Abstract
Background
Sleep disturbances are prevalent and debilitating non-motor symptoms in patients with Parkinson's disease (PD).
Objective
This study aimed to explore sleep architecture and the prevalence of polysomnographic (PSG) sleep findings in PD, examining the associations between sleep parameters and other clinical characteristics.
Methods
The study included 97 PD patients (age: 67.1 ± 7.9) and 42 non-PD controls (age: 64.7 ± 9.7). Participants underwent clinical assessment and video-PSG. Sleep parameters, apnea-hypopnea index (AHI), periodic limb movements index (PLMI), and REM sleep without atonia (RSWA) were obtained. General linear models were used to explore interactions between disease duration and sleep variables in predicting PD symptoms.
Results
Nearly 94% of PD patients showed at least one video-PSG-assessed sleep finding, including AHI-defined obstructive sleep apnea (OSA), periodic limb movements, and RSWA. Sleep alterations correlated with disease severity, with reduced sleep duration and efficiency, higher sleep latency, and higher AHI being associated with worse PD severity. Sleep efficiency was more strongly associated with motor symptoms and disease severity at longer disease duration, while AHI exhibited a stronger relationship with motor symptoms at shorter disease duration. Finally, PD patients showed significant alterations in sleep macrostructure compared to controls, including reduced sleep duration (d = 0.75) and efficiency (d = 1.15) and decreased percentage of stage 3 non-REM sleep (d = 0.37).
Conclusions
The study showed a high prevalence of video-PSG-defined sleep findings in PD, with interactions between disease duration, sleep efficiency, and AHI. The present results support personalized management of sleep disturbances in PD to potentially improve symptoms and reduce the burden of illness.
Plain language summary
Sleep problems are very common in people with Parkinson's disease (PD). Our study aimed to understand the specific sleep issues occurring in people with PD using video-polysomnography, the gold-standard sleep study technique. We focused on understanding the current prevalence of polysomnographic sleep findings and their link to the disease motor burden.
We included 97 people with PD, with an average age of 67 years (23 women and 74 men), sleep monitored overnight using video-polysomnography, which records sleep structure, oxygen saturation parameters, limb movements in sleep, and nocturnal breathing, along with video recording. People with PD was also compared to 42 participants without neurodegenerative disorders of the same age (14 women and 28 men) as controls.
We found that nearly 94% of the participants with PD had at least one polysomnographic sleep finding, such as: obstructive sleep apnea (OSA, breathing problems during sleep identified by a pathological apnea-hypopnea index or AHI), periodic limb movements during sleep (PLMS, frequent movements of the limbs during sleep), REM sleep without atonia (loss of the normal muscle paralysis that occurs during the sleep stage known as rapid eye movement sleep). These sleep disturbances were linked to PD motor severity. We found that lower sleep efficiency (a marker of sleep continuity) was more strongly associated with worse symptoms in people who had PD for a longer time. By contrast, breathing problems during sleep (as measured by AHI) had a greater impact on symptoms when PD was in its earlier stages. Other sleep problems, such as PLMS or REM sleep without atonia, did not show a strong link with symptom severity in our models. Finally, when comparing people with PD to controls, PD patients spent more time awake during the night and had pathological changes in specific sleep stages.
Our findings show that sleep is significantly disturbed in people with PD. These sleep problems can affect the symptoms of the disease differently depending on how long a person has had PD. This suggests that personalized treatments for sleep disturbances could potentially improve symptoms of PD at different stages of the illness.
Keywords
Introduction
In recent years, large multicentric clinical studies have shed light on the prevalence of non-motor symptoms (NMS) in Parkinson's disease (PD), reporting their occurrence in up to 99% of patients.1,2 Among NMS, sleep disturbances stand out as particularly common and distressing symptoms, with high prevalence for REM sleep behavior disorder, restless legs syndrome, insomnia, nocturia, sleep-disordered breathing, and excessive daytime sleepiness. 1 Importantly, sleep disturbances can manifest at any stage of PD, even in the pre-motor stage, but usually worsen in frequency and severity as the disease progresses and motor fluctuations occur, significantly impacting the quality of life of patients.2–5
Polysomnographic (PSG) studies demonstrated nocturnal sleep alterations since the early stages of PD pathology.6–8 Furthermore, PSG findings in de novo PD patients indicated that alterations in sleep stability and continuity might be unresponsive or partially responsive to dopamine-agonist therapy, suggesting a potential non-dopaminergic etiology of the sleep disturbances.7,9 Notably, it has been hypothesized that the neurodegenerative process underlying PD may originate not solely in the nigrostriatal dopaminergic neurons but could also begin in the anterior olfactory structures, gradually spreading in an ascending manner from the midbrain to the forebrain as the disease advances.10–12 Therefore, sleep impairment might be improved by drugs targeting other non-dopaminergic pathways involving glutamate, orexin, or melatonin.13–15
Overall, this complex interplay involving monoaminergic and cholinergic pathways may contribute to the emergence of NMS, including sleep disturbances, and may explain their relative independence from the deterioration of dopaminergic structures. However, although the high prevalence of sleep disturbances in PD patients has been widely documented,16–20 many studies relied only on subjective evaluations using multiple validated questionnaires to include large groups of subjects at different disease stages.4,5,21,22 In contrast, polysomnography-based studies that assess the association between sleep parameters and clinical characteristics of the disease (e.g., functional impairment, disease duration, and dopaminergic therapy) have frequently been limited to small samples of patients and often do not consider the association with motor burden or other clinical features of PD.20,23,24 Furthermore, given the time of acquisition of PSG recordings, the criteria for scoring sleep were not always up to date. Accordingly, recent research has called for additional studies using video-polysomnography (v-PSG) in larger cohorts of PD patients to examine the impact of potentially heterogeneous factors (e.g., disease duration, medication status) on sleep architecture and sleep disturbances. 20
The present study primarily aimed to examine sleep architecture and the prevalence of v-PSG-measured sleep disturbances in a cohort of PD patients, while also analyzing the relationship between sleep parameters and disease characteristics, such as motor impairment, disease stage, and dopaminergic treatment, using standardized visual scoring and PSG-defined thresholds for sleep disturbances. As a secondary objective, and to complement existing literature on PSG sleep architecture in PD,20,23 a group-level comparison with age- and sex-matched non-PD historical controls was also performed.
Methods
Participants
The study included two groups: patients with PD and an age- and sex-matched non-PD control group (CTRL).
Patients with PD were consecutively recruited from the PD Unit of the University Hospital of Rome Tor Vergata. Inclusion criteria required a diagnosis of idiopathic PD according to the UKPD Brain Bank criteria. 25 Upon enrollment, motor symptoms disability was assessed using the Movement Disorder Society Unified PD Rating Scale – part III (MDS-UPDRS-III), with scores ranging from 0 to 108, 26 and PD severity was determined by the modified Hoehn and Yahr stage (H&Y), ranging from 1 to 5.27,28 Disease duration, levodopa daily equivalent dose (LEDD), and body mass index (BMI) were also recorded for each patient. Exclusion criteria included the use of antidepressants, benzodiazepines, or other central nervous system-acting drugs (excluding dopaminergic therapy); thyroid illness; history of malignancies; respiratory diseases; epilepsy; cognitive impairment (Mini Mental State Examination score < 24); clinically significant symptoms of depression (Hamilton Depression Scale score > 16 or Beck Depression Inventory score > 13). 29 These exclusions were made to minimize confounding effects on sleep architecture, as several of these drugs and conditions are known to impact specific sleep stages. For example, serotonergic or noradrenergic antidepressants may suppress REM sleep, benzodiazepines can increase stage 2 of Non-REM sleep, and cholinesterase inhibitors may enhance REM sleep. Moreover, antidepressants have been associated with increased risk of REM sleep without atonia (RSWA) or periodic limb movements in sleep (PLMS) and both benzodiazepines and antipsychotic medications can influence respiratory parameters such as apneas and hypopneas.30–35 The MMSE cut-off score for defining cognitive impairment in PD patients was used since prior PSG studies in PD patients documented that sleep architecture can be affected by cognitive impairment.20,36
CTRL participants were selected from a historical database at the Sleep Medicine Center of the same University Hospital. They were matched to PD participants on age and sex and met the same exclusion criteria. They had no diagnosis of neurological or psychiatric disorders and did not meet criteria for any sleep disorder based on a structured sleep medicine interview and PSG. Only PD participants underwent full v-PSG with respiratory and limb movement channels; therefore, respiratory and PLMS data were not collected for the CTRL group.
The study protocol was approved from the competent Ethical Committee (protocol number 128/23), and written informed consent was obtained from all participants. The study was performed according to the STROBE guidelines for reporting observational studies. 37
Procedures
The study design was cross-sectional. All PD patients underwent clinical examination and overnight v-PSG conducted using an ambulatory dynamic polygraph (SOMNOscreen, SOMNOmedics GmbH, Germany). Electrodes were applied to the scalp following the International 10–20 system, and the PSG montage included eight electroencephalographic (EEG) channels (F4, C4, O2, A2, F3, C3, O1, and A1), two electrooculographic (EOG) channels, and three electromyographic (EMG) channels (mental and anterior tibialis muscles). Oronasal flow, thoracic and abdominal movements, pulse oximetry, and electrocardiography were recorded to assess cardiorespiratory parameters.
The v-PSG for CTRL was performed to examine the overall sleep structure and included only the EEG, EOG, and EMG channels. As mentioned, apnea-hypopnea events and PLMS values were not collected in this group. All control participants did not report or showed clinically significant respiratory or limb movement events during their diagnostic screening.
Sleep technologists expert in sleep medicine scored the v-PSG (MM and RL) and neurologists expert in sleep medicine reviewed the scoring (CL, NM); all of them were blinded to participants’ clinical conditions. Analysis of the sleep studies was performed according to the standard criteria of the American Academy of Sleep Medicine. 38 The following PSG parameters were assessed: total sleep time (TST); sleep efficiency (i.e., the ratio between TST and time spent in bed); sleep latency (the interval between lights off and the onset of sleep); REM sleep latency (the interval between sleep onset and the first epoch of REM sleep); wakefulness after sleep onset (WASO); stages 1 (N1), 2 (N2), and 3 (N3) of non-REM sleep; and REM sleep. All the sleep stages were expressed as a percentage of TST. Apnea and hypopnea events, along with PLMS and RSWA were identified and scored according to standard guidelines. 38 Specifically, the apnea-hypopnea index (AHI, indicating the number of apnea and hypopnea events per hour of sleep) and the PLMS index (PLMI, indicating the number of periodic limb movements per hour of sleep) were calculated. RSWA was defined according to common standard criteria 38 and scored as a dichotomous variable (i.e., present vs. absent), without semi-quantitative measures.
For the purpose of this study, sleep alterations were identified based on PSG data, focusing specifically on respiratory events, PLMS, and RSWA. Although not a sleep disorder, RSWA was considered a polysomnographic finding. Clinically relevant PLMS was defined as a PLMI above 15 movements per hour of sleep based on previous findings. 39 We defined OSA using AHI thresholds only, following PSG-based criteria (i.e., AHI ≥ 5). While a formal diagnosis of obstructive sleep apnea syndrome (OSAS) requires symptoms and clinical evaluation according to the third edition of the International Classification of Sleep Disorders (ICSD-3) criteria, 40 our classification was based solely on AHI: 5–15 = mild, 15–30 = moderate, > 30 = severe. Patients were referred to as having AHI-defined OSA.
Statistical analyses
Categorical variables were summarized as counts and percentages, while continuous variables were presented as means and standard deviations. Differences between the PD group and the CTRL group in sleep variables were evaluated with Welch's t-tests using adjusted p-values obtained with the Benjamini-Hochberg false discovery rate procedure to account for multiple testing, 41 and Cohen's d effect sizes were computed (d = 0.20 was considered a small effect size, d = 0.50 as a medium effect size, and d = 0.80 as a large effect size).42,43 Welch's t-test was preferred over Student's t-test due its robustness against violations of the homogeneity of variances assumption.44,45 Satisfaction of normality assumption was assessed by examining residuals quantile-quantile plots and skewness and kurtosis values (skewness between −2 and 2 and kurtosis between −7 and 7 were considered indicative of approximately normal distribution). 46 Sleep latency and stage N1 percentage exhibited relevant asymmetries in the PD group, and thus a base-10 logarithmic transformation was applied to these variables before conducting t-test.
To compare sleep macrostructure between female and male PD patients, controlling for age and BMI, analysis of covariance (ANCOVA) was performed and omega squared (ω2) effect sizes were obtained. Again, sleep latency and percentage of N1 stage were log-transformed to correct for deviations from normality before performing the analyses.
Bivariate relationships between clinical data and sleep parameters in the PD patients group were explored using Spearman's correlation coefficients. To investigate the linear association between sleep disturbances (considered as antecedent variables) and motor symptoms (considered as consequent variables) controlling for the moderating influence of disease duration, a regression-based moderation analysis was performed.47,48 Generalized linear models (GLMs) were conducted, with each model including sleep efficiency, AHI, PLMI, and RSWA as predictors, disease duration as a moderator, and age, sex, BMI, and LEDD as covariates. Interaction terms between sleep variables and disease duration were computed as the product of centered predictors (e.g., disease duration × sleep efficiency). When significant interactions were found, simple slope analyses were conducted to probe the effect of the predictor at different values of the moderator (i.e., −1 SD, mean, and +1 SD).49,50
All the statistical analyses were conducted with jamovi v. 2.5 51 and R v. 4.4.1, 52 and GLMs and simple slope analyses were performed with the GALMj module for jamovi. P-values below 0.05 were considered significant for all analyses.
Results
Participants characteristics, clinical data, and sleep parameters in PD
The study included 97 PD patients (mean age: 67.1 ± 7.9 y.o., 23.7% female), among whom 30.9% (n = 30) exhibited moderate-to-severe motor impairment, defined as H&Y score > 2. Only four patients were drug-naïve during the study. Clinical characteristics and sleep parameters obtained via v-PSG are summarized in Table 1.
Participants’ demographic characteristics, clinical data, and PSG-measured sleep parameters.
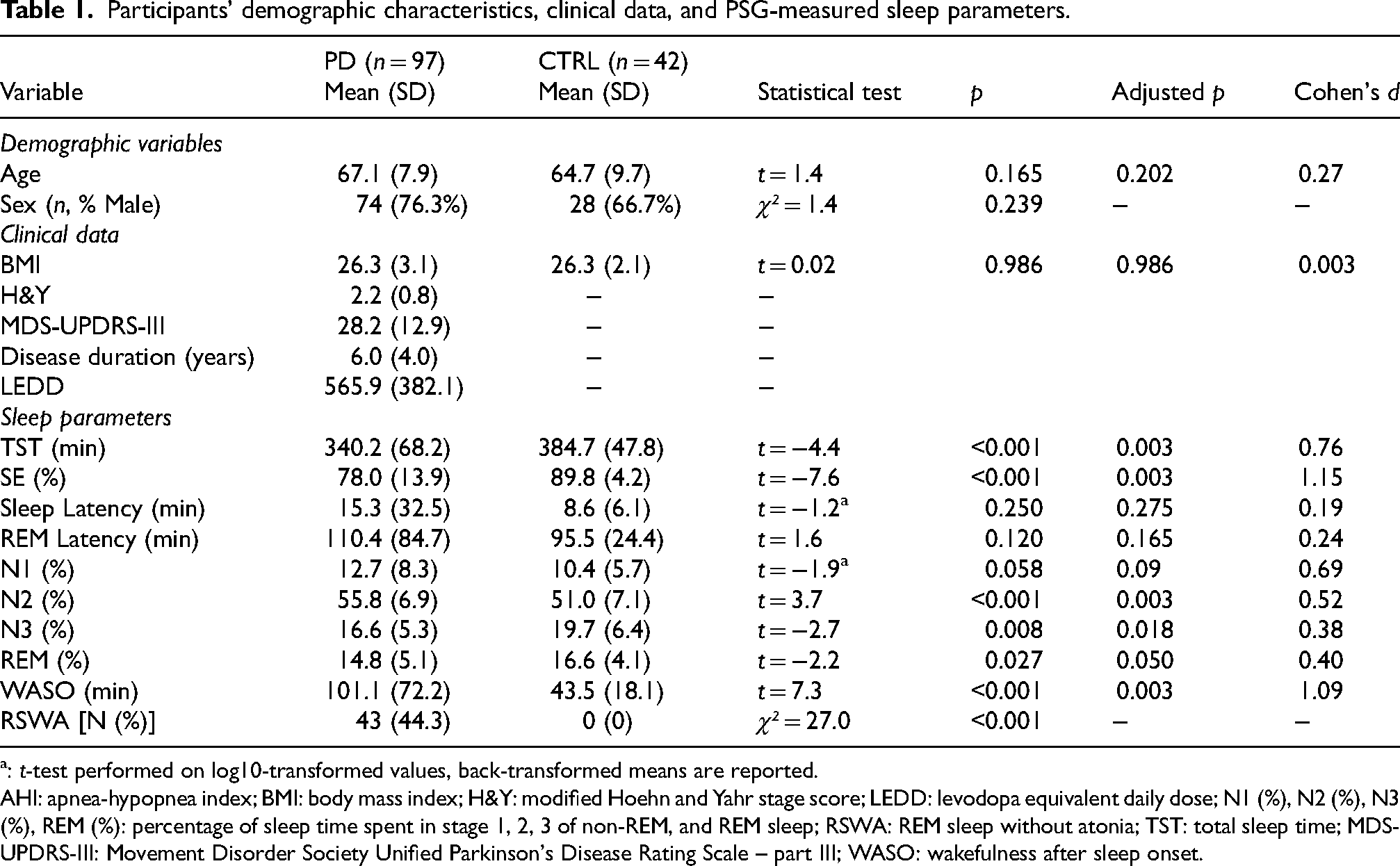
: t-test performed on log10-transformed values, back-transformed means are reported.
AHI: apnea-hypopnea index; BMI: body mass index; H&Y: modified Hoehn and Yahr stage score; LEDD: levodopa equivalent daily dose; N1 (%), N2 (%), N3 (%), REM (%): percentage of sleep time spent in stage 1, 2, 3 of non-REM, and REM sleep; RSWA: REM sleep without atonia; TST: total sleep time; MDS-UPDRS-III: Movement Disorder Society Unified Parkinson's Disease Rating Scale – part III; WASO: wakefulness after sleep onset.
Prevalence of v-PSG sleep findings in PD patients, and associations with demographic and clinical data
Among PD participants, 70.1% (95% CI: 61.0–79.2%, n = 68) were found to have OSA based on PSG (AHI ≥ 5), while 44.3% (95% CI: 34.4–54.2%, n = 43) exhibited RSWA, and 42.3% (95% CI: 32.4–52.1%, n = 41) had clinically-relevant PLMS (PLMI > 15/hour). In total, 93.8% of PD patients (95% CI: 89.0–98.6%, n = 91) demonstrated at least one sleep finding at v-PSG (OSA, RSWA, or PLMS). Mean AHI in the PD group was 14.8 ± 15.6 events/hour, and mean PLMI was 16.1 ± 17.9 movements/hour. Regarding the severity of respiratory events, 39.2% (n = 38) of the PD group had mild OSA (AHI between 5 and 15), 13.4% (n = 13) had moderate OSA (AHI between 15 and 30), and 17.5% (n = 17) had severe OSA (AHI ≥ 30). The prevalence of individual sleep findings and their combinations as assessed by v-PSG is shown in Figure 1.
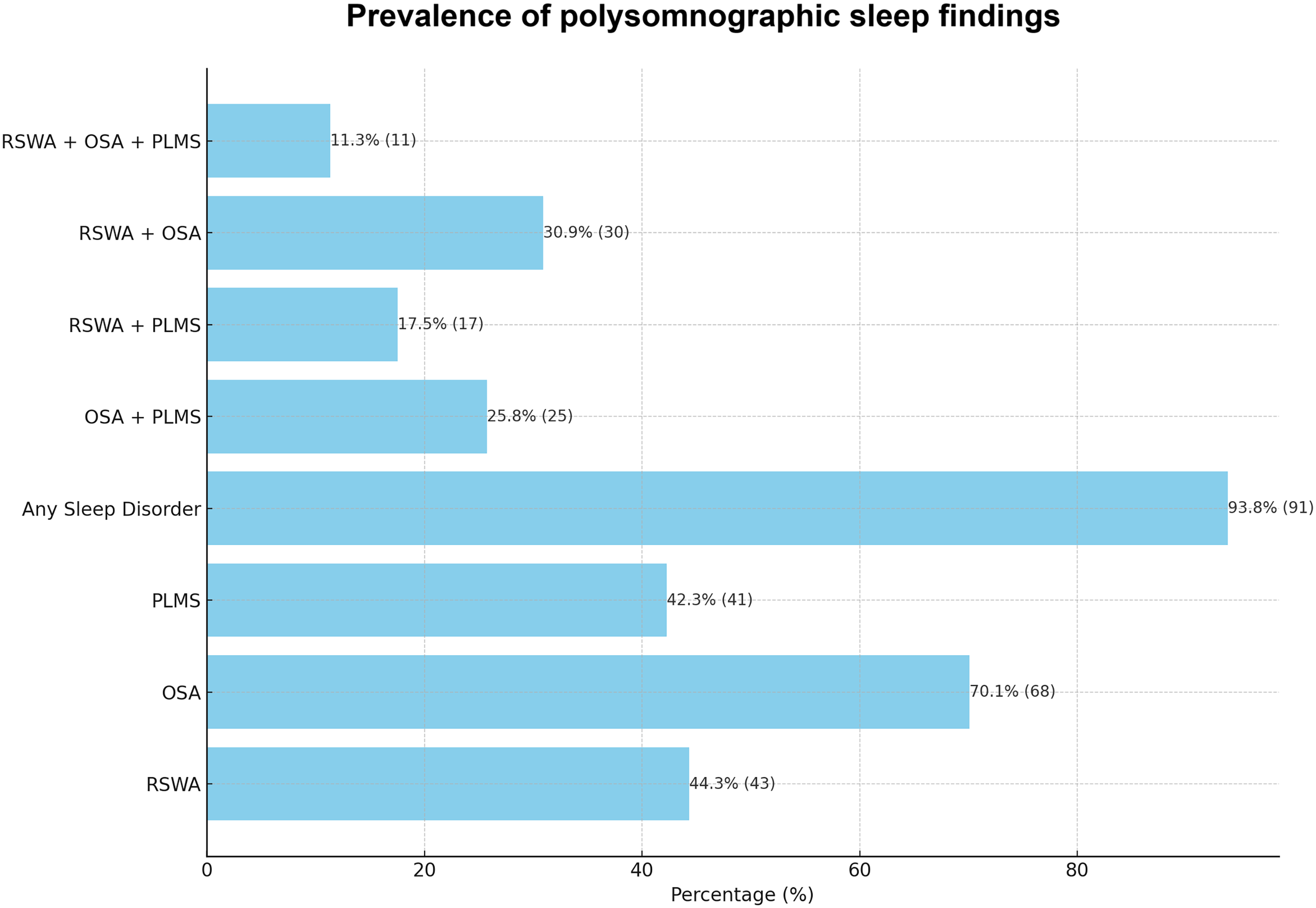
Prevalence of sleep finding assessed with video-polysomnography (percentages, with counts in brackets) in patients with Parkinson's disease (n = 97). RSWA: REM sleep without atonia; OSA: obstructive sleep apnea; PLMS: periodic limb movements of sleep.
No significant differences in age (all p-values above 0.05) were observed between PD participants with or without sleep findings. Similarly, there was no significant difference in the sex distribution between patients with and without RSWA (60.9% of women vs. 54.1% of men; χ² = 0.3, p = 0.57). However, female patients were more likely to have OSA (91.3% vs. 63.5%; χ² = 6.5, p = 0.01), while male patients were more likely to exhibit PLMS (51.4% vs. 13.0%; χ² = 10.6, p = 0.001). Notably, individuals with OSA, clinically-relevant PLMS, or RSWA did not show significant differences in PD severity (H&Y score), motor symptoms (MDS-UPDRS-III), disease duration, or LEDD (all p-values above 0.05). However, worsening OSA severity was associated with older age (rho = 0.22, p = 0.03), higher H&Y scores (rho = 0.44, p < 0.001), higher MDS-UPDRS-III scores (rho = 0.35, p < 0.001), longer disease duration (rho = 0.31, p = 0.002), and higher LEDD (rho = 0.25, p = 0.01).
Associations between sleep parameters, clinical data, and sex
Table 2 illustrates bivariate Spearman's correlations among v-PSG-derived sleep parameters and clinical data in the PD patient group. Older age showed significant associations with reduced sleep efficiency, prolonged sleep latency, higher WASO, increased time spent in N1, higher AHI, and higher PLMI. In contrast, higher H&Y score was correlated with lower TST and sleep efficiency, longer sleep latency and WASO, increased time spent in N1, and higher AHI. Similarly, MDS-UPDRS-III scores showed significant associations with lower TST and sleep efficiency, higher sleep latency and WASO, increased time spent in N1, decreased time spent in N3 and REM, and higher AHI.
Spearman's correlations among demographic, clinical, and PSG sleep variables in PD patients.
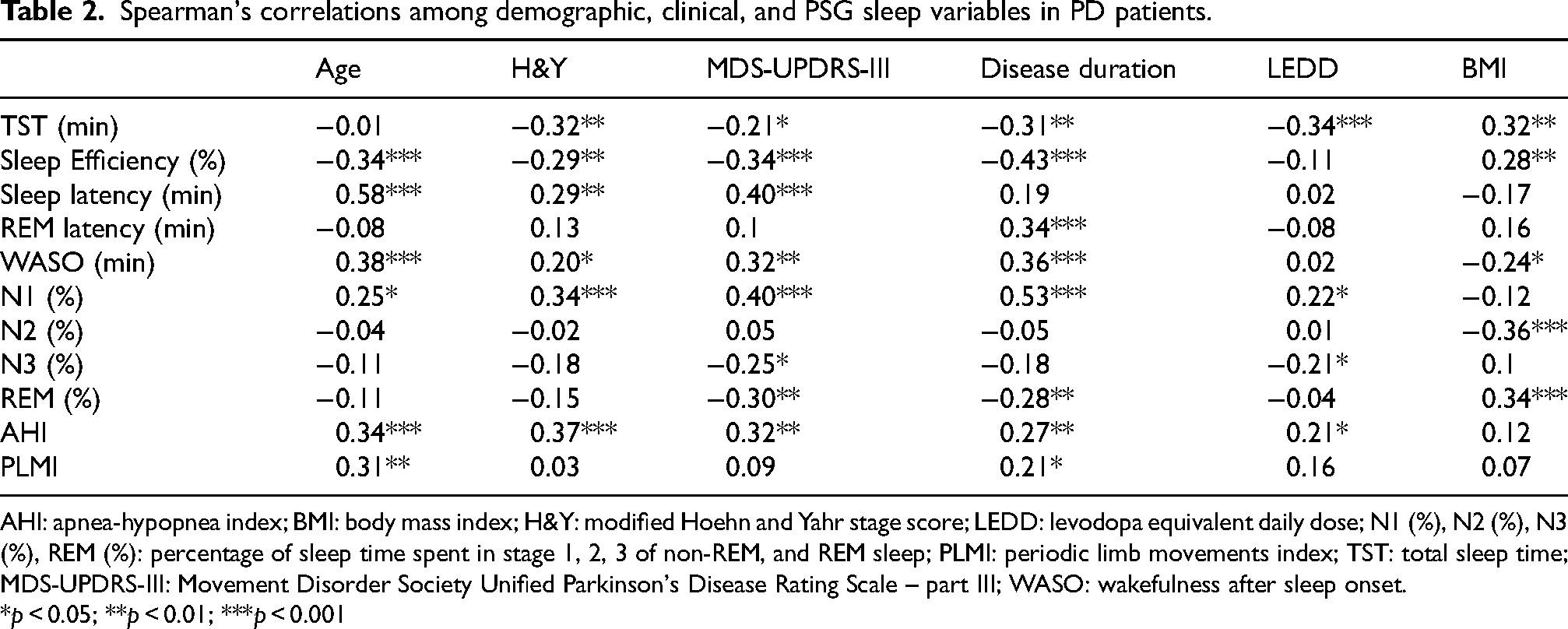
AHI: apnea-hypopnea index; BMI: body mass index; H&Y: modified Hoehn and Yahr stage score; LEDD: levodopa equivalent daily dose; N1 (%), N2 (%), N3 (%), REM (%): percentage of sleep time spent in stage 1, 2, 3 of non-REM, and REM sleep; PLMI: periodic limb movements index; TST: total sleep time; MDS-UPDRS-III: Movement Disorder Society Unified Parkinson's Disease Rating Scale – part III; WASO: wakefulness after sleep onset.
*p < 0.05; **p < 0.01; ***p < 0.001
Disease duration also showed a similar pattern of correlations with sleep variables, demonstrating negative associations with TST, sleep efficiency, and time spent in REM sleep and positive associations with REM sleep latency, WASO, time spent in N1, AHI, and PLMI, indicating that longer disease duration correlated with increased sleep fragmentation and more light sleep and higher AHI and PLMI. Furthermore, a higher LEDD was associated with reduced TST and time spent in N3, as well as increased time spent in N1 and higher AHI. Higher BMI was related to higher TST, sleep efficiency, and REM percentage, and lower WASO and N2 percentage. Finally, no differences were observed in sleep parameters between patients with and without RSWA (all p-values above 0.05).
Sex differences in sleep parameters were also examined in participants with PD. Controlling for the effect of age and BMI, female patients showed higher TST (366.5 ± 51.1 vs. 332.1 ± 71.0; F(1,93) = 4.1, p < 0.05, ω2 = 0.03) and sleep efficiency (82.6 ± 7.1 vs. 76.6 ± 15.1; F(1,93) = 4.9, p < 0.05, ω2 = 0.04), lower WASO (79.4 ± 35.1 vs. 107.8 ± 79.3; F(1,93) = 4.2, p < 0.05, ω2 = 0.03), higher REM latency (152.9 ± 114.7 vs. 96.5 ± 67.6; F(1,89) = 7.5, p < 0.01, ω2 = 0.07), and lower PLMI (7.2 ± 4.3 vs. 18.9 ± 19.6; F(1,93) = 10.8, p = 0.001, ω2 = 0.09).
Comparison between PD patients and CTRL in sleep parameters
Clinical characteristics and sleep parameters obtained via v-PSG for CTRL participants are summarized in Table 1. The CTRL group consisted of 42 age- and sex-matched non-PD participants (mean age: 64.7 ± 9.7 y.o., 33.3% female).
Compared to PD patients, CTRL participants showed significantly higher total sleep time and sleep efficiency, along with a lower WASO, all with large effect sizes. They also exhibited a higher percentage of N3 sleep and a lower percentage of N2 sleep, both with medium effect sizes. A trend in higher REM sleep was documented in CTRL compared to PD patients. None of the CTRL participants showed RSWA.
GLM results: moderating role of disease duration in the relationship between sleep disturbances and PD severity
To investigate the relationship between sleep findings and PD severity and motor symptoms across disease progression, general linear models were estimated including relevant interaction effects. Two separate models were fitted: one with H&Y stage as the dependent variable, and one with MDS-UPDRS-III. Each model included AHI, RSWA, PLMI, and sleep efficiency as predictors, with disease duration as a moderator. Age, sex, BMI, and LEDD were included as covariates. Significant interactions were further explored through simple slope analysis.
In the H&Y model (Table 3), sleep efficiency showed a significant interaction with disease duration (p = 0.018), indicating that reduced sleep efficiency was more strongly associated with higher H&Y scores at longer disease duration (Figure 2, panel A; significant simple slope effect with β = −0.27, p < 0.001). AHI showed a positive main effect on H&Y (p = 0.012), but the AHI × disease duration interaction did not reach significance.
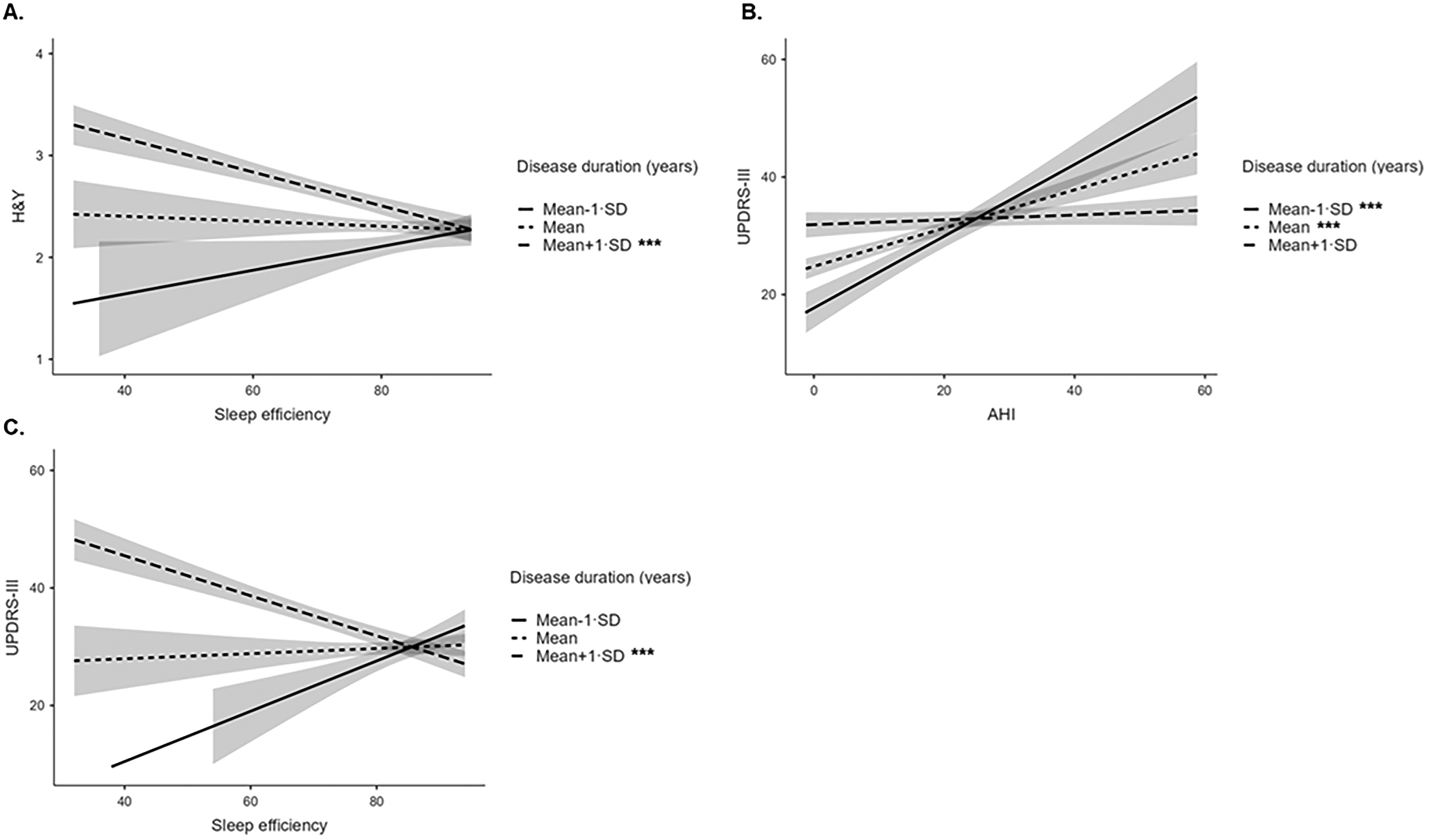
Simple slope plots illustrating the interaction between sleep variables and disease duration in predicting PD severity and motor symptoms. Each panel shows the association between the sleep variable and the clinical outcome at three levels of disease duration: low (−1 SD, solid line), average (mean, short-dashed line), and high (+1 SD, long-dashed line). Shaded areas indicate standard errors. (A) Association between sleep efficiency and H&Y score at different levels of PD duration. (B) Association between AHI and MDS-UPDRS-III score at different levels of PD duration. (C) Association between sleep efficiency and MDS-UPDRS-III scores at different levels of PD duration. PD: Parkinson's disease; AHI: apnea-hypopnea index; H&Y: Hoehn and Yahr score; MDS-UPDRS-III: Movement Disorder Society Unified Parkinson's Disease Rating Scale - part III. ***p < .001.
General linear models predicting PD severity (H&Y score) from disease duration, sleep variables, and their interactions.
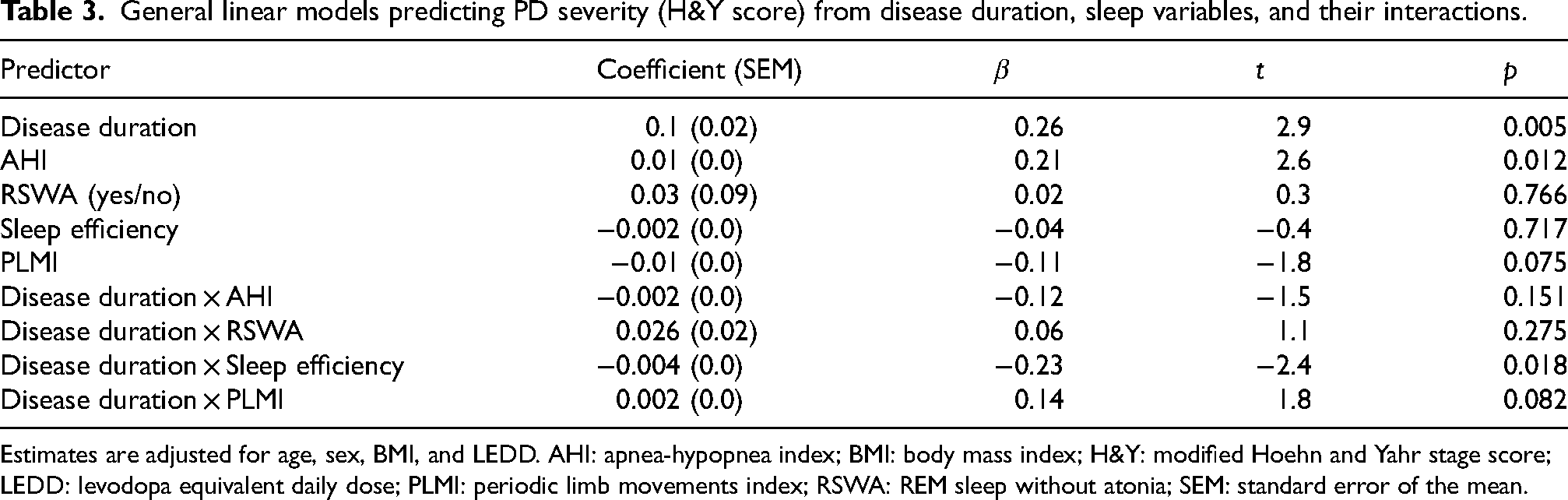
Estimates are adjusted for age, sex, BMI, and LEDD. AHI: apnea-hypopnea index; BMI: body mass index; H&Y: modified Hoehn and Yahr stage score; LEDD: levodopa equivalent daily dose; PLMI: periodic limb movements index; RSWA: REM sleep without atonia; SEM: standard error of the mean.
In the MDS-UPDRS-III model (Table 4), both the AHI × disease duration interaction (p < 0.001) and the sleep efficiency × disease duration interaction (p < 0.001) were significant (Figure 2, panels B–C). These interactions suggested that the relationship between AHI and motor severity was stronger at average and earlier disease stages (significant simple slope effects with β = 0.4, p < 0.001 and β = 0.74, p < 0.001, respectively), while reduced sleep efficiency had a greater impact on motor symptoms in patients with longer disease duration (significant simple slope effect with β = −0.37, p < 0.001). In addition, PLMI showed a significant negative main effect (p = 0.024), indicating that higher PLMI was associated with lower MDS-UPDRS-III scores, although the effect size was modest.
General linear models predicting PD motor symptoms (MDS-UPDRS-III score) from disease duration, sleep variables, and their interactions.
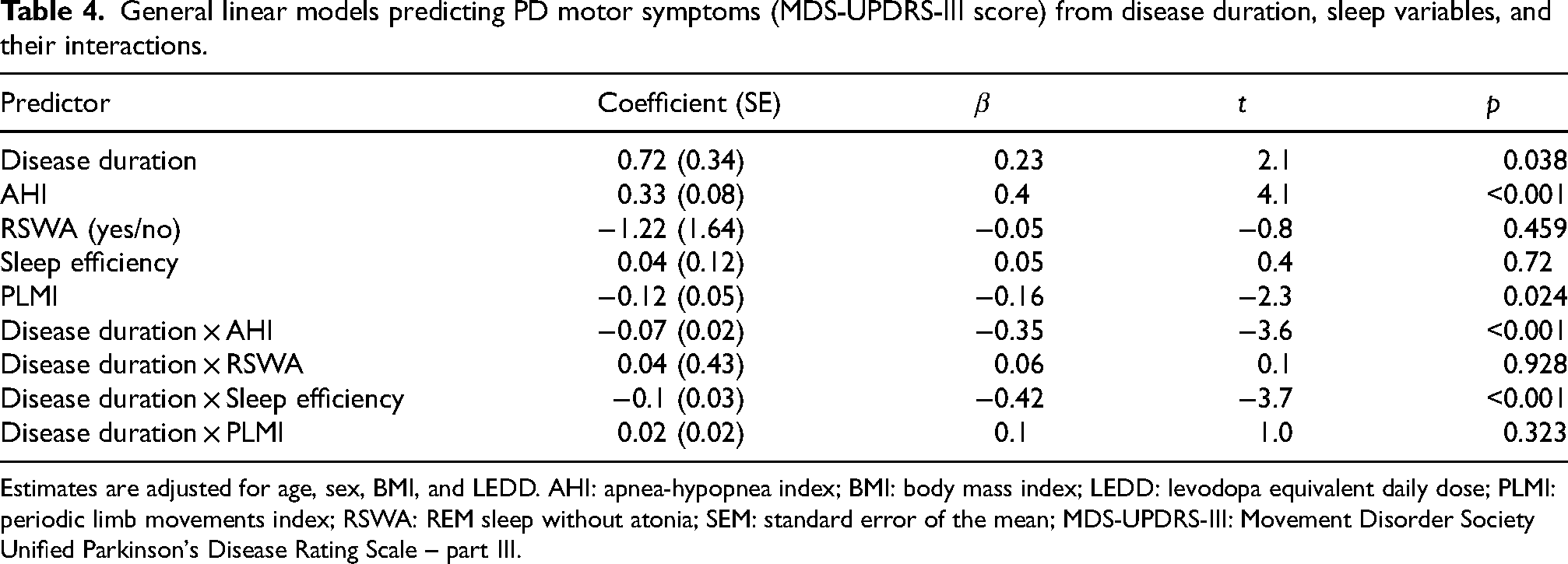
Estimates are adjusted for age, sex, BMI, and LEDD. AHI: apnea-hypopnea index; BMI: body mass index; LEDD: levodopa equivalent daily dose; PLMI: periodic limb movements index; RSWA: REM sleep without atonia; SEM: standard error of the mean; MDS-UPDRS-III: Movement Disorder Society Unified Parkinson's Disease Rating Scale – part III.
Finally, neither RSWA nor PLMI showed significant interactions with disease duration in either model.
Discussion
This study mainly aimed to explore the nocturnal sleep structure and the prevalence of v-PSG determined sleep findings in PD, and to examine the intricate interplay linking the clinical manifestations of PD and the role of antiparkinsonian treatments to v-PSG findings, with a particular focus on the moderating role of disease duration. Consistently, the deterioration of sleep macrostructure (i.e., low sleep efficiency, increased sleep latency and sleep fragmentation, and reduced N3 and REM sleep percentages) showed a significant relationship with longer disease duration and severity of PD motor symptoms, emphasizing the progressive worsening of sleep over the course of PD as well as its impact on patients’ well-being. Additionally, significant sex differences were observed among PD participants, with women exhibiting longer sleep duration, more preserved sleep continuity, and lower PLMS, in line with previous research indicating better sleep quality in aging women compared to men. 53 Regarding the prevalence of v-PSG-evaluated sleep findings, we observed remarkably high rates in our cohort, with up to 94% of PD patients presenting with at least one clinically relevant v-PSG diagnosed sleep finding, including AHI-defined OSA, PLMS, and RSWA. This prevalence rate exceeds those reported in previous questionnaire-based studies,1,19,21 reinforcing the notion that sleep disturbances are a common and possibly debilitating aspect of PD and that PSG-based assessment may be needed for uncovering them. Notably, our investigation found no significant differences in PD severity, motor symptoms, disease duration, or LEDD between patients with or without OSA, RSWA, or clinically relevant PLMS, although OSA severity was shown to correlate with longer disease duration and severity.
Using general linear models with covariates, we examined the role of disease duration as a moderator of the relationship between sleep findings and PD severity. Specifically, the association between sleep efficiency (reflecting sleep continuity) and PD severity was more pronounced in the advanced stages of the disease, indicating that night sleep might become increasingly fragmented and disturbed as PD pathology progresses. Conversely, the association between AHI and motor symptoms was evident only at low and average level of disease duration, highlighting the complex and evolving trajectory of sleep-disordered breathing in PD. Interestingly, PLMI showed a small but statistically significant negative main effect on MDS-UPDRS-III scores, indicating an inverse association with motor symptoms. While this result was unexpected and modest in size, it may reflect sample-specific variation or unmeasured confounding, and it warrants cautious interpretation. Moreover, neither PLMI nor RSWA showed significant interactions with disease duration in the GLM models, suggesting that, although frequent in PD, these disturbances may have weaker associations with motor impairment compared to sleep efficiency and AHI.
Taken together, these results support a bidirectional relationship between PD progression and sleep alterations, confirming and expanding the previously reported association between sleep deterioration and disease duration 54 and supporting the inherent nature of sleep disturbances in the pathophysiology of PD, which might reflect a direct impact of the neurodegenerative process on sleep-regulating brain structures. This observation underscores the importance of early identification and management of sleep disturbances in PD patients as a potential strategy to reduce the burden of the disease and improve overall functioning.
To our knowledge, this is the first study to examine these associations using generalized linear modeling with interaction effects directly probing the mediating role of disease duration. The decision to include only PD patients without cognitive impairment was informed by prior PSG-based studies, which typically included participants with MMSE scores >24. 20 Moreover, we excluded patients taking medications known to alter sleep architecture or contribute to secondary sleep disturbances – including antidepressants, antipsychotics, cholinergic agents, benzodiazepines, and Z-drugs – based on consistent findings in the literature regarding their influence on REM sleep, PLMS, and respiratory indices.31,32,34,35
As a secondary aim, we examined differences in sleep macrostructure between PD patients and age- and sex-matched controls. Our findings are in line with previous systematic and meta-analytic studies reporting altered macrostructure in PD.16,20 Specifically, this study confirmed reduced sleep efficiency, increased sleep fragmentation, and altered N2 and N3 stage proportions in PD patients. However, a direct comparison for AHI and PLMI values was not performed, as historical controls in our database were pre-screened to exclude pathological respiratory events or limb movements.
The present study has other limitations that should be carefully considered when interpreting the results. First, despite the use of moderation analysis to evaluate the mechanisms underlying the associations among the investigated variables, the cross-sectional design limits causal inferences, and the size of the control group restricts the generalizability of our comparisons. Although the study design and the inclusion/exclusion criteria were designed to improve scoring feasibility and avoiding confounding effects of psychiatric, cognitive, and pharmacological factors, this may have reduced the generalizability to patients with more complex or severe PD presentations. Specifically, while the analysis suggests interactions between sleep efficiency, AHI, and disease duration, we cannot infer from our data whether sleep findings exacerbate motor or cognitive symptoms or if worsening motor symptoms lead to greater sleep findings. Longitudinal studies are needed to clarify the directionality of these relationships and explore the long-term effects of managing sleep disturbances on the progression of PD symptoms. Furthermore, due to the exclusion criteria, our sample likely underrepresents PD patients with cognitive impairment, neuropsychiatric symptoms requiring pharmacologic treatment, or advanced-stage disease. As a consequence, the majority of our PD participants had mild-to-moderate disease severity, suggesting the need for further research in both more severely affected populations and drug-naïve patients at the early stages of the disease. Future studies should aim to include a more representative sample and a wider set of covariates (e.g., comorbidity patterns and cardiovascular risk factors) to fully explore the broader impact of additional conditions and medications on sleep findings in PD.
Finally, the study focused primarily on PSG-defined sleep findings and sleep alterations. While PSG is the gold standard for identifying conditions such as OSA, PLMS, and RSWA, it does not capture other important sleep disorders possibly affecting PD patients, such as insomnia, circadian sleep-wake rhythm disturbances, restless legs syndrome, or excessive daytime sleepiness. These disorders often require different diagnostic approaches, such as clinical interviews or additional tests like the Multiple Sleep Latency Test. Additionally, we did not include clinical OSA syndrome as defined by the ICSD-3, 40 since classification in our study was based solely on the AHI, without considering symptoms. Subsequent studies broadening the scope of sleep disturbances studied in PD, incorporating both PSG and non-PSG-defined sleep disorders, are highly warranted to provide a more comprehensive understanding of the full range of sleep disturbances affecting this clinical population.
In conclusion, this study showed the high prevalence of v-PSG-assessed sleep findings in a large cohort of patients with mild-to-moderate PD. General linear models highlighted significant interactions between disease duration, sleep efficiency, and apnea-hypopnea events, suggesting that sleep efficiency may be more closely associated with PD severity at longer disease duration, whereas the relationship between AHI and PD symptoms appears stronger at shorter disease duration. These findings support personalized treatment approaches to manage sleep disturbances and enhance overall sleep quality, and possibly quality of life and motor symptoms, from the early stages of PD, such as targeting PD patients for treatment of sleep-disordered breathing in the early stages of the disease and improving sleep fragmentation with targeted therapeutic pharmacologic and non-pharmacologic options as the disease progresses. Future research should further investigate these relationships using larger and more diverse clinical samples and longitudinal designs.
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: #NEXTGENERATIONEU (NGEU) and partially funded by the Ministry of University and Research (MUR), National Recovery and Resilience Plan (NRRP), project MNESYS (PE0000006) – A Multiscale integrated approach to the study of the nervous system in health and disease (DN. 1553 11.10.2022); European Union (NextGenerationEU) through the Italian Ministry of Health under PNNR-MCNT1-2023-12378408 (CUP E53C24000710006). The views and opinions expressed are those of the authors only and do not necessarily reflect those of the European Union or the European Commission.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
