Abstract
Background:
Fatigue is a common and disabling non-motor symptom in Parkinson’s disease (PD). Autonomic dysfunction is suggested as the possible pathophysiology of fatigue, but it has not been investigated in drug-naïve PD patients.
Objective:
In the present study, the relationship between fatigue and autonomic dysfunction in drug-naïve PD patients was investigated.
Methods:
In the present study, 89 drug-naïve PD patients were analyzed. The Parkinson’s disease fatigue scale (PFS) was used to divide the patients into fatigue (mean PFS≥3.3) and non-fatigue groups (mean PFS < 3.3). The autonomic function test (AFT), Scale for Outcomes in Parkinson’s Disease-Autonomic (SCOPA-AUT), Unified Parkinson’s Disease Rating Scale (UPDRS)-I, -II, -III, modified Hoehn and Yahr (H&Y) scale, Montreal Cognitive Assessment (MoCA), Parkinson’s Disease Questionnaire-39 (PDQ-39), Parkinson’s Disease Sleep Scale (PDSS), and Beck Depress Index (BDI) were performed in all the participants. The AFT results and clinical scales were compared using multiple logistic regression analysis.
Results:
The prevalence of fatigue was 23.6% (n = 21) in drug-naïve PD patients. Total SCOPA-AUT score was higher in the fatigue group than in the non-fatigue group. The fatigue group had lower inspiratory:expiratory (I:E) ratio and Valsalva ratio. The prevalence of abnormal sympathetic skin response and orthostatic hypotension (OH) was 19% and 38.1%, respectively, in the fatigue group. Regression model analysis revealed that SCOPA-AUT and OH were the most related factor of fatigue in drug-naïve PD patients.
Conclusion:
Autonomic dysfunction in drug-naïve PD patients was investigated using a subjective scale as well as objective tests. The results indicated that fatigue is associated with autonomic dysfunction, especially OH, in drug-naïve PD patients.
INTRODUCTION
Patients with Parkinson’s disease (PD) have a wide variety of non-motor symptoms [1]. Fatigue is a common and disabling non-motor symptom in PD and affects 13.7%–56% of the drug-naïve PD population [2–7]. Fatigue contributes to poorer quality of life (QoL), functional capacity, and physical function in patients with PD [8, 9]. Various clinical characteristics are associated with fatigue in PD patients including older age, disease duration, L-dopa equivalent daily dose (LEDD), depression, apathy, anxiety, daytime somnolence, and sleep disturbances [10]. In many studies, fatigue was suggested a primary manifestation of PD rather than a secondary symptom of mood disorders, sleep alteration, medication, or motor symptoms [11]. Despite the clinical importance of fatigue, knowledge regarding the pathophysiology of fatigue in PD is limited. Several theories have been suggested including inflammatory cytokines, abnormalities in the hypothalamic-pituitary-adrenal axis, abnormal basal ganglia output to the cortex, and an imbalance between different neurotransmitters as the pathophysiology of fatigue in PD [11]. As presented in the biological model, autonomic dysfunction, especially cardiovascular dysfunction which is common in PD even in the prodromal phase, is a possible pathophysiology of fatigue [12–15]. In PD, autonomic dysfunction and fatigue are affected by anti-parkinsonian medications or disease progression. In this regard, drug-naïve PD patients are an appropriate study population to investigate the relationship between fatigue and autonomic dysfunction and important for understanding the causal mechanisms and treatment of fatigue in PD patients. However, the association between fatigue and autonomic dysfunction has not been investigated in drug-naïve PD patients. Therefore, in the present study, the relationship between fatigue and autonomic dysfunction was investigated in drug-naïve PD patients in the early stage of the disease.
METHODS
Participants
Drug-naïve PD patients were recruited from the Movement Disorders Clinic of Samsung Medical Center between January 2016 and July 2017. The diagnosis of PD was based on the United Kingdom Parkinson’s Disease Brain Bank Criteria [16]. The patients also fulfilled the following inclusion criteria at the time of the investigation: (1) no current or previous exposure to antiparkinsonian medications, (2) disease duration < 36 months from the onset of motor symptoms of PD, (3) modified Hoehn and Yahr (H&Y) stage < 3, (4) decreased DAT uptake in striatal dopaminergic depletion determined using 18F-radiolabeled N-(3-fluoropropyl)-2β-carboxymethoxy-3β-(4-iodophenyl) nortropane PET, (5) normal cognitive function (Montreal Cognitive Assessment, MoCA≥26). All patients enrolled in the study had been treated at our movement disorder clinic for at least 2 years. Patients who had any of the following conditions were excluded: (1) a red flag sign indicating atypical parkinsonism, except autonomic dysfunction after > 2 years of follow-up [17], (2) lack of response or poor response (<30% decrease in Unified Parkinson’s Disease Rating Scale-III, UPDRS-III) to optimal treatment of L-dopa replacement therapy or the patients did not show sustained response to L-dopa after > 2 years of follow-up [18], (3) cardiovascular disease, peripheral neuropathy, diabetes or musculoskeletal diseases which affect autonomic function, (4) history of relevant head injury or cerebrovascular diseases, major medical diseases, or musculoskeletal diseases (5) taking medications influence autonomic function such as tricyclic antidepressants and alpha-adrenergic antagonists (6) structural brain lesion including putaminal atrophy, T2 slit-like putaminal hyperintensity, cerebellar atrophy, high signal intensity of middle cerebellar peduncle, or hot cross bun on brain MRI conducted at least 12 months after onset of motor symptoms or during the follow-up period. This study was approved by the Institutional Review Board of Samsung Medical Center and all subjects provided written informed consent.
Clinical assessment
Patient fatigue was assessed using Parkinson’s disease fatigue scale (PFS) [19]. PFS is a self -administered questionnaire which contains 16 items for assessing fatigue in PD patients. Items are rated on a 5-point Likert scale and average score of all 16 items gives a total PFS score. An average score ≥ 3.3 is considered as fatigue in patients. Autonomic dysfunction was assessed using two methods, a self-administered questionnaire and objective autonomic function test (AFT). Scale for Outcomes in Parkinson’s Disease-Autonomic (SCOPA-AUT) contains 23 items in 6 regional domains (gastrointestinal, urinary, cardiovascular, thermoregulatory, pupillomotor, and sexual dysfunction) [20]. The maximum score is 69 and a higher score indicates more severe dysautonomia symptom. The UPDRS-I, -II, -III, H&Y stage, MoCA, Parkinson’s Disease Questionnaire-39 (PDQ-39) [21], Parkinson’s Disease Sleep Scale (PDSS) [22], and Beck Depression Inventory (BDI) [23] were also used to assess clinical features. The patients were divided into fatigue (mean PFS≥3.3) and non-fatigue groups (mean PFS < 3.3) and clinical features and AFT results were compared [19].
Autonomic function test
All participants underwent our standardized AFT battery which included heart rate response to deep breathing (HRDB), the Valsalva maneuver, blood pressure (BP) and heart rate (HR) response to head-up tilt test (HUT), and sympathetic skin response (SSR) [24, 25]. The patients discontinued medications which could affect the AFT results at least 24 hours before the test. In addition, the participants were prohibited from drinking beverages containing caffeine and smoking on the day of the test. The electrode and BP cuff were attached to the patient and BP was continuously recorded using the Finometer 1 (FMS, Amsterdam, The Netherlands) electrocardiogram and respiratory movements measured while performing the HRDB test, Valsalva maneuver, and HUT. Orthostatic hypotension (OH) was defined as a fall of at least20 mmHg in systolic BP and/or a 10 mmHg fall in diastolic BP within 3 minutes of HUT. In patients with supine hypertension, a reduction in systolic BP of 30 mm Hg was applied. Delayed orthostatic hypotension was defined as reduction of BP appeared from 3 to 5 minutes of HUT. Postural orthostatic tachycardia syndrome (POTS) was defined as a sustained heart rate increment of ≥ 30 beats/min during HUT in the absence of OH [26]. Symptomatic OH patients was defined as to have more than 1 score in cardiovascular regional domain of SCOPA-AUT among the patients with an obvious OH in the HUT [27, 28]. The AFTs were performed based on a previously described protocol [29].
Statistical analyses
The normality of data was evaluated using the Shapiro-Wilk test. Demographic and clinical features were compared using Student’s t-test, Mann-Whitney U test, chi-square, or Fisher’s exact test depending on the variable. Multiple logistic regression analysis was performed to investigate the relationship between fatigue and demographic and clinical variables. The regression coefficients presented are standardized β and partial correlation coefficients. Corrected p-values for multiple testing were based on the Benjamini-Hochberg method [30]. All tests were two-tailed and the α level was set at p < 0.05. Statistical analyses were performed using IBM SPSS for Windows (version 25.0; IBM Inc., Armonk, NY, USA).
RESULTS
Demographic and clinical features
In the present study, 117 drug-naïve PD patients were evaluated, where 28 patients were excluded, and 89 drug-naïve PD patients were analyzed (Fig. 1). The fatigue group included 21 patients and the non-fatigue group 68 patients. The prevalence of fatigue was 23.6% in drug-naïve PD patients. Demographic and clinical assessments are presented in Table 1. Fatigued patients were older than non-fatigued patients. There were no differences in age, sex, disease duration, prevalence of hypertension, body mass index, UPDRS-III, H&Y stage, or MoCA. The fatigue group had higher UPDRS-I, -II, PDQ-39 SI, and BDI scores than the non-fatigue group. Ten of 21 (47.6%) in fatigue group and 12 of 68 (17.6%) in non-fatigue group had significant depressive symptoms (BDI > 16) [31]. Fatigued patients with PD had lower PDSS which reflects poorer quality of sleep.
Flow chart of participants. PD, Parkinson’s disease; AFT, autonomic function test; L-dopa, levodopa. aPutaminal atrophy, T2 slit-like putaminal hyperintensity, cerebellar atrophy, middle cerebellar peduncle sign, hot cross bun sign, and any other structural lesions. bCardiovascular disease, peripheral neuropathy, diabetics, history of relevant head injury, cerebrovascular diseases, major medical diseases, musculoskeletal disease.
Baseline demographics and clinical characteristic of fatigue and non-fatigue groups
PFS, Parkinson’s Disease Fatigue Scale; UPDRS, United Parkinson’s Disease Rating Scale; H&Y, Hoehn & Yahr; MoCA, Montreal Cognitive Assessment; PDQ-39 SI, Parkinson’s Disease Questionnaire-39 Summary Index; NMSS, Non-Motor Symptoms Scale; PDSS, Parkinson’s Disease Sleep Scale; BDI, Beck Depression Inventory; Expressed as the number of patients (%), the mean±standard deviation (SD).
Autonomic dysfunction and fatigue
Total SCOPA-AUT score was higher in the fatigue group than in non-fatigue group. The regional scores were also higher in the fatigue group, except pupillomotor dysfunction. The fatigue group had lower inspiratory:expiratory (I:E) ratio and Valsalva ratio than the non-fatigue group. The prevalence of abnormal SSR was 19% in the fatigue group (Table 2). Among the 89 drug-naïve PD patients, 18 (20.2%) had OH. In the fatigue group, 8 of 21 patients (38.1%) were diagnosed with OH and 6 were symptomatic OH (≥1 in cardiovascular regional domain of SCOPA-AUT). Conversely, 10 of 68 patients (14.7%) had OH and only 2 patients were symptomatic OH in the non-fatigue group. One patient in non-fatigue group had POST and 1 patient in each group diagnosed with delayed OH. Among the patients with POST and delayed OH, only one delayed OH patient in fatigue group had orthostatic symptom (Fig. 2).
AFT results in fatigue and non-fatigue patients with drug-naïve PD
AFT, autonomic function test; PD, Parkinson’s disease; SCOPA-AUT, Scale for Outcomes in Parkinson’s Disease-Autonomic; HRDB, heart rate response to deep breathing; I:E, inspiratory:expiratory; HUT, head-up tilt test; SSR, sympathetic skin response; OH, orthostatic hypotension; Expressed as the mean±standard deviation (SD). aMultiple comparison was adjusted using the Benjamini-Hochberg method.
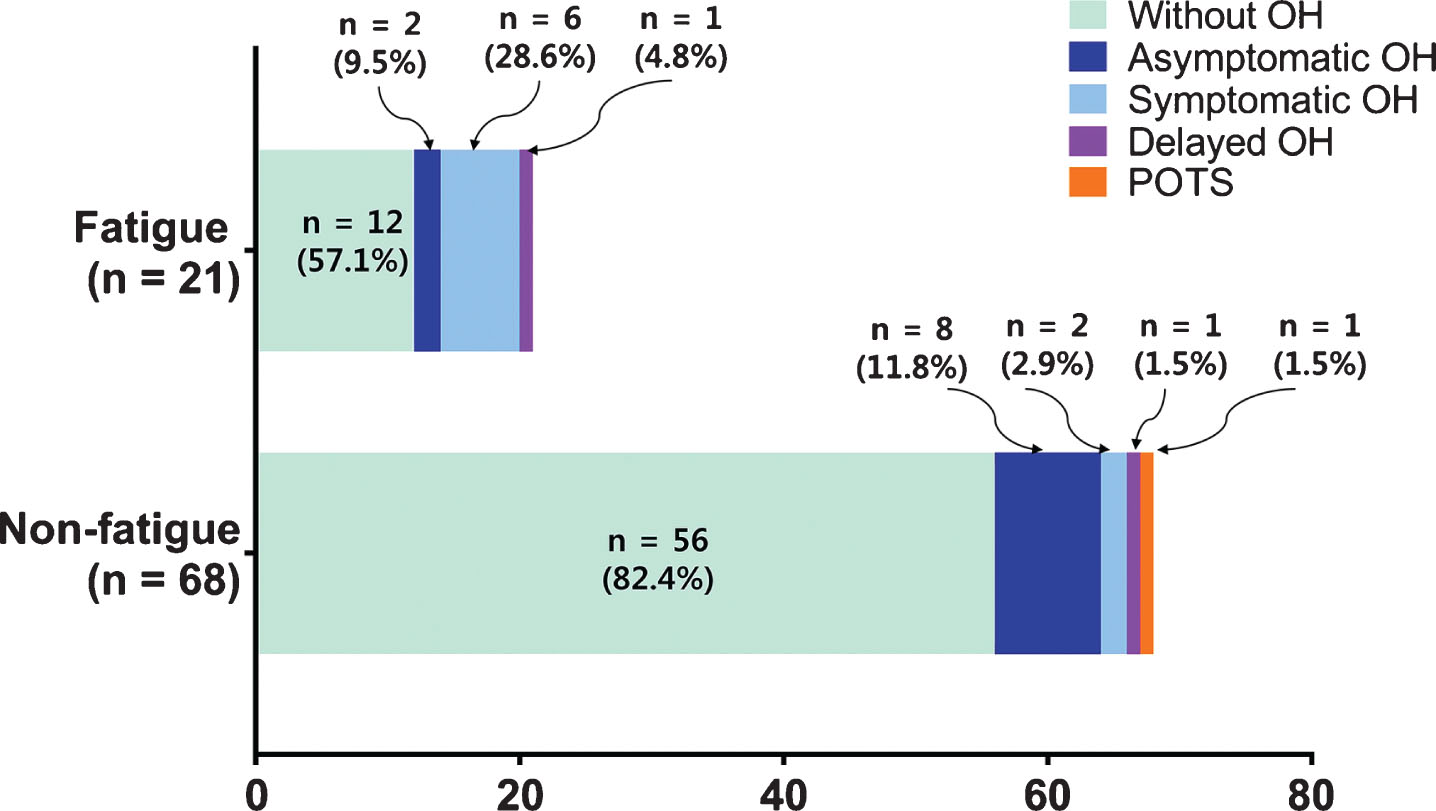
Prevalence of symptomatic OH, asymptomatic OH, delayed OH and POTS in fatigue and non-fatigue groups. OH, orthostatic hypotension; POTS, postural orthostatic tachycardia syndrome.
Multiple logistic regression analysis
Multiple logistic regression analysis was performed with the following independent variables: age, disease duration, UPDRS-I, -II, -III, SCOPA-AUT, OH, BDI, PDQ-39, and PDSS. Regression model analysis showed that SCOPA-AUT and OH were the most related factors of fatigue in drug-naïve PD patients. However, UPDRS-I, -II, -III, BDI, PDQ-39, and PDSS were not significant related factors of fatigue in drug-naïve PD patients (Table 3).
Multiple logistic regression analysis results
CI, confidence interval; UPDRS, Unified Parkinson’s Disease Rating Scale; SCOPA-AUT, Scale for Outcomes in Parkinson’s Disease-Autonomic; OH, orthostatic hypotension; PDQ-39 SI, Parkinson’s Disease Questionnaire-39 Summary Index; PDSS, Parkinson’s Disease Sleep Scale; BDI, Beck Depression Inventory.
DISCUSSION
This is the first study in which the relationship between fatigue and autonomic dysfunction in drug-naïve PD patients was investigated. The study results showed that fatigue is common in drug-naïve PD patients and associated with autonomic dysfunction, especially cardiovascular dysfunction. The association between fatigue and autonomic dysfunction was proven using a subjective scale (SCOPA-AUT) and objective AFT.
Among drug-naïve PD patients, 23.6% had fatigue in the present study and 38.1% of the fatigued patients had OH. Conversely, only 14.7% of non-fatigued patients had OH. In addition, the ratio of symptomatic OH patients was significantly higher in the fatigue group than in the non-fatigue group (fatigue group vs. non-fatigue group, 75% vs. 20%), indicating OH, particularly symptomatic OH, is associated with fatigue in drug-naïve PD patients. Previous studies reported that not only classic OH, but also delayed OH or POST are associated with chronic fatigue [32, 33]. We also found not only classic OH, but also 2 patients with delayed OH and 1 patient with POST in drug naïve PD. Among them, only symptomatic delayed OH were associated with fatigue, but the relationship between delayed OH and fatigue in PD should be interpreted cautiously because of the number of patients was small.
Fatigued patients also had other autonomic dysfunctions based on HRDB, Valsalva maneuver, and SSR tests. The fatigued patients also showed higher SCOPA-AUT score in each regional domain, except pupillomotor dysfunction, which is relatively uncommon in PD [34]. The results from the present study showed autonomic dysfunction and fatigue were strongly correlated, which is in agreement with previous study results from a treated PD population. Nakamura and colleagues reported that fatigued PD patients had cardiac sympathetic denervation and greater pressor responses in the norepinephrine and dobutamine infusion tests [13]. A relationship between fatigue and autonomic dysfunction using SCOPA-AUT or Mayo Clinic Composite Autonomic Symptom Score (COMPASS) was observed in PD patients who were under treatment and not drug naïve [14, 15]. The pathophysiology of the relationship between fatigue and autonomic dysfunction is not fully elucidated. The cardiac sympathetic denervation might manifest clinically as shortness of breath during exercise and a tendency to fatigue [35], or because fatigue and autonomic dysfunction share the same pathologic mechanism, such as deposition of Lewy bodies on the lower raphe nuclei and magnocellular portions of the reticular formation [36]. Further investigations are needed to explain the pathophysiology of the relationship between fatigue and autonomic dysfunction in PD.
Fatigued patients with PD had slightly higher disease duration, LEDD, UPDRS-III score, and H&Y stage [10]. Autonomic dysfunction in PD was associated with older age, LEDD, UPDRS-I, -II and -III scores, and inversely correlated with cognitive function [37–40]. Therefore, drug-naïve PD patients in the early stage of the disease are optimal candidates to help understand the relationship between fatigue and autonomic dysfunction in PD. A few studies have been reported regarding fatigue in drug-naïve PD patients [2–7]; two studies were part of a randomized clinical trial, therefore, participants included in the studies might have been highly motivated [2, 7]. The relationship between fatigue and other clinical characteristics was investigated in drug-naïve PD patients and the results showed that fatigue was associated with higher UPDRS-I, -II, and -III scores, longer disease duration, depression, sleep problems, apathy, and visuospatial ability; however, no definitive conclusions were reached. Furthermore, whether the overlap of fatigue with depression and apathy reflects a diagnostic bias or common pathophysiological mechanisms remains unclear [41]. In the present study, fatigue was also associated with higher UPDRS-I and -II scores, depression, poor QoL, and sleep problems, but was not associated with UPDRS-III score and disease duration. In addition, multiple logistic regression analysis showed that fatigue in drug-naïve PD was strongly correlated with SCOPA-AUT and OH; conversely, UPDRS-I, -II, -III, PDQ-39, PDSS, and BDI did not show any correlation. A previous study proposed a concept of primary (the absence of depression and daytime somnolence) and secondary (the presence of depression or excessive somnolence) fatigue [42]. In accordance with the concept, we performed a subanalysis the patients with primary fatigue. Eleven of 21 in fatigue group and 56 of 68 in non-fatigue group were included in the subanalysis and results were not different from the main result of the study (data not presented). These results support that fatigue in PD is not a secondary phenomenon, but an intrinsic symptom of PD [11]. The results from the present study also indicated autonomic dysfunction might contribute to fatigue in drug-naïve PD patients. Therefore, the high prevalence of fatigue in the early stage of PD is partially explained by the high prevalence of autonomic dysfunction in drug-naïve PD patients. Studies regarding fatigue in drug-naïve PD patients are lacking and only a few hypotheses have been suggested, including inflammatory cytokines, abnormalities in the hypothalamic-pituitary-adrenal axis, abnormal basal ganglia output to the cortex, and an imbalance between different neurotransmitters. In this respect, the present study provides additional information for understanding fatigue in PD patients. Further investigations are needed to understand fatigue in PD, particularly in the early stage of disease.
The present study had several limitations. First, fatigue is multifactorial and has emotional, cognitive, and physical aspects. The PFS, which measures physical aspects of fatigue, was used in the present study. However, PFS is a very specific scale for PD and allows to discriminate fatigued from non-fatigued patients and minimizes the contribution of non-fatigue-specific parkinsonian symptomatology [19]. Second, some of the patients possibly had other pathologies than PD, such as multiple system atrophy (MSA). Severe autonomic dysfunction in the early stage of disease is a hallmark of MSA, especially OH and urinary incontinence, and patients with MSA usually show combined motor and autonomic dysfunction within 2 years from initial symptoms [43]. However, autonomic dysfunction, including OH, is also common in drug-naïve PD patients, even at the prodromal phase [44–47]. In the present study, 20.2% of enrolled patients had OH, which is similar to a previous report [48]. To overcome this limitation and increase diagnostic accuracy, only patients who had a sustained, excellent response to L-dopa replacement therapy for >2 years were included and patients who had any other clinical, radiological, or historical signs indicative of atypical parkinsonism, except autonomic dysfunction, were excluded.
Conclusion
The present study results showed fatigue and autonomic dysfunction are common non-motor symptoms, and in particular symptomatic OH, were strongly correlated in drug-naïve PD patients. The drug-naïve PD patients with fatigue also had higher UPDRS-I and -II scores, depression, poor QoL, and sleep problems. Although pathophysiology of fatigue is largely unknown, the autonomic dysfunction in drug-naïve PD patients in the early stage of disease was investigated using objective tests and subjective scales and the results indicated that fatigue is associated with autonomic dysfunction in PD patients.
CONFLICT OF INTEREST
The authors report no competing interests.
