Abstract
Background:
Olfactory or autonomic dysfunction is one of the earliest prodromal symptoms of Parkinson’s disease (PD). It has not been investigated whether PD patients have different phenotypes depending on the presence of these prodromal symptoms.
Objective:
To investigate whether hyposmia-dominant and dysautonomia-dominant patients with early PD have different clinical manifestations and nigrostriatal degeneration.
Methods:
This cross-sectional study recruited 168 drug-naive PD patients and 34 control subjects. PD patients were classified as patients without hyposmia and dysautonomia (PD–H–D–, n = 51), hyposmia-dominant patients (PD–H+D–, n = 36), dysautonomia-dominant patients (PD–H–D+, n = 33), and patients with hyposmia and dysautonomia (PD–H+D+, n = 48). We then compared the baseline clinical characteristics, striatal specific to non-specific binding ratio (SNBR), neuropsychological performance, and neuropsychiatric symptoms among the groups.
Results:
The PD–H+D–group had a lower SNBR in the ventral striatum (p = 0.013), a greater asymmetric index of striatal SNBRs, and higher prevalence of apathy (p = 0.021) than the PD–H–D+ group. The PD–H–D+ group had older age at onset (p = 0.043) and a higher prevalence of REM sleep behavior disorder (p = 0.041) than the PD–H+D–group. The PD–H+D+ group had higher motor deficits, lower cognitive function, and lower SNBRs in all striatal subregions than the PD–H–D–group. Decreased SNBRs in the anterior caudate, posterior caudate, and ventral striatum were associated with the presence of apathy.
Conclusion:
The present study suggests that hyposmia-dominant and dysautonomia-dominant PD have different clinical characteristics and patterns of striatal dopamine depletion.
INTRODUCTION
Parkinson’s disease (PD) is a multisystem disorder characterized by α-synuclein-containing inclusion bodies in the form of Lewy bodies and Lewy neurites and selective degeneration of dopaminergic neurons in the substantia nigra pars compacta [1]. The distribution of pathologic α-synuclein aggregates in the brain follows highly predictable spatiotemporal patterns [2]. According to Braak staging, α-synuclein pathology originates in the anterior olfactory nucleus and the dorsal motor nucleus of vagus [3]. This pathology then spreads in a rostral direction to the midbrain, eventually reaching the cerebral cortex. Patients with PD develop various motor and nonmotor symptoms depending on the spread of α-synuclein pathology.
Dual-hit of α-synuclein pathology via both the nasal and vagal routes can be the initial event of sporadic PD [4]. Accordingly, olfactory or autonomic dysfunction is one of the earliest prodromal symptoms of PD. Olfactory deficits can precede clinically overt parkinsonian motor symptoms by at least 4 years [5], while substantial autonomic dysfunction is observable from 5 up to 20 years before PD diagnosis [6]. Besides its role as a prodromal marker, olfactory dysfunction is closely associated with cognitive dysfunction and dementia conversion in PD [7]. Moreover, earlier autonomic dysfunction is associated with more rapid disease progression and shorter survival in patients with PD [8]. Collectively, these observations suggest that the initiation site of α-synuclein pathology can not only determine the dominant prodromal nonmotor symptoms but may also affect the pattern of the spreading of pathology and the disease progression.
In the present study, we hypothesized that hyposmia-dominant and dysautonomia-dominant PD may have different clinical features and patterns of nigrostriatal degeneration. Thus, we grouped drug-naïve patients with PD based on the presence of hyposmia and dysautonomia and comparatively analyzed their clinical, neuropsychological, and neuropsychiatric features and patterns of nigrostriatal dopaminergic degeneration.
METHODS
Study participants
We recruited 168 patients with drug-naïve PD who visited the Movement Disorders outpatient clinic at Severance Hospital, Yonsei University Health System, between May 2015 and July 2020. PD was diagnosed based on the clinical diagnostic criteria of the United Kingdom PD Society Brain Bank. Patients underwent baseline assessments that included a neurological examination, olfactory function testing, autonomic function testing, neuropsychological testing, neuropsychiatric testing, brain magnetic resonance imaging (MRI), and N-(3-[18F] fluoropropyl)-2β-carbomethoxy-3β-(4-iodophenyl) nortropane (18F-FP-CIT) positron emission tomography (PET) scanning. Only patients having decreased dopamine transporter (DAT) availability in the posterior putamen were included, which was interpreted by nuclear medicine physicians blinded to the clinical status of the patients. The exclusion criteria for PD participants were as follows: probable PD dementia based on baseline neuropsychological test results; severe white matter hyperintensity, multiple lacunae in the basal ganglia, or hydrocephalus on brain MRI; conditions affecting olfactory function (e.g., nasal fracture, chronic rhinosinusitis, nasal polyps, or a recent history of a common cold) or autonomic function (e.g., autoimmune disease, alcohol abuse, or spinal cord injury); major neurological, psychiatric, or metabolic illnesses; atypical parkinsonism; or history of drug use causing parkinsonian symptoms. Parkinsonian motor symptoms were assessed with the patient in the drug-naïve state at the time of 18F-FP-CIT PET acquisition using the motor subscales of the Unified PD Rating Scale (UPDRS-III). Rapid eye movement sleep behavior disorder (RBD) was assessed using the RBD Screening Questionnaire and diagnosed when patients with PD scored of 6 or more on the RBD Screening Questionnaire [9].
We also enrolled 34 healthy controls in this study who were registered in the normal subject cohort in our clinic. They did not have any subjective symptoms of cognitive impairment or history of neurologic or psychiatric illnesses. All control subjects had normal cognitive function as assessed using the Korean version of the Mini-Mental State Examination (K-MMSE≥26) and performed brain MRI and 18F-FP-CIT PET scanning.
The present study was approved by the Institutional Review Board of Yonsei University Severance Hospital. Written informed consent was obtained from all the participants.
Assessment of olfactory function
Olfactory function was assessed using the Cross-Cultural Smell Identification Test (CCSIT), which consists of 12 smells [10]. Participants were required to scratch the panel, sniff the sample, and select one of four possible answers. Their score was calculated as the sum of the correct responses. Based on previous studies, normosmia was defined as a score of 9–12, mild hyposmia as a score of 7–8, and severe hyposmia as a score of 0–6 [11].
Assessment of autonomic function
Comprehensive autonomic function tests were used to evaluate the sudomotor, cardiovagal, and adrenergic functions of the patients [12]. Sudomotor function was assessed using the Quantitative Sudomotor Axon Reflex Test. Cardiovagal function was assessed using heart-rate response to deep breathing and the Valsalva ratio. Adrenergic function was assessed using beat-to-beat blood pressure, heart-rate response during phases II and IV of the Valsalva maneuver, and the head-up tilt test. The Composite Autonomic Severity Score (CASS), which consists of three subscores (CASS sudomotor, 0–3; CASS cardiovagal, 0–3; CASS adrenergic, 0–4), was calculated using the results of the above tests, as previously described [12]. The CASS results were then used to classify patients as having no dysautonomia (CASS = 0), mild dysautonomia (CASS = 1–3), moderate dysautonomia (CASS = 4–6), and severe dysautonomia (CASS = 7–9).
Grouping of subjects according to autonomic and olfactory dysfunction
The distribution of patients according to their autonomic or olfactory function is provided in Fig. 1A. For the autonomic function test, we set a cut-off score of 3/4 as previously described [13]. Patients with a Composite Autonomic Severity Score (CASS) of 3 or less were classified as having either mild or no evidence of dysautonomia (n = 87) and those with a CASS of 4 or more as having moderate to severe dysautonomia (n = 81). Regarding olfactory function, we set a cut-off score of 6/7 to define definite hyposmia as described in a previous study [11]. Patients with a Cross-Cultural Smell Identification Test (CCSIT) score of 7 or more were classified as having mild or no evidence of hyposmia (n = 84) and those with a CCSIT score of 6 or less as having severe hyposmia (n = 84). PD patients were grouped according to their CASS and CCSIT scores (Fig. 1B) as follows: patients with mild or no evidence of hyposmia and dysautonomia (PD without hyposmia and dysautonomia; PD–H–D–, n = 51); patients with severe hyposmia and mild or no evidence of dysautonomia (hyposmia-dominant PD; PD–H+D–, n = 36); patients with mild or no evidence of hyposmia and moderate to severe dysautonomia (dysautonomia-dominant PD; PD–H–D+, n = 33); and patients with severe hyposmia and moderate to severe dysautonomia (PD with hyposmia and dysautonomia; PD–H+D+, n = 48).
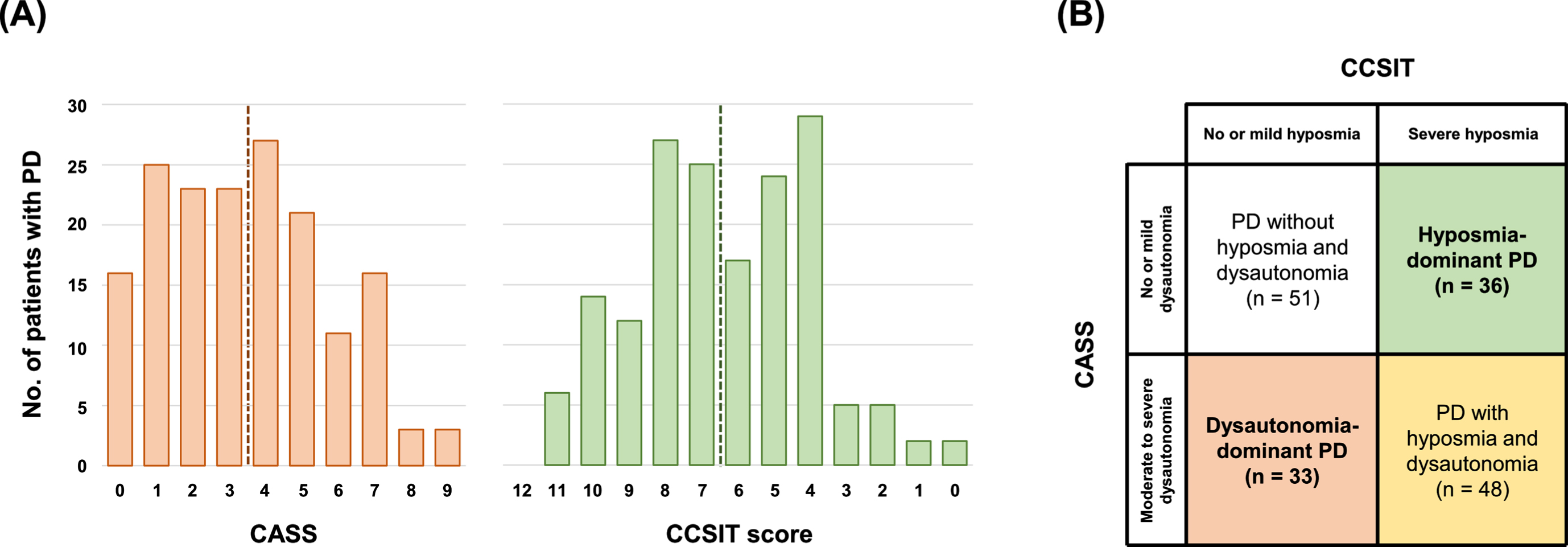
Distribution of autonomic and olfactory function scores and classification of patients with PD. (A) Frequency of CASS and CCSIT scores in all patients with PD. The vertical dotted line indicates the cut-off value for significant hyposmia or dysautonomia. (B) Grouping of patients with PD according to the presence of hyposmia and moderate to severe dysautonomia. PD, Parkinson’s disease; CASS, Composite Autonomic Severity Score; CCSIT, Cross-Cultural Smell Identification Test.
Neuropsychological testing
All participants underwent a standardized neuropsychological battery called the Seoul Neuropsychological Screening Battery [14], which contains the following scorable tests: digit span (forward and backward), repetition, Korean version of the Boston Naming Test, 6-point pentagon drawing test, Rey-Osterrieth Complex Figure Test (RCFT; copying, immediate and 20-minute delayed recall, and recognition), Seoul Verbal Learning Test (SVLT; immediate recall, 20-minute delayed recall, and recognition), contrasting programming and go/no-go test, clock drawing test, phonemic and semantic Controlled Oral Word Association Test, Stroop Test (word and color reading), and K-MMSE. The following 2 tests were designated to represent each of the cognitive domains except language as previously described [15]: digit span task and Stroop test for attention domain; the Korean version of the Boston Naming Test for language domain; RCFT copy and pentagon drawing test for visuospatial domain; SVLT and RCFT for memory domain; and clock drawing test and Controlled Oral Word Association Test for frontal executive domain. Each score was converted into a standardized score (z score) on the basis of the age- and education-specific norms [16]. The scores in each cognitive domain were classified as abnormal when they were below 1.5 standard deviations of the norms. A composite score was calculated for each cognitive domain by dividing the sum of the z scores by the number of tests.
Neuropsychiatric testing
We assessed neuropsychiatric symptoms of patients by interviewing their caregivers using the Neuropsychiatric Inventory (NPI) [17]. The NPI evaluated 10 behavioral and psychological symptoms of dementia: delusions, hallucinations, agitation/aggression, depression/dysphoria, anxiety, elation/ euphoria, apathy/indifference, disinhibition, irritability/lability, and aberrant motor behavior. Each symptom was rated retrospectively on the basis of the patients’ condition. The severity of each manifestation was classified into grades (ranging from 0, absent, to 4, frequent). The NPI score was calculated by multiplying severity by frequency on a scale ranging between 0 and 12. Higher scores indicate greater psychopathology.
PET-CT image acquisition
All subjects were injected with 185 MBq (5 mCi) of 18F-FP-CIT and the PET images were acquired 90 minutes after injection. 18F-FP-CIT PET acquisition was performed on a GE Discovery 600 PET/CT scanner (General Electric Healthcare, Milwaukee, MI, USA). The images were acquired for 15 min following CT scan for attenuation correction. The spiral CT scan was performed with a 0.8 s/rotation at 120 kVp, 10 mA, 3.75 mm slice thickness, 0.625 mm collimation and 9.375 mm table feed per rotation. 18F-FP-CIT PET images were reconstructed using the ordered subset expectation maximization (OSEM) algorithm with 4 iterations and 32 subsets. Gaussian filter with 4 mm full-width at half-maximum (FWHM) was applied into reconstructed PET images, which is a 256 x 256 matrix with 0.98 mm pixel and 0.98 mm slice thickness.
Quantitation of the 18F FP-CIT PET-CT images
Image processing was performed using MATLAB (The MathWorks, Inc, Natick, MA) based software statistical parametric mapping (SPM8) and ITK-SNAP (http://www.itksnap.org). All reconstructed 18F-FP-CIT images were normalized to the 18F-FP-CIT template, which was created using 18F-FP-CIT PET images and T1-weighted MR images of 40 healthy controls as described previously [18]. All healthy controls used to create the FP-CIT template had no previous history of neurologic or psychiatric illness. They showed normal cognitive function on all neuropsychological tests, and exhibited normal findings in neurologic examination, structural MRI, and 18F-FP-CIT PET. Twelve volume-of-interests (VOIs) were drawn on the FP-CIT template as described previously [19]. Briefly, the striatum was divided into the dorsal and ventral portions using the anterior-posterior commissure line on the transverse plane. The ventral portion comprised two subregions: the ventral striatum and ventral putamen. The dorsal portion was divided into the following anterior and posterior subregions along the coronal anterior commissure plane: anterior caudate, posterior caudate, anterior putamen, and posterior putamen. For non-specific binding, mean standardized uptake value was calculated by drawing two occipital VOIs, one on each side. The level of DAT availability in each VOI was calculated in terms of the specific to non-specific binding ratio (SNBR) as follows: (mean standardized uptake of the striatal subregional VOI –mean standardized uptake of the occipital VOI) / (mean standardized uptake of the occipital VOI). The normalized SNBR was defined as follows: (SNBR for patient / mean SNBR for healthy controls)×100. Asymmetric index values of SNBRs for each striatal subregion were also calculated as follows: (less affected –more affected) / (less affected + more affected) [20].
Statistical analyses
The baseline clinical and neuropsychiatric characteristics of the patients were analyzed using analysis of variance for continuous variables and χ2 or Fisher’s exact test for categorical variables. In the comparison of substriatal SNBRs and their asymmetry among the PD and HC groups, analysis of covariance was performed after controlling for age at PD onset and sex. Regarding the striatal subregion which had a significant difference in the analysis of covariance, we further controlled for the SNBR of the posterior putamen to investigate the significant difference at a similar level of PD-specific nigrostriatal dopaminergic degeneration. Since there was a significant difference in age at PD onset (Table 1), we also performed group comparisons of striatal SNBRs reflecting age-dependent declines in DAT availability (6.6%decline per decade) [21]. In the comparison of neuropsychological performance among the PD groups, analysis of covariance was performed after controlling for age at PET scan, sex, and disease duration. The prevalence of neuropsychiatric symptoms was compared using χ2 or Fisher’s exact test as appropriate. Post-hoc subgroup comparisons were performed using the false discovery rate approach. Logistic regression analysis was used to investigate the substriatal SNBR associated with the development of neuropsychiatric symptoms which showed significant differences among the groups, after controlling for age at PD onset, sex, and disease duration. Data were analyzed using SPSS software, ver. 23 (IBM Corporation, Armonk, NY, USA). p-values less than 0.05 were considered significant.
Demographic and clinical characteristics of patients with PD
Values are expressed as mean±standard deviation or number (percentage). p-values represent the significance probability of analysis of variance, χ2 test, or Fisher’s exact test. Post-hoc subgroup analyses were performed using the false discovery rate method. Significant differences in the comparison between the aPD–H–D–vs. PD–H+D–groups, bPD–H–D–vs. PD–H–D+groups, cPD–H–D–vs. PD–H+D+ groups, dPD–H+D–vs. PD–H–D+ groups, ePD–H+D–vs. PD–H+D+ groups, fPD–H–D+ vs. PD–H+D+ groups, and gany PD group vs. HC. D, dysautonomia; H, hyposmia; HC, healthy control; K-MMSE, the Korean version of the Mini-Mental State Examination; PD, Parkinson’s disease; RBD, rapid eye movement sleep behavior disorder; UPDRS, Unified Parkinson’s Disease Rating Scale.
RESULTS
Demographic characteristics
The baseline demographic characteristics of the patients are summarized in Table 1. The PD–H–D+ and PD–H+D+ groups had older age at PD onset than the PD–H–D–and PD–H+D–groups (for PD–H+D–vs. PD–H–D+, p = 0.043). The PD–H–D–group had fewer male subjects and higher K-MMSE total scores than the PD–H+D–and PD–H+D+ groups. The UPDRS-III motor score was higher in the PD–H+D+ group than in the PD–H–D–group. The prevalence of RBD in the PD–H–D+ and PD–H+D+ groups was significantly higher than the PD–H–D–and PD–H+D–groups (for PD–H+D–vs. PD–H–D+, p = 0.041). However, years of education, disease duration, and the proportion of vascular risk factors did not differ among the groups.
Group comparisons of striatal SNBRs and their asymmetry
Compared to the healthy controls, all PD groups showed significantly lower SNBRs and greater asymmetries in all striatal subregions (Supplementary Table 1). In post-hoc analysis, the PD–H+D+ group had significantly lower SNBRs in all striatal subregions than the PD–H–D–and PD–H–D+ groups and in the putaminal subregions than the PD–H+D–group. In particular, the PD–H+D–group had a lower SNBR in the ventral striatum than the PD–H–D+ group (p = 0.013). The significant difference in the ventral striatum remained unchanged even after controlling for the SNBR of the posterior putamen (p = 0.024); however, SNBRs in other striatal subregions, including the caudate and putamen, were comparable between the two PD groups. The comparison of the substriatal SNBRs after reflecting age-dependent decline in the DAT availability also showed similar results (Supplementary Table 1). Heatmaps of the normalized SNBRs in the bilateral striata in each PD group are schematically illustrated in Fig. 2.
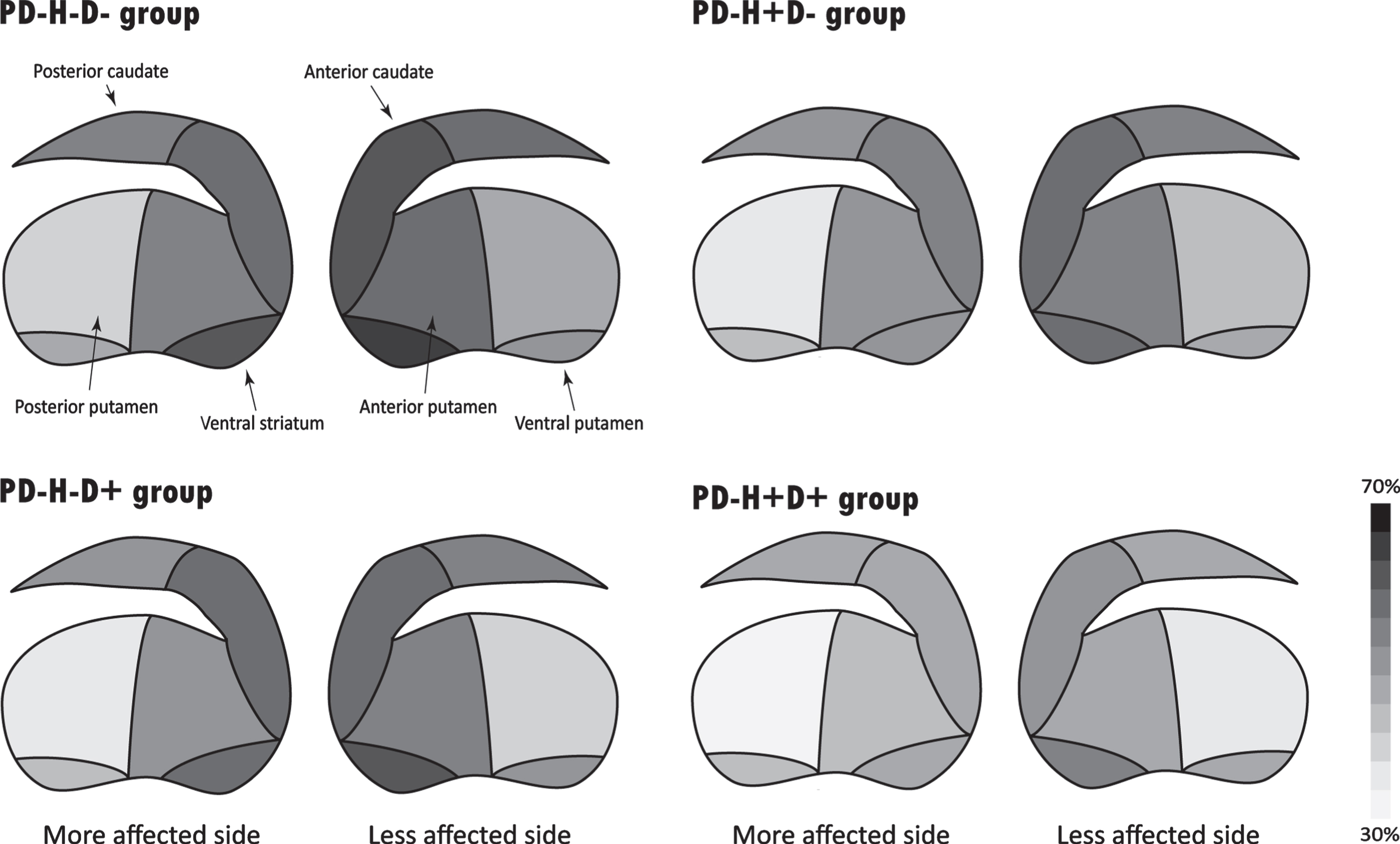
Heatmaps of normalized specific-to-nonspecific binding ratio (SNBR) in the bilateral striata on a scale of 30%(white) to 70%(black). In the comparison between the PD–H+D–and PD–H–D+ groups, the PD–H+D–group had a lower SNBR in the ventral striatum than the PD–H–D+ group and a greater asymmetric index of the SNBRs in all striatal subregions except the posterior caudate than the PD–H–D+ group.
As for asymmetry of the SNBR, the PD–H+D–group showed a significantly greater asymmetric index of the SNBRs in all striatal subregions except the posterior caudate than the PD–H–D+ group, in the anterior caudate than the PD–H–D–group, and in the posterior putamen and ventral putamen than the PD–H+D+ groups (Supplementary Table 2).
Group comparisons of neuropsychological performance and neuropsychiatric symptoms
Among the 13 neuropsychological tests, the PD groups showed significant differences in terms of RCFT copy (p = 0.010) and Stroop color reading (p = 0.024; Table 2). In post-hoc analysis, the PD–H–D–group showed significantly higher RCFT copy scores than the PD–H+D–and PD–H+D+ group and Stroop color reading scores than the PD–H+D+ group. There were no differences in terms of any neuropsychological test when the PD–H+D–and PD–H–D+ groups were directly compared.
Comparison of neuropsychological and neuropsychiatric profiles among the PD groups
Values are expressed as estimated mean (standard error) or number (percentage). p-values represent the significance probability of analysis of covariance adjusted for age at PD onset, sex, and disease duration, χ2 test, or Fisher’s exact test. Post-hoc subgroup analyses were performed using the false discovery rate method. Significant differences in the comparison between the aPD–H–D–vs. PD–H+D–groups, bPD–H–D–vs. PD–H–D+ groups, cPD–H–D–vs. PD–H+D+ groups, dPD–H+D–vs. PD–H–D+ groups, ePD–H+D–vs. PD–H+D+ groups, fPD–H–D+ vs. PD–H+D+ groups. COWAT, Controlled Oral Word Association Test; D, dysautonomia; H, hyposmia; K-BNT, the Korean version of the Boston Naming Test; PD, Parkinson’s disease; RCFT, Rey Complex Figure Test; SVLT, Seoul Verbal Learning Test.
Regarding neuropsychiatric symptoms, only the apathy/indifference item showed different prevalence among the groups (p = 0.012; Table 2). The PD–H+D–and PD–H+D+ groups had a significantly higher prevalence of apathy/indifference than the PD–H–D–and PD–H–D+ groups (for PD–H+D–vs. PD–H–D+, p = 0.024).
In logistic regression analysis, the SNBRs in the ventral striatum (adjusted odds ratio [OR] = 0.35, 95%confidence interval [CI] = 0.10–0.92) as well as the caudate (anterior caudate, adjusted OR = 0.49, CI = 0.26–0.93; posterior caudate, adjusted OR = 0.041, CI = 0.18 –0.94) were significantly associated with the presence of apathy/indifference in patients with PD (Table 3).
Logistic regression analysis of substriatal SNBRs for apathy/indifference in PD
Data are the results of logistic regression analysis with age at PD onset, sex, and disease duration as covariates. PD, Parkinson’s disease; SNBR, specific-to-nonspecific binding ratio.
DISCUSSION
The present study investigated the differences in clinical manifestation and striatal DAT availability among drug-naïve patients with PD grouped by the severity of olfactory and autonomic dysfunction. According to the presence of hyposmia and/or dysautonomia, PD patients showed different characteristics: 1) hyposmia-dominant PD patients had a significantly lower SNBR in the ventral striatum, greater asymmetry in the striatal SNBRs, and a higher prevalence of apathy; 2) dysautonomia-dominant PD patients showed older age at onset and a higher proportion of RBD; 3) compared to those without hyposmia and dysautonomia, PD patients who had both hyposmia and dysautonomia showed a severe phenotype in terms of older age at onset, higher UPDRS motor scores, lower SNBRs in all striatal subregions, visuospatial and frontal executive dysfunction, and apathy; and 4) decreased SNBRs in the caudate and ventral striatum were associated with the presence of apathy. These results suggest that hyposmia-dominant and dysautonomia-dominant PD have different clinical characteristics and patterns of striatal dopamine depletion.
Hyposmia-dominant patients with PD had more profound dopaminergic depletion in the ventral striatum than those with dysautonomia-dominant PD. Previous studies have presented conflicting results regarding DAT correlates of olfactory dysfunction in PD: putamen [22]; caudate [23]; hippocampus and ventral striatum [24]; or no association [7]. However, these studies did not control for the severity of PD in terms of factors such as DAT availability in the posterior putamen or UPDRS motor score. Here, we demonstrated that there is additional dopaminergic impairment in the ventral striatum in patients with hyposmia-dominant PD at a similar level of dopaminergic depletion in the posterior putamen. We can speculate that α-synuclein pathology originating via olfactory route may spread not only into the substantia nigra but also substantially into the ventral tegmental area, resulting in degeneration of the mesolimbic dopaminergic pathway. Inoculation of herpes simplex virus into the rat olfactory bulb resulted in the infection of several regions, including the ventral tegmental area [25]. Evidence from an animal study also suggested a connection between the ventral tegmental area and the olfactory tubercle [26]. In addition, the higher prevalence of apathy observed in hyposmia-dominant patients with PD seems to provide more evidence for the involvement of the ventral striatum. In line with our study, apathetic PD patients in other studies also showed poor performance in the olfactory function test and vice versa [27, 28]. Furthermore, previous studies have reported that apathy is closely associated with dysfunction in the ventral striatum in PD [29]. Collectively, these results suggest that the initiation of PD pathology via the olfactory route would involve the mesolimbic as well as the nigrostriatal dopaminergic pathway from the early PD stage, thus resulting in decreased DAT availability in the ventral striatum and apathy.
Patients with hyposmia-dominant PD showed more significant asymmetry of the striatal SNBR than those with dysautonomia-dominant PD. Lateralized PD motor symptoms during the early stages of the disease are associated with asymmetric dopamine depletion in the substantia nigra and persist for over 5 years [30]. Given that asymmetric spread can be one of the determinants of lateralized nigrostriatal degeneration [31], our data could imply that the olfactory route may be associated with asymmetric dopamine depletion relative to the vagal route. Consistent with our study, previous studies have reported that some early stage PD patients had asymmetric deficits in olfactory function, which may affect the unilateral presentation of parkinsonism [32]. The nasal cycle or an asymmetric nasal passage due to bony septal deformities can explain the preferential entry of diverse environmental pathogens more on one side [33]. On the other hand, parkinsonism caused by the ingestion of neurotoxins (Guadeloupian parkinsonism) is typically symmetric [34]. Anatomically, vagal innervation to the enteric nervous system is bilateral, and there are many sites of decussation along the ascending courses of both sympathetic and parasympathetic nerves. Thus, the degree of contribution of dual-hit pathology may be an important determinant of the asymmetry of parkinsonism in early PD.
Dysautonomia-dominant PD was associated with a higher prevalence of RBD and older age at PD onset when compared to hyposmia-dominant PD. Given the neuroanatomical location, this result is plausible because the main neural substrates for RBD belong to the medulla and lower pons [35] and the dorsal motor nucleus of vagus, which is responsible for autonomic function, is also located nearby in the medulla. Consistent with our results, a previous study reported that autonomic symptoms were more prevalent in the RBD group than in controls [36]. As for the onset age, PD patients with dysautonomia developed parkinsonism around the age of 70, about 5 years later than those without hyposmia. Older age at diagnosis is one of the most important risk factors for autonomic dysfunction in PD [8]. However, this result needs to be interpreted cautiously because autonomic dysfunction can be more prevalent with aging in healthy people [37]. A cohort of patients with autonomic or olfactory dysfunction without PD is necessary to elucidate the association between dysautonomia or hyposmia and later onset of parkinsonian motor symptoms.
As expected, PD with both hyposmia and dysautonomia was associated with severe phenotypes in terms of motor deficit, cognitive dysfunction, neuropsychiatric symptoms, and striatal dopaminergic depletion. Hyposmia has been reported to be associated with cognitive dysfunction and dementia conversion [7], while dysautonomia was associated with more rapid disease progression and shorter survival in PD [8]. Orthostatic hypotension is one of the most important nonmotor symptoms that identify the diffuse/malignant phenotype of patients with PD [38]. Considering Braak’s hypothesis, we can infer from these previous studies that the pathologic burden in either route would affect clinical manifestation and progression in PD. Hits by both the nasal and vagal routes may lead to a greater pathological burden on the subsequent stage of α-synuclein spreading, thereby resulting in severe dopaminergic depletion and motor and nonmotor symptoms. Longitudinal follow-up of patients with PD having both hyposmia and dysautonomia at the time of diagnosis is required to determine whether this group would be a different and malignant subtype.
Recently, the α-Synuclein Origin and Connectome model proposed new PD subtypes: brain-first and body-first PD [39]. This model is somewhat supported by the present results. In brain-first cases, the first pathology appears unilaterally, often in the amygdala, leading to asymmetric dopaminergic degeneration and motor asymmetry. This subtype may correspond to the patients with hyposmia-dominant PD in that they showed more asymmetric DAT loss. In our previous study, we demonstrated that higher asymmetry of dopamine loss in the posterior putamen was associated with slower disease progression [40], and brain-first PD also shows slower motor and cognitive progression. Amygdala, one of the originating sites of initial α-synuclein pathology in the brain-first type, receives diffuse projection from the ventral tegmental area [41], which may be a spreading target from the olfactory route. In contrast to our present results, however, brain-first PD is thought to have less hyposmia, which is still under debate. Since postmortem studies show that the amygdala and olfactory bulb are the two most common sites of ‘single-location’ α-synuclein pathology in incidental Lewy body disease [42] and since there is no actual data of olfactory function test between the brain-first and body-first subtypes, further studies are necessary to address this issue.
This study had several limitations. First, in this cross-sectional study, all baseline evaluations were performed at the time of PD diagnosis, and we cannot confirm whether the olfactory or autonomic dysfunction were prodromal in all patients. Second, the CASS reflects only cardiac and sudomotor autonomic function and not overall autonomic functions. A comprehensive evaluation of the autonomic nervous system will be required to more accurately assess dysautonomia in PD. Third, although the cut-off values used for determining hyposmia and dysautonomia were based on previous studies, they could be considered arbitrary and borderline. Fourth, the diagnosis of RBD was not based on polysomnographic study. The questionnaire-based diagnosis of RBD in this study may be insufficient for diagnosing RBD in de novo PD because of its low sensitivity [43]. Alternatively, when we applied another cut-off score of 6/7 as a sensitivity analysis, there was still a significant difference in the prevalence of RBD between the PD–H+D–and PD–H–D+ groups (data not shown). Nevertheless, the results should be interpreted cautiously. Fifth, although we tried to compensate for age-dependent declines in striatal DAT availability by adjusting for age or applying 6.6%decline per decade, these are not optimal methods. Data on DAT availability in a large number of normal subjects spanning a wide age range will be necessary. Finally, PD progression does not follow the Braak staging scheme in all cases. Given that around 30%of patients develop PD without impaired olfaction and autonomic dysfunction in this study, factors other than dual-hit and stereotypic spreading may also affect PD pathogenesis and progression. Thus, cautious interpretation is necessary before generalizing these results.
In conclusion, the present study demonstrated the differences in clinical and dopamine depletion patterns between hyposmia- and dysautonomia-dominant PD, suggesting that pathologic propagation originating either from the lower brainstem or the olfactory system may be an important determinant of clinical heterogeneity in early PD.
Footnotes
ACKNOWLEDGMENTS
This work was supported by a grant from the Korea Health Technology R&D Project through the Korean Healthy Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HU21C0053) awarded to Phil Hyu Lee.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
