Abstract
Background:
Autonomic dysfunction and depression are common non-motor symptoms of Parkinson’s disease (PD) that confer poorer prognosis. These PD symptoms may have overlapping pathophysiologic underpinnings.
Objective:
To investigate associations between autonomic and depression symptoms in early PD, and their evolution over time.
Methods:
We obtained data from the Parkinson’s Progression Markers Initiative, a prospective open-access database of early PD. Regression analyses were used to model effects of depression on autonomic symptoms in controls and in PD at baseline, visit 6 (24 months after baseline), and visit 12 (60 months after baseline), correcting for multiple comparisons.
Results:
Data from 421 people with PD at baseline, 360 at visit 6, 300 at visit 12, and 193 controls were included. When controlling for age, depression, and anti-hypertensive medications, depression predicted autonomic symptoms in all groups. Accounting for motor symptoms did not alter these associations. When comparing groups, the influence of depression on autonomic symptoms was stronger in all PD groups compared to controls, and strongest in PD at visit 12. Depression predicted the presence of orthostatic hypotension only in the PD group at visit 12.
Conclusion:
We demonstrated the important impact of depression on autonomic symptoms in early and middle stages of PD, which are independent of motor symptoms. Though the physiologic basis of these two PD symptoms are not fully understood, our findings add to pathologic evidence of a shared mechanistic substrate, separate from that responsible for PD motor symptoms. These findings may influence clinical management and development of novel therapies.
INTRODUCTION
Parkinson’s disease (PD) is defined by the presence of specific motor symptoms, including rigidity, bradykinesia, and tremor [1]. Depression and autonomic nervous system dysfunction (AuD) are common and often debilitating non-motor symptoms (NMS) of PD, which impact over 50% of people with PD and portend a poorer prognosis [2–7]. Depression and AuD increase in prevalence with increasing age and PD disease duration [8, 9], but the pathophysiology of these two NMS remains poorly understood. AuD and depression in PD are often refractory to treatment with the limited arsenal of medications and interventions currently available [8].
Despite the increasing prevalence and severity of all PD symptoms with longer disease duration, studies investigating associations between AuD and other motor and non-motor symptoms show conflicting results [10–12]. This may indicate the presence of symptom clusters in PD, rather than a common pathophysiologic underpinning to all symptoms of the disease [12]. This is particularly important as the current treatment of PD symptoms often gravitates around dopaminergic pathways, known to be responsible for motor symptoms, or around conventional treatment for those symptoms in non-PD cohorts [8, 13]. Identifying alternative systems responsible for specific PD symptom clusters is vital to the development of targeted and effective therapies. Available evidence points to significant overlap between AuD and depression in PD, both pathologically and by the shared involvement of norepinephrine neurotransmission [14–20], yet currently these symptoms are treated in isolation. Prior studies attempting to delineate clinical associations between various PD NMS have had difficulty disentangling the effects of disease duration and age from true overlapping pathophysiology of these symptoms, at least in part due to the heterogeneity of the PD study samples, recruitment bias, and/or lack of longitudinal data [11, 21]. A clearer understanding of the overlap of these two influential NMS may lead to a synergistic approach to treatment. This in turn may improve the rate of diagnosis of these symptoms, enhance planning of treatment regimens, and advance the development of effective treatment targets for NMS in PD.
To help address this gap, we investigated longitudinal associations between depression symptoms and AuD in a large prospective cohort of people with early PD and controls. We hypothesized that depression symptoms associate with AuD in PD at early stages of disease, with increased association as the disease progresses and pathologic burden increases. We hypothesized that these findings would be independent of age and motor symptom severity. This finding in a prospective clinical sample of people with PD would strengthen prior pathological and neuroimaging data implicating overlapping pathophysiology of depression and AuD in PD [14–18, 20].
METHODS
Subjects
Data for the current study were obtained from the Parkinson’s Progression Markers Initiative (PPMI) [22], a prospective longitudinal database of early PD (baseline visit is within 2 years of disease onset). PPMI participants include people with Parkinson’s disease as well as neurologically healthy controls who undergo validated clinical evaluations, symptom questionnaires, and collection of neuroimaging and biofluid samples. Participants must not be taking medication for PD within 6 months of the baseline visit to be included in the study. The PPMI study was approved by local Institutional Review Boards of all participating centers, and informed consent was obtained from all participants prior to enrollment into the study. For up-to-date information on the study, visit https://www.ppmi-info.org. As PPMI is an ongoing prospective study, we used data that was collected as of 2/1/2021.
We used data from the baseline visit, visit 6 (24 months after baseline visit), and visit 12 (5 years after baseline visit) from all PD participants with confirmed abnormalities on 123I-ioflupane SPECT or DATScan (indicating presence of dopaminergic deficits) without genetic variants known to be associated with PD diagnosis, and who had complete data for all variables used in these analyses. Participants with genetic variants known to be associated with PD were excluded due to variable neuropathologic findings among those with genetic variants [23]. We used data from all controls without missing data from baseline in all examined variables. Controls who were found to have genetic variants known to be associated with PD were excluded from this analysis due to the possibility of subclinical PD.
Clinical data
Demographic data, including date of birth and gender, was obtained at the initial screening visit. Medication data, orthostatic vital signs, clinical evaluations, and questionnaires performed at each analysis time point were extracted from the database. Autonomic symptoms were assessed using the SCales for Outcomes in PArkinson’s – autonomic (SCOPA-AUT) [24], a validated questionnaire assessing gastrointestinal (7 questions), urinary (6 questions), cardiovascular (3 questions), pupillary (1 question), thermoregulatory (4 questions), and sexual symptoms (2 questions) in PD. Depression symptoms were assessed using the Geriatric Depression Scale (GDS), a validated depression symptoms screen [25]. Motor symptoms were assessed using the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part III motor exam [26]. For each scale, higher scores indicate higher burden of symptoms. Orthostatic hypotension was defined as a reduction in systolic blood pressure of at least 20 mmHg or a reduction in diastolic blood pressure of at least 10 mmHg between supine and standing measurements.
Statistical analysis
All statistical analyses were performed with SAS 9.4 statistical software (SAS Institute Inc., Car, NC, USA). Descriptive statistics were calculated for all variables. For continuous variables, mean, range, and standard deviation are presented. For binomial variables, percentages are presented. Symptom rating scale scores failed test of normality using Shapiro-Wilk test. Mann-Whitney-U test was used to compare means between two groups (PD, no PD; gender; presence of orthostatic hypotension (OH), use of depression and antihypertensive medications). Spearman correlation was used to identify associations between continuous and ordinal variables. Linear regression was used for prediction modeling of the effects of depression symptoms and motor symptoms on autonomic symptoms while controlling for confounding variables. Logistic regression was used for prediction modeling of the effects of depression and motor symptoms on OH while controlling for confounders. Interaction terms were added to the regression models to investigate differences between groups (control vs. PD at baseline vs. PD at visit 6 vs. PD at visit 12). p < 0.05 was the cutoff for statistical significance. Bonferroni correction was applied to correct for multiple comparisons in final regression models.
RESULTS
Demographic and clinical data
We included data from 421 PD participants at baseline, 360 PD participants at visit 6 (24 months), 300 PD participants at visit 12 (5 years), and 193 controls. Sexual symptoms were not included in the total SCOPA-AUT calculations for the purposes of these analyses due to high and unequal proportions of non-responders in each group (68/421 (16.2%) PD participants at baseline, 68/360 (18.9%) PD participants at visit 6, 63/300 (21.0%) of PD participants at visit 12, and 28/193 (14.5%) of control participants). Demographic data and descriptive statistics for all variables used in the analysis are presented in Table 1.
Demographics and descriptive statistics
SCOPA-AUT, SCales for Outcomes in PArkinson’s – Autonomic; SCOPA-GI, Gastrointestinal symptoms; SCOPA-Uri, Urinary symptoms; SCOPA-CV, Cardiovascular symptoms; #p < 0.05 compared to controls, *p < 0.001 compared to controls.
Total SCOPA-AUT (U(419) = 43342.5, Z = –7.865, p < 0.0001), as well as subscores in gastrointestinal symptoms (U(419) = 39948.5, Z = –9.820, p < 0.0001), urinary symptoms (U(419) = 50427.0, Z = –4.411, p < 0.0001), and cardiovascular symptoms (U(419) = 51588.5, Z = –4.836, p < 0.0001), were higher in PD at baseline compared with controls. Total SCOPA-AUT (U(358) = 33753.0, Z = –11.028, p < 0.0001), gastrointestinal symptoms (U(358) = 30981.0, Z = –12.816, p < 0.0001), urinary symptoms (U(358) = 41858.0, Z = –6.538, p < 0.0001), cardiovascular symptoms (U(358) = 43788.5, Z = –6.54, p < 0.0001), and the prevalence of OH (U(358) = 51464.0, Z = –1.848, p = 0.032) were higher in PD at visit 6 compared with controls. Total SCOPA-AUT (U(298) = 28981.0, Z = –12.127, p < 0.0001), gastrointestinal symptoms (U(298) = 26267.5, Z = –14.120, p < 0.0001), urinary symptoms (U(298) = 35568.5, Z = –7.889, p < 0.0001), cardiovascular symptoms (U(298) = 38797.5, Z = –6.935, p < 0.0001), and the prevalence of OH (U(298) = 45,363.5, Z = –2.40, p = 0.008) were higher in PD at visit 12 compared with controls (Fig. 1; Supplementary Figure 1). When comparing autonomic symptoms in PD at different time points, total SCOPA-AUT, as well as gastrointestinal, urinary, and cardiovascular subscores were higher at visit 6 and visit 12 compared to baseline, and higher in visit 12 compared to visit 6. Though prevalence of OH is higher with increasing PD disease duration in this cohort, these differences were not statistically significant between PD groups (Table 1).
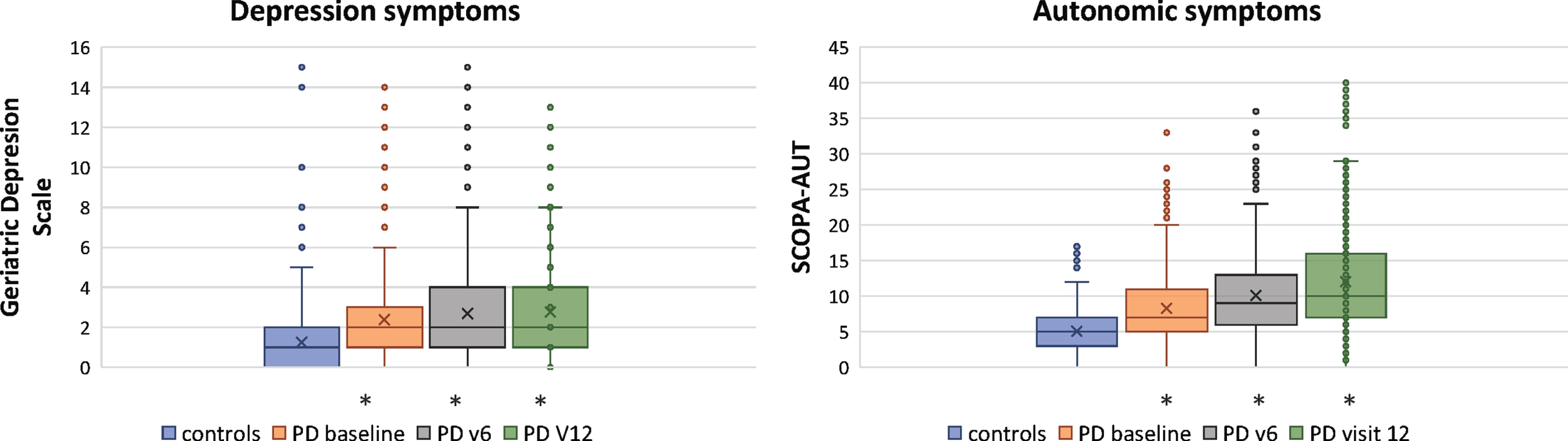
Burden of autonomic and depression symptoms in Parkinson’s disease. α SCOPA-AUT, SCales for Outcomes in PArkinson’s disease – autonomic; *p < 0.001 PD vs. control.
GDS was higher in PD at baseline (U(419) = 45,220.0, Z = –7.091, p < 0.0001), at visit 6 (U(358) = 41,071.5, Z = –7.093, p < 0.0001), and at visit 12 (U(298) = 36,097.0, Z = –7.672, p < 0.0001) compared to controls. There was no difference in GDS between PD at baseline and PD at visit 6 (U(358) = 143,299.5, Z = 0.821, p = 0.206), or between PD at visit 6 and PD at visit 12 (U(298) = 101,195.5, Z = 0.850, p = 0.198). GDS was higher in PD at visit 12 compared to PD at baseline (U(298) = 112,916.0, Z = 1.700, p = 0.045) (Fig. 1). Motor symptoms were more severe in PD at all time points compared to controls, and the severity of motor symptoms in PD increased at each consecutive time point (Table 1).
Associations between clinical variables
Symptoms of depression were significantly associated with total autonomic symptom burden in PD at baseline (r(419) = 0.366, p < 0.0001), at visit 6 (r(358) = 0.382, p < 0.0001), at visit 12 (r(298) = 0.523, p < 0.0001), and in controls (r(191) = 0.220, p = 0.002), with stronger associations in PD groups compared to controls (Fig. 2). When evaluating sub-categories of the SCOPA-AUT, depression symptoms were associated with cardiovascular symptoms, urinary symptoms, and gastrointestinal symptoms in PD at baseline, visit 6, and visit 12, as well as in controls (Supplementary Figure 2).
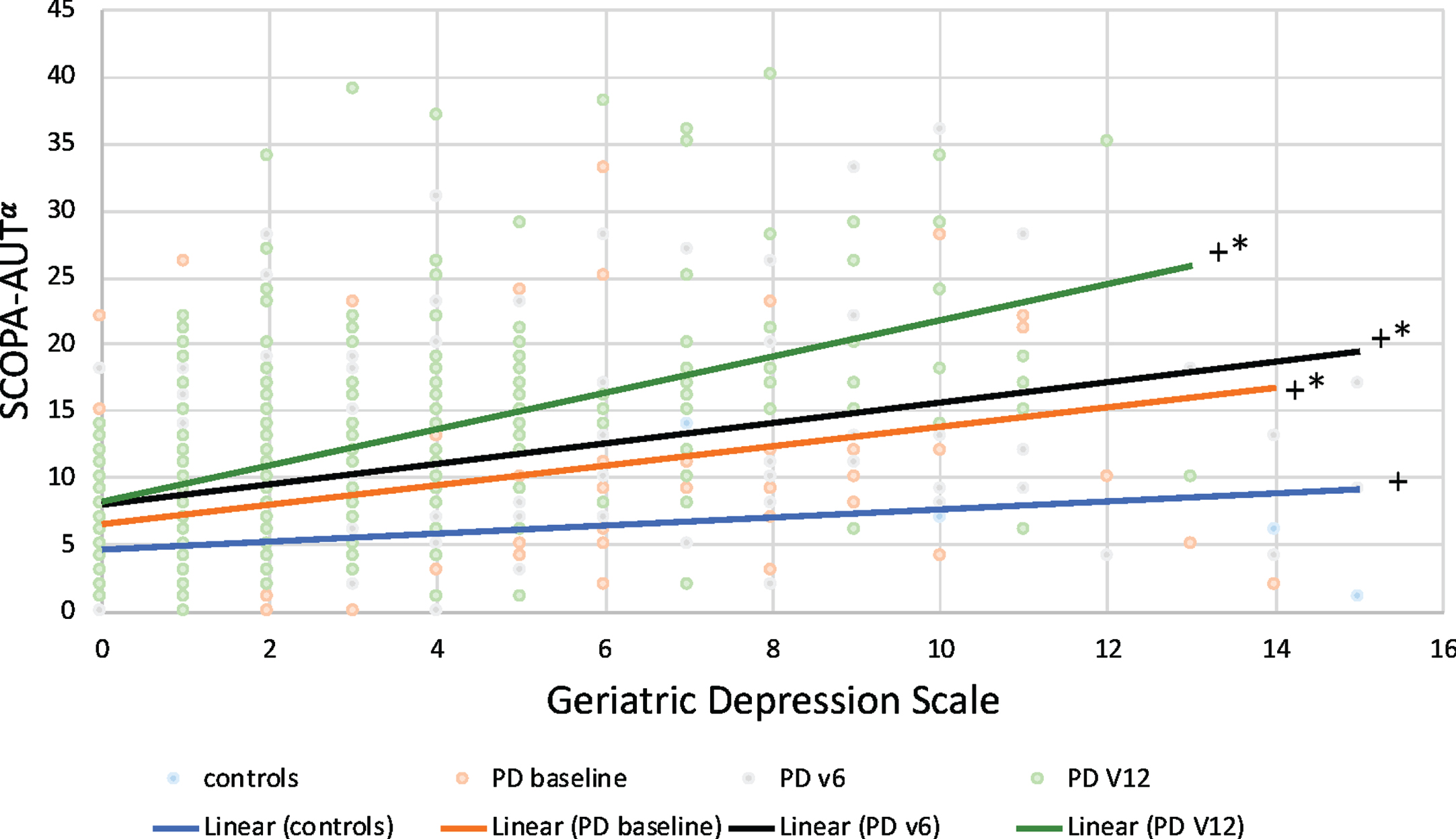
Associations between autonomic symptoms and depression symptoms in Parkinson’s disease. α SCOPA-AUT, SCales for Outcomes in PArkinson’s disease – autonomic; +p < 0.05 for association between SCOPA-AUT and GDS; *p < 0.05, PD vs. controls.
Interestingly, though there was a positive trend, the presence of OH did not correlate with autonomic symptoms in the control or baseline PD group. The presence of OH did correlate with total autonomic symptoms (U(358) = 12,169.50, Z = 2.35, p = 0.0097) and urinary symptoms (U(358) = 12,212.00, Z = 2.42, p = 0.0080) in the PD group at visit 6, but not with cardiovascular or gastrointestinal symptoms after correction for multiple comparisons. The presence of OH strongly correlated with total autonomic symptom burden (U(298) = 11,248.0, Z = 5.12, p < 0.0001) as well as cardiovascular (U(298) = 10,227.0, Z = 3.73, p = 0.0001), gastrointestinal (U(298) = 10,971.5, Z = 4.67, p < 0.0001), and urinary subscores (U(298) = 10,843.0, Z = 4.43, p < 0.0001) at visit 12.
Covariate analysis
There were correlations between age and autonomic symptoms in PD at baseline (r(419) = 0.184, p < 0.0001), at visit 6 (r(358) = 0.151, p = 0.004), and at visit 12 (r(298) = 0.219, p < 0.0001), as well as in controls (r(191) = 0.301, p < 0.0001). PD participants with OH had higher burden of motor symptoms at baseline (U(419) = 14,132.0, Z = 1.687, p = 0.046), visit 6 (U(358) = 13,119.0, Z = 3.652, p = 0.0001), and at visit 12 (U(298) = 9,260.5, Z = 1.691, p = 0.046). There was no association between presence of OH and motor scores in controls. There were no associations between depression symptoms and motor scores in controls, or in PD at baseline or visit 6. There was an association between depression symptoms and motor scores at visit 12 (r(298) = 0.233, p < 0.0001).
66/193 (34.2%) of control participants, 148/421 (35.2%) of PD participants at baseline, 125/360 (34.7%) of PD participants at visit 6, and 100/300 (33.3%) of PD participants at visit 12 were taking medications that lower blood pressure. 8/193 (4.1%) of control participants, 26/421 (6.2%) of PD participants at baseline, 21/360 (5.8%) of participants at visit 6, and 20/300 (6.7%) of PD participants at visit 12 were taking medications to treat depression. We next examined whether taking medications to treat depression, or taking medications that lower blood pressure and may therefore worsen autonomic symptoms, may influence these results. There were associations between antihypertensive medication use and autonomic symptoms in controls (U(193) = 7,458.0, Z = 2.88, p = 0.002), but not in PD at baseline, visit 6, or visit 12. Depression symptom scores were higher in those taking antidepressant medications among controls (U(193) = 1,057.0, Z = 1.92, p = 0.028), PD at baseline (U(419) = 6,894.0, Z = 2.38, p = 0.0088), PD at visit 6 (U(358) = 5,472.5, Z = 3.22, p = 0.0007), and PD at visit 12 (U(298) = 4,012.5, Z = 2.71, p = 0.004).
Predictors of autonomic symptoms
When controlling for age, depression medication use, and anti-hypertensive medication use, and correcting for multiple comparisons, depression symptoms predicted autonomic symptoms in PD at baseline (β= 0.76, p < 0.001), visit 6 (β= 0.76, p < 0.001), and at visit 12 (β= 1.33, p < 0.001), as well as in controls (β= 0.29, p = 0.018) (Table 2).
Regression models of depression symptoms predicting autonomic dysfunction1
1Each dependent variable modeled separately. All models are corrected for age, depression medication use, and antihypertensive medication use. SCOPA-tot, SCales for Outcomes in PArkinson’s – Autonomic total score; OR, Odds ratio. *p < 0.05 for interaction, PD vs. controls.
To understand whether depression also predicted physiologic measures of autonomic failure, we next evaluated whether depression symptoms predicted the presence of orthostatic hypotension (OH). Depression did predict OH in PD at visit 12 (OR = 1.138, p = 0.025), but not in PD at baseline, PD at visit 6, or in controls (Table 2).
We next looked at subcategories of the SCOPA-AUT scale to understand whether certain symptom groups may be driving these associations, correcting for multiple comparisons. We found that depression symptoms predicted gastrointestinal symptoms, urinary symptoms, and cardiovascular symptoms in PD participants at all time points, with strongest associations between depression symptoms and gastrointestinal and genitourinary symptoms. Depressive symptoms did not predict symptom subscores of the SCOPA-AUT in controls (Supplementary Table 1).
When comparing the influence of depression symptoms on autonomic symptoms between all groups, we found that GDS and total burden of autonomic symptoms were more strongly linked in PD at baseline (β= 0.197, p = 0.0034), PD at visit 6 (β= 0.451, p = 0.0135), and PD at visit 12 (β= 0.909, p < 0.0001) compared to controls. This link is also stronger in PD at visit 12 compared to PD at baseline (β= 0.493, p = 0.002) and PD at visit 6 (β= 0.461, p = 0.004).
To understand the specificity of our findings, we used regression analyses to model prediction of autonomic symptoms based on severity of motor dysfunction, as measured by the MDS-UPDRS. We found that MDS-UPDRS part III motor score did not predict autonomic symptoms in controls or in PD at baseline visit but did predict autonomic symptoms at visit 6 (β= 0.093, p < 0.001) and at visit 12 (β= 0.161, p < 0.001). Due to this finding, we repeated the regression models to evaluate the influence of depression symptoms on autonomic symptoms, controlling for MDS-UPDRS part III as well as age and medication use. We found no significant differences compared to the model that did not include motor scores (β= 0.746, p < 0.0001 in PD at baseline; β= 0.743, p < 0.001 at visit 6; β= 1.21, p < 0.001 at visit 12; β= 0.290, p = 0.008 in controls).
DISCUSSION
Using prospective longitudinal data in a large cohort of early PD participants, we found that symptoms of depression predict AuD in PD more strongly than in controls, and that this effect intensifies by five years of disease duration. Importantly, depression symptoms predicted autonomic symptoms independently of the severity of motor symptoms, another measure of Parkinson’s disease severity.
Our study supports and advances the findings of previous studies investigating associations between autonomic and depression symptoms in PD [2, 12]. Previous studies have demonstrated that PD subjects with the highest burden of autonomic symptoms also have the highest burden of depression symptoms, psychotic symptoms, motor dysfunction, and cognitive impairment [11]. Autonomic symptoms in PD have previously been found to correlate with age and with symptoms of depression and anxiety, independent of motor symptom severity, suggesting clustering of these symptoms in PD [12]. Associations of autonomic symptoms with mood symptoms and age are also found in non-PD cohorts [27, 28], thus it has been unclear whether this association in PD represents a unique pathologic phenomenon. We add to the existing literature by demonstrating that associations between autonomic and depression symptoms in PD are stronger than those in controls, and that they increase with disease duration and therefore PD related pathologic burden. We again demonstrated that these associations are independent of age and the severity of motor symptoms, which also increase with increasing disease duration. This is particularly important as there are known associations between autonomic dysfunction and age in PD and non-PD adults [28], and between all non-motor symptoms of PD with advancing disease duration [8, 9]. Our results indicate that autonomic and depression symptoms in people with PD may share unique pathophysiologic underpinnings, independent of dopaminergic pathways implicated in PD motor symptoms.
Several lines of evidence point to overlapping pathophysiologic mechanisms in PD related depression and autonomic dysfunction. For example, the hypothalamus, anterior cingulate cortex, medial prefrontal cortex, and insula are important nodes in both the central autonomic network (CAN), where they modulate autonomic nervous system activity [29, 30], as well as in the limbic system, where they regulate emotion [31]. These structures are also preferentially affected by PD pathology [14, 32]. Interestingly, other structures implicated in Major Depression Disorder, such as the dorsolateral prefrontal cortex, remain relatively spared by PD pathology [14]. This may indicate that the brain structures and networks involved in PD depression differ from those involved in Major Depression Disorder. Further evidence for overlapping pathophysiology between autonomic and depression symptoms comes from neuroimaging studies. Reduced norepinephrine (NE) neurotransmission is found in both depression and autonomic dysfunction in PD [15–17]. Furthermore, depressive symptoms in PD appear to respond better to agents that alter NE transmission [18–20, 33–36], and treatment of OH in PD with agents that enhance NE neurotransmission may be more effective than more traditional treatments of OH [13, 37].
There was no significant association between autonomic symptoms and the presence of OH in the PD group at baseline. This finding highlights the potential limitations of the measures used to evaluate autonomic dysfunction in this study. The SCOPA-AUT is a validated tool used to assess autonomic symptoms in PD, however the questionnaire is disproportionally weighted towards gastrointestinal and urinary symptoms. For example, there are 7 questions in the gastrointestinal subdomain (21 possible points), 6 questions for the urinary subdomain (18 possible points), but only 3 questions for cardiovascular symptoms (9 possible points). This may underrepresent associations with cardiovascular autonomic symptoms, and overrepresent the impacts of gastrointestinal and urinary symptoms on total autonomic symptom burden. Similarly, OH, measured as a single orthostatic blood pressure measurement, is the only objective measure of autonomic dysfunction available in this database. OH is the most severe form of adrenergic cardiovascular autonomic failure. Though OH is common, affecting about 50% of people with PD at some point in their disease course [3], it is more common with increasing disease duration [3, 4] and as expected was uncommon in this cohort of early PD. In PPMI, standing blood pressure during vital sign testing is obtained after 1–3 minutes of standing, which may not provide sufficient sensitivity for the detection of OH (if measured before 3 minutes) or delayed OH (considered a precursor to “classic” neurogenic OH, and evidence of autonomic failure) [38–40]. The lack of ability to detect delayed OH, the low prevalence of OH in early PD, as well as the potential underrepresentation of cardiovascular symptoms in the SCOPA-AUT, may help to explain the poor correlation between autonomic symptoms and the presence of OH in the baseline PD group. Despite the low prevalence of OH in this study, there was a robust association between presence of OH and symptoms of depression at visit 12. Future studies may benefit from more sensitive objective measures of autonomic dysfunction. Another potential limitation in the current study is the evidence that a higher burden of depression may influence perception and report of autonomic symptoms, rather than reflect true overlapping pathophysiology [41]. Though this may potentially confound our results, the presence of stronger correlations between autonomic and depressive symptoms in the PD groups compared with the control group reduces the impact of this potential confounder. Furthermore, controlling for motor symptom severity did not alter the strength of the associations between autonomic and depression symptoms in PD, arguing against overall PD symptom burden as a confounder.
It is important to point out that the burden of depression symptoms among those taking antidepressant medications in the PD groups was much higher than that among those not taking antidepressant medication. This may be a clue that depression symptoms are undertreated, or may be refractory to treatment, in people with PD. As symptoms of depression portend a poorer disease prognosis in PD, this is particularly concerning [6]. Though this was not a primary area of investigation in the current study, it does demonstrate the need for more targeted therapies for these symptoms.
This study provides important insight into key non-motor symptoms of PD. Development of targeted treatments for these symptoms is needed. Treatment of AuD and depression in PD should be considered as a potential synergistic target for new interventions such as brain stimulation for the modulation of the shared cortical network substrate.
Footnotes
ACKNOWLEDGMENTS
PPMI, a public-private partnership, is funded by the Michael J. Fox Foundation for Parkinson’s Research funding partners 4D Pharma, Abbvie, Acurex Therapeutics, Allergan, Amathus Therapeutics, ASAP, Avid Radiopharmaceuticals, Bial Biotech, Biogen, BioLegend, Bristol-Myers Squibb, Calico, Celgene, Dacapo Brain Science, Denali, The Edmond J. Safra Foundaiton, GE Healthcare, Genentech, GlaxoSmithKline, Golub Capital, Handl Therapeutics, Insitro, Janssen Neuroscience, Lilly, Lundbeck, Merck, Meso Scale Discovery, Neurocrine Biosciences, Pfizer, Piramal, Prevail, Roche, Sanofi Genzyme, Servier, Takeda, Teva, UCB, Verily, and Voyager Therapeutics
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
