Abstract
Background:
REM behavior disorder (RBD) can occur in the context of neurodegenerative alpha-synucleinopathies, such as Parkinson’s disease (PD). PD patients with RBD (PD-pRBD) represent more severe symptoms and signs compared with those without RBD (PD-nRBD). On another note, autonomic dysfunction in PD patients is categorized as one of the most prominent non-motor symptoms and has been lately the field of interest in research.
Objective:
In the current study, we longitudinally studied autonomic dysfunction in PD-pRBD and PD-nRBD groups.
Method:
This study was conducted on 420 drug-naïve PD patients selected from the Parkinson’s Progression Markers Initiative database. The RBD Screening Questionnaire was used to define the presence of probable RBD. SCOPA-AUT was used to assess autonomic dysfunction. Additionally, dopamine transporter deficits on [123I] FP-CIT SPECT imaging was performed for all of the patients.
Results:
Out of 420 PD patients, 158 individuals (37.6%) were considered to have probable RBD (PD-pRBD) and others without RBD (PD-nRBD). Except for pupillomotor function, all the autonomic symptoms were significantly more severe in PD-pRBD group. In PD-nRBD group, caudate striatal binding ratio was negatively correlated with SCOPA-AUT scores, while no significant correlation was observed in PD-pRBD group. Finally, there was a significant difference considering the longitudinal changes of SCOPA-AUT total between PD-pRBD and PD-nRBD groups, suggesting a more severe autonomic decline in PD-pRBD patients.
Conclusion:
Our results indicate that PD-pRBD patients have more severe autonomic dysfunction. These results support the theory that PD patients can be categorized based on the clinical presentation, possibly representing differences in the disease pathophysiology.
INTRODUCTION
Parkinson’s disease (PD) is mostly recognized by motor impairments, including rigidity, bradykinesia, resting tremor and postural instability [1]. How-ever, there is also a broad spectrum of non-motor symptoms (NMS), including cognitive, sleep, and autonomic symptoms. For instance, rapid eye movement (REM) sleep behavior disorder (RBD) is observed in about 42% of PD patients [2], albeit it has an estimated prevalence of 0.5% in the general population [3]. Dysautonomia is also a common phenomenon in PD patients as 30–40% of the patients complain of relevant symptoms [4]. Though, there is not much evidence concerning the association of RBD and dysautonomic symptoms in PD.
RBD has characteristic clinical features, including the absence of atonia, repeated episodes of vocalization, and abnormal behavior during REM sleep [5]. RBD can occur either as an isolated pheno-menon or because of neurodegenerative alpha-synuc-leinopathies, such as PD, as the underlying cause [6, 7]. However, almost 80% of patients with isolated RBD (iRBD) also develop parkinsonism after an average of 14 years [8]. Numerous studies have confirmed the alteration in the autonomic function of patients with iRBD [9–13]. On the other hand, there is little but growing evidence that PD patients with RBD generally experience higher grades of motor and non-motor symptoms in comparison with those without RBD [14–17]. This issue has been making RBD as an appropriate clinical feature for subtyping a disease as heterogeneous as PD in the purpose of achieving a better estimation on practical and research outcomes of the disease. However, the exact pathophysiology remains unclear and needs further attention.
Autonomic dysfunction in PD patients is one of the main NMS and has been lately a field of interest in this area. Autonomic symptoms have a broad range, including gastrointestinal, cardiovascular, urogenital, thermoregulatory and pupillomotor abnormalities [18], and the Scales for Outcomes in Parkinson’s Disease–Autonomic (SCOPA-AUT) questionnaire could play an important role as a tool for a comprehensive assessment of these symptoms in PD patients. Despite the common incidence of autonomic dysfunction in PD patients, the treatment options have remained limited [19]. Thereby, there is an ongoing effort to discover the etiology of autonomic disturbance in PD patients. On this manner, few studies have explored the association between the SCOPA-AUT subscales and striatal dopaminergic activity and reported conflicting findings [20, 21].
Several studies have addressed a direct relationship between the severity of dysautonomic manifestations and the presence of RBD in PD patients. Concerning the link between autonomic symptoms and RBD in PD, it is hypothesized that scattering of REM-modulator neurons near the central auto-nomic network (CAN) might be responsible for the concurrence of autonomic dysfunctions and sleep disorders, such as RBD [22]. However, as far as we are concerned, previous studies could not certainly determine the influence of RBD on the change of autonomic symptoms in PD patients across time; therefore, we designed a longitudinal study on autonomic dysfunction in PD patients with RBD (PD-pRBD) compared with those without RBD (PD-nRBD). We aimed to investigate whether the auto-nomic dysfunction in PD patients is associated with the striatal dopaminergic activity to provide novel insight into the involved pathophysiology.
METHODS
Participants
This study was conducted on 420 drug-naïve PD patients included in the Parkinson’s Progression Markers Initiative (PPMI) database (ppmi-info.org/data). Patients presented with two out of three symp-toms (rigidity, resting tremor and bradykinesia) or asymmetric resting tremor/bradykinesia at the scr-eening visit. Dopamine transporter deficits on [123I] FP-CIT SPECT imaging was used to confirm the diagnosis for all patients.
The RBD Screening Questionnaire (RBDSQ) was used to define the presence of probable RBD. RBDSQ has been validated and translated in several languages, showing to be an appropriate alternative approach to video-polysomnography due to its high sensitivity and time-effectiveness [23–26]. This questionnaire consists of 10 items evaluating sleep behavior. Patients are asked to answer “yes” or “no” to each question. A patient with an RBDSQ score of five or above is considered to have probable RBD.
Clinical evaluation
Hoehn and Yahr (H&Y) scale and Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) Part III were used to assess motor symptoms of participants. Montreal Cognitive Assessment (MoCA) and the University of Pennsylvania Smell Identification Test (UPSIT) were used to evaluate global cognitive status and olfactory dysfunction, respectively. SCOPA-AUT was used to assess autonomic dysfunction at baseline and three further visits with intervals of one year. SCOPA-AUT was introduced by Visser et al. [27] in 2004 and is currently the most widely used questionnaire for this issue in PD patients. SCOPA-AUT consists of 25 questions exploring clinical autonomic symptoms in six categories, including gastrointestinal (seven items), urinary (six items), cardiovascular (three items), thermoregulatory (four items), pupillomotor (one item) and sexual function (two items for women and three items for men) [27]. Results from baseline visit, year one, year two and year three were used to assess longitudinal changes in SCOPA-AUT scores.
Dopaminergic imaging
After administrating an injection of 185 MBq [123I] FP-CIT (4±0.5 hours), SPECT images were taken from participants. Subsequently, the scans were analyzed using the imaging technical operations manual (http://ppmi-info.org/). Briefly, SPECT images were spatially normalized using an Ioflupane template. Eight of the most prominent axial slices that contained the striatum were gathered, and a standardized volume of interest (VOI) template was applied. Using the occipital region as the reference tissue, VOI analyses were done on the left and right putamen and caudate. Using the formula: (striatal region VOI count density)/(occipital cortex count density)–1, striatal binding ratios (SBRs) of left and right putamen and caudate were calculated. Considering the motor dominancy, contralateral SBRs were calculated and compared between study groups.
Statistical analysis
IBM SPSS Statistics for Mac, version 22 (IBM Corp., Armonk, N.Y., USA) and RStudio (R version: R-3.6.1) were used for statistical analysis. Probability graphics and the Shapiro-Wilk test were used to check the compliance of variables with the normal distribution. Student’s t-test was used to evaluate numeric variables across PD groups. Pearson’s chi-square was used to assess nominal variables across two subgroups. Spearman’s correlation was used to test the association between SCOPA-AUT scores and SBR results, and corrplot package in R was used for the graphs [28].
To further describe the significant association between RBD and longitudinal autonomic dysfunction, a mixed between-within subjects analysis of variance was conducted. To eliminate the effect of confounding variables, the model was adjusted for age, gender, education and UPSIT score. Finally, p values less than 0.05 were considered statistically significant.
RESULTS
Clinical characteristics
Out of 420 PD patients, 158 individuals (37.6%) were considered to have probable RBD (PD-pRBD) and others without RBD (PD-nRBD). PD patients with RBD and those without were not different considering age, gender, disease duration, education, UPDRS part III score, H&Y stage and MoCA score (Table 1). PD-pRBD patients exhibited a substantially greater loss of smell function considering UPSIT test (p = 0.048). No significant differences were observed in [123I] FP-CIT uptake in the caudate (p = 0.911) or putamen (p = 0.553), between PD-pRBD and PD-nRBD groups. However, SCOPA-AUT total score and almost all its subscores were significantly greater in PD patients with RBD compared with PD-nRBD group (Table 2). Except for sexuality and pupillomotor subscores (p = 0.008 and 0.148, respectively), p values less than 0.001 were observed for SCOPA-AUT subscores and total score.
Baseline demographic variables and SBRs in PD patients
SCOPA-AUT, Scales for Outcomes in Parkinson’s Disease-Autonomic; PD, Parkinson’s disease; SD, standard deviation; RBD, rapid eye movement sleep; PD-pRBD, Parkinson’s disease patients with probable RBD, PD-nRBD, Parkinson’s disease patients without probable RBD. *Calculated by independent t-test.
SCOPA-AUT and its sub scores in PD patients with and without probable BD
SCOPA-AUT, Scales for Outcomes in Parkinson’s Disease-Autonomic; PD, Parkinson’s disease; SD, standard deviation; RBD, rapid eye movement sleep; PD-pRBD, Parkinson’s disease patients with probable RBD, PD-nRBD, Parkinson’s disease patients without probable RBD. *Calculated using chi-square test.
SCOPA-AUT and DAT scan correlation
Considering the association between SCOPA-AUT scores and DAT scan results, no significant correlation was observed in PD-pRBD group (Fig. 1), where all p values were greater than 0.05 and all correlation coefficients were less than 0.15. However, significant associations were observed in PD-nRBD group (Fig. 2). Caudate SBR was negatively correlated with SCOPA-AUT total score and gastrointestinal, urinary, pupillomotor and sexuality subscores (p values ≤0.001; correlation coefficients = –0.223, –0.204, –0.131, –0.202 and –0.246, respectively). Considering putamen SBR, significant correlations were observed with SCOPA-AUT total score, urinary and sexuality subscores (p values < 0.05; correlation coefficients = –0.144, –0.127 and –0.142, respectively).
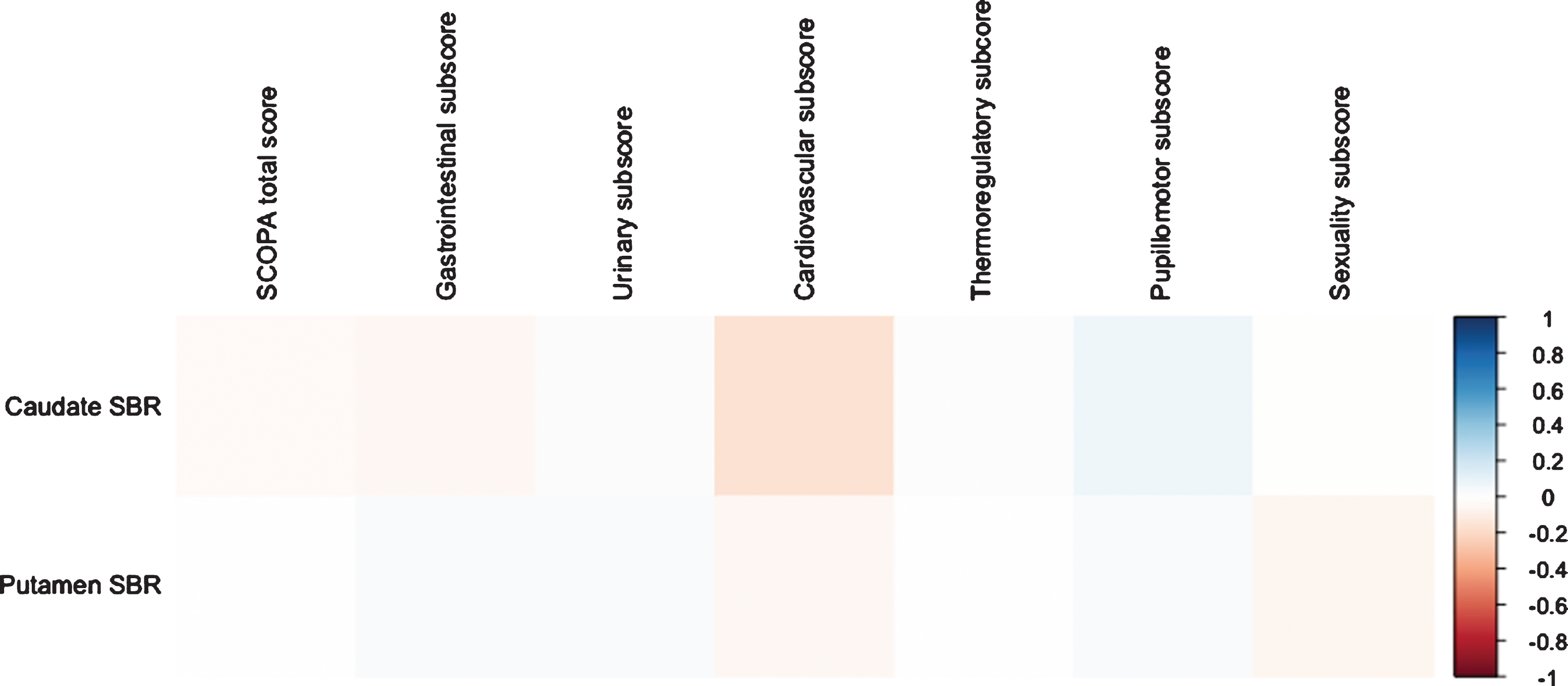
Correlation plot of SCOPA-AUT scores and DAT scan results in PD-pRBD group.
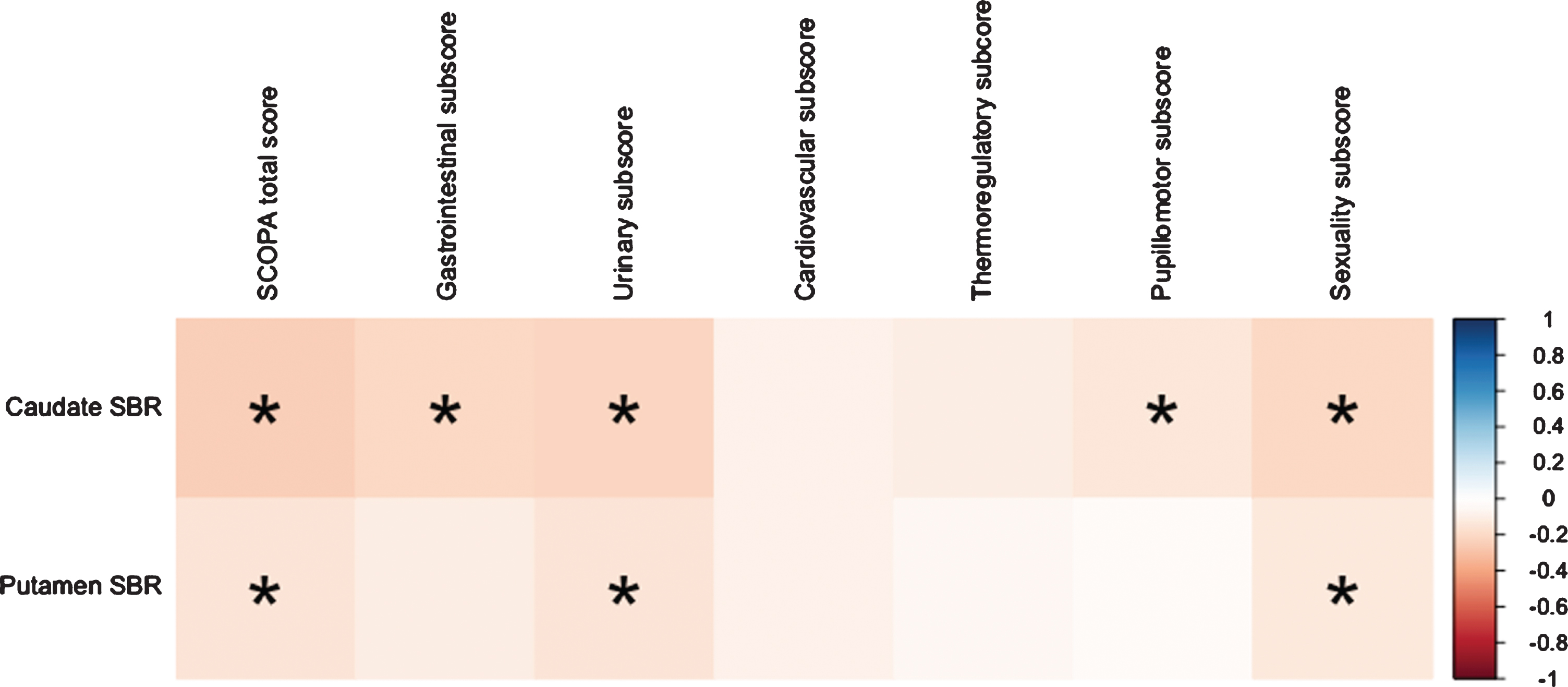
Correlation plot of SCOPA-AUT scores and DAT scan results in PD-nRBD group.
Longitudinal changes of SCOPA-AUT score
A mixed between-within subjects analysis of variance was conducted to assess the impact of RBD (PD-nRBD vs. PD-pRBD) on the total SCOPA-AUT score across four time periods (baseline, year-1, year-2 and year-3). There was a substantial main effect for time (p values less than 0.001), with both groups showing an increase in total SCOPA-AUT score across the four time periods (Fig. 3). The main effect comparing the presence and absence of RBD in PD patients was significant (z value = 6.03, p value < 0.001), suggesting greater risk of SCOPA-AUT score worsening in PD-pRBD patients.
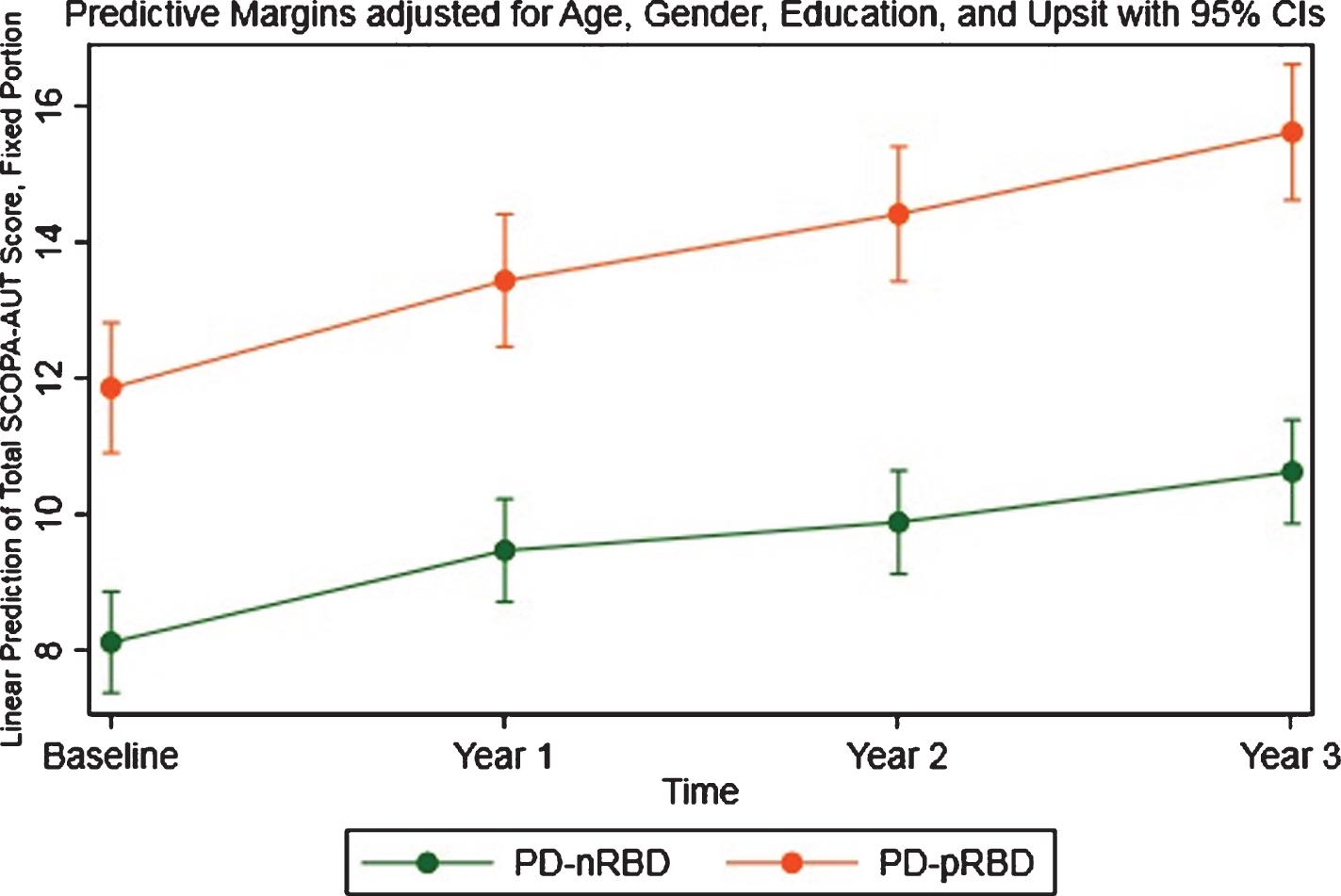
Longitudinal changes of SCOPA-AUT total score in PD-pRBD and PD-nRBD groups.
DISCUSSION
A wide range of sleep disturbances is evident in patients with PD, including insomnia, RBD, sleep disordered breathing, excessive daytime sleepiness and fatigue, vivid dreaming, restless leg syndrome, periodic leg movements of sleep, and sleep attacks [29]. RBD shares some similarities with other sleep disturbances and therefore, RBDSQ was used in the current study to distinguish those who have probable RBD from patients with general sleep disturbances. In the next step, we provided evidence about how autonomic features differ in drug-naïve PD-pRBD and PD-nRBD patients, and how they are correlated with striatal dopamine reduction. Although a recent study evaluated the autonomic dysfunction association with RBD in PD patients, and discovered no significant correlation [30], our results showed that except for pupillomotor function, all of the autonomic symptoms are significantly more severe in PD-pRBD group. We believe this conflict could be justified according to the presence of a considerably higher number of patients in our study, specific inclusion of drug-naïve patients, and the use of adjustments for confounding factors in our analysis. Using DAT scan, we identified that the level of dopamine activity in the caudate and putamen are negatively associated with autonomic dysfunctions, considering the total score, urinary gastrointestinal and sexuality subscores in PD-nRBD group. However, no significant correlation between SBR and autonomic features was observed in PD-pRBD group. Finally, there was a significant difference considering the longitudinal changes of SCOPA-AUT total score between PD-pRBD and PD-nRBD groups, suggesting faster progression of autonomic symptoms in the presence of RBD.
Neural activity in the brainstem seems to be responsible for controlling REM sleep. In detail, REM-on glutamatergic neurons are located in the periventricular grey matter and sublaterodorsal nucleus while REM-off neurons could be found in the in the ventrolateral part of the periaqueductal grey matter, the lateral pontine tegmentum and the pedunculopontine nucleus [31, 32]. Although the exact pathophysiology of RBD remains unclear, there have been theories regarding the significant association between involvements of the aforementioned regions and RBD presentation [33]. Moreover, autopsy studies of RBD associated with PD or Dementia with Lewy bodies (DLB) have reported a great accumulation of synuclein in the subcortical structures and brainstem nuclei [34–36].
In addition to the sleep neuronal circuits, the brainstem encompasses the CAN that plays the main role in the control of autonomic functions. The CAN is composed of several interconnected regions, including the periacqueductal grey in the midbrain, the parabrachial nucleus of the pons and several other regions in the medulla (e.g., the dorsal motor vagal nucleus and the nucleus ambiguous) [37]. Involvement of the CAN components has been extensively observed at early stages of synuclein-related neurodegeneration, justifying the autonomic dysfunction in PD course [16]. Besides, involved brain areas in patients with RBD have considerable anatomical overlap with the CAN, which might explain the concurrence of sleep problems and autonomic dysfunction.
There have been a remarkable number of studies evaluating the correlation between the presence of iRBD and autonomic dysfunction. The investigation was first started in 1996, when Ferini-Strambi and colleagues discovered an altered autonomic function of the cardiovascular system during wakefulness in iRBD subjects [38]. Over time, various studies have supported the dysfunction of the cardiovascular system [11, 39–41]. The main limitation of these studies stems from their outcome of interest since they have mainly focused on a single symptom or subdivision of the autonomic nervous system. Several other studies have resolved this limitation, using questionnaires, such as SCOPA-AUT to assess different aspects of autonomic function in iRBD patients. A prospective case-control cohort had assessed the difference of autonomic dysfunction between 318 iRBD patients and 318 age- and gender- matched controls through SCOPA-AUT and found more prominent urinary, gastrointestinal, and cardiovascular symptoms in iRBD patients compared with the control participants [27], which is relatively in accordance with the findings of another study, carried on by Ferini-Strambi et al. [9]. Of note, data on autonomic dysfunction in iRBD patients is not completely homogenous; Aguirre-Mardones et al. found no significant statistical differences compared with healthy controls in domains of the SCOPA-AUT, despite observing dysautonomic symptoms in examination [10]. Overall, most of the mentioned studies confirm the role of RBD presence in developing autonomic dysfunction.
A large body of evidence addresses autonomic dysfunction in PD patients [4, 43]. Nonetheless, the heterogeneous pattern of this kind of symptoms in PD has recently directed the researchers to examine these symptoms in distinct subtypes of the disease. As mentioned above, RBD is tightly associated with autonomic dysfunction and therefore, subtyping PD patients based on the presence of RBD might clarify the exact position of the autonomic system in PD. However, the existing literature is inconsistent and the number of studies in this field is limited. In a cross-sectional study, Leclair-Visonneau et al. compared the severity of autonomic symptoms between PD-pRBD and PD-nRBD patients through SCOPA-AUT and reported no significant difference [30], while Pagano et al. indicated significantly higher grades of autonomic dysfunction in PD-pRBD group [44]. On this manner, our results suggested those PD patients with RBD comorbidity exhibit faster progression of autonomic symptoms over long-term. After adjusting for covariates including age, gender, education and UPSIT score, it was indicated that more severe autonomic decline is observed in PD-pRBD patients. This might be due justified by the connected circuits responsible for these commodities in PD patients, which were discussed previously.
In this regard, previous reports have evaluated different phenotypes of PD. For instance, excessive daytime sleepiness in PD patients was associated with higher depression and autonomic dysfunction scores [45]. Other studies have suggested that the phenotype with sleep disturbances might be associated with some of motor problems such as freezing of gait [46]. However, baseline motor and cognitive performance (assessed by UPDRS part III and MOCA scores) in our patients were not significantly different across PD-nRBD and PD-pRBD groups. Considering the majority of previous studies and results of the current study, it can be suggested that phenotype with sleep disturbances might be more associated with comorbidities such as autonomic dysfunction.
Several previous investigations have evaluated the correlation between SCOPA-AUT scores and SBR in PD patients to realize whether more advanced autonomic dysfunction is related to lower striatal dopamine activity. Winge and colleagues indicated that the severity of bladder dysfunction is correlated with the relative degeneration of the caudate nucleus [47]. In a recent study on PPMI database, Kim and colleagues showed that striatal dopaminergic activity is associated with urinary symptoms, but not with other subscales of the SCOPA-AUT questionnaire [21]. Contrary to Winge’s study, an association was found between urinary symptoms and SBR of the putamen, but not the caudate nucleus. Moreover, Hinkle and colleagues discovered a negative relationship between caudate SBR and gastrointestinal, but not urinary autonomic function [20]. However, by categorizing PD patients based on the concurrence of RBD, we observed significant associations between striatal dopamine uptake and autonomic dysfunction in the PD-nRBD group. Therefore, the discrepancy among previous studies could be justified by the heterogeneity of PD patients based on their underlying disease pathophysiology.
Braak et al. suggested that an ascending pattern of α-synuclein spread causes neural damage in PD, from peripheral neurons to as far as brainstem, midbrain and finally higher cortical structures [48], explaining the cognitive dysfunction that happens in late stages of the disease course. This hypothesis is supported by several studies suggesting a trans-synaptic mechanism for PD progression from the gastrointestinal system to the central nervous system (CNS). Pan-Montojo et al. observed that intragastric administration of low dose rotenone would lead to the accumulation of α-synuclein in the dorsal motor nucleus of the vagus, spinal cord and substantia nigra of wild mice [49]. In another study by Ulusoy and colleagues, it was indicated that the injection of adeno-associated viral vectors in the vagus nerve of rats would cause α-synuclein accumulation in medullary neurons [50]. In contrast, some studies have shown that ascending aggregation of Lewy bodies is not observed in up to 50 % of patients with PD [51, 52]. Eventually, an alternative theory was proposed as “threshold theory” in 2016. This theory explains that disease pathologies simultaneously hit different neurons and regions in the CNS and peripheral nervous system, and the disease progression starts in regions with lower functional reserve, which are not able to retain network compensation [53]. The theory is supported by studies indicating a widespread neural expression of α-synuclein [54, 55]. Putting these claims together, we hypothesize that both theories might explain the disease course, depending on the subset of patients. This could justify SBR being correlated with autonomic dysfunction only in the PD-nRBD group as the presence of RBD could represent different pathophysiology.
Several limitations should be noted considering the results of the current study. first, we have categorized our patients based on having probable RBD using RBDSQ. Although RBDSQ is a validated questionnaire and showed decent performance in the original study, it is not considered as the gold standard and later studies have raised doubts about its sensitivity and specificity [56]. Since video-polysomnography is not performed in the PPMI cohort, we were not able to categorize our patients based on definite RBD diagnosis. This classification bias might be one of the reasons behind the findings different with corresponding results of other studies such as the lower prevalence of RBD in our study, and future studies using video-polysomnography could confirm the results of the current study. Second, although all PD patients included in our study were drug-naïve, longitudinal analysis was not adjusted for taken medications and co-morbidities due to issues with heterogeneity of the data.
In conclusion, our results indicate that the presence of RBD in PD patients gives rise to more serious autonomic dysfunction. These results support the hypothesis that PD patients can be categorized based on their clinical manifestations, possibly representing differences in the disease pathophysiology. Further longitudinal studies evaluating the pathophysiology of the autonomic nervous system in PD patients might confirm these hypotheses.
CONFLICT OF INTEREST
Professor Zahra Vahabi received funding in form of research grant from Tehran University of Medical Sciences for her project in the Department of Geriatric Medicine.
Other authors have nothing to disclose.
