Abstract
Background:
Studies on olfaction in LRRK2-associated Parkinson’s disease (LRRK2-PD) have yielded variable results. The impact of smell dysfunction upon daily life activities have been rarely assessed in PD.
Objective:
To characterize the olfactory deficit in LRRK2-PD and its impact on daily life activities.
Methods:
Twenty-four LRRK2-PD, 40 idiopathic PD (IPD), and 49 age-sex-matched controls were interviewed about olfactory characteristics and the impact of smell on daily life activities. The Barcelona Smell Identification test (BAST-24) and the Spanish-version of the 40-item University of Pennsylvania smell test (UPSIT) were applied.
Results:
Nineteen (79.2%) LRRK2-PD patients reported subjective smell impairment with a low impact upon daily living activities. UPSIT score was higher in LRRK2-PD than in IPD (22.54±7.98 vs 18.84±6.03; p = 0.042). All IPD and 95.8% LRRK2-PD patients had hyposmia/anosmia, assessed by means of the UPSIT. No differences were found between LRRK2-PD and IPD regarding smell detection, memory or forced-choice identification.
Conclusion:
Most LRRK2-PD patients reported subjective smell impairment and presented hyposmia, according to validated smell tests, with a low impact of the smell dysfunction on daily life activities.
INTRODUCTION
Olfaction has been studied frequently in Parkinson’s disease (PD), although the impact of hyposmia upon the patient’s daily life activities has rarely been assessed. Partial loss of smell (hyposmia) is commonly found in idiopathic PD (IPD) but in some forms of PD, such as genetic PD, it may occur rarely [1]. The study of olfaction in LRRK2 associated PD (LRRK2-PD), an autosomal dominant adult onset form of PD, has yielded variable results [2–5]. This form of genetic PD is of particular interest since, in manifest cases, hyposmia could separate those patients with Lewy type synuclein pathology from those without [6], which could have therapeutic implications. Furthermore, smell loss could antedate the onset of classical motor symptoms, as is thought to be the case in IPD, and constitute a marker of the prodromal stage [7, 8].
The aim of the current study was to characterize olfactory function in LRRK2-PD as well as in a group of IPD subjects employing both the UPSIT and the Barcelona Smell Identification test (BAST-24) [9], a subjective olfactory test validated in the Spanish population which evaluates different smell characteristics. In addition, we evaluated the impact of hyposmia upon the patient’s daily life activities, an aspect of smell loss which has not been evaluated previously in LRRK2-PD.
METHODS
This is a cross-sectional study. Patients attending the Movement Disorders Unit of Hospital Clínic de Barcelona between May 2013 and May 2015 were prospectively included.
Participants
Twenty-four consecutive LRRK2-PD patients among a cohort of LRRK2-PD patients already identified at our center were included, irrespective of any specific complaint or clinical feature. PD was diagnosed according to United Kingdom Brain Bank (UKBB) criteria [10]. Patients have been screened for LRRK2 mutations as previously described [11].
Forty subjects, clinically diagnosed with PD according to UKBB criteria and comparable to the LRRK2-PD patients in terms of age and gender were also recruited irrespective of any specific complaint or clinical feature. All of them were negative for LRRK2 mutations.
Controls consisted of 49 community subjects, again comparable in age and gender with the LRRK2-PD and IPD participants. They were non-blood relatives or friends of the PD patients that participated in the study. All had no relevant neurological diseases, had no family history of PD and reported lack of motor and cognitive problems.
Exclusion criteria in all study groups were significant hearing or visual impairment that interfered the understanding of the tests, and any disease that could affect the sense of smell, such as trauma brain injury (TBI), postviral loss of smell, chronic rhinosinusitis with/without nasal polyps, or allergic rhinitis. Subjects with a score lower than 24 in the Mini-Mental State Examination (MMSE) were also excluded.
Clinical assessments
Demographic and medical data was collected. In patients, age at diagnosis of PD were recorded. Patients were assessed through the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) [12], Schwab & England scale and Hoehn and Yahr stage [13] in On state. Medications at the time of the evaluation were also recorded. All participants were asked about TBI, symptoms and signs of rhinologic disease, other causes of smell dysfunction, and smoking habits or exposure.
Participants underwent a semi-structured interview to assess the subjective perception of their sense of smell. In this interview patients were first asked to answer “yes” or “no” to the question “Are you experiencing problems with your sense of smell?”. A conducted interview was then performed in those patients declaring to have a smell impairment in order to characterize a potential impact of the impairment on a number of daily living activities. The following fields were evaluated: 1) safety, 2) hygiene, 3) food handling and cooking, and 4) work and free time. In each field questions were included related to: safety (1.1 - Does the smell loss make it difficult to notice if the food is in a good state? 1.2 - Does the loss of smell make it difficult to detect the gas? 1.3 - Does the loss of smell make it difficult to keep the trashcan cleaned?), hygiene (2.1 - Does the loss of smell make it difficult to keep an adequate personal hygiene? 2.2 - Does the loss of smell make it difficult to smell your own corporal odor? 2.3 - Does the loss of smell make it difficult to smell the corporal odor of other people?), food and cooking (3.1 - Does the loss of smell make it difficult to perceive the odor or taste of food? 3.2 - Does the loss of smell make it difficult to notice if the food is in a bad state? 3.3 - Does the loss of smell make it difficult to buy food? 3.4 - Does the loss of smell make it difficult to cook?), and work and free time (4.1 - Does the loss of smell make it difficult to carry on your work? 4.2 - Does the loss of smell make it difficult to enjoy your free time?). Patients were asked to respond with a “yes” or “no” answer to each of the previous questions.
Olfactory evaluation
Subjective olfactometry
Olfactory evaluation was performed in On-state in all patients. The Barcelona Smell Test 24 (BAST-24) was applied to all participants. This is a twenty-four chemical compounds test validated in the Spanish population [9]. The first 20 odors mainly assess the first cranial nerve (1st CN, olfactory) and the last 4 odors mainly assess the fifth cranial nerve (5th CN, trigeminal) [9]. In the current study only the first 20 odors referring to the 1st CN were assessed. The smell test was performed in a quiet, noise isolated, well-ventilated room. All the odorants were located in hermetic glass jars. The explorer and volunteers did not wear perfumes the day of examination. The odorant jar should stay at 1 cm of the nose and with no contact with explorer’s finger and patient’s face.
After being exposed 5 seconds to each odor, participants were asked by the investigator to answer the following questions: 1) to test smell detection: “did you smell some- thing?”; 2) to test smell memory/recognition: “did you recognize this odor?”; and 3) to test forced-choice identification: “which of these four odors did you smell?”. The first two questions had two possible answers: yes (1) or no (0), while the third question had four forced multiple-choice answers. The test was repeated for each of the 20 odors.
For all three smell characteristics (detection, memory/recognition, and forced-choice identification), the total score was 0 to 20 (0–100%). Approximately 30 minutes were needed to test the entire battery of odors.
For comparative purposes, the smell function was also evaluated with the Spanish-version of the University of Pennsylvania Smell Identification Test (UPSIT), applied in a different day from the BAST-24 to avoid interference. The UPSIT is a standardized scratch-and-sniff test where 40 microencapsulated odors are asked to be identified in a forced-choice format. The identification score is the total number of correct answers, from 0 to 40 points. Higher scores indicate better olfactory identification.
Study participants were classified, according to their UPSIT scores, age and gender, with normal (normosmia), partial(hyposmia), or total (anosmia) loss of smell (Doty et al. [14]). Thirteen study participants were classified as normosmic, 72 as hyposmic and 25 as anosmic.
The Ethics Committee at the Hospital Clinic of Barcelona approved the study and all participants gave written informed consent.
Statistical analysis
Data were presented as frequencies and mean (SD) for discrete and continuous variables, respectively. Differences between proportions were compared using the Pearson’s chi-squared test or Fisher’s exact test (if the application conditions of the former were not met). For quantitative variables, Kruskal-Wallis equality of populations rank test was used to compare the study groups.
The significance level was set at 0.05. The analysis was carried out using SPSS version 20.0 (IBM, NYC, USA) and Stata [15]. Graphing was done using the R programming language [16].
RESULTS
Demographic and clinical data
Thirteen (54.2%) LRRK2-PD patients, 27(67.5%) IPD patients, and 28 (57.1%) controls were males (Table 1). Disease duration was similar between LRRK2-PD and IPD patients (7.3±5.6 and 6.8±6.5 years, respectively). MDS-UPDRS I, II and III scores, Hoehn and Yahr stages were also comparable between groups. The MMSE score were similar between patients and controls (p = 0.666).
Demographic and clinical data from Parkinson’s disease patients and healthy controls
aMean±standard deviation; bn (%). *Kruskal-Wallis equality of populations rank test. ¶Pearson’s chi-squared test. IPD, idiopathic Parkinson’s disease; LRRK2-PD, LRRK2 associated Parkinson’s disease; UPDRS-I, II, III and IV, Unified Parkinson’s disease Rating Scale, parts I, II, III and IV; MMSE, Mini-Mental State Examination.
Olfaction interview and impact of smell impairment in daily living activities
Forty-seven (73.4%) patients (LRRK2-PD plus IPD) and 4 (8.2%) controls reported having lost smell. Nineteen (79.2%) LRRK2-PD patients and 28 (70.0%) IPD patients reported a subjective impairment in their sense of smell (p = 0.425).
Fourteen (58.3%) LRRK2-PD patients and 11 (27.5%) IPD patients reported that the smell loss started at the same time or after the onset of the motor symptoms (p = 0.001). Seventeen (89.5%) LRRK2-PD patients, 25 (89.3%) IPD patients and all the healthy controls considered that the loss of smell occurred gradually.
No differences were found between LRRK2-PD and IPD regarding impact of smell loss in any activity of daily living (Fig. 1), except for the item “to detect gas” where IPD patients showed more difficulty (54.0% vs. 21.0%; p = 0.0497). Only in three (25%) questions more than 50% of patients responded that the smell loss provoked any difficulty in the activity investigated.
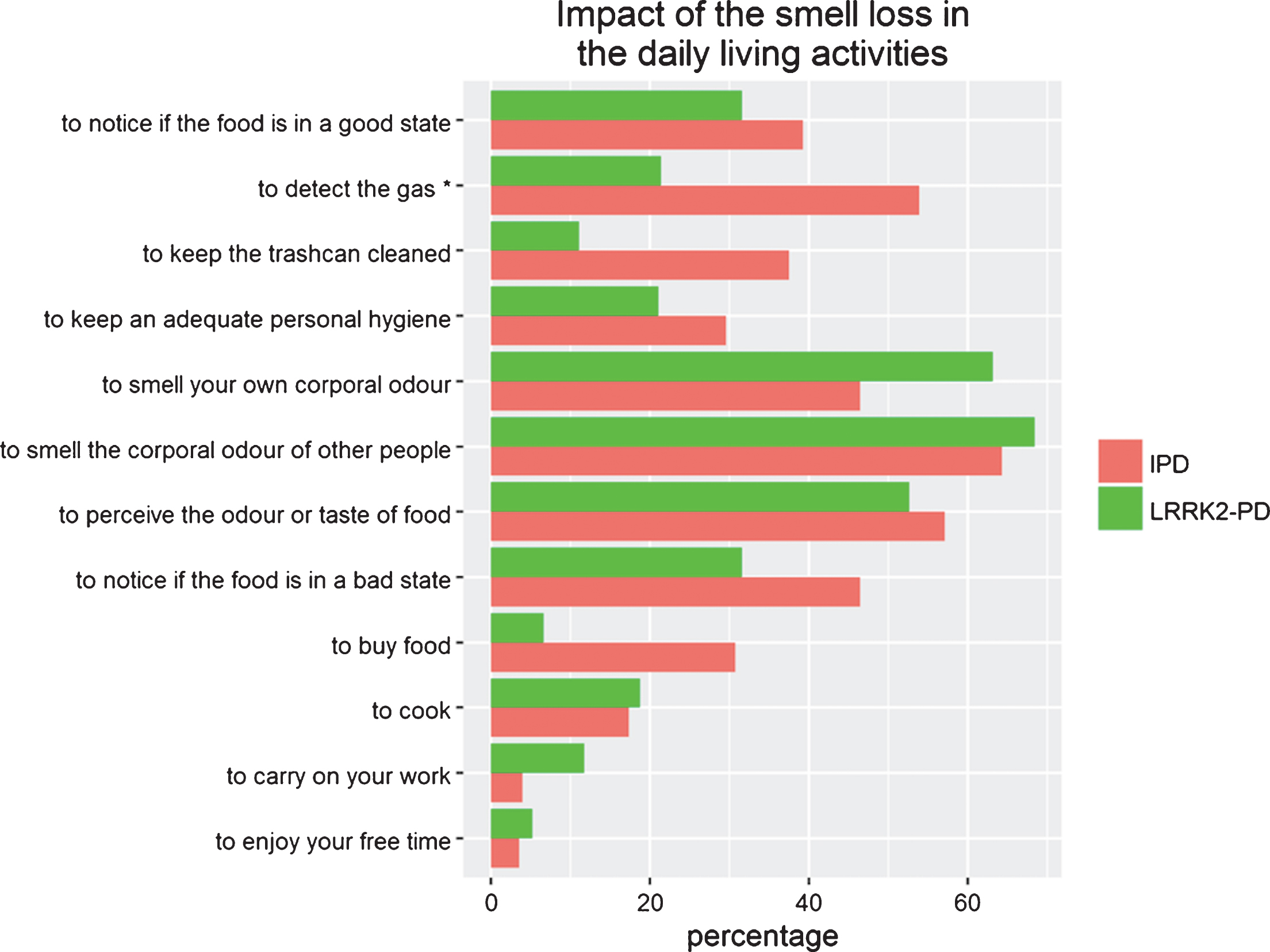
Impact of smell loss in Parkinson’s disease patient’s daily life activities. Impact of the loss of smell in the patient’s daily life activities. IPD, idiopathic Parkinson’s disease; LRRK2-PD, LRRK2 associated Parkinson’s disease. Bars represent the percentage of patients who respond “Yes” to the questions included in Table 1.
Olfactory tests
Concerning to BAST, for the first twenty odors and IPD patients scored lower than controls on smell detection (95.8±8.3% for LRRK2-PD, 91.7±18.8% for IPD, and 99.7±1.6% for controls; p = 0.001), memory/recognition (53.3±28.5% for LRRK2-PD, 47.5±28.2% for IPD, and 68.2±24.7% for controls; p = 0.002) and forced-choice identification (46.2±21.2% for LRRK2-PD, 37.9±17.7% for IPD, and 65.7±14.2% for controls; p = 0.001) (Table 2). No differences were found between LRRK2-PD and IPD subjects regarding smell detection (p = 0.501), memory/recognition (p = 0.505), or forced-choice identification (p = 0.157).
BAST-24 subjective olfactometry outcomes in patients with idiopathic (IPD) and genetic (LRRK2-PD) Parkinson’s disease versus healthy controls
anumber of odours (mean±standard deviation). bpercentage (mean±standard deviation). IPD, idiopathic Parkinson’s disease; LRRK2-PD, LRRK2 associated Parkinson’s disease. Data were presented as frequencies and mean (SD) for discrete and continuous variables, respectively. Comparison were performed with the Kruskal-Wallis test.
UPSIT scores were higher in LRRK2-PD than in IPD (22.5±8.0 vs. 18.8±6.0; p = 0.042). Controls showed the highest UPSIT scores (30.3±4.7). All the IPD patients and 23 (95.8%) of the LRRK2-PD patients had hyposmia or anosmia, assessed by means of the UPSIT. The distribution of normosmic, hyposmic, or anosmic subjects was similar between LRRK2 and IPD patients (p = 0.145), but different between patients (LRRK2 and IPD together) and controls (p < 0.001).
A strong correlation was found between the forced-choice identification of the BAST-24 test and the UPSIT in all the study groups (Fig. 2). A significant correlation was also found between the smell detection of the BAST-24 and the UPSIT in the IPD group (r = 0.424; p = 0.009). No correlations were found between smell detection and UPSIT in LRRK2-PD or controls neither between smell memory and UPSIT scores in any study group.
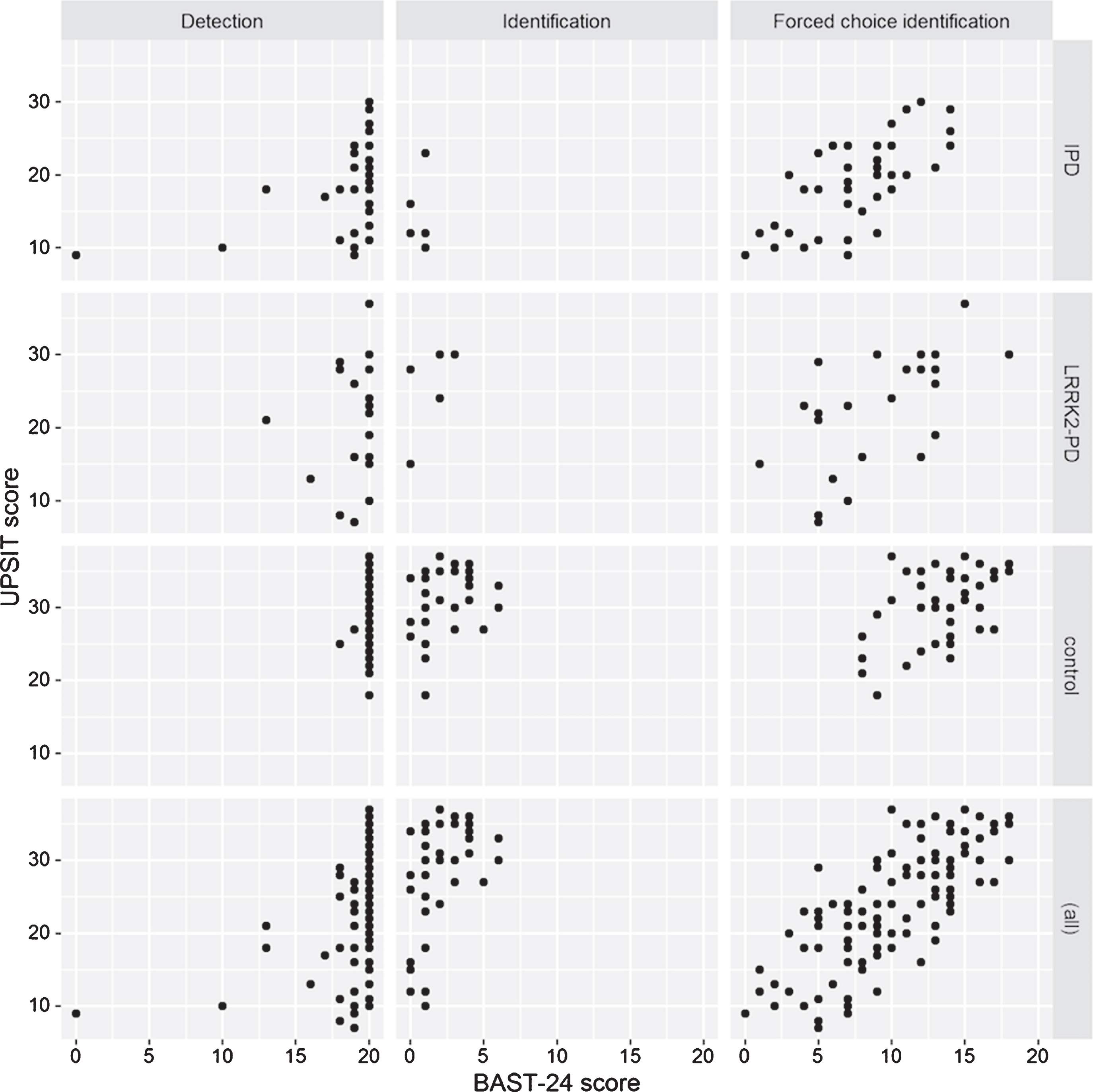
Correlations between BAST and UPSIT subjective olfactometry outcomes in all the study population.
DISCUSSION
The majority of LRRK2-PD patients in this study reported subjective impairment in their sense of smell, assessed by means of a semi-structured interview, and also had hyposmia, assessed by the UPSIT. However, the impact of the olfactory loss in daily living activities in this group of genetic PD was low, similar to IPD. No differences were found between LRRK2-PD and IPD subjects regarding smell detection, memory, or forced-choice identification assessed with the BAST-24 test.
The impact of smell disturbances upon the patient’s wellbeing, activities of daily life or quality of life has not been evaluated before in PD, unlike for other non-motor symptoms, such as fatigue, depression or pain, that have been shown to have a negative impact on quality of life in PD patients [18, 19]. In particular, the repercussion of smell dysfunction in LRRK2-PD patients has not previously reported. Our study suggests that the impact of hyposmia was generally low and it was striking that despite a prominent smell loss detected by the UPSIT, the majority of patients did not seem to be too disturbed by such symptom. Perhaps this apparent low concern reflects the gradual development of smell loss and its chronicity. It is also possible that the questionnaire we used may not have been sensitive enough to detect the impact of the olfactory deficit. The Questionnaire of Olfactory Disorders [20] that specifically addresses olfactory dysfunction and its impact on a patient’s daily life was not used since it is not validated in Spain.
In line with other studies most of our LRRK2-PD patients had smell loss compared to controls but the difference in mean UPSIT scores between LRRK2-PD and IPD was small. The frequency of patients with smell loss and the distribution of normosmic, hyposmic and anosmic subjects was similar between LRRK2 and IPD patients and no differences were found between LRRK2-PD and IPD regarding smell characteristics using the BAST-24 test. Several studies have reported that smell loss in LRRK2-PD was less intense than in IPD but others have not [5]. The variable results obtained in olfactory testing in the LRRK2-PD population may be attributed on the one hand to the subjectivity of the different methods used to assess loss of smell, the small sample size of some studies, or different ethnicities and cultural aspects of the subjects studied. Also, it could reflect the heterogeneous pathology of LRRK2-PD carrying the G2019S mutation, with some cases lacking central nervous system alpha-synuclein aggregates. Information on smell is infrequently reported in LRRK2-PD studies and is missing in more than 90% of reported patients [17]. Additional studies are needed to clarify the frequency and severity of hyposmia in LRRK2-PD.
The most frequent smell tests used in PD are the UPSIT and the Sniffin’ sticks test [21]. These tests, however, may not be available to most clinicians, are time-consuming, expensive, and their reliability has been estimated to be poor. The BAST-24 test, validated for the Spanish population, evaluates different smell characteristics, not evaluated with the usual smell tests used in PD. In our study, BAST-24 scores were lower in PD patients compared to controls, suggesting that it could be a useful test to assess olfaction in PD, but it did not differentiate between LRRK2-PD and IPD subjects. The BAST-24 test is, however, time-consuming, which makes it a difficult tool for smell screening in routine clinical practice. A bed-side, fast, and easily-applicable tool to detect those patients with olfactory deficit in daily practice is not yet available. The short version of the UPSIT test or a recently developed 6-item self-administered hyposmia rating scale [22] are less time consuming than the standard UPSIT or the Sniffin’ sticks test. Additional studies are however needed to confirm the reliability of these tests in different populations before they can be considered as reference in the clinical setting.
Some limitations of our study should be mentioned. First, the lack of a validated tool to assess the impact of smell dysfunction on quality of life or on daily function. Also, the relatively low sample size could interfere in the detection of differences between study groups.
In summary, the majority of LRRK2-PD patients from our study reported subjective smell impairment and presented hyposmia when evaluated with validated smell tests, but with a low impact upon daily living activities. No differences were found between LRRK2-PD and IPD regarding the frequency of hyposmia or anosmia. Some LRRK2-PD patients reported that the onset of smell loss started before the onset of motor symptoms but further studies are needed to clarify if smell loss occurs in the prodromal phase of LRRK2-PD.
CONFLICT OF INTEREST
AC has received funding from “Barcelona Institute for Global Health (ISGlobal)”.
YC has received funding, research support and/or honoraria in the last 12 months from Union Chimique Belge (UCB pharma), Teva, Abbvie, Merz, Piramal Imaging, Esteve, Zambon and Bial.
MJ Martí received honoraria for lecturing or advisory boards from AbbVie and Merz. Received research grants from CIBERNED, Fondo Investigación Sanitaria, Fundació la Marato de TV3 and the Michel J Fox Foundation.
JM reports personal fees and/or research grants from Allakos, Sanofi-Genzyme & Regeneron, MYLAN-MEDA Pharma, URIACH Group, ALK-Abelló, Menarini Group, MSD, and Genentech-Roche-Novartis outside the submitted work and for the preceding 12 months.
ET received honoraria for consultancy from Novartis, TEVA, Bial, Accorda, Boehringer Ingelheim, UCB, Solvay, Lundbeck, Abbvie and BIOGEN and has received funding for research from Spanish Network for Research on Neurodegenerative Disorders (CIBERNED)- Instituto Carlos III (ISCIII), and The Michael J. Fox Foundation for Parkinson’s Research (MJFF)
Dolores Vilas, María Quintana, Claustre Pont-Sunyer, Meritxell Santos, and Francesc Valldeoriola do not have financial disclosures.
Footnotes
ACKNOWLEDGMENTS
The authors are grateful to the patients and healthy volunteers that participated in this study. They also acknowledge the support of the CERCA Program of Generalitat de Catalunya.
