Abstract
Background:
Treatment of patients with late-stage parkinsonism is often sub-optimal.
Objective:
To test the effectiveness of recommendations by a movement disorder specialist with expertise in late-stage parkinsonism.
Methods:
Ninety-one patients with late-stage parkinsonism considered undertreated were included in apragmatic a pragmatic multi-center randomized-controlled trial with six-month follow-up. The intervention group received a letter with treatment recommendations to their primary clinician based on an extensive clinical assessment. Controls received care as usual. The primary outcome was the Unified Parkinson Disease Rating Scale (UPDRS)part-II (Activities of Daily Living). Other outcomes included quality-of-life (PDQ-8), mental health (UPDRS-I), motor function (UPDRS-III), treatment complications (UPDRS-IV), cognition (Mini-mental-state-examination), non-motor symptoms (Non-Motor-Symptoms-scale), health status (EQ-5D-5L) and levodopa-equivalent-daily-dose (LEDD). We also assessed adherence to recommendations. In addition to intention-to-treat analyses, a per-protocol analysis was conducted.
Results:
Sample size calculation required 288 patients, but only 91 patients could be included. Treating physicians followed recommendations fully in 16 (28%) and partially in 21 (36%) patients. The intention-to-treat analysis showed no difference in primary outcome (between-group difference = –1.2, p = 0.45), but there was greater improvement for PDQ-8 in the intervention group (between-group difference = –3.7, p = 0.02). The per-protocol analysis confirmed these findings, and showed less deterioration in UPDRS-part I, greater improvement on UPDRS-total score and greater increase in LEDD in the intervention group.
Conclusions:
The findings suggest that therapeutic gains may be reached even in this vulnerable group of patients with late-stage parkinsonism, but also emphasize that specialist recommendations need to be accompanied by better strategies to implement these to further improve outcomes.
Keywords
INTRODUCTION
Late-stage parkinsonism (LSP) is characterized by a high burden of motor and non-motor symptoms, resulting independence in daily functioning, low quality of life and, ultimately, an increased risk of institutionalization and death [1, 2]. Studies suggest that treatment in LSP is often suboptimal [1, 4]. In a Dutch nursing home population, 44% of patients reported to be “off” most of the day [4] and received a seemingly too low dose of dopaminergic treatment. Also, patients in this study perceived their professional care givers as having insufficient knowledge of Parkinson’s disease (PD) [3].
As treatment in LSP is more complex than in earlier disease stages [1], movement disorders experts are potentially well equipped to address this complexity as they frequently treat patients with PD. However, LSP-patients are underrepresented in their patient population, as they are often unable to travel for appointments with a neurologist or for hospital-based assessments of their condition. The feasibility of implementation and effectiveness of a movement disorder expert’s advice in this population, communicated to the patient’s primary physician in a letter, has not been tested.
The primary aim of this European pragmatic study was to evaluate the effect of recommendations made by movement disorder experts in a population of undertreated LSP-patients on clinically relevant outcomes measures, such as activities of daily living, motor symptoms, non-motor symptoms and quality of life.
METHODS
Study design
This study is part of the Care of Late-Stage Parkinsonism-study (CLaSP-study) [5]. To assess the effect of the intervention, we designed a multi-center pragmatic parallel randomized controlled trial that allowed us to observe the effectiveness embedded within existing clinical care routines in four European countries (UK, France, Sweden and the Netherlands). Centres in two other countries, who participated in the CLaSP-study, did not participate in this trial due to organizational and regulatory issues. To establish an estimation of impact, the study had a pragmatic design and was executed in routine clinical practice [6]. The study recruitment was set up to be as inclusive as possible. Allocation to the intervention versus control group followed a 3:1 ratio to ensure that as many patients as possible could potentially benefit from the intervention. The study consisted of a baseline measurement following inclusion of the patient in the study, and a follow-up measurement after six months.
Study sample
Patients recruitment took place from January 2015 until December 2017. Last follow-up measurement was June 2018. Undertreated LSP-patients formed the target population. As these patients normally do not access expert research centres, recruitment was set-up to include care-pathways outside of routine recruitment pathways like expert clinics. Care settings included in the recruitment were nursing homes (France, Sweden, the Netherlands), general practices (UK), non-research centre hospitals (Sweden, the Netherlands), patient-advocate organizations (UK) and PD patient registries (Sweden). Patients with a disease duration of 7 years or longer were invited for participation if they either had disease stage Hoehn and Yahr stage ≥4 or a Schwab and England-score≤50%. This allowed for inclusion of patients with disability not only due to motor but also non-motor problems, such as dementia, neuropsychiatric symptoms and autonomic dysfunction [7]. Under-treatment was defined by the presence of any insufficiently treated symptoms or problems (for full set of possible symptoms and problems see Table 1). PD and atypical parkinsonian disorders were diagnosed using established clinical criteria [8–10]. Patients with atypical parkinsonism were purposely not excluded as their care needs are likely comparable to those of patients with late-stage PD [11–13]. Exclusion criteria were:1. a diagnosis of normal pressure hydrocephalus or drug-induced parkinsonism (except if parkinsonism persisted after discontinuation of the causative drug for at least 6 months), 2. dementia prior to or at time of parkinsonism diagnosis; 3. having seen a movement disorder specialist recently (≤4 months); and 4. the patient was unable to comply with changes to treatments (for example unable to attend physiotherapy in their region).
Definition of undertreated LSP patients
Intervention
Our intervention consisted of a letter with specific recommendations to optimize treatment and care, formulated by a movement disorder expert, based on a comprehensive clinical assessment by the researchers, as part of the CLaSP protocol [5]. The researchers assessed the symptoms and discussed these with the movement disorder expert, who drafted the letter. Each study centre assigned one expert to write this letter. To align the recommendations between the centres, the experts used an extensive, predesigned study guideline. During a face-to-face meeting, the group of movement disorder experts in the study developed this consensus-based recommendation guideline based on combined treatment recommendations of multiple European and International guidelines [14–19] (see Supplementary Material). The guideline covered four distinct domains: 1. dopaminergic treatment, 2. non-dopaminergic treatment, 3. mental health medications, and 4. allied health care, social services and nursing care.
For each patient, the letter with recommendations was sent to the physician who was identified by the patient as being the physician responsible for the parkinsonism treatment, i.e., the primary physician. The movement disorder expert drafted the letter after the CLaSP baseline assessment, considering current and previous disease factors, review of medications and current medical and social care arrangements. The movement disorder expert sent the letter to the primary physician with the invitation to contact the expert if the recommendations were unclear or additional advice was needed. The decision to implement the recommendations remained with the patient’s primary physician. Patients in the control group received care as usual during the follow-up period, but had the possibility to receive a letter with recommendations from the expert after the follow-up assessment, i.e., outside the current study window. For ethical reasons, if the assessments revealed issues requiring urgent treatment, these were to be communicated to the primary physicians also in control group.
Outcomes
The primary outcome was the Unified Parkinson’s Disease Rating Scale –part II: activities of daily living (UPDRS-II) [20]at 6 months, and secondary outcomes were quality of life (Parkinson Disease Quality of Life Questionnaire 8-items version; PDQ-8), mental health (UPDRS-I), motor function (UPDRS-III), complications of therapy (UPDRS-IV), total UPDRS score (UPDRS-total), cognition (Mini-Mental State Examination; MMSE), non-motor symptoms (Non-Motor Symptoms scale; NMSs) and health status (EQ-5D-5L). We also assessed the levodopa equivalent daily dose (LEDD) [21–26]. We chose activities of daily living as the primary outcome, because it contributes to the disease burden of patient and caregiver, and to adverse outcomes like nursing home placement [27–29]. Outcomes were assessed twice: at baseline and at the primary end-point after six months. Assessors visited the patients mostly at home, but if possible, patients came to the study centre. Process information was collected to assess implementation of the treatment recommendations and barriers to implementation. During the follow-up meeting the assessor discussed the treatment recommendations with the patient and scored recommendations as completely followed, partially followed, not followed, or unknown. The assessor contacted the primary physician for an interview to find out if recommendations were followed, and to assess barriers for implementations. For the latter, we used a structured questionnaire based on the Cabana model [30–32], which identifies barriers in knowledge, attitude or behavior for guideline adherence among neurologist and GP [31, 32]. Barriers listed in the original questionnaire that were not applicable to our intervention were removed, leaving a comprehensive list of eleven items (see Table 4).
Randomization and concealment of allocation
Permuted block randomization was used, stratified by country, presence of dementia and residency (nursing home or similar/ home). Randomization was performed centrally at the Coordinating Centre for Clinical Trials (Marburg, Germany). Assessors and patients were not blinded.
Statistical analysis
A power calculation was performed to estimate the target sample size, based on the primary outcome: UPDRS –part II: activities of daily living [33]. An independent sample t-test was used and the assumptions were a difference in change of 4.8 points between both treatment groups, a standard deviation of 10 points for difference in change and non-participating and dropout rates of 20% each. 288 patients had to be included to achieve a power of 80% with a two-sided significance level of 5%. The current study was terminated at the end of the funding period, prior to reaching the target sample size.
Missing data were substituted with an imputation strategy, preferably according to the user guidelines of each measurement instrument. As such, we used the validated protocol for handling missing data of the UPDRS [34], by which imputations were allowed if the number of missing items did not exceed 1 for the UPDRS-I, 1 for the UPDRS-II and 7 for the UPDRS-III. No imputation was allowed for UPDRS-IV. Imputation for NMS items is possible if less than 15 items were missing. The case-specific mean of completed items was used for imputation of missing UPDRS and NMS items. No valid imputation strategies exist for the other questionnaires and analyses were performed on the available data.
For the intention-to-treat analysis, we performed multivariate linear regression analyses with the outcome measures at follow-up as dependent variables and the group (intervention or care as usual) and baseline score of the outcome measure as the independent variables, correcting for relevant covariates (i.e., presence of dementia, presence of informal caregiver, residency, age, gender and disease duration). We present the covariate-adjusted mean difference between treatment groups and the 95% confidence intervals. We also performed an exploratory perprotocol analysis, only including in the intervention group those patients in whom the recommendations were completely or partially followed; all others were included in the control group. Descriptives are presented with mean and standard deviation for normally-distributed variables and with median and interquartile range for non-normally distributed variables. Critical p-value for statistical significance was set at 0.05. All analyses were performed using Statistical Package of Social Sciences, version 22.
Standards protocol approvals, regulations, and patient consent
This study was in compliance with the Helsinki Declaration (World Medical Association Declaration of Helsinki 1997). Detailed oral and written information was given to the patients and their informant to ensure that the patients fully understood the potential risks and benefits of the study. Written consent was given by patients or, if patients lacked capacity, by a legal guardian, in accordance with local ethical and legal regulations. The study protocol was approved by the local ethics committees of all participating study sites (London: Camden and Islington NRES Committee 14/LO/0612, Lisbon: Centro HospitalarLisboa Norte, DIRCLN-19SET2014–275, Lund: EPN Regional aetikprovnings namnden (EPN Regional Ethics Name) JPND NC 559–002). Nijmegen: Radboud universitairmedisch centrum, Concernstaf Kwaliteiten Veiligheid, Commissie Mensgebonden Onderzoek Regio Arnhem-Nijmegen (Radboud university medical center, Group staff Quality and Safety Human Research Committee, Arnhem-Nijmegen region, DJ/CMO300). Inclusion was possible if patients gave their written informed consent. The protocol was registered at ClinicalTrials.gov as NCT02333175 on 07/01/2015.
Data availability
Anonymized data can be shared with qualified investigators on reasonable request.
RESULTS
Out of the 477 patients in the overall CLaSP study in the participating centres with ethical approval, 167 had not received care by a movement disorder specialist within the last four months. Out of these 91 could be included, of whom 70 were randomized to the intervention group and 21 to the control group. Reasons for non-inclusion, including four who declined participation, are listed in Fig. 1. Patients in the intervention group did not differ in baseline characteristics from controls except for the presence of an informal caregiver that was more present in the intervention group in the per protocol group allocation (Table 2). Overall, 58 (83%) patients in the intervention group and 18 (86%) patients in the control group completed the trial (Fig. 1).
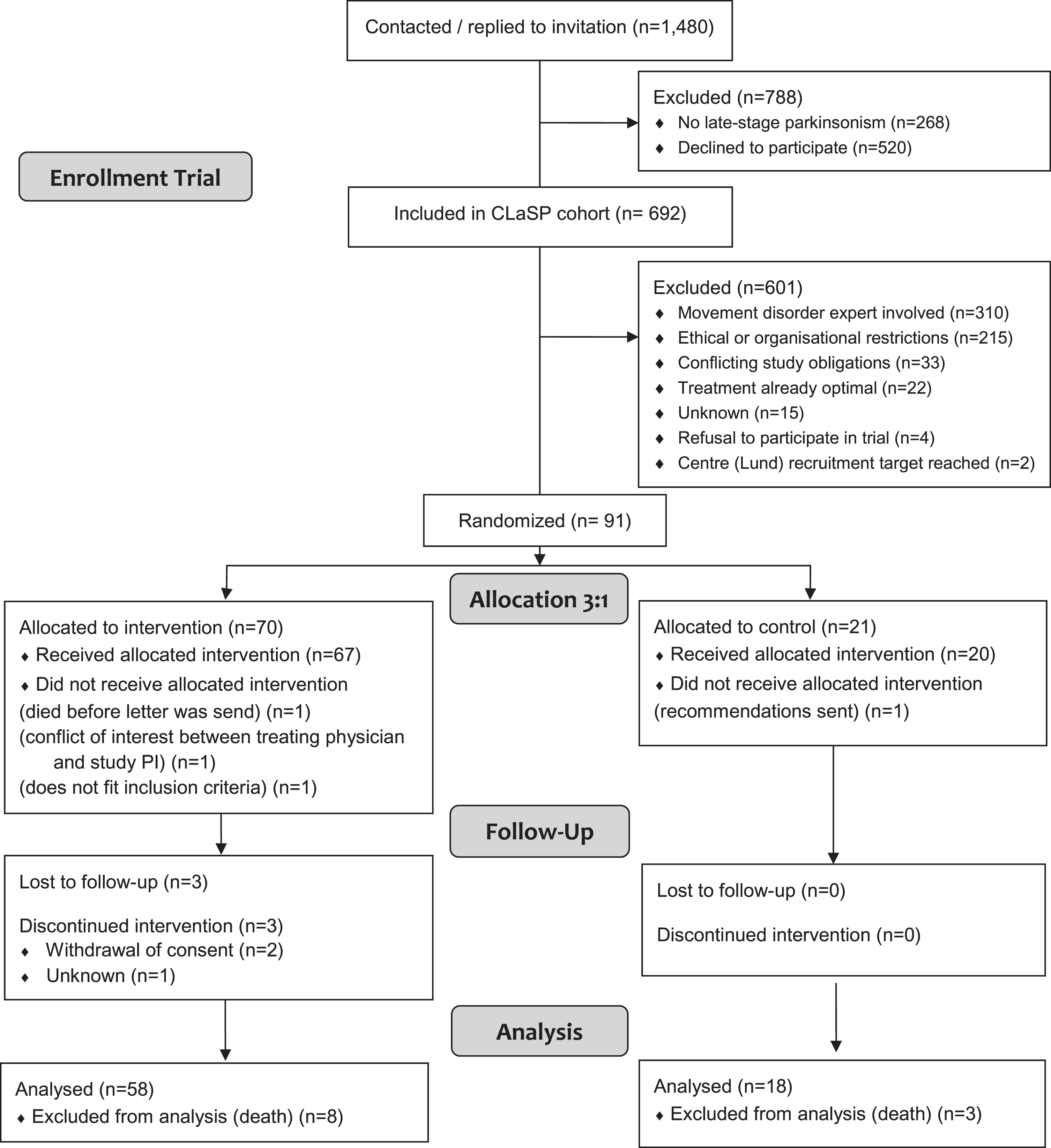
Flow diagram of recruitment of cohort and trial population.
Univariate comparative analysis of baseline characteristics
IQR, interquartile range; SD, standard deviation.
In the intention-to-treat analysis, there was no difference in change in the UPDRS ADL scores, i.e., the primary outcome measure, between the intervention and control group at six months (between-group difference = –1.2, 95% CI = –4.2 to 1.8, p = 0.45). The group difference in UPDRS motor and total score showed a trend towards improvement (between-group difference = –5.1, 95% CI = –10.7 to 0.6, p = 0.08). Quality of life had improved at six months for patients in the intervention group, but had worsened in controls (PDQ-8, between-group difference = –3.7, 95% CI = –6.7 to –0.9, p = 0.01; Fig. 2a and Supplementary Material). All other secondary outcomes showed no group differences.
The per-protocol analysis (Fig. 2b and Supplementary Material) confirmed these findings, showing no between-group difference in the UPDRS ADL score, but again a difference in PDQ-8 in favour of the intervention group (between-group difference = –2.7, 95% CI = –5.1 to –0.3, p = 0.03). The difference in UPDRS total score as well as part I scores also reached significance (UPDRS total: between-group difference = –7.4, 95% CI = –14.6 to –0.2, p = 0.04; UPDRS part I: between-group difference=–1.1, 95% CI = –2.2 to –0.4, p = 0.04), with a trend for part III scores (between-group difference = –4.2, 95% CI = –9.2 to 0.8, p = 0.10). Finally, patients in the intervention group had a larger increase in LEDD (between-group difference = 165 mg, 95% CI = 51 to 279, p = 0.01). A sensitivity analysis with presence of a caregiver as a covariate in the per-protocol analysis gave similar results (data not shown). Different definitions of the per protocol groups did not change the main results (Supplementary Materials).
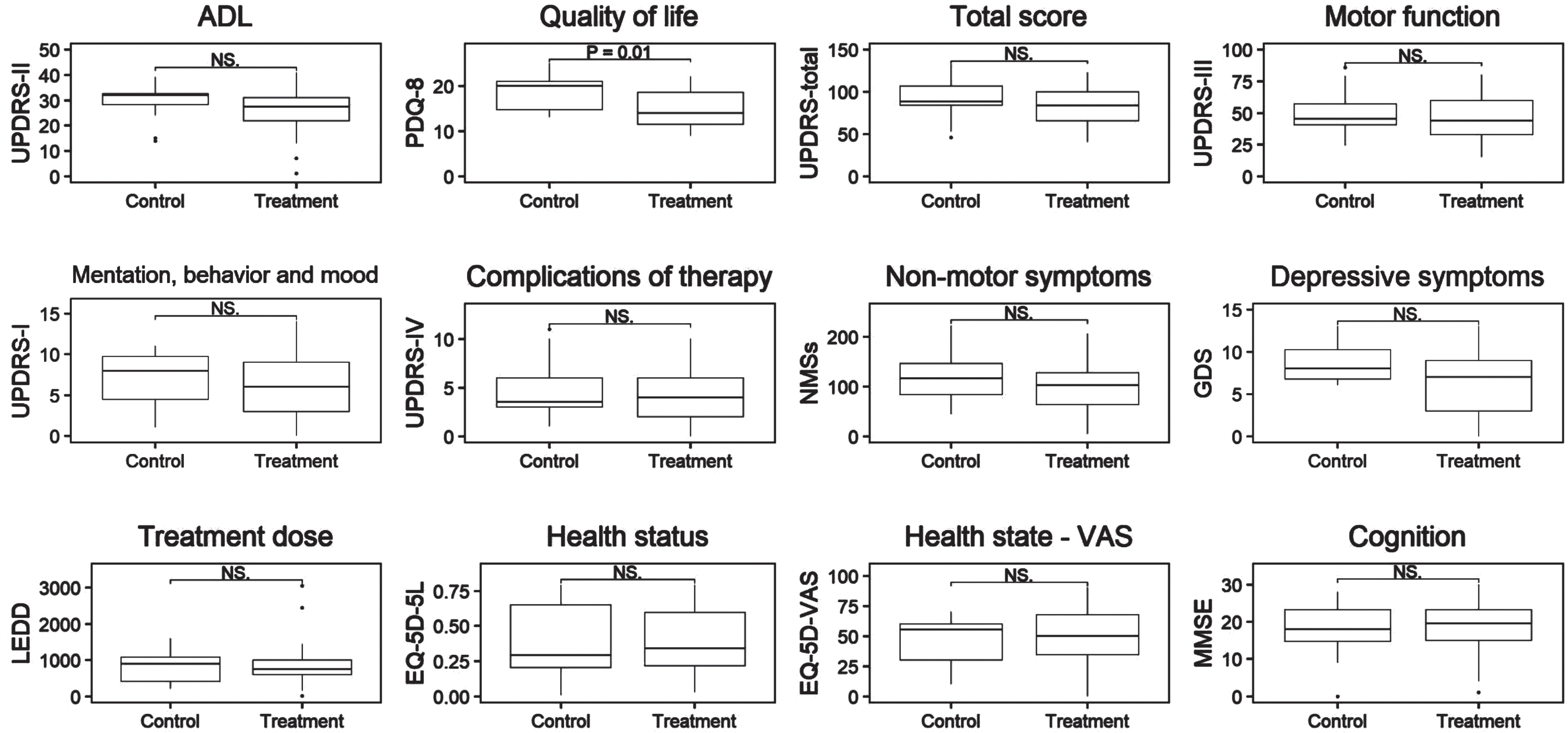
Intention-to-treat analysis CLaSP-trial. Shown are boxplots of primary and secondary outcome measures at follow-up. UPDRS, Unified Parkinson Disease Rating scale; MMSE, Mini-Mental State Examination; NMSs, non-motor symptoms scale; GDS, Geriatric Depression Scale –15 items; PDQ, Parkinson Disease Questionnaire; EQ-5D-5L, EuroQol-5 dimensions; VAS, Visual Analogue Scale; LEDD, Levodopa Equivalent Daily Dose; NS., non-significant.
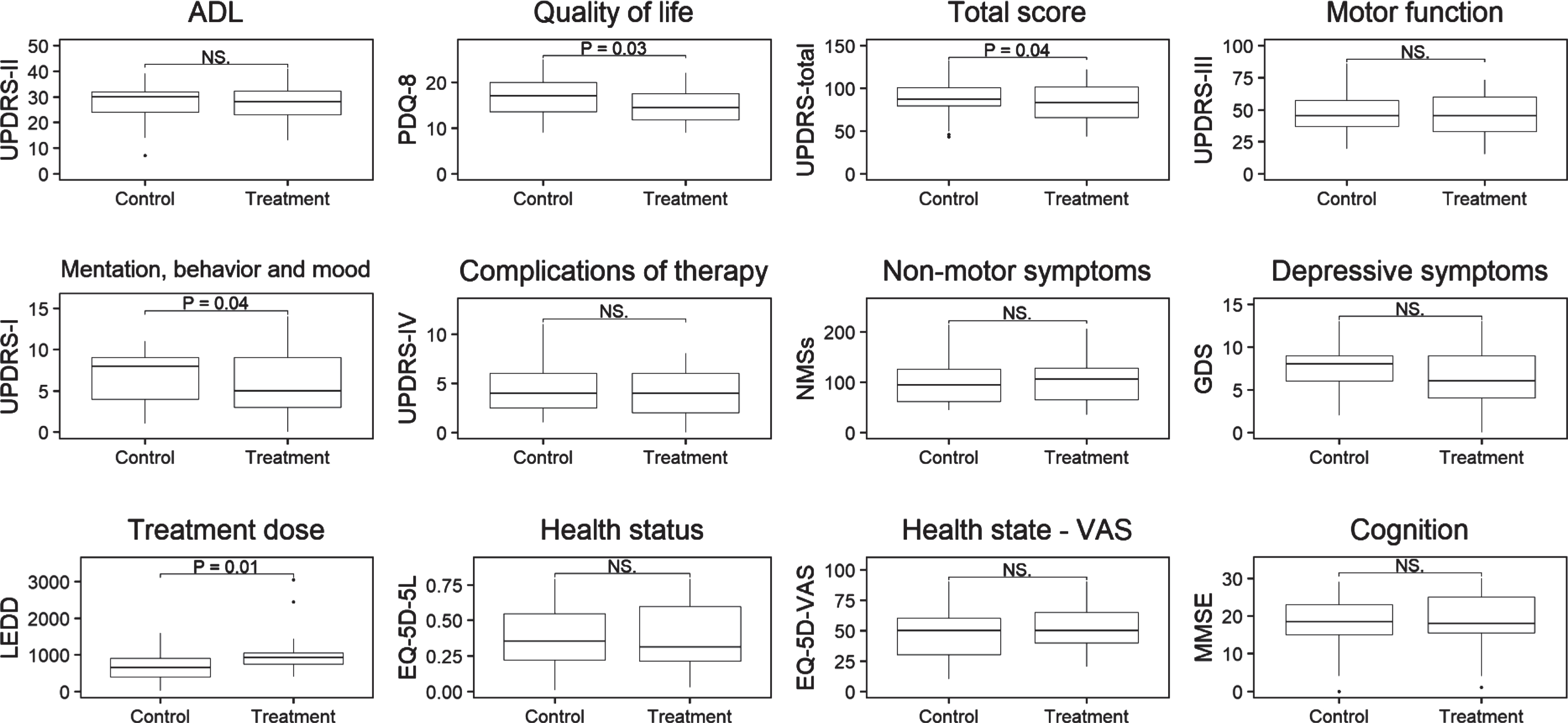
Per-protocol analysis CLaSP-trial. Shown are boxplots of primary and secondary outcome measures at follow-up. UPDRS, Unified Parkinson Disease Rating scale; MMSE, Mini-Mental State Examination; NMSs, non-motor symptoms scale; GDS, Geriatric Depression Scale –15 items; PDQ, Parkinson Disease Questionnaire; EQ-5D-5L, EuroQol-5 dimensions; VAS, Visual Analogue Scale; LEDD, Levodopa Equivalent Daily Dose; NS., non-significant.
Process analysis of implementation
The primary physicians receiving the letter with recommendations followed these recommendations completely in only 16 (28%) patients and partially in 21 (36%). Recommendations were not followed in 18 (31%) and remained unclear in 3 (5%) patients. The extent to which recommendations were followed, differed per type of recommendation and ranged from 15% for referral to physiotherapist (complete or partially followed: 5/33) to 50% for recommendations about dopaminergic treatment (complete or partially followed: 20/40). In total, 36 recipients of the letter with recommendations were contacted to assess barriers for implementing recommendations. As the main reason for not following the recommendations, the physicians reported to have experienced an inability to reconcile patient preferences with the recommendation (10/36 = 28%), lack of time (8/36 = 22%) and lack of improved outcome expectancy (7/36 = 19%) (Table 3). In addition to the items from the Cabana model, the open question retrieved eightadditional barriers, reported in total 12 times (33%; see Table 4, item 14). The most frequent additional barrier to not following recommendation was a change in physician (5/36 = 15%).
Performance of recommendation letter as implementation strategy
aIf multiple recommendations were given in one domain, the results of the best followed recommendations is reported.
Barriers to implementation of recommendations as reported by letter recipients
Proportions of the barriers per recipients are shown following a structured interview using the Cabana model and one open-ended question allowing other barriers to be mentioned. Multiple barriers could be report per recipient.
DISCUSSION
Despite not reaching the required sample size, this pragmatic trial is the largest study to date in the underserved and poorly studied population of LSP. A letter to the primary physician with recommendations to optimize treatment by a specialist, based on standardised assessments by a trained assessor, did not improve the primary outcome measure of UPDRS ADL score compared to care as usual, but there was a significant improvement in quality of life scores in both the intention-to-treat and per-protocol-analysis, with an effect size exceeding the minimally important benefit [35]. In addition, there was a trend towards better outcome on the UPDRS part III as well as UPDRS total scores in the intention-to-treat population, and a significant improvement on the UPDRS total and part I scores in the per-protocol analysis, together with a greater increase in LEDD, suggesting that adjustment of dopaminergic medication partially mediated the observed effects. This notion is also in line with earlier work showing dopaminergic under treatment in LSP patients [3, 4], and also with other studies showing that levodopa improved motor and non-motor features in LSP patients [36–38]. The significant difference between intervention and control group in the PDQ-8 suggests that the intervention had a positive impact on the patients’ overall quality of life that was not captured by the UPDRS-ADL part. Several other studies on complex multidisciplinary interventions in PD failed to show an impact on ADL-measures, indicating that these outcomes may not be sensitive enough to capture relevant change in these situations [39–42]. A quality of life measure may be a more appropriate tool, particularly in the advanced, complex stage of PD, where treatment is increasingly aiming to optimize quality of life instead of pursuing improvement of objective functioning. Social elements of the disease, like feeling embarrassed by symptoms or having trouble in personal relations, are represented in our quality of life measurement but not in the other outcome measures, which could explain the lack of finding on other outcome measures in the intention-to-treat analysis. For the same reason, patient-reported outcome measures, such as quality of life measures, are increasingly used as primary outcome measures in large trials [43, 44]. Finding a change on such patient-reported measure, like quality of life measures, but not on clinician-completed outcome measures has also been reported by other trials [45, 46], indicating that patients may report improvements that are not appreciated in assessment by others, including using standardised assessment tools.
It is also noteworthy that our process evaluation revealed that whilst physicians followed recommendations to at least some degree in most patients (64% either completely or partially), many other recommendations were not followed and several barriers to implementing the recommendations were identified. These findings indicate suboptimal implementation of the advice of movement disorder experts communicated in a letter, as typically done in standard outpatient settings, and that other medical consultation models may be more appropriate for this population. This is in line with previous studies, with more elaborate interventions, that reported low adherence in interventions aiming at improving quality of disease management in elderly populations [47–51]. Perhaps the most important result of this trial is that we identified several barriers for the implementation of the advice. The most common reasons were difficulty in reconciling the advice with the patient’s preference, lack of time, lack of improved outcome expectancy, change in primary physicians and lack of agreement with the advice. This may in part be related to the constraints of the trial, with standardised recommendations, assessments rather than ongoing care, and assessment of complex patients with a trained study assessor rather than the movement disorder specialist who made the recommendations following discussion. It has previously been shown that understanding the medicine-taking behavior of patients should be the first step in optimizing therapy, which requires knowledge and consideration of a patient’s personal beliefs about their medicines [52]. However, it may also suggest that recommendations by the specialist require greater interaction with the primary care physician to adjust to the circumstances of their care, availability and access to treatments such occupational therapy, a PD specialist nurse, and wishes of patients with LSP, or that the healthcare system is ill-equipped to implement the intervention. Further work is needed to explore this and future research should take note of these barriers in developing more elaborate interventions which are better suited to the local health care system.
The pragmatic design of this trial had limitations which may have affected our findings. Primarily due to lack of ethics approval in two participating countries and many patients already receiving specialist care, we did not reach the targeted study sample size. We extended the study recruitment window and developed several new strategies to boost recruitment, but this population remains difficult to include in clinical trials. As a consequence, we cannot draw any firm conclusions on the impact of our intervention on the primary outcome measure. In addition, we conducted the study in several countries across Europe with different health care provisions and these differences could have concealed a greater effect. Furthermore, we included patients with all types of parkinsonism, not all of whom would respond to anti parkinsonian medication changes. However, only three individuals who completed the trial did not have a diagnosis of PD and the main results were comparable when we only analysed typical patients. The movement disorder specialist had limited contact with participants, as assessments were done by trained staff, recommendations were standardised, availability and ease of access, and the beliefs on their treatment were not assessed behaviour. As discuss above, these are is likely to have affected to the implementation of the recommendations [52]. In addition, movement disorder experts had limited contact with most of participants’ healthcare providers, and greater interaction may have improved adherence to the recommendations. Nevertheless, our methodology mirrored typical daily practice in current healthcare systems, where infrequent specialist appointments with recommendation letters for other involved healthcare providers, are typical forms of intervention, and continuity of care by a specialist, good interaction with primary physicians, and sufficient time in primary care are often not available. Our results suggest that in order to achieve the best results with significant improvement of outcomes for activities of daily living and quality of life, specialist recommendations need to be accompanied by strategies to increase implementation. Close interaction with primary physicians, sufficient time for discussion with patients and their carers on preferences, wishes and beliefs and the benefits of the recommended treatments, and long term follow-up with continuity of care may be helpful to achieve this. LSP poses particular challenges to provision and participation in care, including cognitive deficits, low mood, apathy or fatigue which can limit participation in some non-pharmacological interventions [53], and there are limitations in ability to attend appointments and high caregiver burden [12, 55]. Novel approaches to providing specialist input for this population, including community-based support, palliative care models with neurological input, online support and other modalities may be required to maximise the benefit from specialist recommendations to improve quality of life and disability [56].
In summary, whilst there was no improvement of ADL on the UPDRS-ADL part in this study, which was limited by under recruitment and limited implementation of recommendations, we found that specialist recommendations communicated by letter had a positive impact on quality of life in patients with LSP. Our results also demonstrate the limitations in implementation of treatment recommendations communicated by letter to the primary treating physicians in this complex and vulnerable patient group.
CONFLICT OF INTEREST
The authors have no conflicts of interest to report related to the present study.
Footnotes
ACKNOWLEDGMENTS
We would like to thank patients and their families participating in the study and all staff members in the recruiting centres who participated in the CLaSP consortium. Special thanks to the following members of the consortium: Brice Laurens, Umberto Spampinato, Alexandra Foubert-Samier, Sylvain Vergnet, Jeanette Härnberg and Inga-LillSvensson. We would further like to thank the European Commission for funding this study (Joint Programme –Neurodegenerative Disease Research “European research projects for the evaluation of health care policies, strategies and interventions for Neurodegenerative Diseases”; through national funding bodies in all six countries: Economic and Social Research Council ES/L009250/1; BMBF, Marburg, Germany 01ED1403A, Munich, Germany 01ED1403B, Bordeaux, France: ANR-13-JPHC-0001-07, Lisbon, Portugal: HC/0002/2012, Lund, Sweden: HC-559-002, Nijmegen, Holland, 733051003. We would like to thank the following organisations for co-funding: Groenhuysen organisation, Stichting Beroepsopleiding Huisartsen, National Institute for Health Research UCL/UCLH Biomedical Research Centre.
We would like to thank our funding sources: the European Commission AS was supported by the National Institute for Health Research UCL/UCLH Biomedical Research Centre. DH was supported by Groenhuysen Organisation and Stichting Beroepsopleiding Huisartsen. SL has received honoraria for lectures from UCB, AbbVie. We received no industry-sponsored funds.
