidobacterium at baseline may be predictive of progression of PD in 2 years [20]. In the present study, we found an association between progression of cognitive dysfunction and the phylum Actinobacteria, to which the genus Bifidobacterium belongs to. Aho and colleagues (2019) reported a 2-year follow-up of their previous study [22] and found that PD patients who progressed were more likely to have a Firmicutes-dominated enterotype than stable patients or control subjects, as well as lower abundance of Lachnospiracee in PD patients compared to healthy controls, although no differences were found after stratifying the PD cohort according to the trend of disease progression [21]. Moreover, Aho and colleagues (2019) did not replicate the findings reported by Minato and colleagues (2017) concerning the prognostic role of Bifidobacterium [20, 21]. These discrepancies might be due to either methodological differences or the lack of a strong microbial signal associated with PD progression. Among methodological differences, (i) baseline assessment was performed by Aho and colleagues on PD patients with an average disease duration of 6.5 years and on pharmacological therapy [21]; (ii) we analyzed the progression of clinical variables over time and stratified the patients according to the relative abundance of the different families and genera of microbiota at baseline.
Non-motor features including cognitive functions
More severe non-motor symptoms at the NMSQ were associated with lower abundance of several genera of the Firmicutes phylum, such as Roseburia, unclassified Lachnospiraceae (Lachnospiraceae family), and Faecalibacterium (Ruminococcaceae family). Our data are in line with a number of case-control studies showing lower relative abundance of the phylum Firmicutes and the Ruminococcacee family in PD patients [4]. In line with our data, a reduction in the relative abundance of Faecalibacterium has been previously reported in PD patients with severe disease as compared to those with mild symptoms [23].
Among non-motor symptoms, we investigated the incident impairment of global cognitive function using the MMSE scale at baseline and at 3-year follow-up. Changes regarding the phylum Firmicutes, and in particular the reduction in the relative abundance of the genus Roseburia, was also related to a reduction in the average 3-year MMSE scores. This was in line with our previous study, which had found an association between a reduction in bacteria of the Lachnospiracee family and greater cognitive impairment [5]. Lower abundance of Faecalibacterium, was associated with faster progression of overall non-motor symptoms (Fig. 2C) and, in particular, with greater impairment of cognitive functions (Fig. 3C). Besides Faecalibacterium, another genus of the Ruminococcaceae family has been found to be associated with the evolution of cognitive decline, but with an opposite pattern. Indeed, patients with higher abundance of Oscillospira (in contrast with the lower abundance of Faecalibacterium) at baseline were associated with lower cognitive performance at follow-up (Fig. 3D). The correlation between increase in Oscillospira and PD is confirmed by previous case-control studies [5, 25]. Furthermore, the evolution of cognitive dysfunction was associated with greater abundance of the phylum Actinobacteria at baseline. There is considerable evidence in the literature supporting a correlation between higher concentrations of Actinobacteria and PD [5, 25–27], and a correlation with disease duration [21] and severity [20]. Taken together, these results suggest a potential role of gut dysbiosis in the development of cognitive impairment in PD. This is consistent with recent studies showing gut microbial metabolites in the cerebrospinal fluid of patients diagnosed with Alzheimer’s disease (AD), a finding that suggests that the microbiome may be involved in the pathogenesis of the disease [28, 29]. Intriguingly, there are similarities between the gut microbial profile in AD and our cohort of PD patients, such as reduced abundance of Firmicutes, Ruminococcacee and Lachnospiracee family [28, 30], and increased relative abundance of the phylum Actinobacteria [30]. Furthermore, the worse MMSE scores in patients with lower abundance of Roseburia further strengthens the hypothesis of a potential role of Lachnospiraceae in the pathogenesis of cognitive dysfunction in PD. Consistently, lower concentrations of Roseburia were associated with greater NMSQ scores referring to the memory-attentional domain. Interestingly, the putative influence of the infant gut microbiota on hippocampal neurogenesis in adults could raise the hypothesis that different conformations of the microbiota from the early stages of development could determine a greater or lesser predisposition to develop neurodegenerative diseases with hippocampal involvement in adulthood [31]. The significance of the correlation found between greater abundance of Oscillospira and worse performance at MOCA and Trail Making A would instead indicate a greater predisposition to the development of cognitive deficits belonging more to the frontal-non-executive sphere, which is more frequently implicated in the cognitive impairment associated with PD.
Although the exact mechanisms underlying the relationship between gut dysbiosis and more severe PD clinical features are yet to be fully elucidated, several microbiota-related mechanisms may influence neurodegeneration, such as neuroinflammation, oxidative phosphorylation, biosynthesis of molecules, LPS, ubiquinone and short-chain fatty acids (SCFA) [4, 32]. There are numerous lines of evidence correlating the depletion of SCFA or SCFA-producing bacteria not only with PD, but also with AD [4, 32], suggesting transversal effects of abnormalities in SCFA-producing bacteria taxa on the course of different neurodegenerative diseases. The beneficial effects played by these bacterial taxa and by SCFA on the central nervous system functions involve several mechanisms (including immune, endocrine, vagal and other humoral pathways) that promote normal microglial development, suppressing inflammation, and stimulating vagal signaling and production of glucagon-like peptide-1 by intestinal L-cells, with beneficial effects in animal models of PD as well as in AD and PD patients [4, 33]. The seemingly deleterious effect played by a greater abundance of Lactobacillaceae may be related to several mechanisms: (i) directly influencing the secretion of α-synuclein by enteric nervous system neurons [34], (ii) modulating inflammatory response by increasing Lipopolysaccharide Binding Proteins [4, 20], and (iii) modulating the levels of the intestinal hormone ghrelin [35], which exerts a regulatory role in nigrostriatal dopaminergic function [36, 37], Anyway, the increase in fermentative taxa may also reflect the increase in intestinal permeability.
Strengths, limitations, and future perspectives
The major limitation of the present study is its relatively small sample size. After correcting for multiple comparisons all the association found were no longer significant. Therefore, the present study should be considered exploratory, but also a useful proof-of-concept for a larger observational multicentric study enabling the identification of targeted intestinal microbiota therapies with potential ‘disease-modifying’ effect. Several studies have shown that dietary measures, as well as the administration of prebiotics and probiotics, are able to normalize gut dysbiosis [38–42] and could be useful in PD. Interestingly, the disease-promoting role of various infectious pathogens hypothesized in the pathogenesis of AD has raised considerable interest in possible disease-modifying therapies [29, 43–46]. Nonetheless, a cohort of de novo PD regularly followed up for a period of 3 years is relatively difficult to gather. Indeed, the present study represents the first prospective 3-year follow-up study starting from patients with naïve PD, in whom an intestinal microbiota is not yet altered by possible confounding factors, such as drug therapies. The homogeneous examination of motor and non-motor features at a single tertiary care center enabled also to limit assessment bias.
Conclusions
The current study represents the first prospective 3-year follow-up study exploring the prognostic role of gut microbiota at the onset of PD. The present data, showing an association between microbial composition and clinical progression of the disease, provide the proof-of-concept that intestinal microbiota-directed interventions could be a promising novel therapy for PD, potentially able to modify the disease’s course at least in a subset of susceptible individuals. Although epidemiologic evidence does not support a neuroprotective role of nonsteroidal anti-inflammatory drugs [47], it remains possible that subgroups of patients with different patterns of immune activation could be targeted with selective immunomodulatory strategies (such as those with an abnormal microbial pattern at the onset of PD), or probiotics, or even fecal transplantation (ClinicalTrials.gov Identifier: NCT03876327). Studies on larger populations, in which the effects of multiple established confounding factors can be accounted for, are necessary to validate the results of the present study.
CONFLICT OF INTEREST
RC has received fees for speaking at conferences from Zambon, Bial, UCB, and Lusofarmaco; has received fees for consultancies from Roche, Z-Cube; has received congress sponsorship from Zambon, and Boston Scientific. CF is in the Editorial Board of Parkinson Disease and Neurological Sciences; Coordinator of PRIN 2017, Grant: 2017CY3J3W, received grants and compensations for consulting services from Biogen, DOCPharma, Merck, Roche. All the other authors report no disclosures.
Footnotes
ACKNOWLEDGMENTS
The authors wish to thank all the clinicians of the Parkinson Institute for data acquisition and Jennifer S. Hartwig, MD, for assistance with editing of the manuscript. This study was supported by the “Fondazione Grigioni per il Morbo di Parkinson”, Milano, Italy.
