Abstract
Background:
The impact of concurrent osteoarthritis on mobility and mortality in individuals with Parkinson’s disease is unknown.
Objective:
We sought to understand to what extent osteoarthritis severity influenced mobility across time and how osteoarthritis severity could affect mortality in individuals with Parkinson’s disease.
Methods:
In a retrospective observational longitudinal study, data from the Parkinson’s Foundation Quality Improvement Initiative was analyzed. We included 2,274 persons with Parkinson’s disease. The main outcomes were the effects of osteoarthritis severity on functional mobility and mortality. The Timed Up and Go test measured functional mobility performance. Mortality was measured as the osteoarthritis group effect on survival time in years.
Results:
More individuals with symptomatic osteoarthritis reported at least monthly falls compared to the other groups (14.5% vs. 7.2% without reported osteoarthritis and 8.4% asymptomatic/minimal osteoarthritis, p = 0.0004). The symptomatic group contained significantly more individuals with low functional mobility (TUG≥12 seconds) at baseline (51.5% vs. 29.0% and 36.1%, p < 0.0001). The odds of having low functional mobility for individuals with symptomatic osteoarthritis was 1.63 times compared to those without reported osteoarthritis (p < 0.0004); and was 1.57 times compared to those with asymptomatic/minimal osteoarthritis (p = 0.0026) after controlling pre-specified covariates. Similar results hold at the time of follow-up while changes in functional mobility were not significant across groups, suggesting that osteoarthritis likely does not accelerate the changes in functional mobility across time. Coexisting symptomatic osteoarthritis and Parkinson’s disease seem to additively increase the risk of mortality (p = 0.007).
Conclusion:
Our results highlight the impact and potential additive effects of symptomatic osteoarthritis in persons with Parkinson’s disease.
INTRODUCTION
Gait impairment is a prominent feature of Parkinson’s disease that worsens with time. Persons with Parkinson’s disease exhibit deficits across a variety of gait activities: initiating and terminating, while turning, performing concurrent motor or cognitive tasks, crossing obstacles, and performing gross mobility-related activities of daily living [1–5]. Additionally, these gait impairments and impaired balance control issues place persons with Parkinson’s disease at an increased risk of falling [1, 6]. Thus, it is not surprising that gait function restoration is a primary aim of treatment plans for persons with Parkinson’s disease [7].
Diverse factors contribute to the presence of mobility impairment. While the mechanisms of Parkinson’s disease represent the primary cause of the observed gait impairment, it is likely that factors also impact gait performance. For example, postural instability was only weakly correlated with time to complete the task and self-reported experience of mobility on a health-related quality of life instrument [8]. The low correlation suggests that individual factors, such as comorbidities, may play a contributing factor in mobility performance. To date, the area of comorbid factors in Parkinson’s disease and their influence on mobility and gait are largely unknown.
The coexistence of other disorders alongside Parkinson’s disease can complicate the clinical evaluation, decision-making, and ultimately treatment that clinicians provide to patients. Recent work [9] suggests that arthritis is the most prevalent comorbidity in Parkinson’s disease. Osteoarthritis prominently affects the joints of the hips, knees, and spine, leading to joint inflammation, wear, and stiffness [10, 11]. Neurologically healthy individuals with lower extremity osteoarthritis frequently exhibit slower gait speed, shortened step lengths, decreased single limb support, and decreased mobility compared to healthy controls [12, 13]. Similar to persons with Parkinson’s disease, individuals with osteoarthritis also exhibit an increase in experiencing falls, disease severity, gait dysfunction, and mobility impairment throughout aging [14–16].
Insight into the impact of osteoarthritis on Parkinson’s disease is critical to determine the extra burden that osteoarthritis may contribute to gait impairment. Using data collected during the Parkinson’s Foundation Quality Improvement Initiative (PF-QII), we sought to understand to what extent osteoarthritis symptom severity influenced functional mobility performance in individuals with Parkinson’s disease. Further, understanding the comparative mortality in individuals with Parkinson’s disease who also have osteoarthritis compared to individuals without osteoarthritis may help identify those at higher risk of death and aid in the determination of specific clinical decision-making to enhance patient survival. Therefore, we also sought to quantify the mortality rate among individuals diagnosed with Parkinson’s disease with and without osteoarthritis in relation to disease status, age, sex, disease characteristics, and comorbid conditions. We hypothesized that increased osteoarthritis severity would be related to lower functional mobility performance and an increased likelihood of falling—regardless of age, body mass, sex, and cognition. Further, we hypothesized that the impact of osteoarthritis severity on functional mobility performance would increase over time. Specifically, we expected individuals with more severe osteoarthritis to have a higher rate of functional mobility performance worsening across time compared to individuals with less severe or no osteoarthritis. Finally, we hypothesized that increased osteoarthritis severity would be related to a higher mortality rate, even when controlling for age, body mass, sex, and cognition.
METHODS
We obtained data from a large cohort of individuals with Parkinson’s disease across 21 participating Parkinson’s Foundation Centers of Excellence within the United States and Canada who were part of the PF-QII. This initiative was a multicenter effort that has resulted in a large standardized clinical practice and longitudinal registry. See Okun et al. [17] for a full description of the database. All participants signed an informed consent. We included data from 2,274 persons with Parkinson’s disease with available timed up and go (TUG) and osteoarthritis information. As of May 2016, we had 8,502 patients participated in the PF-QII study. 6,228 participants were excluded for the following reasons: not being diagnosed as Parkinson’s disease (n = 109), missing TUG or osteoarthritis information at baseline (n = 444), no follow-up data (n = 3,211), missing TUG or osteoarthritis information at follow-up (n = 310), Parkinson’s disease certainty <90% (n = 500), known cognitive problems or were missing cognitive testing results (n = 1,624), or non-community dwelling (n = 30). For the mobility analysis, we used data collected at baseline and the first follow-up (range: 9 to 27 months after baseline). For the mortality analysis, we included data from all visits (range: 9 to 84 months after baseline). All participants were taking medication during the evaluations. Demographic information of the participants included in the analysis is displayed in Table 1.
Comparison of baseline characteristics across osteoarthritis (OA) severity groups.
Each participant underwent a neurologic examination by a neurologist. Parkinson’s disease severity was scored using standard Hoehn and Yahr staging [18]. We determined disease duration by the difference of the date of diagnosis of idiopathic Parkinson’s disease to the date of the study exam. The presence and severity of osteoarthritis were self-reported and scored as absent, asymptomatic/minimal, moderate, severe, or very severe. The severity of osteoarthritis was further divided using predictive analyses into bins: zero (absent), one (asymptomatic/minimal), and two (symptomatic = moderate, severe, very severe).
Functional mobility was the primary outcome variable for the first aim. Functional mobility performance was assessed using the TUG test with a hand-held stopwatch, and we categorized individuals’ performance as either “High” or “Low” functional mobility. We defined high functional mobility performance as performing the TUG in 12 seconds or less, and low as over 12 seconds [19]. During the TUG test, individuals were instructed to stand up from a chair, walk forward at a comfortable speed for three meters, then turn around, walk back to the chair and sit down. The evaluation was timed in seconds from the command, “Go,” until the participant made contact sitting back unto the chair. If the individual could not perform the task without using their hands to push off, the participant was allowed to perform the test while using their hands or an assistive device to push off the chair. We designated all individuals who could not complete the task without using one hand or both to push off as “Low” functional mobility.
Frequency of falls (with response categories of None, Rare, Monthly, Weekly and Daily) were recorded via self-reported history over the previous three months from each participant. Fall data were further combined into either “none/rare” or “at least monthly” for the final analysis. During the exam visit, quality of life was evaluated using the Parkinson’s Disease Questionnaire-39 (PDQ-39). The PDQ-39 is a self-report questionnaire that assesses eight dimensions (activities of daily living, functional mobility, emotional well-being, stigma, social support, cognition, communication, and bodily discomfort) of health-related quality of life over the last 30 days. Standardized cognition was measured using immediate and delayed word recall and verbal fluency. For immediate recall, participants were asked to remember five words (face, velvet, church, daisy, red) that were orally provided to them. The individual was instructed to repeat the words aloud, and the number of correct responses was recorded. For delayed recall, following a minimum of 90 seconds after the immediate recall task and after performing a task such as the TUG, participants were asked to list the same five words without any cues. Finally, participants were asked to list as many animals as they could think of in one minute. All correct responses were counted and recorded.
Association of baseline characteristics with osteoarthritis severity
To assess differences in demographics and characteristics of Parkinson’s disease across groups in aim one and aim two, we used Analysis of Variance (ANOVA) for continuous variables. A chi-squared test was used to assess differences among categorical variables.
Mobility
To evaluate differences in TUG performance at baseline and the first follow-up (Low vs. High functional mobility) among osteoarthritis groups, we fitted generalized linear mixed effect models considering center as a random effect while controlling for pre-specified covariates. Sex, age, body mass, physical therapy use, exercise program participation, use of anti-depressant medication, presence of comorbidity, and standardized cognition (immediate five-word recall, verbal fluency, delayed five-word recall) were added as covariates to the analysis based on their known relationship with gait speed or their potential to limit mobility [19, 20]. In addition, we assessed the TUG category change (worse, same, or better) from baseline to the follow-up in osteoarthritis groups using a chi-square test.
Mortality
To investigate the effect of osteoarthritis on mortality, we investigated the osteoarthritis group effect on survival time (in years) while controlling for pre-specified covariates (sex, age, body mass, physical therapy use, exercise program participation, use of anti-depressant medication, presence of comorbidity, and cognition). Kaplan-Meier estimates of survival were calculated for each group. Survival curves were compared with a log-rank test. To determine if osteoarthritis group was an independent predictor of survival time, a Cox proportional hazards regression analysis was conducted, to control for the pre-specified covariates.
RESULTS
Mobility
Association of baseline characteristics with osteoarthritis severity
Demographic and disease characteristics of individuals in the absent, asymptomatic/minimal, and symptomatic osteoarthritis groups are displayed in Table 1. At baseline, 1,324 individuals (58.2%) reported no symptoms of osteoarthritis, 560 (24.6%) reported asymptomatic/minimal osteoarthritis, and 390 (17.2%) reported symptomatic osteoarthritis (moderate to very severe). Further, more individuals with symptomatic osteoarthritis reported at least monthly falls compared to the other groups (14.5% vs. 7.2% without symptomatic osteoarthritis and 8.4% asymptomatic/minimal osteoarthritis, p = 0.0004).
Primary outcomes by osteoarthritis severity over time
Individuals with symptomatic osteoarthritis had worse TUG time (13.3±6.7 s vs. 10.5±4.6 s and 11.4±5.6 s) and PDQ-39 total scores (26.1±14.8 vs. 19.3±13.3 and 21.2±13.6) than the other two groups (without symptomatic osteoarthritis, asymptomatic/minimal osteoarthritis) (all p values < 0.0001). Both TUG and PDQ-39 total scores became worse within each group at the follow-up testing compared to the baseline testing (all p values < 0.001). However, the changes in TUG and PDQ-39 were not statistically significant across the three groups (p = 0.056 and p = 0.215, respectively).
The symptomatic group contained significantly more individuals with low functional mobility (TUG≥12 s) at baseline (51.5% vs. 29.0% and 36.1%, p < 0.0001). Both the asymptomatic/minimal osteoarthritis and symptomatic groups contained significantly more individuals with low functional mobility (TUG≥12 s), at the follow-up (both p < 0.0001) (Fig. 1). The differences among groups in change from baseline to follow-up were not significant (p = 0.458).
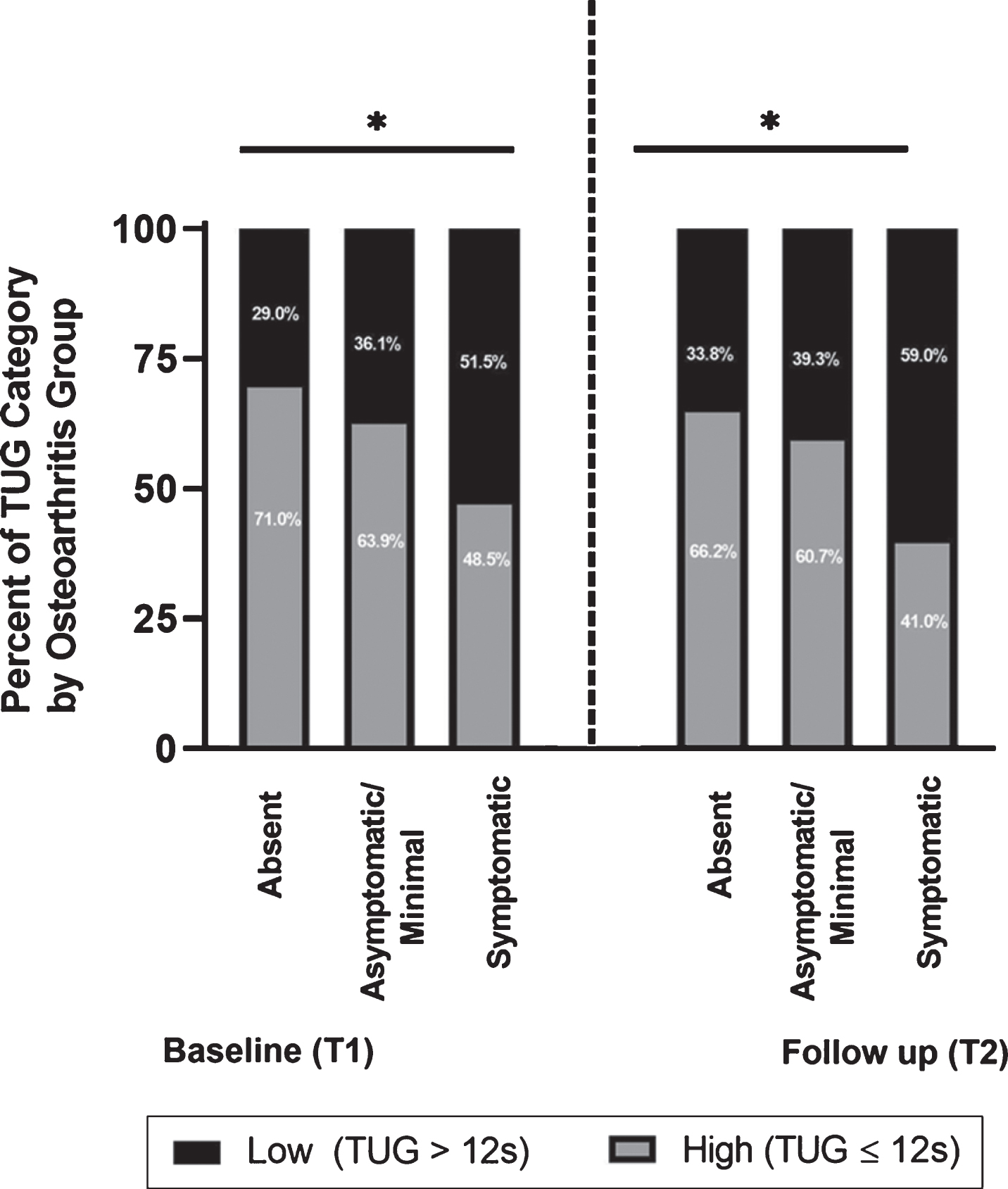
More patients with osteoarthritis were in the lower functional mobility performance category at baseline and follow-up.
We completed a follow-up test to investigate the strategies individuals were using to perform the test to better understand the functional mobility results. For example, an individual could stand up without pushing off, or could push-off of the arms of the chair or may have used an assistive device. During the TUG test, individuals with symptomatic osteoarthritis were less likely to stand up and walk without pushing off than individuals in the other two groups at baseline and follow-up (78.2% vs. 90.5% and 86.8%, all p-values < 0.0001). Additionally, individuals with symptomatic osteoarthritis performed worse over time compared to the other two groups at baseline and follow-up (all p-values < 0.0001).
Evaluation of differences among osteoarthritis groups while controlling for pre-specified covariates
The difference in performance between groups remained statistically significant after covariate adjustment (sex, age, body mass, physical therapy use, exercise program participation, use of anti-depressant medication, presence of comorbidity, and cognition) based on the generalized linear mixed model. Specifically, at the baseline visit, the odds of having low functional mobility for individuals with symptomatic osteoarthritis were 1.63 times compared to those without osteoarthritis (p < 0.0004); and were 1.57 times compared to those with asymptomatic/minimal osteoarthritis (p = 0.0026). Also, female sex, age at first visit, increased body mass, physical therapy use, use of anti-depressant, and presence of comorbidity were all significantly associated with higher odds of low functional mobility performance, while participating in exercise and higher standardized cognition scores in immediate five-word recall and verbal fluency were significantly associated with lower odds of low functional mobility performance (See Table 2 for details). In addition, there was significant variation across centers in low functional mobility performance (p = 0.014). The between group differences were similar at the follow-up, with an odds ratio of 1.91 for those with symptomatic osteoarthritis compared to those without osteoarthritis (p < 0.0001); and an odds ratio of 2.07 for those with symptomatic osteoarthritis compared to those with asymptomatic/minimal osteoarthritis (p < 0.0001). Again, at the follow-up, the random center was significant as well (p = 0.0085), and the same covariate effects remain statistically significant (see Table 2 for details).
Analysis Results from Generalized Linear Mixed Models for Probability of Low Functional Mobility Skill at Baseline and at the Follow-up.
Mortality
Evaluation of survival time among modified osteoarthritis groups while controlling for pre-specified covariates
Survival time between participants without osteoarthritis and those with asymptomatic/minimal osteoarthritis did not significantly differ. Consequently, we combined participants without osteoarthritis and those with asymptomatic/minimal osteoarthritis to create one group for the Cox regression analysis. Survival probability was higher in the absent/asymptomatic/minimal group compared to the symptomatic group (5-year survival probability of 93.2% vs. 87.5%, p = 0.007, Fig. 2). As reported in Table 3, after controlling for the pre-specified covariates, the group effect on survival time was not statistically significant (Hazard ratio of 1.33, p = 0.248).
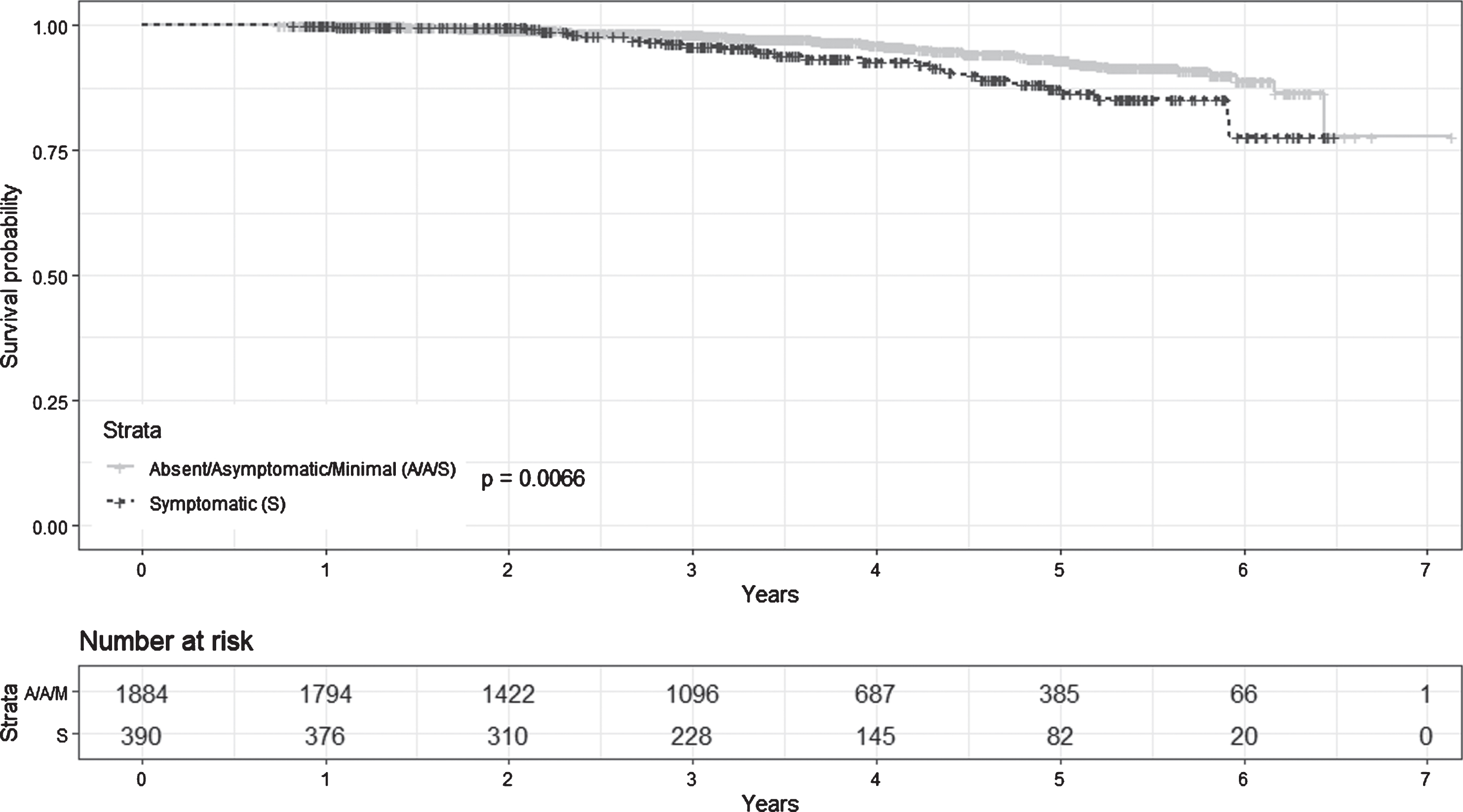
Plots of survival probability estimates by group: patients with absent/asymptomatic/minimal osteoarthritis, and patients with symptomatic osteoarthritis. Censored observations were indicated by ‘+’.
Cox proportional hazards regression analysis results on factors associated with survival time.
DISCUSSION
We investigated the impact of osteoarthritis severity on mobility function and mortality in individuals with Parkinson’s disease. The current study presents three major findings that are central to understanding mobility disability in Parkinson’s disease with osteoarthritis. First, the rate of falls was highest among those with symptomatic osteoarthritis compared to the groups without symptomatic osteoarthritis or who had asymptomatic/minimal osteoarthritis. Second, functional mobility was worse in individuals with symptomatic osteoarthritis when compared to the groups who did not have osteoarthritis or who were asymptomatic. Finally, five-year survival probability was significantly lower in individuals with symptomatic osteoarthritis compared to those who did not have osteoarthritis or who were asymptomatic. Our findings provide new insight and enhance our understanding of mobility and mortality in individuals diagnosed with Parkinson’s disease and osteoarthritis.
Our results suggest that falls, mortality, and functional mobility are worse in individuals with co-occurring Parkinson’s disease and osteoarthritis. Our analysis revealed that 18.3% of individuals with symptomatic osteoarthritis and Parkinson’s disease fall at least once a month, compared to 11% of individuals with Parkinson’s disease and without symptoms of osteoarthritis. Falls occur at a frequency of 60.5% in individuals with Parkinson’s disease living in the community (one fall per year) [21]. Furthermore, individuals with osteoarthritis fall 25% more than age-matched healthy controls, and 50% of people with osteoarthritis experience at least one fall a year [22, 23]. Mounting evidence indicates that falls are a severe impairment in Parkinson’s disease and osteoarthritis, and the results of this study suggest this is particularly concerning when the two diseases coexist [1, 21].
The pharmacological and non-pharmacological therapies used to treat Parkinson’s disease are not expected to be effective in addressing the mechanism/symptoms of osteoarthritis that contribute to the increased risk for falls, low functional mobility, and high mortality rate. Likewise, therapies, (such as non-steroidal anti-inflammatory drugs), used to target osteoarthritis may also be ineffective treatments for addressing the symptoms of Parkinson’s disease. However, it is possible that an individual who has both Parkinson’s disease and osteoarthritis must also overcome the resulting interaction of the two diseases. Our study did not investigate the effects of treatment for Parkinson’s disease and osteoarthritis. Further research should examine how collectively treating both Parkinson’s disease and osteoarthritis may influence falling, mobility, and mortality among individuals. This work could shed light on types of therapy (both pharmacological and non-pharmacological) that may lead to improvements in mobility and reduced fall risk in individuals with Parkinson’s disease and osteoarthritis. Additional work is also required to understand if independent treatment of both Parkinson’s disease and osteoarthritis would prove enough; or if therapy should focus on the interaction of the two diseases.
Current practice for diagnosis of Parkinson’s disease relies on patients’ symptoms, neurologic examination results, and response to medication (carbidopa-levodopa) over time to confirm the diagnosis of disease [24]. The Hoehn and Yahr rating scale is a commonly used evaluation tool that emphasizes mobility to assess the impact of Parkinson’s disease [18]. The Hoehn and Yahr describes simple staging from zero to five that estimates clinical function and assesses functional deficits and objective signs (impairment) of Parkinson’s disease. It is worth noting that if osteoarthritis is present, its symptoms (e.g., stiffness and slowness in gait) likely resemble symptoms of Parkinson’s disease. Therefore, a lack of concern for comorbid factors (e.g., osteoarthritis) may lead to inflation of the Hoehn and Yahr score, resulting in a rating of a more severe disease state than the true Parkinson’s disease severity. Indeed, we observed more severe Parkinson’s disease ratings in the symptomatic osteoarthritis group compared to the asymptomatic and minimal groups. While we cannot ascertain to what extent osteoarthritis inflated the disease scores in our study, it is possible that these scores were higher because of osteoarthritis symptoms rather than just degradation due to Parkinson’s disease. Future work should investigate the impact of osteoarthritis on diagnosis and disease severity of Parkinson’s disease.
This study was not without limitations. Our primary measure of mobility, the TUG, was scored using a hand held stopwatch, rather than an automated timer. Further, scoring of osteoarthritis was based upon self-report of the individual. This limitation stemmed from the data collection approach of the PF-QII. However, self-reported arthritis has been shown to have high reliability, sensitivity (84%) and moderate specificity (71%) [25]. Because symptoms of osteoarthritis could be confounded by Parkinson’s disease signs, future work should require that all participants undergo a radiographic examination to determine the presence and severity of osteoarthritis. Finally, including an additional group of individuals who were age-matched and only diagnosed with osteoarthritis could provide further information regarding mobility and mortality.
Conclusions
This study provides evidence that individuals with Parkinson’s disease and symptomatic osteoarthritis have an increased risk of falling, mobility impairment, and mortality. These results are present even when controlling for sex, age, body mass, and cognition. Future work should aim to understand how comorbidities such as osteoarthritis influence the diagnosis and treatment of individuals with Parkinson’s disease. Additionally, individuals with coexisting Parkinson’s disease and osteoarthritis may have different risk profiles than individuals with Parkinson’s disease alone. These results highlight the need for an interdisciplinary healthcare approach to disease management. Further studies that lead to better prognoses and early identification of individuals at risk for developing both Parkinson’s disease and osteoarthritis may help guide the field.
FINANCIAL DISCLOSURE/CONFLICT OF INTEREST RELATED TO MANUSCRIPT
Dr. Wu has received research funding from the Parkinson’s Foundation. Dr. Okun serves as a consultant for the Parkinson’s Foundation and has received research grants from the Parkinson’s Foundation.
FINANCIAL DISCLOSURES
JAR: Dr. Roper has received research funding from the Department of Defense.
ACS: Dr. Schmitt has received research funding from the American Society of Biomechanics and the Elaine C Pidgeon Neurology Research Fund.
YH: none.
SW: Dr. Wu has received research funding from the Parkinson’s Foundation.
HG: none.
PS: Dr. Schmidt is an advisor on two studies conducted by Acadia Pharmaceuticals, Inc., for which he is not compensated. He is an advisor to and receives a grant from the Marlene and Paolo Fresco Foundation, in Fiesole, Italy. He is on the board of directors for the Huntington’s Study Group, the Davis Phinney Foundation, and Night Nurse Triage, Inc.
MSO: Dr. Okun serves as a consultant for the Parkinson’s Foundation, and has received research grants from NIH, Parkinson’s Foundation, the Michael J. Fox Foundation, the Parkinson Alliance, Smallwood Foundation, the Bachmann-Strauss Foundation, the Tourette Syndrome Association, and the UF Foundation. Dr. Okun’s DBS research is supported by: NIH R01 NR014852 and R01NS096008. Dr. Okun is PI of the NIH R25NS108939 Training Grant. Dr. Okun has received royalties for publications with Demos, Manson, Amazon, Smashwords, Books4Patients, Perseus, Robert Rose, Oxford and Cambridge (movement disorders books). Dr. Okun is an associate editor for New England Journal of Medicine Journal Watch Neurology. Dr. Okun has participated in CME and educational activities on movement disorders sponsored by the Academy for Healthcare Learning, PeerView, Prime, QuantiaMD, WebMD/Medscape, Medicus, MedNet, Einstein, MedNet, Henry Stewart, American Academy of Neurology, Movement Disorders Society and by Vanderbilt University. The institution and not Dr. Okun receives grants from Medtronic, Abbvie, Boston Scientific, Abbott and Allergan and the PI has no financial interest in these grants. Dr. Okun has participated as a site PI and/or co-I for several NIH, foundation, and industry sponsored trials over the years but has not received honoraria. Research projects at the University of Florida receive device and drug donations.
CJH: Dr. Hass has received research funding from NIH and the Michael J. Fox Foundation.
Footnotes
ACKNOWLEDGMENTS
Editing assistance was provided by Leslie Poston of A Draft Supreme, LLC.
Samuel Wu had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
