Abstract
Keywords
Introduction
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease, characterized by progressive degeneration of motor neurons in the spinal cord, brain stem, and motor cortex, leading to progressive muscle atrophy and weakness. Patients usually die from respiratory failure, on average within 5 years of symptom onset. 1 The progression of symptoms leads to increased functional limitations in activities of daily living, participation restrictions, 2 and a decline in health-related quality of life (HRQoL). 3 Because there is no cure for the disease, ALS care is based on treatment of symptoms and optimizing daily activities, participation, and QoL, provided in a multidisciplinary setting. 4
Early-stage ALS patients show a skeletal muscle mass similar to that of healthy individuals but with a reduced peak oxygen uptake, most likely caused by deconditioning as a result of a reduction in habitual activities. 5 Sustained deconditioning may lead to a vicious cycle of further physical inactivity and deconditioning. 6 A reduced aerobic capacity in patients with ALS may hamper the ability to perform activities of daily living and social activities that require sustained aerobic metabolism and, subsequently, may reduce HRQoL. 7
Aerobic exercises have been proven to increase aerobic capacity in various muscle diseases.8,9 Prescribing exercise programs for patients with ALS has been controversial because of concern that damage to muscle tissues potentially leads to a faster progressive decline. 10 A Cochrane review 6 included 2 small randomized controlled trials (RCTs)11,12 on strengthening exercise training in ALS patients and showed a weighted mean improvement in the ALS Function Rating Scale (ALSFRS) in the exercise compared with the control group. The authors concluded that RCTs examining aerobic exercise in this population were scarce. Recently, a single RCT showed a reduction of functional decline (ALSFRS–Revised) as a result of a program including active exercises combined with cycle ergometer activity. The sample size of the cycle intervention group (n = 10) was, however, too small to allow unequivocal conclusions about the effectiveness of aerobic exercise to be drawn. 13 A prospective controlled study reported that patients who participated in endurance exercises while on bi-level positive airway pressure (BiPAP) had significantly greater functional impact measure scores, slower Spinal Norris score decline, and less forced vital capacity (FVC) decline compared with those who did not exercise. 14 Neither aerobic nor strengthening exercise studies reported any adverse effects. Larger controlled studies to elucidate the effect of aerobic exercise in patients with ALS are needed.
The primary objective of the RCT was to study the effectiveness of aerobic exercise therapy (AET) in addition to usual care (UC), compared with UC alone, on disease-specific and generic HRQoL in early-stage, ambulatory ALS patients. The secondary objective was to assess the effects of AET, in addition to UC, on impairments, activity limitations, and participation restrictions 15 to gain insight into the potential working mechanisms of the AET intervention.
Methods
Study Design and Participants
This stratified, multicenter, assessor-blinded RCT, with equal allocation across treatment arms and 4 follow-up visits in the course of 10 months, was nested in a larger study: the FACTS-2-ALS trial (acronym for Fitness And Cognitive behavioral TherapieS for Fatigue and ACTivitieS). The FACTS-2-ALS trial examined the effects of both AET and cognitive behavioral therapy (CBT) on HRQoL in patients with ALS. Contamination in the trial was not an issue because neither patients in the AET+UC nor those in the UC group received CBT. The design of this study and the results of the CBT study have already been published elsewhere.16,17
Patients were recruited from 5 rehabilitation centers or rehabilitation departments of academic hospitals in the Netherlands. Eligible patients met the following criteria: age between 18 and 80 years; FVC of at least 80%; possible, laboratory-supported probable, probable, or definite ALS according to the revised version of the El Escorial criteria 18 ; diagnostic phase completed; life expectancy longer than 1 year; ability to walk with or without walking aid (≥10 minutes); and ability to cycle on a cycle ergometer (≥15 minutes). Patients who already exercised (≥2 hours a week) were excluded because the treatment effect was expected to be minimal. Other exclusion criteria were the following: severe cognitive impairment (whether or not related to ALS, preventing completion of the AET), disabling comorbidity, and psychiatric disorder, all assessed using the Cumulative Illness Rating Scale, 19 and insufficient mastery of the Dutch language.
Changes after trial commencement from the original protocol 17 were widening of the age range from 18 to 70 years, to 18 to 80 years, to allow inclusion of additional patients who could benefit from the intervention, and adding 1 rehabilitation center to further empower the study.
Standard Protocol Approvals, Registrations, and Patient Consents
The RCT was registered at the Netherlands National Trial Register (NTR1616). The study protocol was approved by the Medical Ethics Committee of the University Medical Center Utrecht (08-459/O). All participating centers granted approval to participate, and all patients provided written informed consent.
Randomization
Randomization was performed centrally by an investigator (JHV) not involved in testing or treatment. Patients were randomized (1:1) to either AET+UC (intervention group) or UC alone (control group) according to the minimization method described by Pocock, 20 without a dynamic element, for the following factors: age, site of symptom onset (bulbar or spinal), center, and gender.
The assessors who performed the outcome assessments were blinded to the group allocation. Patients and intervention physiotherapists could not be masked to group allocation because the intervention involved attendance at appointments and home exercises, but allocation was masked from data entry researchers and from the trial statistician (RPAvE) until the database had been closed.
We used a biphasic randomization model with postponed information, which allowed us to inform patients in the UC group about their part of the study only, and the remaining information was given to them at the end of the study to prevent disappointment of withholding treatment and, subsequently, seeking a similar intervention. 21 First, a rehabilitation physician checked the eligibility criteria, and potential patients were provided with written and oral information about a longitudinal study, including 4 repeated measurements. Eligible patients were asked by the investigator (ACvG) to give signed consent for 4 repeated measurements (first permission). Second, patients randomized to AET received written and oral information about the exercise trial and were asked by the investigator (ACvG) to take part in the training study and to sign the informed consent for participating in AET (second permission). Patients were explicitly instructed and reminded not to reveal their treatment allocation to the single-blinded assessor.
Procedures
Interventions
Aerobic exercise therapy
Within 2 weeks of randomization, AET was started. It was designed specifically to maintain or enhance the patient’s aerobic capacity. AET lasted 16 weeks and consisted of (1) a home-based training program twice a week and (2) an individual training session once a week at a rehabilitation center or rehabilitation department of an academic hospital, supervised by specially trained intervention physiotherapists.
(1) The home-based training consisted of individual tailored AET on a cycle ergometer (Kettler X7, Heinz Kettler, Germany) and a step board (Kettler, Heinz Kettler, Germany). The heart rate was continuously monitored with a heart rate belt and watch (FR50, Garmin, USA). The training intensity was gradually increased from 50% (moderate) to 75% (vigorous) heart rate reserve (HRR), 22 and training duration was gradually increased from 20 to 35 minutes per session. All patients received identical training equipment at home for the duration of the intervention.
(2) The 1-hour supervised individual training sessions at the center consisted of workstations; each session was divided into a 5-minute warming-up, 30 minutes individually tailored aerobic exercises (cycle ergometer, step board, and treadmill), 20 minutes muscle strengthening exercises (quadriceps, biceps, and triceps), and a 5-minute cooling down. Training intensity of aerobic exercises was gradually increased from 50% to 75% HRR, and training intensity of muscle strengthening exercises was gradually increased from 40% to 50% of the maximum strength of different muscle groups (quadriceps, biceps, and triceps). The maximum strength, defined as 1 repetition maximum (1-RM), was estimated using a submaximal test. 23 Only muscle groups with a Medical Research Council score ≥3 were trained. 24 Each exercise was repeated 10 to 15 times.
To monitor patients’ safety, training intensity was adjusted according to percentages of the HRR and 1-RM, but also according to adverse events and to perceived exertion on the Borg Ratings of Perceived Exertion (RPE) Scale. 25 All adverse events (such as severe muscle fatigue, cramps, pain, or other events) were recorded in an individual logbook and were monitored until they abated or until a stable situation had been reached. Training aimed at patients reporting a Borg score of between 11 (fairly light) and 14 (moderately heavy); if a patient scored >14 in 2 successive training sessions, the exercise intensity was reduced by 5% HRR, and subsequently increased by 5% HRR after training twice with a Borg RPE score ≤14. This adjustment prevented patients from training with an intensity exceeding 75% of HRR, 26 thus avoiding the risk of overloading. On the other hand, if patients perceived little training stimulus (Borg RPE score < 11) on 2 consecutive occasions, the exercise intensity was increased by 5% HRR. Because muscle strength was expected to deteriorate over time in patients with ALS, the 1-RM was estimated at baseline and again after 5 and 10 weeks of training to ensure that patients exercised at an adequate training intensity. During the weekly supervised sessions, the physiotherapist verified compliance by reading the data recorded by the heart rate watch and by reviewing the logbook. In addition, the patient was given training instructions for the following week.
Usual Care
All patients received UC. UC in the Netherlands is neuropalliative care by multidisciplinary, secondary care teams. Such teams consist of a rehabilitation medicine consultant, an occupational therapist, physical therapist, speech therapist, dietician, social worker, psychologist, and consultant physicians. 27 Neither allocation group was restricted in their daily activities.
At baseline, demographic and clinical characteristics were collected, including the ALSFRS-R as measure of global functioning and disease progression (Table 1). 28 Patients in the AET and UC group were assessed at study entry (pretreatment), at 4 months (postintervention), at 3 months follow-up, and at 6 months follow-up. Measurements were performed by a single-blinded assessor at the location of the AET intervention. Patients were asked to complete the questionnaires at home.
Baseline Characteristics of Intention-to-Treat Groups, Per-Protocol Versus Discontinued Protocol Groups, and Propensity-Matched Cohorts. a
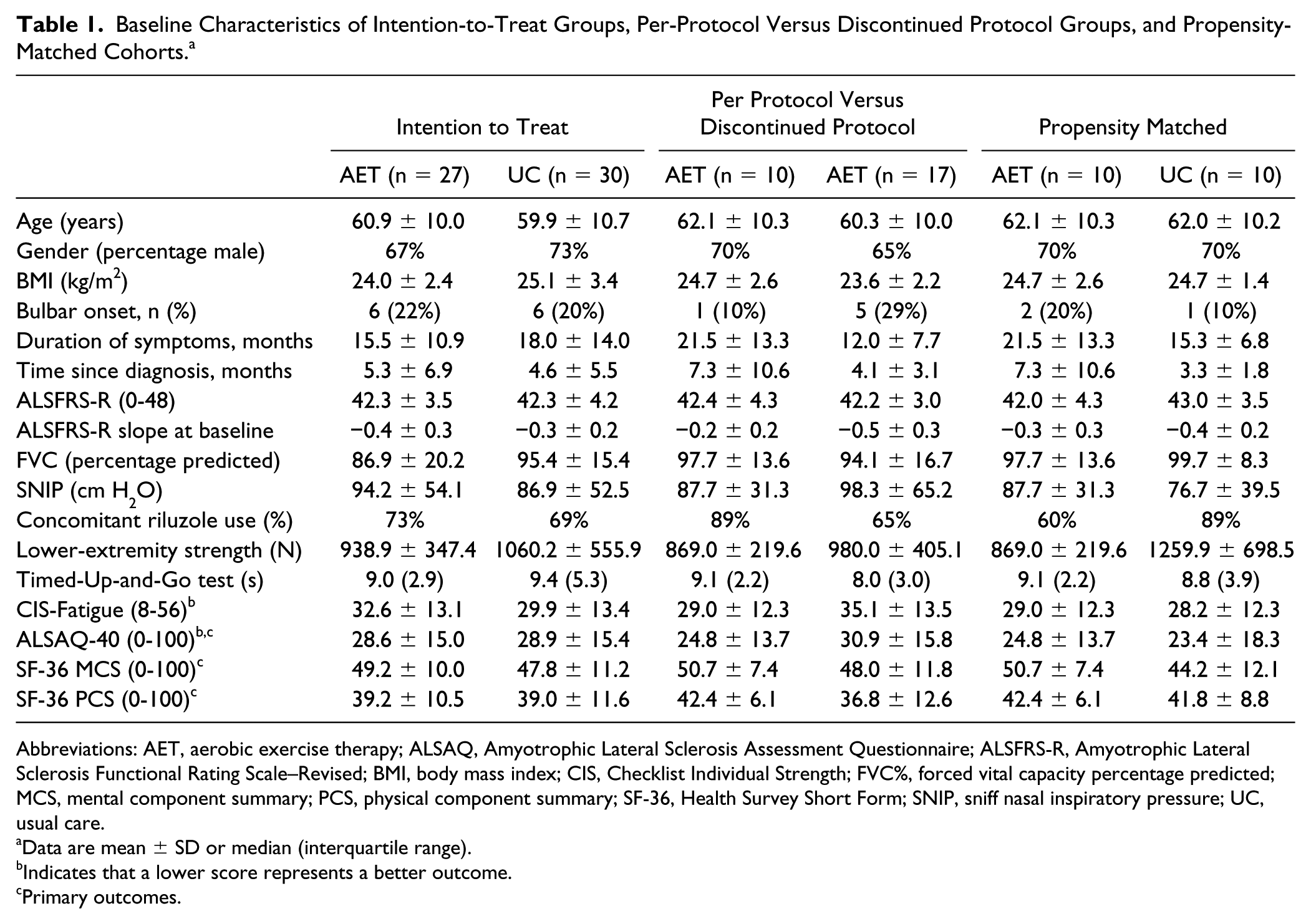
Abbreviations: AET, aerobic exercise therapy; ALSAQ, Amyotrophic Lateral Sclerosis Assessment Questionnaire; ALSFRS-R, Amyotrophic Lateral Sclerosis Functional Rating Scale–Revised; BMI, body mass index; CIS, Checklist Individual Strength; FVC%, forced vital capacity percentage predicted; MCS, mental component summary; PCS, physical component summary; SF-36, Health Survey Short Form; SNIP, sniff nasal inspiratory pressure; UC, usual care.
Data are mean ± SD or median (interquartile range).
Indicates that a lower score represents a better outcome.
Primary outcomes.
Outcomes
The primary outcome was the change in disease-specific and generic HRQoL during the follow-up period of 6 months. Disease-specific HRQoL was measured using the Amyotrophic Lateral Sclerosis Assessment Questionnaire (ALSAQ-40). 29 Generic HRQoL was assessed with the short-form survey (SF-36) using both mental component summary (MCS) scores and physical component summary (PCS) scores. 30
The secondary outcomes were categorized into the domains of “body functions and structures/impairments,” “activity/activity limitations,” and “participation/participation restrictions,” in accordance with the International Classification of Functioning, Disability and Health conceptual framework 15 and were selected to gain insight into the potential working mechanisms of the AET intervention.
Body functions and structures/impairments: aerobic capacity (maximal oxygen uptake [L/min] estimated with the Åstrand-Ryhming [ÅR] test) 31 ; lower- and upper-extremity strength (sum score in Newtons assessed using a MicroFET hand-held dynamometer [Jamar, Biometrics Ltd, USA]); grip strength (assessed using a hydraulic hand-held dynamometer [Jamar, Biometrics Ltd, USA]); fatigue severity (the Checklist Individual Strength subscale fatigue [CIS-fatigue]) 32 ; FVC percentage predicted (FVC%) 33 and sniff nasal inspiratory pressure (SNIP) 34 measured using spirometry (MicroRPM, Pt Medical, the Netherlands); pain intensity (Visual Analogue Scale); body mass index (BMI); and self-reported sleep disturbances (assessed using the Nottingham Health Profile Sleep Subscale [NHP-Sleep]). 35
Activity/activity limitations: Physical activity (in Metabolic Equivalent of Task per day estimated with the LASA Physical Activity Questionnaire [LAPAQ]), 36 global functioning (ALSFRS-R), 28 functional mobility (the Timed Up and Go test [TUG]), 37 and stair test. 38
Participation/participation restrictions: Sickness Impact Profile 68 (SIP-68), 39 Impact on Participation and Autonomy questionnaire (IPA). 40
Deviations from the original protocol 17 were removal of the LAPAQ questionnaire, 36 because patients failed to complete it adequately, and removal of the ÅR test at follow-up assessments because of a diminished feasibility of the ÅR test. 7
Data Analysis
A detailed description of the sample size calculation for this study is given in a protocol article. 17 In short, we based our expected treatment effect on 2 previous clinical trials.11,12 These trials did not use our primary outcome (ALSAQ-40) but rather the ALSFRS. Therefore, we standardized their effect size on the ALSFRS and assumed identical effects on the ALSAQ-40. To detect an effect size of 0.8 (SD of 1) with 80% power and a 2-sided α of 5%, we would need 26 patients per treatment arm.
We compared sociodemographic characteristics as well as the clinical characteristics of both groups with 2-tailed independent t test (for continuous variables) and χ 2 tests (for dichotomous and categorical variables). All continuous outcomes were visually assessed for normality and if necessary transformed. Primary and secondary outcomes were analyzed on an intention-to-treat (ITT) principle blinded for group allocation; all patients with follow-up data were included in the analysis according to the group to which they had been randomized, whether or not they received their allocated intervention and whether or not they fulfilled the follow-up assessment at 10 months. We analyzed the change over time of continuous longitudinal measures of both the primary and secondary outcomes. The effect of AET was expressed as the mean difference in the rate of decline (slope) for the 2 treatment groups during the total follow-up period of 6 months. The slope over time was estimated using a 2-level linear mixed model (LMM) in which longitudinal data (level I) were clustered within patients (level II). Additionally, a random intercept and random slope for time were incorporated for each patient. Each model was corrected for the baseline score in accordance with a standard analysis of covariance model. Because of the use of a minimization method during randomization, all models were additionally corrected for gender, site of onset (bulbar vs other), age, and center. 20 We fitted 2 models, one model without and one model with the interaction term between treatment allocation and time (indicating whether the slope over time differs between the treatment groups). The models were fitted using maximum likelihood estimation and compared with a likelihood ratio test. When we estimated coefficients, we fitted the LMM with restricted maximum likelihood. Survival probabilities in both treatment arms were estimated using Kaplan-Meier curves, which were compared using the log-rank statistic.
Additionally, per-protocol (PP) analyses were performed including only patients who attended 75% or more of the treatment sessions (≥36 sessions) 41 and were not lost to follow-up at 10 months. To correct for baseline inequalities, we did a propensity-matched analysis in which every patient receiving AET was 1:1 matched with a control patient receiving UC alone. We matched patients in treatment groups by nearest propensity score based on their age, type of onset, slope, FVC, BMI, and gender because these variables are consistently reported as prognostic factors related to survival. 42 An α ≤5% was considered significant. LMM were fitted using the lmer function in the R package lme4 (version 1.1-11). 43
Results
Figure 1 shows the trial profile. Between February 26, 2010, and January 1, 2015, 325 consecutive patients were screened for eligibility, of whom 176 were excluded based on not meeting the inclusion criteria (FVC<80% [n = 50]; age ≥80 years [n = 38]; walking disability [n = 31]; and already active in sports ≥2 hours a week—ie, performing aerobic exercises such as running/jogging, swimming, cycling, and walking [n = 4]). Of the 149 remaining patients, 15 were enrolled in CBT, 16 and 77 patients declined to participate in a longitudinal study (first permission). The most cited reason for not participating was the distance required to travel to the assessment location (n = 30) and feeling overburdened (n = 22). A total of 57 patients were randomized: 27 to AET+UC and 30 to UC. Baseline characteristics were similar between groups (Table 1).
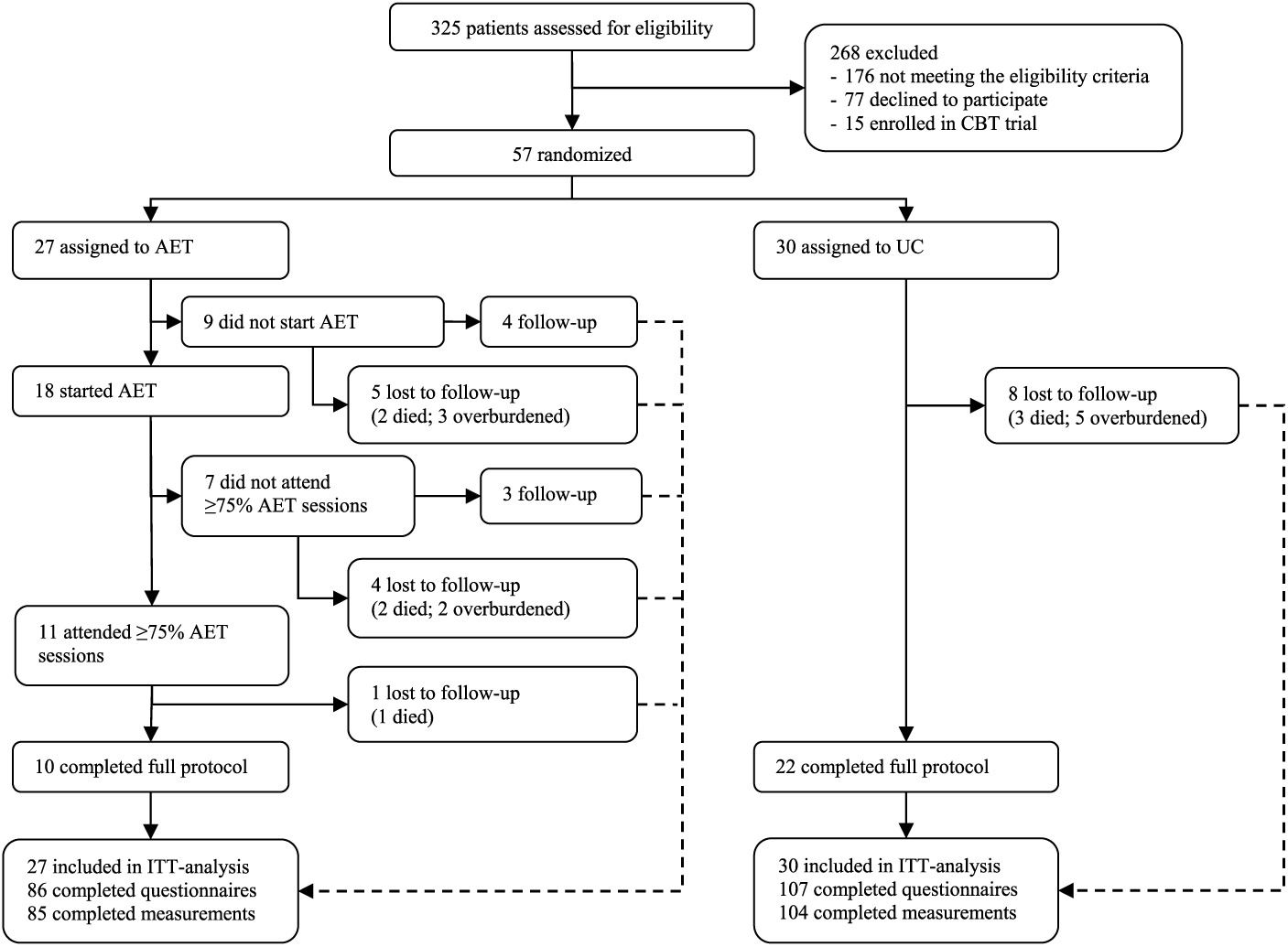
Trial profile.
Of 27 patients who were randomized to AET, 9 (33%) decided not to participate (did not give their second permission), because of alternative physical activities (n = 7) or rapid disease progression (n = 2). Patients who embarked on AET (1) were more often male than patients who did not start AET, (2) had a longer duration of symptom onset, (3) were more often <45 or >65 years of age, (4) were more bulbar affected, and (5) more often used riluzole (results not shown).
Of 18 patients, 7 (39%) did not attend 75% of the AET sessions. Reasons for nonattendance were the following: psychosocial problems (n = 1), clavicular fracture as a result of a fall (n = 2), time constraints (n = 1), perceived lack of benefit from AET (n = 1), percutaneous endoscopic gastrostomy procedure (n = 1), and long holiday (n = 1). A total of 11 AET patients attended ≥75% of the AET sessions (41%); of these, 1 patient was lost to follow-up (deceased); hence, 10 AET patients completed the study protocol. A total of 22 patients (73%) in the UC group completed the study protocol. Five patients assigned to the AET group and 3 assigned to the UC group died during the study period. All deaths were a result of respiratory failure; none was considered to be related to AET.
The median number of overall AET sessions received for the ITT sample was 26 (range 0-47; prescribed: 48). The median number of training weeks was 12 (range 0-19; prescribed: 16). Five adverse events were reported after a total of 614 training sessions (0.8%): 1 patient experienced myalgia lasting 4 days on 1 occasion after the 1-RM test; 2 patients described an increase of fasciculations once after AET; and 2 patients had (nocturnal) cramps once, with one of them also experiencing extreme fatigue on 4 occasions and restless legs once. No serious adverse events were reported.
Outcomes
Results of Intention-to-Treat Analyses
No significant difference in primary outcomes, and disease-specific and generic HRQoL, as reflected by the mean slope differences between groups for ALSAQ-40 and SF-36 (MCS and PCS) score, respectively, was detected between the AET group and the UC group up to 6 months after the training period (Table 2). With respect to the secondary outcomes on impairments, activity limitations, and participation restrictions up to 6 months (Tables 2 and 3), again, no significant differences were found.
Slopes and P Values for Monthly Changes per Outcome in Intention-to-Treat Groups, Corrected for Age, Gender, Site of Onset and Center.
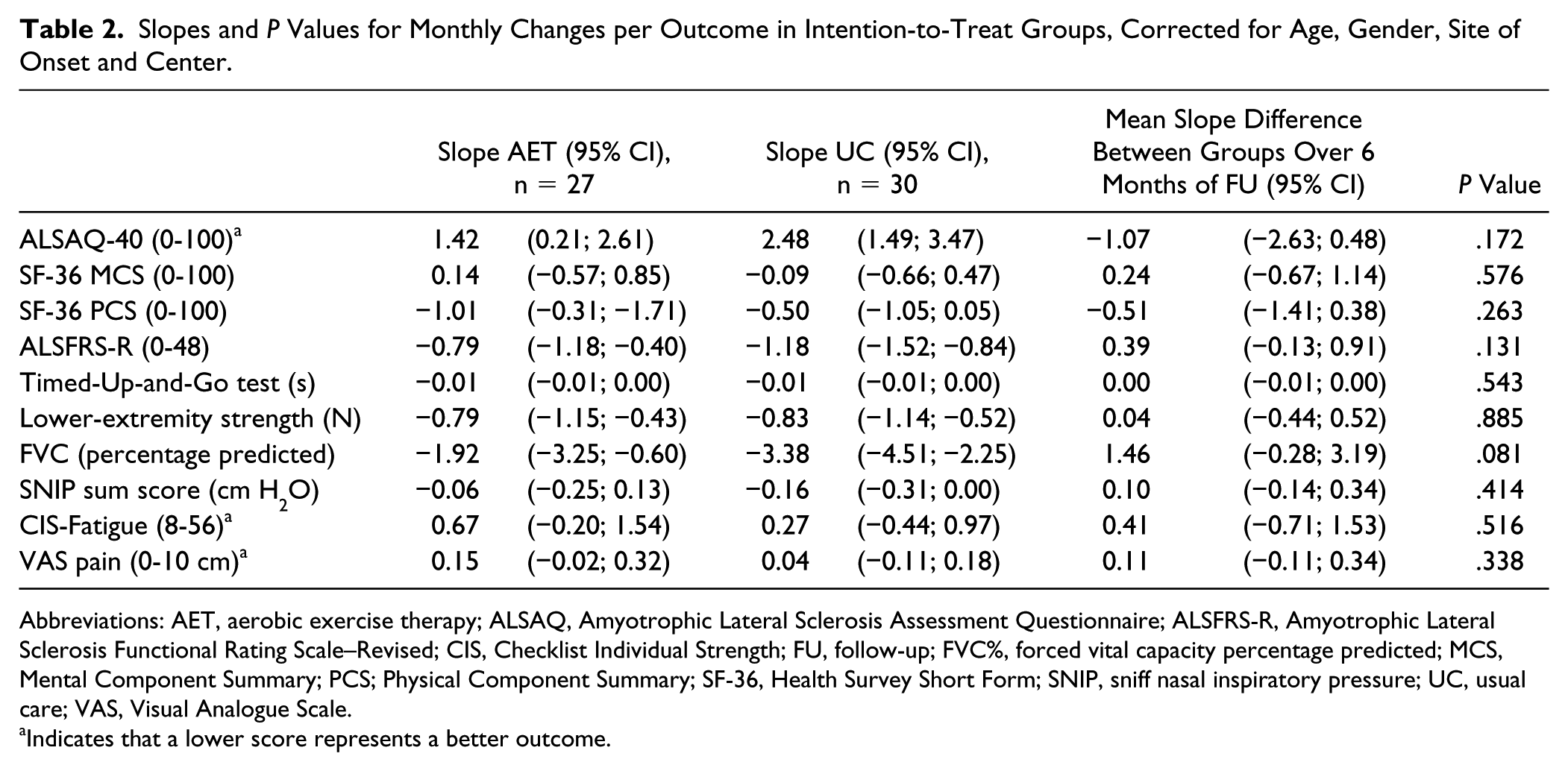
Abbreviations: AET, aerobic exercise therapy; ALSAQ, Amyotrophic Lateral Sclerosis Assessment Questionnaire; ALSFRS-R, Amyotrophic Lateral Sclerosis Functional Rating Scale–Revised; CIS, Checklist Individual Strength; FU, follow-up; FVC%, forced vital capacity percentage predicted; MCS, Mental Component Summary; PCS; Physical Component Summary; SF-36, Health Survey Short Form; SNIP, sniff nasal inspiratory pressure; UC, usual care; VAS, Visual Analogue Scale.
Indicates that a lower score represents a better outcome.
Slopes and P Values for Monthly Changes per Outcome in Intention-To-Treat Groups, Corrected for Age, Gender, Site of Onset and Center.
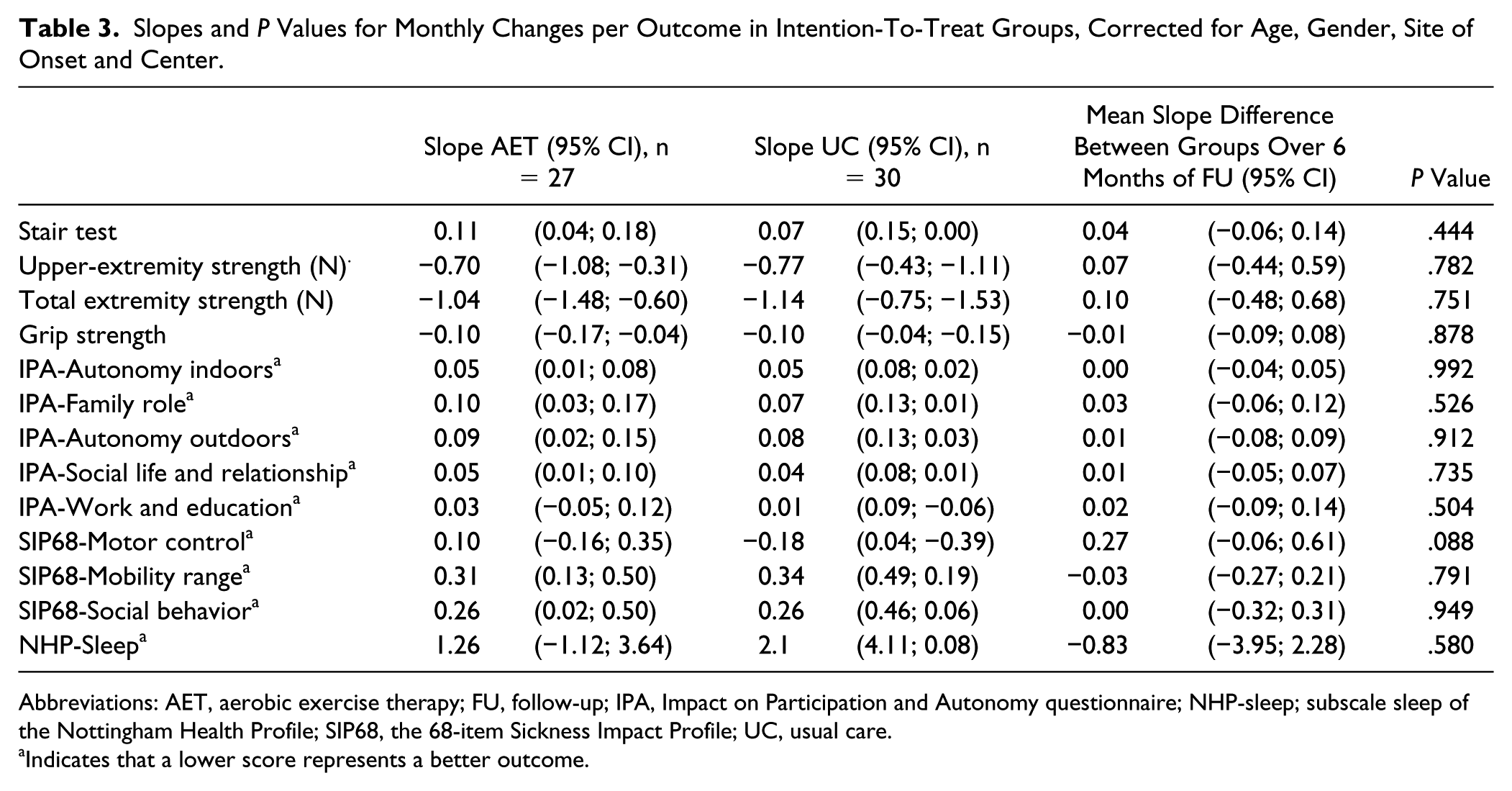
Abbreviations: AET, aerobic exercise therapy; FU, follow-up; IPA, Impact on Participation and Autonomy questionnaire; NHP-sleep; subscale sleep of the Nottingham Health Profile; SIP68, the 68-item Sickness Impact Profile; UC, usual care.
Indicates that a lower score represents a better outcome.
Results of Per-Protocol Analyses
Patients (n = 10) who completed the protocol (attending ≥75% of AET sessions and not lost to follow-up at 10 months) had a less steep ALSFRS-R slope at inclusion (−0.2 vs −0.5, P = .049; Table 1) and a longer median survival time since randomization (28.0 vs 52.2 months, P = .01) than patients who discontinued the protocol (n = 17).
PP analysis was performed with a propensity-matched cohort of 10 versus 10 patients from the AET and UC group, respectively, who completed the protocol and who were well balanced in terms of age, type of onset, slope, FVC, BMI, and gender (Table 1). The slope of disease-specific HRQoL (ALSAQ-40) up to 6 months for patients in the AET PP group was 0.3 (95% CI = −1.1 to 1.6) points per month; this did differ significantly from the UC PP group, with a slope of 2.1 points monthly (95% CI = 0.8 to 3.5; P = .046), in favor of the AET PP group. No significant difference in generic HRQoL, as reflected by the SF-36 (MCS and PCS) score, was detected between the groups.
Regarding the secondary outcomes, the rate of decline in FVC% up to 6 months did differ significantly between groups, in favor of the AET PP group (mean slope difference = 1.7; 95% CI = −0.1 to 3.5; P = .048). A trend was found in the mean slope difference in ALSFR-R scores (mean slope difference of 0.5; 95% CI = −0.1 to 1.1), but this difference was not statistically significant (P = .065; Tables 4 and 5).
Slopes and P Values for Monthly Changes per Outcome in the Propensity-Matched Cohort, Matched for Age, Type of Onset, Slope, FVC, BMI, and Gender.
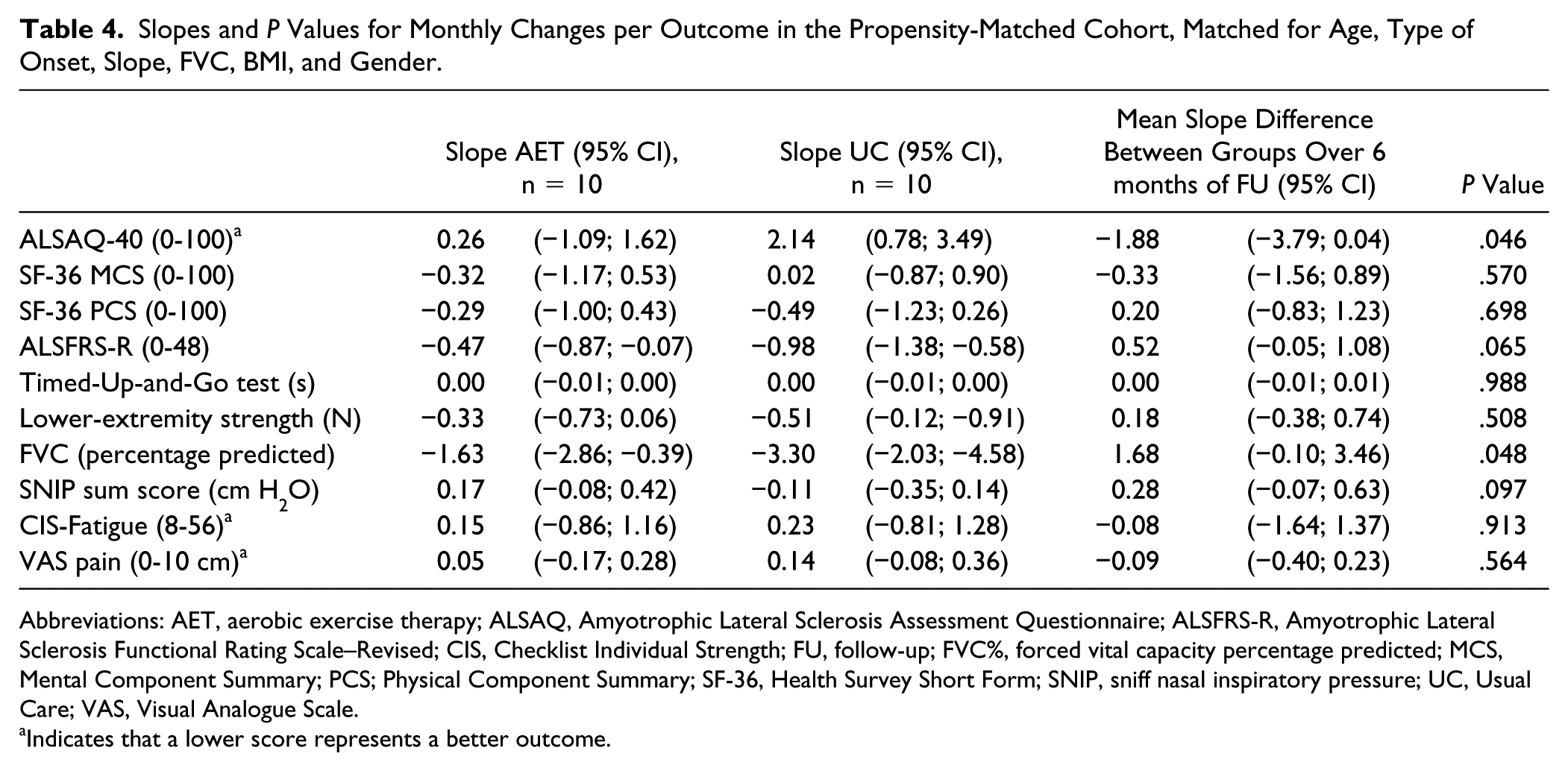
Abbreviations: AET, aerobic exercise therapy; ALSAQ, Amyotrophic Lateral Sclerosis Assessment Questionnaire; ALSFRS-R, Amyotrophic Lateral Sclerosis Functional Rating Scale–Revised; CIS, Checklist Individual Strength; FU, follow-up; FVC%, forced vital capacity percentage predicted; MCS, Mental Component Summary; PCS; Physical Component Summary; SF-36, Health Survey Short Form; SNIP, sniff nasal inspiratory pressure; UC, Usual Care; VAS, Visual Analogue Scale.
Indicates that a lower score represents a better outcome.
Slopes and P Values for Monthly Changes per Outcome in Propensity-Matched Cohort, Matched for Age, Type of Onset, Slope, FVC, BMI, and Gender.
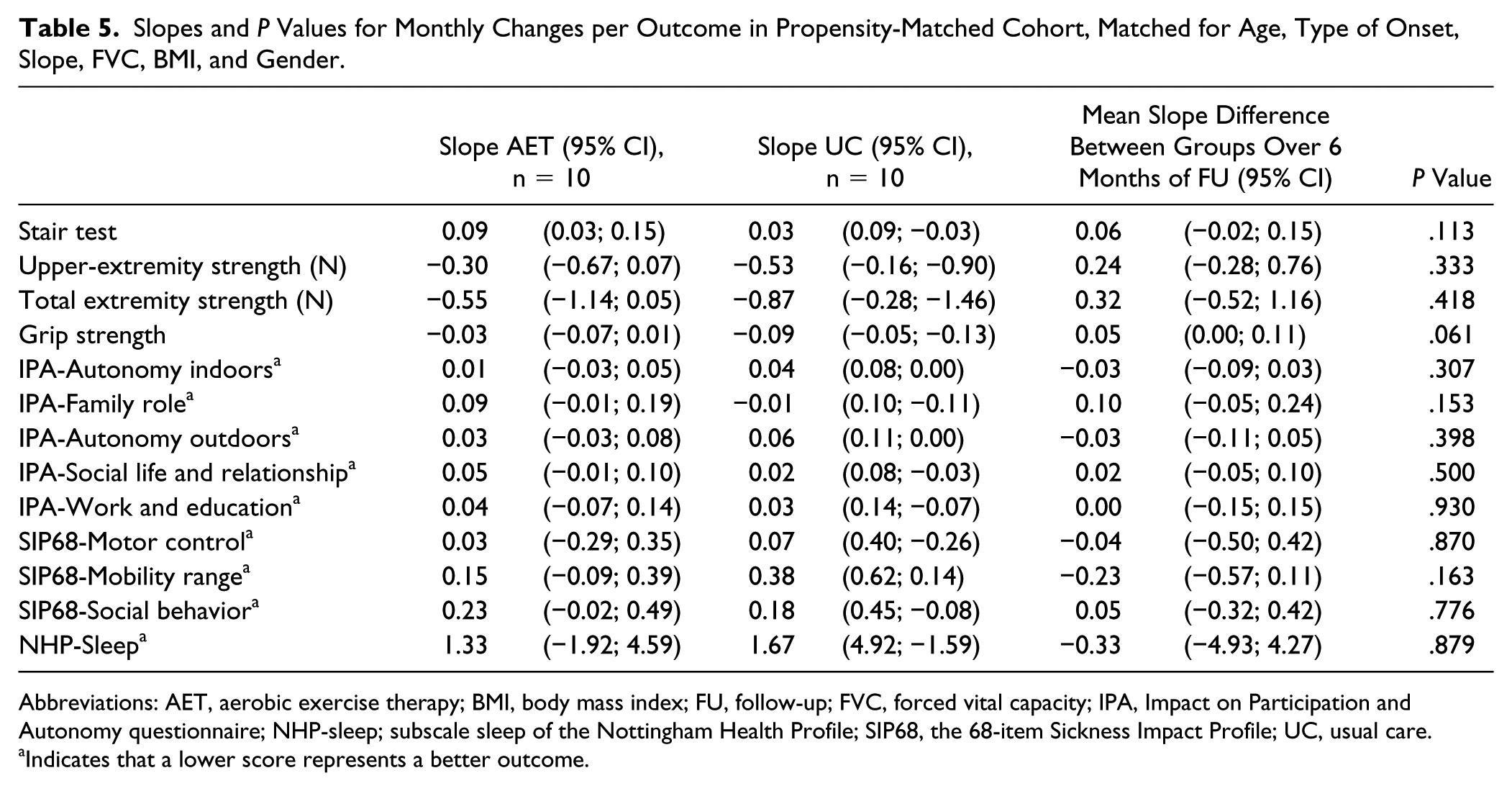
Abbreviations: AET, aerobic exercise therapy; BMI, body mass index; FU, follow-up; FVC, forced vital capacity; IPA, Impact on Participation and Autonomy questionnaire; NHP-sleep; subscale sleep of the Nottingham Health Profile; SIP68, the 68-item Sickness Impact Profile; UC, usual care.
Indicates that a lower score represents a better outcome.
Discussion
The primary objective of the FACTS-2-ALS trial was to study the effectiveness of AET in addition to UC, compared with UC alone, on disease-specific and generic HRQoL in early-stage, ambulatory patients with ALS. No evidence was found for a beneficial effect of AET in addition to UC either on disease-specific or on generic HRQoL over a 6-month follow-up period.
The secondary objective was to assess the effects of AET, in addition to UC, on impairments (aerobic capacity, muscle strength, FVC, SNIP, fatigue, pain), activity limitations (TUG, stair test), and participation restrictions (IPA, SIP68) and to gain further insight into the potential working mechanisms of the AET intervention. Again, there were no effects on the secondary outcomes.
Based on the percentage of patients who were willing (67%) and able (41%) to participate in our AET program, we conclude that AET in addition to UC was not generally feasible for ambulatory ALS patients with relatively good respiratory function. AET was only feasible in a subgroup of ambulatory ALS patients, characterized by a significantly slower disease progression and a significantly longer survival probability.
The absence of effects on HRQoL in the AET group is in line with the aerobic exercise study by Lunetta et al, 13 which also reported no effects on QoL. Our results were, however, inconsistent with previous aerobic and strengthening exercise studies, all of which found a significantly slower decline in global function in the exercise group.13,14 Considering lung function, previous aerobic exercise studies have yielded conflicting results; a significant reduction in the rate of decline in FVC was shown by Pinto, whereas Lunetta et al did not find any effects on FVC after training.13,14
None of the previous aerobic and strengthening exercise studies described the disease progression rate of the included patients at baseline, which could have influenced the outcomes. Dissimilarity in results may also be related to the differences in type, duration, frequency, and intensity of the exercise programs. For example, in the study by Pinto et al, 14 patients had compromised FVC and needed to use BiPAP when completing the aerobic exercise. Training intensity of the treatment program of Lunetta et al 13 was fixed at 60% of patients’ maximal power output for the whole treatment period, not describing adaptations if patients were under- or overtrained during the treatment period.
Only a subgroup of patients was able to complete the AET protocol and was considered to have received enough training stimulus (≥75% training sessions). These patients were characterized by a relatively slow disease progression and a longer survival probability. In this subgroup, we found significant effects on disease-specific HRQoL (ALSAQ-40) and lung function (FVC) at the 6-month follow-up. Also, all secondary outcomes, except for the MCS of the SF-36, showed a positive but not significant trend in favor of the AET PP. These findings may support the idea of a potential protective effect of AET among the “slow progressors.”
The question arises as to whether a physical exercise program of moderate to vigorous intensity and an increase in aerobic capacity are really necessary to optimize HRQoL in patients with ALS. For example, results of a trial in Parkinson’s disease showed that a physically active lifestyle had also beneficial effects on HRQoL. 44 The recommendations on physical activity for the healthy population have been described in the World Health Organization (WHO) Global Recommendations on Physical Activity for Health. 45 This standard is probably also more feasible for patients with ALS.
So far, the effects of exercise on disease progression in the context of the different clinical subtypes and prognostic factors in ALS have not been addressed. As survival for patients with ALS varies greatly, ranging from several months to more than 10 years, stratification of patients by clinical subtype and prognostic factors may strengthen treatment effects and minimize between-subject variability by establishing treatment groups that are more homogeneous with respect to disease progression. 46 Recently, Westeneng et al 47 published a model that can reliably predict survival outcomes at an individual patient level, which allows future ALS studies to improve the potential for stratification of patients for trials, and timing of interventions, making exercise prescription more efficient.
We could not establish our hypothesis that AET can increase or preserve aerobic capacity, and subsequently preserve global function and HRQoL, because aerobic capacity could not be measured at follow-up. 7 In a previous ALS study, however, a strong relationship between aerobic capacity and respiratory muscle function was shown. 5 Moreover, respiratory function, in terms of VC and SNIP, has proven to be moderately to strongly and independently correlated with global function (ALSFRS) and HRQoL.48,49 Our findings among the subgroup who completed the AET protocol seemed in line with these studies because they showed a significantly slower rate of decline in FVC, a trend in preservation of global function (ALSRS-R; P = .065), and a significant maintenance of HRQoL (ALSAQ-40) compared with the UC group. Considering that part of the decline in global function and HRQoL in ALS is possibly a result of a diminished FVC, and AET is not feasible in the general ALS population, respiratory muscle training may in fact be a less burdensome alternative for slowing respiratory decline and maintaining global function and HRQoL. 50
It is difficult to compare our findings on the feasibility of an exercise program in addition to UC in ALS with previous studies11-14 because these reported a willingness to participate of 100%, probably because of a selection bias caused by selecting only patients who were interested in training. Our proportion of 67% is more in accordance with participation rates in exercise programs among incurable cancer patients, of whom 63% were willing to participate in a structured exercise program. 51 In all, 41% of the patients were able to attend 75% or more treatment sessions of our AET program. Compliance in terms of “percentages of training sessions attended,” remained unclear in previous exercise programs11,13,14 and was only partly quantified by DalBello. 12
It may be worthwhile to consider telemedicine systems, to provide supervision while exercising at home and, thereby, increase participation and compliance in exercise programs by including patients who normally decline to participate because of the long travel time to the center. Telemedicine has already been applied for interventions in studies involving patients with ALS52,53 or other neuromuscular diseases with chronic respiratory insufficiency. 54 It has been shown that ALS patients who were managed with telemedicine received the same quality of care 52 as those patients who received standard of care in a medical center. In patients with fascioscapulohumeral muscular dystrophy, the telemedicine system has been shown to be user-friendly and efficient for the home treatment of respiratory physiotherapy and also reduced hospital admissions. 54
Because of the extremely small number of reported adverse events and the absence of serious adverse events, we can conclude that an AET program tailored to the individual patient, with a moderate to vigorous intensity, can be performed without harm in the ALS population in general: increase in cramps, fasciculations, pain, and fatigue was not beyond expectation as a result of typical disease progression. This was in accordance with previous exercise studies among ALS patients, which did not report any (serious) adverse effects.11,13,14
This study is the first aerobic exercise RCT in ALS in ALS that describes the disease progression rate progression rate of the included patients in terms of the ALSFRS-R slope at baseline. This enabled us to analyze which subgroup of patients responded more positively to AET. We used a biphasic randomization model with postponed information. 19 Hence, because patients allocated to the UC group were not aware of the existence of the AET, disappointment or resentful demoralization, or a Hawthorne effect, became less of a problem in the UC group. Another strength of this study is that we used a standardized AET program tailored to the individual patient to avoid the risk of overloading and undertraining. 17 Furthermore, this is the first ALS exercise RCT, which monitored compliance with the protocol using heart rate monitors in addition to logbooks. We tried to overcome a decrease in compliance by maintaining weekly supervised training sessions at the centers; during these, the intervention physiotherapists encouraged the patients to continue the AET.
Generalizability of our study results to the ALS population at large may be considered a limitation because only 57 of the 325 screened patients (17.5%) in 5 centers, took part. Although loss to follow-up was minimized by home assessment if patients were no longer able to visit the clinic, a high drop-out rate in both treatment groups could not be prevented, which resulted in a small sample size at 6 months of follow-up. This might also be a reason for the negative study. Post hoc analysis revealed that the post hoc power was 41% (n = 30 per group). The present study was underpowered because of underestimation of the variation in the design phase of the trial. To detect a difference of 9.1 points (SD = 20) on the ALSAQ-40 with 80% power, we would have needed 77 patients per treatment arm.
Although the small sample size lowered the statistical power of the results, our findings are encouraging for patients with a slow disease progression because all primary and secondary outcomes, except for mental functioning, favored the AET group. Patients in the UC group were not restricted in terms of aerobic exercise activities. As we did not monitor which patients started aerobic exercises on their own, we cannot rule out contamination in the UC group; however, we assumed that our postponed design would prevent patients seeking a similar aerobic intervention.
Conclusions/Implications
AET is not recommended to be included in UC. Because only 10 patients completed the protocol, the study was underpowered to detect a significant benefit from a combination of AET and UC, compared with UC alone, in preserving body functions, activities, participation, and HRQoL. Moreover, AET+UC is not feasible for every ambulatory ALS patient.
Therefore, in the early-stage of ALS, we would recommend that ALS patients preserve a physically active lifestyle according to the WHO Global Recommendations on Physical Activity for Health. 45 Clinicians in multidisciplinary ALS teams have to stimulate and facilitate ALS patients to preserve their physically active lifestyle.
Furthermore, AET did not seem harmful and PP analyses showed that AET may preserve respiratory function and disease-specific HRQoL in ALS patients with a relatively slow disease progression and a longer survival probability. Therefore, AET can be offered in this subgroup of patients.
Future research should confirm the subgroup benefits. To select “slow progressors,” survival prediction models could be used to establish treatment groups that are more homogeneous with respect to disease progression. Applying telemedicine systems may increase patient comfort and tolerance and consequently decrease drop-out rates.
Footnotes
Acknowledgements
The authors thank the patients who participated in this trial.
Authors’ Note
Johanna M. A. Visser-Meily and Leonard H. van den Berg are principal investigators and contributed equally to this work. Clinical trial registration number: Netherlands National Trial Register (NTR): 1616.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Netherlands Organization for Health Research and Development (ZonMw) and the Prinses Beatrix Spierfonds (PBS). The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report.
