Abstract
Parkinson’s disease (PD) patients experience a range of non-motor symptoms that are believed to be related to disease pathophysiology, many of which are treatable by medications. Among newly-diagnosed PD participants in the Parkinson’s Progression Markers Initiative study, we describe (1) the frequency of medication use for common non-motor symptoms, and (2) when non-motor symptomatic treatment was initiated relative to PD diagnosis. Non-motor medication use was reported by 73% of participants, most commonly for depression, constipation, and anxiety. Treatment of some non-motor symptoms, notably depression, antedated diagnosis. These data may be useful for studies of non-motor symptoms in PD.
INTRODUCTION
Non-motor symptoms significantly contribute to decreased quality of life in people with Parkinson’s disease (PD) [1–4]. A recent MDS-commissioned review detailed non-motor symptoms treatments, efficacy, safety, and practice implications [5]. Using data from the Parkinson’s Progression Markers Initiative (PPMI) observational study, we report medication use for common non-motor symptoms (anxiety, bladder control, cognition, constipation, depression, fatigue, hypotension, psychosis, sexual dysfunction, sialorrhea, and sleep disturbance) in the newly-diagnosed PD (de novo) cohort, for which there are few treatment restrictions.
METHODS
Data source
Data were obtained from the PPMI database (accessed 1/7/2020). PPMI is a 33-site (United States [21], Europe [10], Australia [1], and Israel [1]), longitudinal, observational study to identify biomarkers of PD development and progression [6]. Additional details can be found at https://www.ppmi-info.org/. Time in the study was derived from the ‘Vital Signs’ data, measures taken at every study visit. Data described herein detail the entire period of observation at the time of download, including pre-study medication initiation. Medications were reported at every study visit.
Drug cleaning and query
Drug records were curated from the file ‘Concomitant_Medications’ by converting free-text entries to a single generic drug name using Lexicomp (Wolters Kluwer) or Micromedex (IBM). Drug indications were confirmed using Micromedex or drug prescribing information. The following non-motor drugs were identified in the dataset: (1) Anxiety: alprazolam, bromazepam, buspirone, clonazepam, delorazepam, diazepam, hydroxyzine, lorazepam, medazepam, opipramol, oxazepam; (2) Bladder control: darifenacin, fesoterodine, mirabegron, oxybutynin, propiverine, solifenacin, tolterodine, trospium; (3) Cognition: donepezil, galantamine, memantine, rivastigmine; (4) Constipation: bisacodyl, linaclotide, lubiprostone, and over-the-counter (OTC) products; (5) Depression: agomelatine, amitriptyline, amitriptyline-medazepam, brexpiprazole, bupropion, citalopram, desipramine, desvenlafaxine, doxepin, duloxetine, escitalopram, fluoxetine, fluvoxamine, imipramine, maprotiline, mirtazapine, nefazodone, nortriptyline, paroxetine, reboxetine, sertraline, tranylcypromine, trazodone, venlafaxine, vilazodone, vortioxetine; (6) Fatigue: armodafinil, modafinil; (7) Hypotension: droxidopa, fludrocortisone, midodrine; (8) Psychosis: aripiprazole, clozapine, lurasidone, olanzapine, pimavanserin, prochlorperazine, quetiapine, risperidone, ziprasidone; (9) Sexual dysfunction: sildenafil, tadalafil, vardenafil; (10) Sialorrhea: atropine, glycopyrrolate, rimabotulinumtoxinB; (11) Sleep: brotizolam, clomethiazole, doxepin, eszopiclone, flurazepam, melatonin, phenobarbital, suvorexant, temazepam, triazolam, zaleplon, zolpidem, zopiclone.
Medication restrictions for the PPMI de novo cohort include (1) medications that may interfere with DaTSCAN or VMAT-2 PET for a period of time before imaging, (2) PD motor medications at baseline, within 60 days before baseline, and prior use ≥60 days, (3) investigational drugs/devices within 60 days before baseline, and (4) anticoagulants that would interfere with lumbar puncture. Additionally, de novo participants could not have a diagnosis of dementia at baseline (as determined by a site investigator) and were not expected to require motor medications within six months of baseline.
RESULTS
The average duration of follow-up for the de novo cohort from screening was 6.1±2.0 years (range 0–9.2). Out of the 422 de novo PD participants identified, 308 (73%) reported at least one of the non-motor drug categories of interest. The time to initiate pharmacologic treatment for a specific non-motor symptom relative to PD diagnosis is displayed in Fig. 1. Symptomatic treatment of depression was most common overall, with 50 initiating an antidepressant before and 123 participants at or after PD diagnosis. Constipation was the next most commonly treated non-motor symptom and included 31 participants initiating constipation medication use before PD diagnosis and 103 at or after diagnosis. Sexual dysfunction had the highest proportion of treatment initiation to occur before PD diagnosis (26 before diagnosis; 30 at or after diagnosis). Conversely, treatment of cognitive dysfunction, psychosis, orthostatic hypotension, and sialorrhea was always initiated after PD diagnosis in this cohort. The top three drugs used for each symptom category are detailed in Table 1. The least commonly-treated symptoms were fatigue (n = 8) and sialorrhea (n = 3). However, fatigue treatment did not consider caffeine and amphetamine (or derivatives).
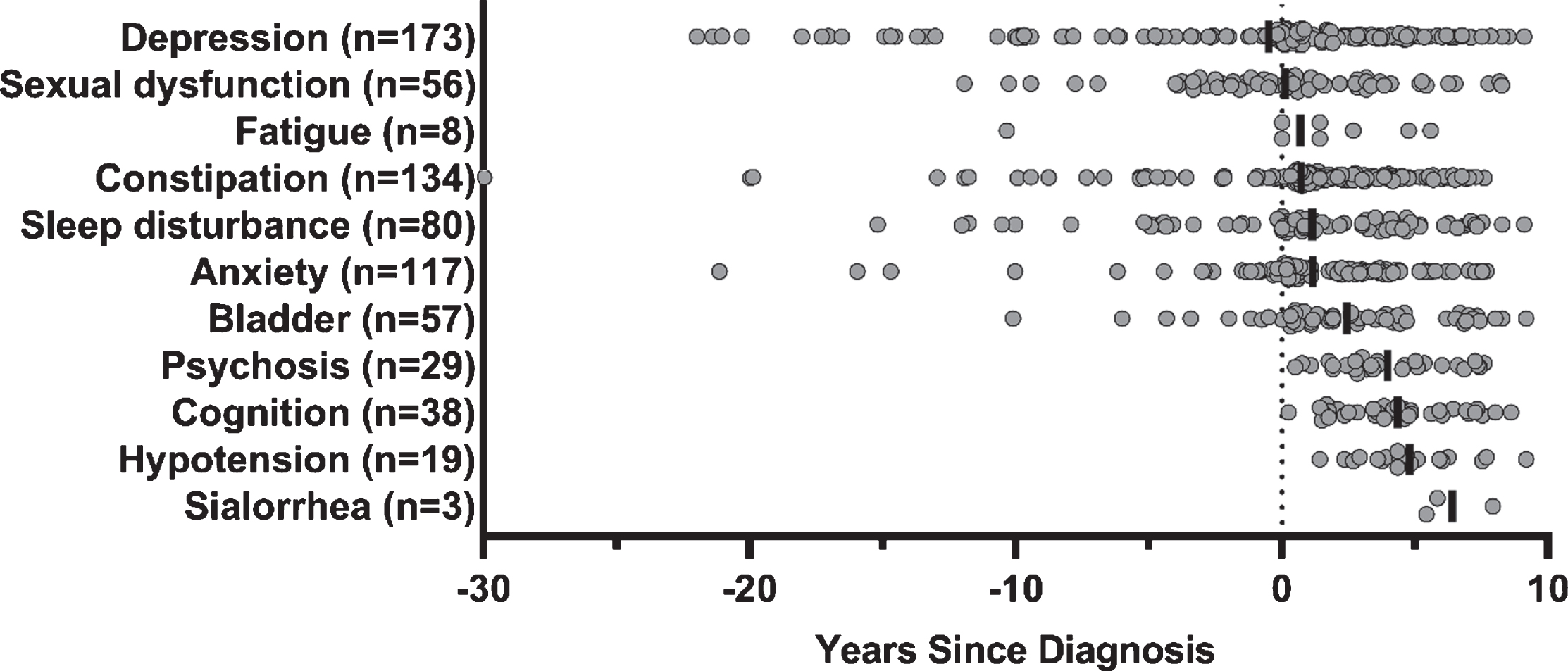
Time to initiate non-motor symptom treatment, relative to Parkinson’s disease diagnosis. Non-motor symptomatic treatment initiation, prior to study participation and throughout follow-up. Vertical solid black line denotes average years to initiate treatment relative to Parkinson’s disease diagnosis, and circles denote an individual participant’s first treatment for a given non-motor symptom. Depression (–0.5±6.2), sexual dysfunction (0.1±4.4), fatigue (0.7±4.9), constipation (0.7±5.4), sleep disturbance (1.1±4.9), anxiety (1.2±4.0), bladder (2.4±3.6), psychosis (4.0±2.1), cognition (4.3±2.1), hypotension (4.8±2.0), and sialorrhea (6.4±1.3). Two records (one for escitalopram; one for a PEG 3350-based laxative) were removed from the figure due to no available start date. Over the duration of study follow-up, 110 of the 308 (35.7%) were treated for one non-motor symptom category, 89 (28.9%) for two, 50 (16.2%) for three, 35 (11.4%) for four, 15 (4.9%) for five, five (1.6%) for six, and three (1.0%) for seven, and one (0.3%) for eight symptom categories.
Top three drugs reported, by Parkinson’s disease non-motor symptom
Individual drug report ‘n’ may exceed the total number of participants treated for the non-motor symptom, as some subjects reported using more than one drug within the symptom category, and medication use may have changed during the study. Due to the low incidence of drug use in this cohort, data for fatigue and sialorrhea are not displayed. *Docusate and PEG 3350 use were equally reported. †Olanzapine, pimavanserin, and prochlorperazine were each reported by one participant.
Anxiety and depression
Anxiolytics were reported by 117 participants. Clonazepam was used by 60 participants (14.2%), followed by alprazolam (n = 32, 7.6%), lorazepam (n = 29, 6.9%), diazepam (n = 16, 3.8%), hydroxyzine (n = 6, 1.4%), bromazepam (n = 2, 0.5%), opipramol (n = 2, 0.5%), and buspirone (n = 1, 0.2%). Antidepressant use was reported by 174 participants. The most common were citalopram (n = 41, 9.7%), escitalopram (n = 34, 8.1%), sertraline (n = 33, 7.8%), trazodone (n = 26, 6.2%), bupropion (n = 25, 5.9%), mirtazapine (n = 25, 5.9%), venlafaxine (n = 23, 5.5%), fluoxetine, (n = 15, 3.6%), amitriptyline (n = 12, 2.8%), duloxetine (n = 12, 2.8%), and paroxetine (n = 11, 2.6%). Other drugs were each reported by <1% of the de novo PD cohort. Importantly, several pharmacotherapies are indicated for both anxiety and depression, and some can address other PD-related symptoms including sleep disturbance, pain, and urinary incontinence.
Constipation
We identified 135 participants who took constipation medications. The most common treatments were PEG 3350-containing or docusate-containing (each n = 44, 10.4%), psyllium (n = 26, 6.2%), senna/sennosides (n = 24, 5.7%), and bisacodyl (n = 13, 3.1%). Use of prescription drugs linaclotide (n = 7, 1.7%) and lubiprostone (n = 3, 0.7%) was less common, compared to OTC treatments.
Sleep
Eighty participants reported sleep medications, and one additional participant reported doxepin but as an antidepressant. The most commonly used drugs were melatonin (n = 49, 11.6%), zolpidem (n = 27, 6.4%), temazepam (n = 7, 1.7%), and eszopiclone (n = 5, 1.2%). Several OTC medications contain hypnotic active ingredients (e.g., diphenhydramine, doxylamine). However, as it can be difficult to determine whether the drug is being used for sleep or other purposes, we only reported melatonin.
Urinary urgency and incontinence
Fifty-seven participants (40 male) reported a drug for urinary dysfunction. Mirabegron was the most commonly used (n = 27, 6.4%), followed by solifenacin (n = 20, 4.7%), oxybutynin (n = 16, 3.8%), and trospium (n = 12, 2.8%). Other drugs were each reported by <1% of participants. However, at least 83 participants reported taking pharmacotherapies indicated for benign prostatic hyperplasia (e.g., alfuzosin), which may also alleviate urinary symptoms.
Sexual dysfunction
Fifty-six participants (all male) reported erectile dysfunction-treating drugs. Sildenafil was the most commonly reported (n = 39, 9.2%), followed by tadalafil (n = 22, 5.2%), and vardenafil (n = 4, 0.9%). For female participants, it is challenging to determine whether a drug is being used for sexual dysfunction, contraception, menopause, or other symptoms. At least 22 participants (5.2%) reported taking a drug containing estrogen, estradiol, or estriol; none reported taking flibanserin or ospemifene.
Cognition
Thirty-eight participants reported antidementia medication use. The most commonly reported medication was donepezil (n = 23), taken by 5.5% of all de novo participants. Rivastigmine was used by 19 participants (4.5%), followed by memantine (n = 7, 1.7%) and galantamine (n = 1, 0.2%). Interestingly, rivastigmine the only drug specifically indicated for PD-dementia.
Psychosis
Twenty-nine participants reported antipsychotics, with quetiapine taken by 24 people (5.7%) and clozapine by four (0.9%). Other drugs were each reported by one person, though the indicated use for prochlorperazine was related to vertigo, nausea, and dizziness.
Orthostatic hypotension
Nineteen participants reported the use of agents for orthostatic hypotension; midodrine was used by 13 (3.1%), followed by fludrocortisone (n = 11, 2.6%) and droxidopa (n = 3, 0.7%). The low incidence of droxidopa use may be due to it being a relatively newer drug, approved by the Food and Drug Administration (FDA) in 2014.
DISCUSSION
Treatment analysis can provide insight into the timing, frequency, and nature of the treatments utilized. For instance, there was more treatment heterogeneity for depression compared to the treatment of other symptoms; the top three antidepressants combined (citalopram, escitalopram, and sertraline) made up 62.1% of all antidepressants used in this cohort. In contrast, 82.8% of participants taking an antipsychotic reported quetiapine, despite numerous drug options and an FDA-approved drug specifically for PD psychosis (pimavanserin). The source of the medication information prior to formal reconciliation as part of a clinical trial or electronic health record (EHR) should be taken into consideration, though it may not necessarily negate the accuracy of the data [7–9].
There are limitations to our approach, namely our emphasis on prescription medications indicated to treat specific symptoms, which does not take into consideration (1) off-label use of these medications or (2) off-label use of other drugs (prescription or OTC) for these symptoms. Given the data, we believed this approach would provide an overview of treatment utilization while minimizing assumptions. A detailing of OTC interventions for motor and non-motor symptoms is the subject of ongoing research. Further, while pain is receiving growing recognition as an important non-motor symptom, we were unable to reliably distinguish the cause for using prescription and OTC pain relievers and had to omit it from this manuscript.
When studying manifest or prodromal PD, non-motor symptom treatment may serve as a proxy for symptom onset date. These non-motor symptoms are not unique to PD, and future, more in-depth analyses could quantify symptom prevalence between groups and determine differences in treatment utilization. Performing analyses in larger cohorts or local patient populations may allow for cohort comparisons, provide more generalizable information, or aid in the selection of study inclusion/exclusion criteria. Further, treatment utilization in longitudinal study cohorts may reveal differences in prescribing patterns across geographic regions, as drugs are approved or discontinued, or as new research becomes available.
Lastly, unlike motor assessments, which often account for motor (i.e., dopaminergic) drugs and timing (e.g., MDS-UPDRS III on/off scores), most non-motor assessments do not consider the use of treatments that can affect symptoms. Pharmacological and non-pharmacological treatments and duration of use could be taken into consideration as covariates when researching non-motor symptom-related outcomes or used to quantify treatment effect.
CONFLICT OF INTEREST
Dr. Shoulson, Dr. Kieburtz, Dr. Venuto, and Dr. Javidnia receive and/or have received research support from the Michael J Fox Foundation for Parkinson’s Research (New York, NY). The authors have no other conflicts of interest to report.
Footnotes
ACKNOWLEDGMENTS
Research reported in this publication was supported by the National Institute of Neurological Disorders and Stroke of the National Institutes of Health under Award Numbers P50NS108676 and T32 NS007338. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Dr. Javidnia was supported by The Michael J. Fox Foundation Fellowship in Regulatory Science. The authors would like to thank Dr. Joe Kane for his guidance on psychiatric medications. Data used in the preparation of this article were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (www.ppmi-info.org/data). For up-to-date information on the study, visit ![]() . PPMI—a public-private partnership—is funded by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including AbbVie, Allergan, Amathus Therapeutics, Avid Radiopharmaceuticals, Biogen, BioLegend, Bristol-Myers Squibb, Celgene, Denali, GE Healthcare, Genentech, GlaxoSmithKline, Janssen Neuroscience, Eli Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi Genzyme, Servier, Takeda, Teva, UCB, Verily, and Voyager Therapeutics.
. PPMI—a public-private partnership—is funded by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including AbbVie, Allergan, Amathus Therapeutics, Avid Radiopharmaceuticals, Biogen, BioLegend, Bristol-Myers Squibb, Celgene, Denali, GE Healthcare, Genentech, GlaxoSmithKline, Janssen Neuroscience, Eli Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi Genzyme, Servier, Takeda, Teva, UCB, Verily, and Voyager Therapeutics.
