Abstract
Background:
Pain is highly prevalent in patients with Parkinson’s disease (PD), but underlying pathophysiological mechanisms are largely unclear. Alterations in somatosensory processing might contribute to sensory abnormalities in PD.
Objective:
This study investigated sensory processing in PD patients.
Methods:
We used the standardized “Quantitative Sensory Testing” (QST) protocol (German Research Network on Neuropathic Pain) to investigate 13 somatosensory parameters in 19 PD patients naïve to dopaminergic medication and 19 healthy controls matched for age, gender, and handedness. We tested for differences in sensory parameters between i) drug-naïve PD patients and healthy controls, ii) patients’ more and less affected body side, and iii) for an association of somatosensory parameters with disease-specific factors.
Results:
We did not observe any significant group differences in somatosensory parameters between PD patients and healthy subjects. In PD patients, QST mean z-scores did not differ between the predominantly and the less affected body side, PD patients with and without PD-specific chronic pain or between different PD subtypes. Age, but not PD disease severity, was associated with a greater loss of function in thermal and mechanical detection thresholds.
Conclusions:
Somatosensory processing, as assessed with the well-established QST protocol, was normal in drug-naïve PD patients. Thus, somatosensory abnormalities previously reported in medicated PD patients might rather be a result of dopaminergic medication, or may occur later in the course of the disease or with increasing age.
INTRODUCTION
Pain and somatosensory abnormalities are highly prevalent in patients with Parkinson’s disease (PD) [1–3]. Pain can precede motor symptoms [4, 5] and has been reported to affect up to about 90% of PD patients [6] and to significantly impair patients’ quality of life [7, 8]. In addition to pain, PD can affect other somatosensory domains, such as thermal and tactile perception or proprioception with the majority of findings pointing to perception deficits as indicated by increased somatosensory thresholds [1, 2].
The neurophysiological mechanisms underlying pain and somatosensory changes in PD are only poorly understood. So far, the existing literature mainly focussed on medicated PD patients and yielded mixed results regarding changes in somatosensory processing and possible causes of the high prevalence of pain in PD.
Overall, pain perception seems to be aberrant in PD. Medicated PD patients showed increased thermal pain intensity ratings [9] and reduced thermal pain thresholds as compared to healthy subjects [10–12]. Furthermore, pain thresholds assessed using mechanical [13], electrical [11, 14], and laser pinprick stimuli [12] were reduced in medicated PD patients. Pain thresholds were also found to be lower in the unmedicated off-state compared to the medicated on-state [15].
Studies on thermal detection thresholds have yielded inconsistent findings. Some studies showed increased thermal detection thresholds [13, 17], while others have reported normal [10] or lower (i.e., increased sensitivity) thermal thresholds in treated PD patients [9].
Tactile thresholds have been found to be increased in PD (e.g., [13], for review see [2]), yet there are also studies reporting normal tactile thresholds. PD patients have also been reported to show alterations in proprioception including decreased proprioception accuracy and increased proprioceptive thresholds (for review, see [2]).
However, results might be confounded because all studies examined somatosensory thresholds in currently medicated PD patients, either under ongoing medication (on-state) or after a brief washout phase of dopaminergic medication (off-state). Considering the rather long half-life time of dopamine agonists [18] and the fact that central activity of levodopa can last up to 8 days despite its short half-life time [19], it cannot be ruled out that a residual influence of dopaminergic medication on somatosensory processing is maintained. This could lead to changes or normalization of somatosensory thresholds in PD and altered pain perception due to dopamine-related anti-nociceptive effects (for review, see [1]).
To rule out potential confounds related to dopaminergic medication and to provide a comprehensive, standardized method to test somatosensory thresholds, we examined drug-naïve PD patients and healthy subjects using the well-established Quantitative Sensory Testing (QST) protocol published by the German Research Network on Neuropathic Pain (DFNS) [20–22]. We investigated whether (a) somatosensory detection thresholds and pain thresholds differed between drug-naïve PD patients and healthy controls, (b) QST parameters differed between the predominantly and the less affected body side in drug-naïve PD patients, and whether (c) clinical parameters, such as PD severity, correlated with somatosensory thresholds.
MATERIALS AND METHODS
Participants
The study was conducted in the Department of Neurology at the University Medical Center Hamburg. PD patients were recruited from our movement disorders outpatient clinic between February 2011 and February 2013. Patients had to fulfill the following inclusion criteria: (1) diagnosis of idiopathic PD according to the criteria of the UK PD Society Brain Bank [23], (2) Hoehn & Yahr scale <stage III [24], (3) naïve to dopaminergic medication (= patients had never taken any dopaminergic medication), (4) no severe cognitive impairment (Parkinson Neuropsychometric Dementia Assessment (PANDA) [25] score ≥ 15), (5) age 40–90 years, (6) no major depression as indicated by Hospital Anxiety and Depression scale (HADS) [26] or clinical interview, (7) no acute pain or analgesic medication during the last 24 hours, (8) no history of chronic pain disorders (PD-specific chronic pain [27] was allowed), (9) no regular use of prescription analgesics, tranquilizers, antidepressants, pain modulating anticonvulsants (e.g. gabapentin or pregabaline), (10) no pregnancy, (11) being right-handed. Criteria (3), (5), (6), (7), (8), (9), (10), and (11) were also applied to the healthy controls.
35 PD patients were screened for study participation. 16 of these patients were not enrolled in the study as they did not fulfill the inclusion criteria (6 had already started dopaminergic medication at assessment day, 8 were on medication (criterion 9) or suffered from comorbidities that did not fit inclusion criteria, 1 with incomplete questionnaires, and 1 patient decided to withdraw from participation.
To test for alterations of QST parameters in the PD group, a control group of healthy participants matched for age, gender, and handedness was recruited locally by advertisement. Only right-handed subjects were included in the study.
The study was conducted in accordance with the Declaration of Helsinki and approved by the local Ethics Committee of the Medical Council in Hamburg (PV3644). All participants gave written informed consent and were free to withdraw from the study at any time.
Neuropsychiatric assessment
All participants first underwent a clinical interview to ensure that all inclusion criteria were met. In the drug-naïve PD group, a clinician experienced in movement disorders re-assessed the PD diagnosis and determined the patients’ Hoehn and Yahr stage [24] and Unified Parkinson’s Disease Rating Scale (UPDRS [28]) score (total and motor score (part III)).
PD patients completed the PANDA [25] and HADS [26] as depression has been shown to modulate pain perception [29]. PD subtypes were clinically classified according to the German AWMF Guidelines (www.awmf.org) as tremor-dominant, akinetic-rigid, or mixed depending on the motor symptom that was predominant at symptom onset and over the course of disease.
Quantitative Sensory Testing
QST was performed using an established protocol and the standard equipment according to the DFNS [20, 21]. All QST experimenters were trained to perform QST by the DFNS and our lab has been officially certified by Certkom e.V. (http://www.certkom.com/).
All participants underwent testing of their left and right dorsal hand. In the patient group, the test side was defined as the more affected body side. The less affected body side served as control side. All 13 standard QST parameters were determined: cold (CDT) and warm detection thresholds (WDT), thermal sensory limen (THS; perception of changing temperatures from warm to cold and vice versa), paradoxical heat sensations (PHS; perception of cold as heat), cold (CPT) and heat pain thresholds (HPT), mechanical detection (MDT) and mechanical pain thresholds (MPT), mechanical pain sensitivity (MPS; sensitivity to pinprick stimuli), pressure pain threshold (PPT), vibration detection thresholds (VDT), dynamic mechanical allodynia (DMA; experience of pain during innocuous dynamic tactile stimulation), and the wind-up ratio (WUR; temporal pain summation). The room temperature was kept between 20°C and 25°C. Please see supplementary material for details.
Statistical analysis
Statistical analyses were performed using the Equista software provided by the DFNS (http://www.neuro.med.tu-muenchen.de/dfns/arzt/qstform.html) and the IBM SPSS software version 20.0 (http://www-01.ibm.com/software/analytics/spss/). Data were tested for normal distribution using the Kolmogorov-Smirnov-Test and for homogeneity of variance using Levene’s test. As described in previous studies [30], test scores of both groups were log-transformed using Equista to establish normal distribution and mapped onto the distribution of the DFNS reference group [21, 22] using z-transformation (z-score Participant = (QST Participant – QST Reference) / standard deviation Reference). This method assured comparability of QST results as the z-scores included adjustments to sex, age, and the tested body site of the published reference group [21, 22]. Initially, z-scores for both test sites were calculated separately and compared in both groups. A mean z-score >0 indicates higher sensitivity (gain of function), while a mean z-score < 0 signals lower sensitivity (loss of function) to the external stimulus applied. Z-scores exceeding 95% of the confidence interval of the reference group (±1.96 standard deviation (SD)) are considered pathologic. All scores were normally distributed and showed homogeneity of variances. For PHS and DMA only, raw data was compared between groups (according to the protocol). In both groups, results for both tested sites were contrasted using paired t-test.
For comparisons of PD patients and healthy controls, mean z-scores (z mean = mean (z test side + z control side)) were contrasted using two sample t-test. The comparison of WUR scores was based on 17 PD patients and their matched controls, as 2 PD patients did not perceive the pinprick stimuli as painful. Furthermore, group mean z-scores and – for exploratory purposes – individual z-scores were compared to the published reference data [21, 22]. Finally, individual mean QST z-scores were correlated with age, clinical scores, and questionnaire scores mentioned above using Pearson correlation coefficient. All results were corrected for multiple comparisons applying Bonferroni correction (pBonf = 0.05/13 = 0.0038 ≈ 0.004).
Further, within the PD group, z-scores were contrasted between patients with and without PD-related chronic pain (classified according to Ford as musculoskeletal pain, radicular, or neuropathic pain, dystonia-related pain, akathitic discomfort, or central pain [31]) using paired t-test and between different PD subtypes using a one-factorial ANOVA and Mann-Whitney-U test.
RESULTS
19 drug-naïve PD patients (mean age 64.8 ± standard deviation (SD) 10.0 years, range 42-82 years) and 19 healthy controls (mean age 64.8 ± SD 9.9 years, range 42–80 years) matched for age (t (36) =0.000, p=1.000), gender (each group: 12 male, 7 female, χ2 = 0.000), and handedness (all participants were right-handed) were included in the study. Characteristics and results of neuropsychiatric assessment of PD patients are given in Table 1.
Clinical characteristics and scores of drug-naïve patients with Parkinson’s disease (PD).
Clinical characteristics and scores of drug-naïve PD patients are given as mean values with standard deviation and minimum-maximum in brackets.
In the control group, there were no differences between QST z-scores of the left and right body side (all p-values >0.05). QST z-scores of the PD group revealed a significant difference in MDT (t (18) =-2.570, p=0.019) with a greater loss of function on the predominantly affected compared to the less affected body side (see Fig. 1). Furthermore, patients showed a side difference in WUR (t (18) =2.150, p=0.047) with greater WUR z-scores on the predominantly compared to the less affected body side. However, both analyses (MDT and WUR) did not survive Bonferroni correction for multiple comparisons (pBonferroni= 0.05/11 = 0.004) and mean z-scores lay within the 95% confidence interval of the reference group [21, 22]. DMA was found in two healthy subjects. PHS occurred in six patients and five healthy controls. There was no body side difference in DMA and PHS for either of the groups (p>0.05).
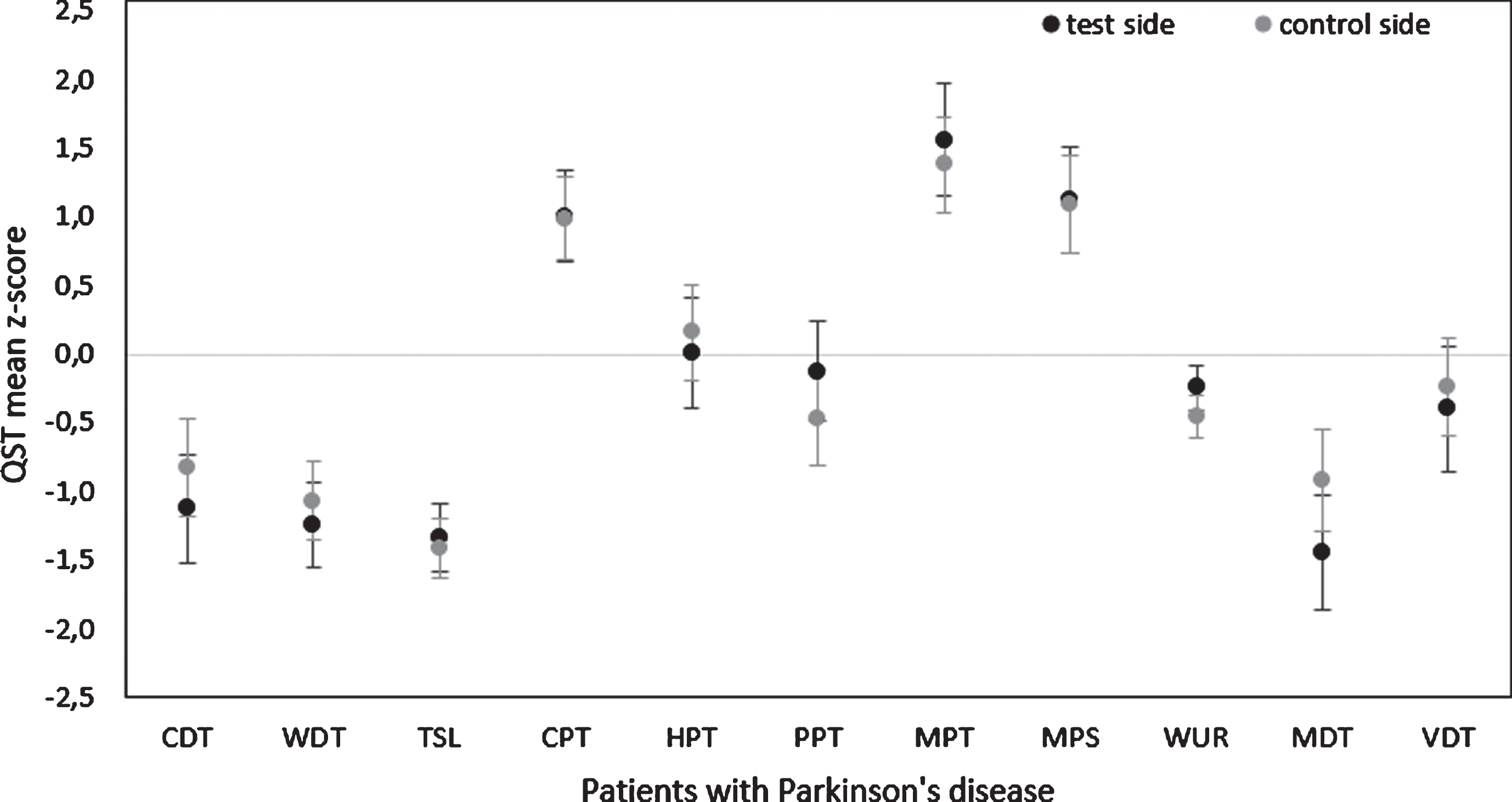
Body side differences of Quantitative Sensory Testing (QST) parameters in patients with Parkinson’s disease. Results are given as mean z-scores of patients with Parkinson’s disease for the predominantly affected test side (black) and the less affected control side (light grey) including standard error of means (SEM). Z-scores exceeding 95% of the confidence interval of the DFNS reference group are considered as pathologic (± 1.96 standard deviation). CDT, cold detection threshold; WDT, warm detection threshold; TSL, thermal sensory limen; CPT, cold pain threshold; HPT, heat pain threshold; PPT, pressure pain threshold; MPT, mechanical pain threshold; MPS, mechanical pain sensitivity; WUR, wind-up ratio (data of 2 patients are missing); MDT, mechanical detection threshold; VDT, vibration detection threshold.
As there were no body side differences for no QST parameter in either of the two groups, further group analyses were conducted using the mean bilateral z-scores of both groups. Group comparisons of single QST parameters’ mean z-scores revealed a significant difference in WUR (t (32) =2.422, p=0.021) with lower WUR z-scores in PD patients compared to controls (see Fig. 2 and Supplementary Table 1). However, these did not survive Bonferroni correction and mean z-scores were within the reference range (see Fig. 2, for individual scores see Fig. 3a and 3b). PD patients did not significantly differ from healthy controls in the remaining QST parameters, the number of PHS (p>0.05) and DMA raw values (p>0.05; see Supplementary Table 1), which were within the DFNS reference range (see Supplementary Table 1 in [22]). Taken together, no specific pathologic pattern pointing towards a certain neurobiological mechanism, such as peripheral nerve fiber dysfunction or peripheral/central sensitization [32], has been observed neither for the PD nor the control group.
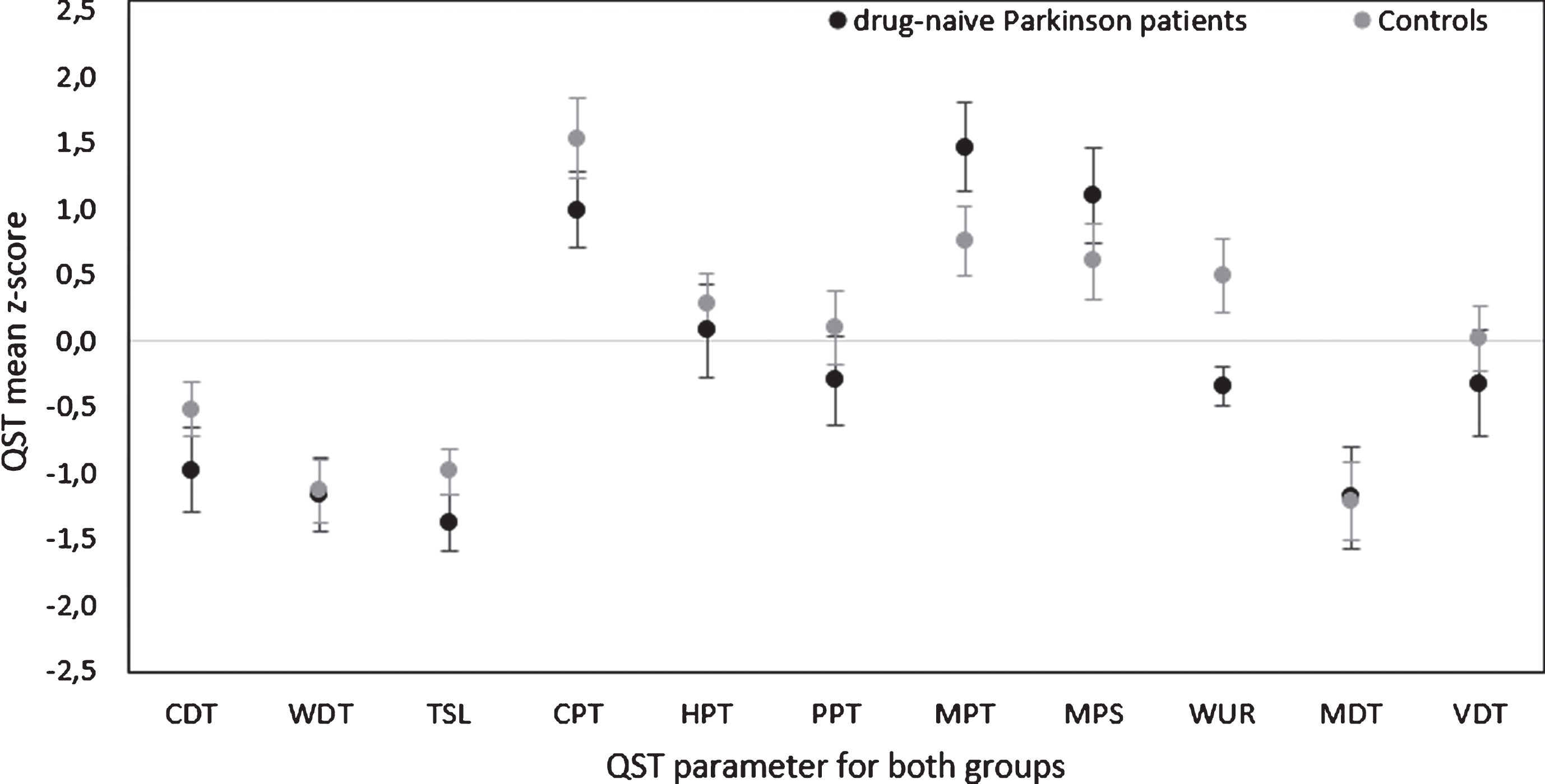
Group comparison of bilateral Quantitative Sensory Testing (QST) parameters between drug-naïve patients with Parkinson’s disease and healthy subjects. Results of Quantitative Sensory Testing are given as bilateral mean z-scores of patients with Parkinson’s disease (black) and healthy controls (light grey) including standard error of means (SEM). Z-scores exceeding 95% of the confidence interval of the DFNS reference group are considered as pathologic (±1.96 standard deviation). Abbreviations: see Figure 1. For wind-up ratio (WUR) data of 2 patients are missing.
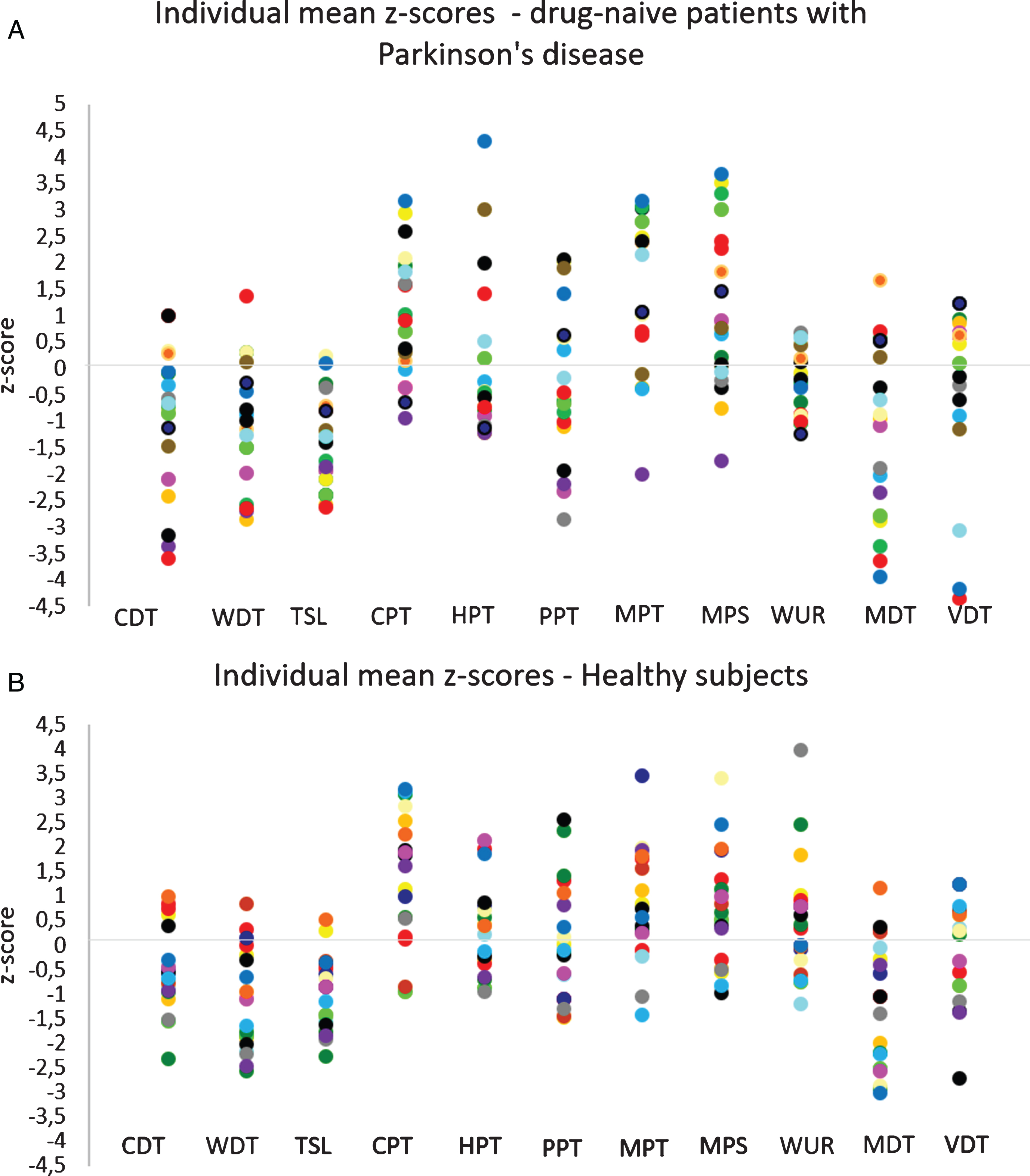
Individual results of Quantitative Sensory Testing. Given as mean z-scores of patients with Parkinson’s disease (A) and healthy subjects (B). Z-scores exceeding 95% of the confidence interval of the DFNS reference group are considered as pathologic (±1.96 standard deviation). Abbreviations: see Figure 1.
QST parameters did not differ between those drug-naïve PD patients with PD-related chronic pain (n=8) and those without chronic pain (n=11; all p-values >0.05). Furthermore, an exploratory one-factorial ANOVA revealed no significant differences between z-scores of the three different motor subtypes. Mann-Whitney-U test also revealed no difference between tremor-dominant PD patients (n=11) and a pooled group of akinetic and mixed type PD patients (n=8, all p-values >0.05).
The PD group showed a negative correlation with age for WDT (r= -0.637, p=0.003) and MDT (r= -0.677, p= 0.001). This indicates a decrease in thermal and mechanical detection thresholds with progressing age in PD patients. In healthy subjects, no correlation between individual z-scores and clinical or cognitive scores reached statistical significance after correction for multiple testing. Nevertheless, we observed a negative correlation of the mean z-scores for CDT (r= -0.623, p=0.004) and MDT (r= -0.608, p=0.006) with age. However, these results did not survive the threshold for multiple testing and thus must be interpreted with caution but. Overall, results indicate a thermal and mechanical sensitivity decrease with increasing age in both groups. QST parameters in the PD group did not correlate with the UPDRS total and motor score or the PANDA and HADS-D score (all p>0.05).
DISCUSSION
This study investigated somatosensory processing in drug-naïve PD patients using the well-established, comprehensive QST protocol (DFNS). Our results indicate that somatosensory processing is not altered in this group of drug-naïve PD patients compared to age- and gender-matched healthy controls. Furthermore, somatosensory processing did not differ between the predominantly affected and the control side in the patient group. There were no differences between PD subtypes. For PD patients, age correlated negatively with warm and mechanical detection thresholds, i.e. thresholds were lower with increasing age.
In contrast to previous findings on sensory detection and pain thresholds in medicated PD patients, our non-medicated patients neither showed reduced thermal and mechanical pain thresholds (e.g., [10-12]) nor increased tactile detection thresholds (e.g., [2, 13]) compared to healthy controls. Likewise, the more affected body side did not differ from the less affected side in these measures [13]. These findings indicate that changes in somatosensory thresholds either occur (a) later in the course of the disease, (b) with increasing age or (c) as a result of dopaminergic medication. Interestingly, regarding (a), we did not find a significant correlation between QST parameters and disease severity, which suggests that disease progression alone is insufficient in explaining changes in somatosensory function. However, it should be noted that our sample was rather homogenous in disease severity. With view to age (b), an age-dependent decline of descending pain modulation mechanisms has been found in healthy participants [33] as well as in PD patients [34]. This might explain reduced pain thresholds [10-12] and higher pain sensitivity ratings [9] in elderly PD patients. Our findings of reduced thermal and mechanical thresholds with age in PD patients indicate a detection hypersensitivity for mechanical and warm stimuli that could also be observed in elderly healthy subjects. We can only speculate about the nature of this increasing hypersensitivity with increasing age, as the value of QST to distinguish central and peripheral mechanisms is limited [32]. However, it is tempting to assume that central mechanisms, such as central sensitization rather than peripheral mechanisms, contribute to this phenomenon. Besides the speculative role of central and peripheral mechanisms and with view to (c), it has not been clarified yet, to what extent somatosensory abnormalities found in medicated PD patients might be related to the intake of dopaminergic medication or not. Previous studies have reported abnormal somatosensory thresholds in medicated PD patients in the on or off-state (review [1, 2]). Studies exploring the influence of dopamine on pain perception thresholds and neural pain processing in PD indicate that dopaminergic pathways are involved in nociceptive processing [1, 35]. Some even presume concentration-dependent pro- or antinociceptive effects of dopamine. Regarding other somatosensory domains, literature on the influence of dopamine is sparse. Studies on dopaminergic effects on proprioception in PD yielded contradictory results. While there is evidence that dopaminergic medication has no effect on proprioception [36] or can even worsen proprioceptive abilities [37], others reported an enhancement of curvature sensitivity in the on-medication compared to the off-medication state [38]. Studies on tactile perception focused mainly on somatosensory temporal discrimination thresholds (review [2]) or vibration temporal judgement orders and reported positive dopaminergic treatment effects. In our unmedicated patient group, we did not identify any consistent specific pathological QST pattern. However, since the effect of dopaminergic medication seems to be unclear even after a washout phase, our drug-naïve patients cannot directly be compared to previously medicated patients. Therefore, further studies should investigate drug-naïve PD patients using QST before and after initiating dopaminergic medication. Moreover, the complete QST protocol should also be used in medicated PD patients to compare on- and off-state.
To our knowledge, there are only few studies investigating sensory aspects in drug-naïve PD patients. Besides one study showing normal electrophysiologically determined peripheral nerve function [39], other studies mainly focussed on single or few somatosensory thresholds, such as temporal discrimination thresholds, (which were normal in drug-naïve PD patients [40]), warm sense detection (increased) and heat pain thresholds (normal) [41], or a combination of tactile (increased), thermal (increased) and mechanical pain thresholds (impaired) [42]. As opposed to this, the advantage of our study is the simultaneous assessment of various somatosensory parameters using the previously validated, standardized QST method.
We did not find a correlation between disease severity (measured using the UPDRS motor score) or the symptom duration given in months with QST parameters. Interestingly, in another study examining a mixed patient group of drug-naïve and medicated PD patients, somatosensory temporal discrimination threshold abnormalities correlated with the duration and severity of PD [40]. Due to our highly selected patient group of drug-naïve PD patients, there might be a selection bias towards patients with a relatively short disease duration and less severe disease severity (as the disease severity progresses with disease duration). This rather low variation in disease severity and duration might have impaired the sensitivity to detect a correlation between these disease-related variables and QST parameters. Future research should consider longitudinal data collection in PD patients to clarify this question.
In the PD sample investigated here, QST parameters did not differ significantly between the predominantly affected and the less affected body side, which is consistent with previous studies [10, 16]. Furthermore, QST parameters of drug-naïve PD patients with and without chronic pain were comparable, which is in line with previous findings regarding innocuous [10] and nocuous [14] somatosensory stimuli.
Taken together, we did not find any differences in QST parameters between drug-naïve PD patients and healthy controls. However, we did find a negative relationship between MDT and WDT and age in PD patients indicating increasing sensitivity with age. Interestingly, previous studies also reported increasing thermal [43] and tactile [44] thresholds in elderly in general, but MDT and WDT increase might be more pronounced in PD compared to healthy subjects.
Our findings have to be seen in the light of two limitations. (1) The HADS scores were only gathered in PD patients but not in healthy controls. However, an interview conducted by an experienced neurologist with a strong background in (neuro-) psychiatry did not indicate major depression in any of the healthy controls. (2) Our study comprised a relatively small sample size of drug-naïve PD patients and matched healthy controls. It should be noted that this very homogenous sample of drug-naïve PD patients is notoriously difficult to recruit and unique, given that an early beginning of dopaminergic therapy is recommended in PD patients (e.g., in the German AWMF guidelines for PD: http://www.dgn.org/leitlinien/11-leitlinien-der-dgn/2346-ll-09-2012-parkinson-syndrome-diagnostik-und-therapie). Although none of our PD patients had ever taken any dopaminergic medication, our patient collective was relatively old and disease duration quite long (see Table 1).
Conclusion
This study investigated somatosensory processing in drug-naïve PD patients using the comprehensive, established QST test battery (DFNS). We did not observe significant differences in QST parameters of drug-naïve PD patients compared to healthy subjects. However, we did find a negative relationship between MDT (mechanical) and WDT (thermal) and age in PD patients (and by trend in controls), indicating heightened sensitivity with increasing age, which seems to be more pronounced in PD and might potentially be mediated by central sensitization processes associated with PD. Our findings suggest that somatosensory abnormalities previously reported in medicated patients might be a result of dopaminergic medication or may occur later in the course of the disease or with increasing age.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
