Abstract
Background and Objective:
To investigate the predictive value of striatal dopamine turnover in patients with de novo Parkinson’s disease (PD) for later occurrence of major non-motor health outcomes.
Methods:
This retrospective, observer-blinded cohort study followed up 29 patients with de novo PD for a median of 10.7 years, who completed 18Fluorodopa PET imaging to measure striatal effective distribution volume ratio (EDVR, inverse of dopamine turnover) prior to antiparkinsonian treatment. Outcomes were assessed with a battery of non-motor, health-related quality-of-life and non-motor fluctuation (WOQ-19) measures and survival.
Results:
During follow-up, 52% of patients developed wearing-off, 43% neuropsychiatric fluctuations, 35% sensory fluctuations, 32% dementia, 46% depression, 30% psychosis, and PD-related mortality was 26%. Patients with wearing-off and neuropsychiatric fluctuations showed significantly lower baseline EDVR (higher dopamine turnover) in the putamen but not in the caudate nucleus than those without these fluctuations. Consistently, baseline EDVR in the putamen predicted development of wearing-off and neuropsychiatric fluctuations with a lower risk with higher EDVR (lower dopamine turnover), whereas EDVR in caudate nucleus did not correlate with these fluctuations. No relationships were observed between baseline PET measures and the presence of other major health outcomes including survival.
Conclusions:
Lower putaminal dopamine turnover in de novo PD is associated with reduced risk for later neuropsychiatric fluctuations comprising a disease-intrinsic predisposing factor for their development, similar as reported for levodopa-induced motor complications. Striatal (putaminal/caudate) dopamine turnover is not predictive for other long-term major health outcomes. These results should be treated as hypothesis generating and require confirmation.
Keywords
INTRODUCTION
The progression of Parkinson’s disease (PD) is heterogeneous and includes not only motor and non-motor symptom progression, but also the development of levodopa-induced motor and non-motor complications such as wearing-off and levodopa-induced dyskinesia [1]. Individual prediction of disease progression would thus be helpful for therapeutic management in PD.
Dopamine transporter (DAT) imaging has demonstrated that the magnitude of striatal DAT binding in early PD is independently predictive for long-term motor and non-motor outcomes [2]. Patients with lower striatal DAT binding showed a higher risk for the development of motor-related disability as well as cognitive impairment, psychosis, and depression. Moreover, striatal DAT binding in early PD has been reported to be predictive for the development of levodopa-induced dyskinesia [3]. A recent retrospective cohort study did however not observe any correlation of striatal DAT binding with survival [4]. While these studies addressed DAT binding as a measure of structural integrity of dopaminergic nerve terminals, similar studies using 18Fluorodopa PET to estimate dopamine metabolism as a potential predictor for long-term outcomes are largely missing. A recent study in untreated early PD patients suggested that striatal 18Fluorodopa uptake does not predict mortality [5].
Striatal dopamine metabolism as measured by 18Fluorodopa uptake and dopamine effective distribution volume ratio (EDVR) as the inverse of dopamine turnover using PET is highly dynamic and underlie marked compensatory changes in early PD in response to the nigro-striatal dopaminergic neurodegenerative processes with relative upregulation of 18Fluorodopa uptake and dopamine turnover [6–8]. These disease-induced changes are largely determined by age and MAOB (rs1799836) polymorphism, but not gender [7, 9]. Our recent longitudinal PET study has indicated that putaminal dopamine turnover in de novo PD is associated with the development of late-stage motor complications [10]. However, the importance of these compensatory changes for disease progression and late-stage non-motor complications remains unknown. We thus analyzed here the predictive value of striatal EDVR for later occurrence of major non-motor health outcomes such as dementia, depression, non-motor fluctuations and survival in a PD cohort that was followed retrospectively after enrollment in a de novo PD clinical trial [8]. For long-term assessment of motor and non-motor fluctuations, we used the Wearing-Off Questionnaire with eight motor and eleven non-motor items (WOQ-19) [11–13] as a self-administered questionnaire with adequate clinimetric properties to detect wearing-off phenomena in PD, which is recommended by the Movement Disorders Society task force [14, 15].
SUBJECTS AND METHODS
Study participants
This retrospective, observer-blinded long-term cohort study followed up 35 patients with de novo PD, who had been initially enrolled in a randomized, parallel-group clinical study between 2005 and 2007 and had a complete and interpretable 18Fluorodopa PET imaging dataset and at least one follow-up visit after the randomized study phase [8] (see Supplementary Figure 1 for study flow-chart). The study had approval from the ethics committee at TU Dresden (EK91052003) and the German Federal Institute for Drugs and Medical Devices (BfArM), and was registered with ClinicalTrials.gov (NCT00153972). For the present follow-up study, we received renewed approval from the ethics committee. Written informed consent was obtained from all participants. Since the initial randomized trial, the majority of patients visited our outpatient clinic every 3-12 months, for remaining patients data were collected from medical records of treating neurologists or by telephone visits. We excluded three patients with evidence for other diagnoses than PD from PET imaging data or during the clinical follow-up (two patients were diagnosed with essential tremor [both had normal putaminal Kocc values], and one with Lewy-body dementia [LBD]).
Clinical assessments
Motor symptoms were estimated using Unified Parkinson’s Disease Rating Scale (UPDRS) part III motor score, activities of daily living (ADL) using UPDRS ADL part II score and the Schwab & England (S&E) ADL score [16], cognition using Montreal Cognitive Assessment (MoCA), impulse control disorder (ICD) using the Questionnaire for impulsive-compulsive disorders in PD rating scale (QUIP-RS) [17] and depression using Beck’s Depression Inventory (BDI) or Geriatric Depression Scale (GDS) [18]. Non-motor symptoms were investigated by Non-Motor Symptom Scale (NMSS) [19] and health-related quality of life (hr-QoL) by PD Quality of Life (PDQ-8) questionnaire [20]. We used the following defined clinically important cut-offs: Dementia (MoCA score≤21) [21], psychosis (UPDRS thought disorder item score≥2), depressive syndromes (BDI≥13 or GDS≥5) [18], and falling (UPDRS falling item score≥1). Motor fluctuations (wearing-off) and non-motor fluctuations were assessed using the WOQ-19 [11]. We classified NMS as proposed by Seki and colleagues [22] into the three domains neuropsychiatric (4 items in WOQ-19: anxiety, mood changes, panic attacks and cloudy mind/dullness of thinking), autonomic (3 items: sweating, abdominal discomfort and experiencing hot and cold), and sensory category (3 items: numbness, pain and aching). All clinical ratings were assessed by movement disorder trained physicians blinded to the PET data (M.L., J.M., M.W., W.H, D.H.). Medication regimes of all patients were reconstructed using medical records and prescription plans. Levodopa equivalent doses (LED) were calculated according to [23].
Acquisition and analyses of 18Fluorodopa PET data
PET measurements were carried out in drug-naïve participants using a 3D ECAT EXACT HR+ system (Siemens/CTI, Knoxville, USA) as described previously [8, 24]. Briefly, we applied a tissue reference model, in which the activity curve of the occipital cortex served as input function for calculation of the tissue input uptake rate constant Kocc and EDVR [8–10]. Data from the left and the right side were averaged for each scan. For a detailed description of data analyses including analyses of the tissue reference region serving as input function, refer to [9].
Statistical analyses
Group comparisons of clinical and PET data were made with unpaired t-test or Mann-Whitney U test (continuous variables), or with χ2 test or Fisher exact test (discrete variables), as appropriate. For analyses of the associations between baseline PET data and long-term outcomes, we initially selected caudate nucleus/putamen EDVR data as continuous variables. Then we dichotomized EDVR imaging data into halves based on their distributions and designated the subgroup with low EDVR (high dopamine turnover) as reference. Since we did not detect significant differences of all major clinical and demographic data between the groups, we first used χ2 test or Fisher exact test as appropriate to estimate Odd’s ratios (ORs) for dichotomous and unpaired two-sided t-test for continuous outcome measures. The results were then confirmed by applying multiple logistic regression analyses for dichotomous and multiple linear regression analyses for continuous variables. To compare PET data as continuous variables we performed ANCOVA analyses. In each of the models, we controlled for the candidate co-variates age at PET scan, gender, PD symptom duration at PET scan (baseline), LED at last assessment, time period of follow-up since baseline and baseline UPDRS part II/III sum score as a measure of disease severity. For survival analyses, we performed univariate and multivariate Cox proportional hazards models to estimate hazard ratios (HRs) with 95% confidence intervals (95% CIs) and p values for pairwise comparisons. The proportional hazards assumption was tested using log-log plots. Similar to previous studies [4, 25–27], gender, age and symptom duration at PET imaging, and baseline UPDRS part II/III sum score were entered into the multivariate Cox proportional hazards model to adjust for relevant covariates. Dichotomized PET imaging measures were then added to the selected models. Analyses were conducted using SPSS, version 23.0 (SPSS, Chicago, IL), and all p values were two-sided and values of less than 0.05 were deemed statistically significant (not adjusted for multiple comparisons).
RESULTS
Study participants and long-term clinical outcome
Twenty-nine of the 35 subjects (83%) in the primary efficacy cohort (cohort with complete PET dataset) had at least one follow-up visit after the randomized study and no evidence for other diagnoses than PD, and were included in the main analyses (see Supplementary Figure 1 for study flow-chart). Table 1 shows the characteristics of the cohort at baseline and at most recent visit at a mean±SD follow-up time of 10.7±1.9 (IQR: 11.0–12.0) years after baseline. At baseline visit, subjects had a mean±SD age of 60.8±9.6 years, UPDRSIII motor scores of 19.5±7.5, and median [IQR] S&E ADL score of 90% [90–90%] and were not taking any antiparkinson medication. Sixty-nine percent were male. Table 1 shows mean (of left and right) striatal 18Fluorodopa uptake and EDVR.
Cohort characteristics at baseline (time of PET scan) and last visit
Data are mean (SD), median (interquartile range, IQR), or number (%). EDVR, Effective distribution volume ratio; PET, positron emission tomography; UPDRS, Unified Parkinson’s disease rating scale; ADL, Activities of daily living; Kocc,18Fluorodopa uptake. &Disease duration was defined as the time since PD had first been diagnosed. #Levodopa equivalent dose (LED) was calculated according to Tomlinson and co-workers [23]. ¶significantly different from controls with p < 0.05 (normal ranges defined as mean values ± 2 SDs from our previous study [24]).
Comparisons of clinical parameters at baseline with those at most recent visit show the expected clinical progression of PD (Table 1). The relatively small change in UPDRS scores is the result of the fact that, at baseline, subjects were not taking dopaminergic therapy, but were on optimized dopaminergic therapy at follow-up visits. Survival analysis was performed in all patients with sufficient clinical and PET data and thus included three additional patients who died prior to first clinical follow-up (cohort characteristics in Supplementary Table 1).
We used WOQ-19 assessment as outcome measure to determine late stage motor and non-motor complications. WOQ-19 scoring revealed that all patients showed at least one kind of motor symptoms, and 19 out of 21 patients (90%) showed at least one kind of NMS (Supplementary Table 2). Neuropsychiatric symptoms were the most frequent NMS (75% of patients) followed by sensory (70%) and autonomic symptoms (30%). The number of patients who presented with motor fluctuations was 11 (52%) and with non-motor fluctuations 12 (57%). We did not observe any differences in major baseline measures (age at PET scan, gender, PD symptom duration at PET scan, UPDRS part II/III sum score), LED at last assessment and time period of follow-up since baseline between the groups of patients with motor fluctuations or non-motor symptoms/fluctuations and those without these symptoms/fluctuations (p > 0.05, unpaired two-sided t-test). Consistently, multiple logistic regressions using a stepwise selection process did not reveal relevant clinical and demographic predictors for long-term WOQ-19 motor and non-motor outcomes.
Striatal dopamine turnover measured by 18Fluorodopa PET imaging at baseline
Similar to our previous reports [8, 10], Pearson correlation test revealed that EDVR in the putamen (inverse of DA turnover) was negatively correlated with baseline UPDRSIII motor scores and UPDRSII/III sum scores as a measure of disease severity (r = –0.334, p = 0.046 and –0.344, p = 0.048). Caudate nucleus EDVR did not correlate with UPDRS baseline measures (UPDRSIII: r = –0.001, p = 0.997 and UPDRSII/III sum score: r = 0.026, p = 0.892). Pearson correlation tests and subsequent multiple linear regressions using a stepwise selection process showed no further associations of striatal EDVR measures with baseline clinical and demographic factors (gender, age, symptom duration) were observed.
Predictive value of baseline striatal EDVR (inverse of dopamine turnover) for later motor fluctuations
Comparison of continuous baseline EDVR measures between individuals with and without motor fluctuations as measured by WOQ-19 using unpaired two-sided t-test or ANCOVA to adjust for candidate covariates revealed higher putaminal EDVR (lower dopamine turnover) in patients without motor wearing-off, whereas EDVR in the caudate nucleus did not differ between groups (Fig. 1, Table 2). Adjustment only for baseline UPDRSII/III sum scores as the single covariate associated with putaminal EDVR revealed similar results with significant differences of putaminal EDVR between individuals with and without motor wearing-off (F-value: 4.870; p = 0.041). For group-wise estimation of the risk for long-term motor fluctuations, we dichotomized patients into halves based on the distribution of striatal EDVR and adopted those in the lower half as reference group. Since we did not observe any influence of clinical/demographic factors for WOQ-19 long-term outcome prediction, we initially performed χ2 or Fisher exact tests to estimate unadjusted ORs for the associations of striatal EDVR and WOQ-19 (Fig. 2, Table 3): Similar to the analysis of continuous EDVR measures, we detected that higher putaminal EDVR (lower dopamine turnover) at baseline was significantly associated with lower risk for motor wearing-off (Table 3). Adjustment for candidate covariates or only for baseline UPDRSII/III sum scores by using multiple regression models again revealed a similar association of putaminal EDVR with the occurrence of motor wearing-off (Table 3). For example, patients in the upper half of putaminal EDVR had an OR of 0.01 (95% CI: 0.00–0.87; p = 0.047) for developing wearing-off as compared to the lower half (reference value) when adjusted to major candidate covariates and an OR of 0.01 (95% CI: 0.00–0.84; p = 0.042) when adjusted only for baseline UPDRSII/III sum scores.
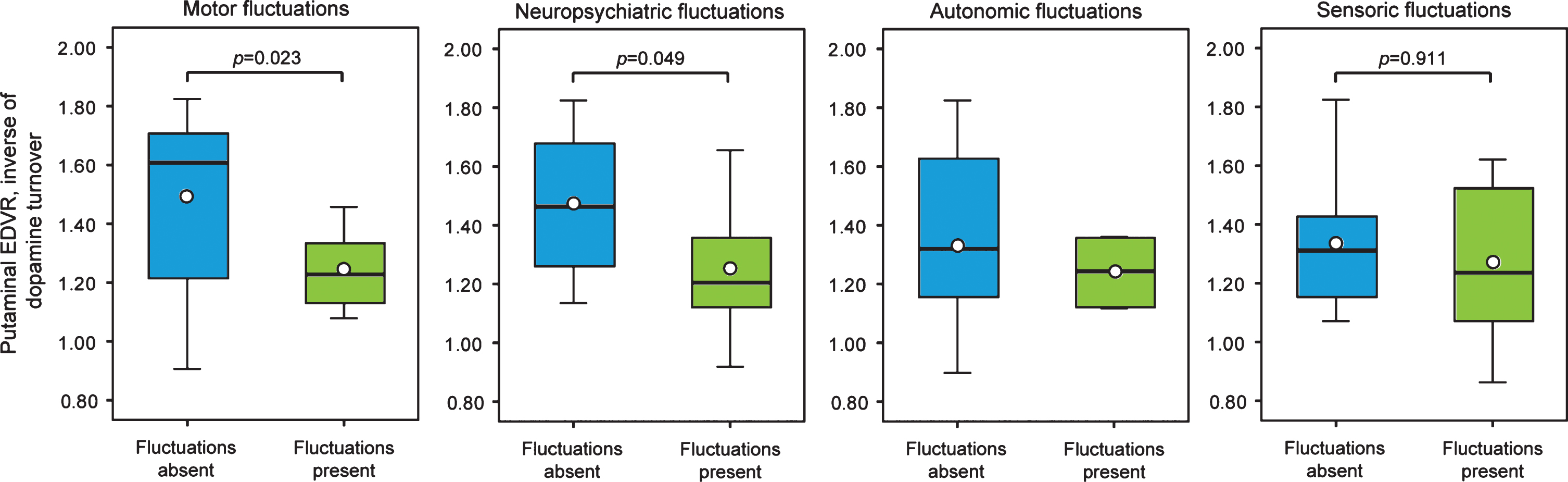
Putaminal dopamine turnover as measured by 18Fluorodpa PET imaging with respect to long-term motor and non-motor outcome. The boxplots show putaminal effective distribution volume ratio (EDVR) as the inverse of dopamine turnover with respect to occurrence of motor and non-motor fluctuations. In the plots, boxes represent the interquartile range, horizontal lines the medians, dots the mean values and the antennas the range excluding outside values (defined as values beyond lower/upper quartile±1.5 times interquartile range). Non-motor symptoms were categorized into three categories (neuropsychiatric, autonomic, and sensory symptoms). Displayed are unadjusted p-values from unpaired two-sided t-tests (for adjusted values, refer to Table 2; note that statistics for autonomic fluctuations were not calculated due to only two patients reporting autonomic fluctuations).
Comparison of patients with and without late-stage complications with regard to baseline striatal EDVR (inverse of dopamine turnover)
Data are mean values±standard deviations. EDVR, Effective distribution volume ratio from 18Fluorodopa positron emission tomography; WOQ-19, Wearing-Off Questionnaire with 19 items. aResults from unpaired two-sided t-tests. bResults from separate ANCOVA analyses adjusted the candidate covariates age at PET scanning, gender, symptoms duration at PET scanning, levodopa-equivalent dose (LED) at last assessment, time period of follow-up since PET and baseline UPDRS part II/III sum score as a measure of disease severity. Note that patient group with autonomic fluctuations (n = 2) were rather small (thus no statistics were calculated for autonomic fluctuations).
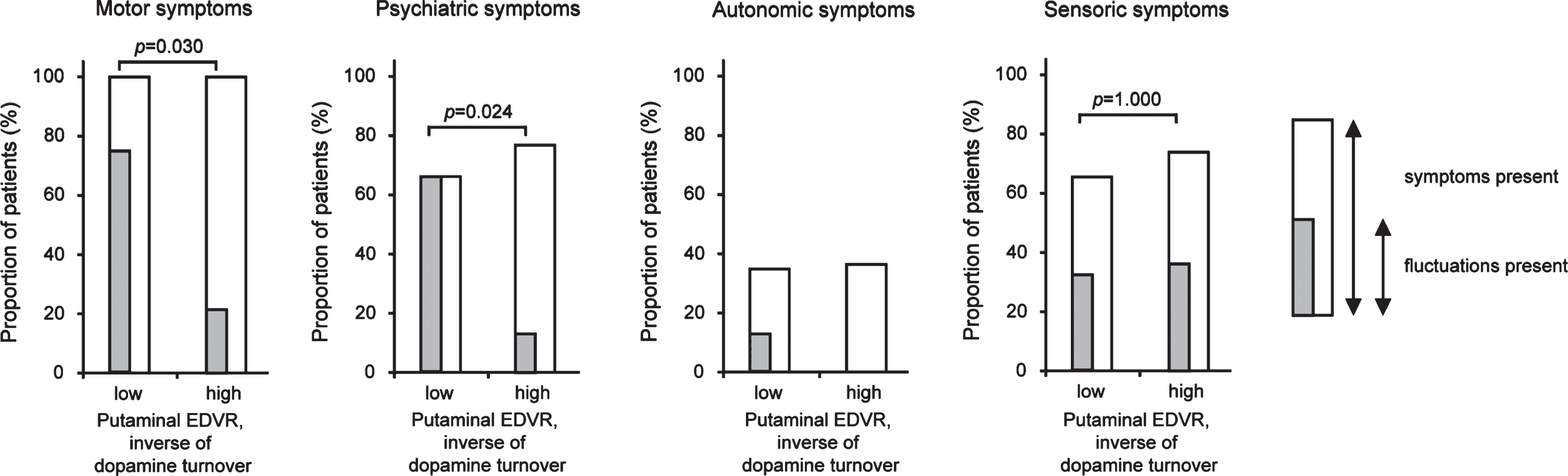
Long-term motor and non-motor outcome with respect to putaminal dopamine turnover as measured by 18Fluorodpa PET imaging. Percentages of patients presenting with motor and non-motor symptoms and/or their fluctuations as assessed by the WOQ-19 questionnaire with respect to putaminal effective distribution volume ratio (EDVR) as the inverse of dopamine turnover. Non-motor symptoms were categorized into three categories (neuropsychiatric, autonomic, and sensory symptoms). Displayed are unadjusted p-values for symptom fluctuations from χ2 or Fisher exact tests as appropriate (for adjusted values, refer to Table 2; note that statistics for autonomic fluctuations were not calculated due to only two patients reporting autonomic fluctuations).
ORs for motor and non-motor fluctuations by dichotomized baseline striatal EDVR (inverse of dopamine turnover)
EDVR, Effective distribution volume ratio from 18Fluorodopa positron emission tomography; OR, Odds ratio; 95% CI, 95% confidence interval; WOQ-19, Wearing-off questionnaire with 19 items. aAn OR < 1 indicates smaller risk for the respective outcome in the groups with high EDVR (low dopamine turnover). bAdjustment of OR for relevant covariates were performed by separate logistic regressions entering age at PET scanning, gender, symptoms duration at PET scanning, levodopa-equivalent dose (LED) at last assessment, time period of follow-up since PET scanning and baseline UPDRS part II/III sum score as a measure of disease severity. An OR < 1 indicates smaller risk for the respective outcome in the groups with high EDVR (low dopamine turnover). cAll patients report motor symptoms in WOQ-19 questionnaire and thus statistics are not calculated. dData for autonomic fluctuations were not calculable due to too few patients reporting autonomic fluctuations in WOQ-19 questionnaire (2 out of 20 patients [10%]).
Predictive value of baseline striatal EDVR (inverse of dopamine turnover) for later non-motor complications
Comparison of continuous baseline EDVR measures between individuals with and without subsequent non-motor symptoms/fluctuations (WOQ-19) using unpaired two-sided t-test or ANCOVA to adjust for candidate covariates revealed lower putaminal EDVR (higher DA turnover) in patients with neuropsychiatric fluctuations (Fig. 1, Table 2). Adjustment only for baseline UPDRSII/III sum scores as the single covariate associated with putaminal EDVR revealed similar results (F-value: 7.351; p = 0.014). No such relationships were observed for autonomic and sensory fluctuations, and caudate nucleus EDVR did not correlate with any non-motor complication measures.
Group-wise estimation of the risk for long-term non-motor complications and other major health outcomes revealed that higher putaminal EDVR (lower dopamine turnover) at baseline was significantly associated with lower risk for neuropsychiatric fluctuations (Table 3): Patients in the upper half of EDVR had an OR of 0.01 (95% CI: 0.00–0.77; p = 0.037) for having neuropsychiatric fluctuations compared to the lower half (reference value) when adjusted to major candidate covariates. Adjustment only for baseline UPDRSII/III sum scores revealed an OR of 0.01 (95% CI: 0.00–0.64; p = 0.033). No further relationships between putaminal EDVR and other WOQ-19 outcome measures were detected and no associations between caudate nucleus EDVR and long-term WOQ-19 outcomes were observed.
Statistical analyses of the association of dichotomized striatal EDVR values and other major health outcomes did neither reveal any relationship between baseline striatal EDVR and the occurrence of other long-term health outcome measures such as dementia, depression, psychosis, ICD or falling (Supplementary Table 3) nor with UPDRSIII motor score, ADL scores (UPDRS part II and S&E scale), NMSS scale or PDQ-8 hr-QoL scale at last assessment (Supplementary Table 4).
Predictive values of baseline striatal EDVR (inverse of dopamine turnover) for survival
The overall mortality was 28% in the survival study cohort (9 out of 32 patients), but we excluded one patient from survival analysis due to a PD-independent cause of death (tonsillar carcinoma) leading to PD-related mortality of 26% (no censored observations). Multiple logistic regression models as described in the methods section did not show any relationship of striatal (caudate nucleus or putaminal) EDVR with survival (adj. p > 0.05). Additional univariate Cox proportional hazard models revealed only age at PET scan to be associated with survival (HR for one increase in age = 1.16 [95% CI: 1.05–1.29]; p = 0.005), but not gender, symptom duration at PET imaging, Hoehn&Yahr stage, and baseline UPDRS ADL and motor scores (p > 0.05). In keeping with univariate analysis, multivariate Cox proportional hazard models of relevant covariates showed the same result (adjusted HR for one year increase in age = 1.17 [95% CI: 1.02–1.33]; p = 0.022). The results of the multivariate Cox proportional hazard model analysis entering EDVR and age at PET scan as relevant covariate revealed no significant associations between PET measures and survival (Fig. 3).
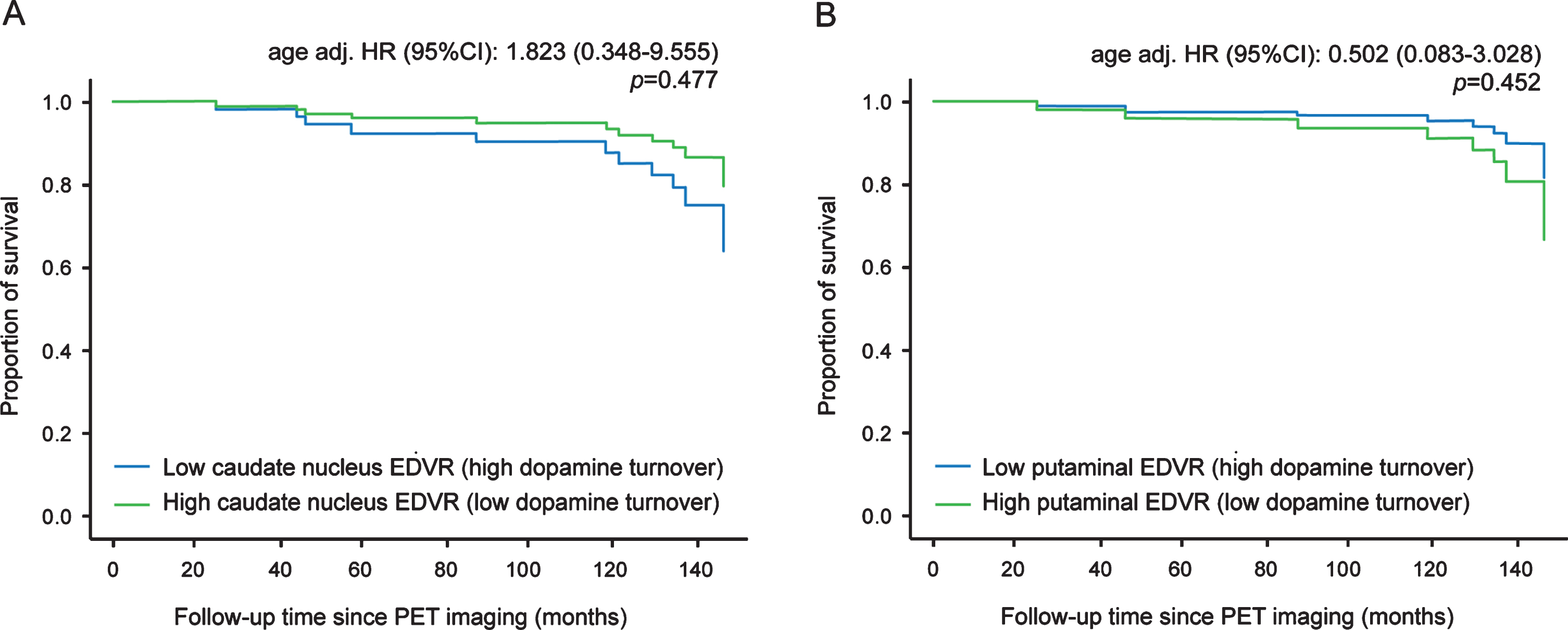
Survival with respect to striatal dopamine turnover as measured by 18Fluorodpa PET imaging. The graphs show the independent impact of caudate nucleus (A) and putaminal EDVR (B) on survival after adjusting for age in the multivariate Cox regression model (adjusted HR for one year increase in age = 1.17 [95% CI: 1.02–1.33]; p value: 0.022; the other candidate variables [gender, symptom duration at PET time, UPDRS part II/III sum score] did not show significant prediction of survival time [p value≥0.05] and were thus not included into the models. Hazard ratios (HR) with 95% confidence intervals (95% CI) and p values are from Multivariate Cox proportional hazard models with age as covariate (HR < 1 indicates smaller risk for death in the groups with high EDVR [low dopamine turnover]).
DISCUSSION
This retrospective longitudinal study shows patients with motor and neuropsychiatric fluctuations showed higher putaminal dopamine turnover in early de novo PD than those without this late-stage complication. Consistently, lower putaminal dopamine turnover in early PD is associated with a smaller risk to develop motor and neuropsychiatric fluctuations after long-term follow-up. No associations were observed for putaminal dopamine turnover and fluctuations of autonomic and sensory symptoms or other major health outcomes such as the dementia, depression, hr-QoL and survival. The association of motor and neuropsychiatric fluctuations and dopamine turnover is exclusively observed in putamen but not in caudate nucleus.
The frequencies of long-term health outcomes and the PD-related mortality of 26% after a disease duration of ∼11 years are expected from reports on larger PD cohorts [4, 29]. Although our cohort is rather small due to the technological complexity of EDVR estimation, it is thus representative for both early de novo PD [2, 30] as well as long-term longitudinal followed-up PD [1].
The extended 18Fluorodopa PET protocol allows the direct measurement of striatal dopamine turnover and revealed that elevated dopamine turnover occurs very early in PD [6–8]. These changes have not only been discussed as an early disease-intrinsic compensatory mechanism [7], but dopamine turnover in putamen, but not in caudate nucleus, has also been shown to predict the development of later motor complications (present study using WOQ-19 assessment and [10]). In agreement, longitudinal DAT binding studies suggested that lower DAT activity in the putamen, which is directly correlated with higher dopamine turnover [31], is associated with a higher risk for levodopa-induced dyskinesia [3]. Mechanistically, dopamine turnover is—together with other factors—responsible for dopamine haemostasis with low dopamine turnover leading to a more sustained dopamine action with less prominent fluctuations in synaptic dopamine concentration, which is considered a major factor responsible for the development of motor complications [7].
We here expand this knowledge by showing a similar association of putaminal dopamine turnover with later development of neuropsychiatric but not autonomic and sensory fluctuations. These results imply different mechanisms mediating the development of the various non-motor fluctuations with dopaminergic factors being essential for neuropsychiatric fluctuations in a very similar manner as compared to motor complications [28]. This is in agreement to the observation that fluctuations in mood and cognition occur in parallel to and with similar timing and kinetics as changes of motor states [32, 33]. The involvement of dopaminergic systems—particularly the meso-cortico-limbic system—in neuropsychiatric fluctuations is furthermore supported by various investigations showing a dose-dependent improvement of mood symptoms by levodopa infusion [28, 32]. Dopaminergic modulation of cognitive function has also been reported in PD with even greater oscillations in cognitive performance in patients with motor fluctuations [28, 34]. Since we do not observe any associations of 18Fluorodopa PET data on autonomic and sensory fluctuations, our data do not support a critical role of dopaminergic factors in their development [28].
We did not observe any association of striatal dopamine turnover with other major non-motor outcomes and survival. In agreement, a recent study was not able to show predictive value of DAT binding for dementia development, but only for future cognitive worsening [35]. This is in contrast to other studies using DAT imaging in early PD as a predictor for later occurrence of major non-motor symptoms including dementia, depression, falls and psychosis, with data on dementia prediction being confirmed in several cohorts [2, 36]. This discrepancy likely reflects the differences of the two measures: In early PD, striatal DAT binding does not only reflect dopaminergic degeneration, but is also influenced by changes of DAT expression as a compensatory mechanism to increase dopamine turnover and its synaptic availability [31, 37]. In contrast, dopamine turnover is considered a direct functional measure of the presynaptic compensatory changes in dopamine metabolism not only correlating with the extent of the degenerative process, but also depending on other genetic and demographic factors [7, 9].
Moreover, the association of DAT binding with a very broad range of non-motor health outcomes suggests a lack of specificity and implies that DAT imaging findings might reflect a more-widespread pathology [2]. Another candidate reason might be the different time periods of follow-up with only 2 to 5 years in previous studies but ∼11 years in the present one. This is of particular interest for dementia, because dopaminergic factors might be more relevant for cognitive dysfunction in the early compared to the later disease stages, when cholinergic factors become more important [38].
Our data on striatal dopamine turnover and prediction of mortality are in good agreement with previous data using DAT binding or 18Fluorodopa uptake in early PD patients showing no association of these imaging measures and survival of PD patients in larger cohorts [4, 5]. This implies that the early compensatory changes of dopamine metabolism [6] are, at least, not the only factor limiting the predictive value of dopaminergic imaging measures for mortality.
Although this is the first study investigating the predictive value of striatal dopamine turnover for later occurrence of major non-motor health outcomes, some important limitations need to be addressed: First, the complexity of the EDVR PET technology resulted in a relatively small cohort size based on one University center. This is somehow compensated by the homogenous study cohort at study entry (PET scanning) and the long-term and tight follow-up, which might however limit the generalizability of our results to a less homogenous population. Secondly, except for survival we only have reliable data on the presence of the various non-motor outcomes at the respective follow-up assessment, but not on the exact time point of symptom occurrence. As in similar studies [2], we calculated the predictive values of PET data for all these outcomes using logistic/linear regression models with data of the last available assessment [10]. Third, we used WOQ-19 data from our retrospective dataset to determine the occurrence of wearing-off phenomena. This scale is a self-questionnaire and thus depends on the patient’s understanding and perception of wearing-off. This scale display good sensitivity to detect wearing-off, but specificity might be limited [14, 15]. However, the present results on motor wearing-off closely match our data from medical records reflecting clinical examination data [10]. Fourth, our retrospective study design only allows indirect adjustment for differences in baseline characteristics by multivariate analysis. However, we adjusted for all major candidate covariates including age, symptom duration, disease severity at baseline and follow-up time period to largely exclude that inhomogeneity of the study cohort influence prediction of PET measures. Furthermore, by using the total LED as a covariate, we addressed the influence of different medication regimes during the observation period. Finally, we estimated striatal dopamine turnover (EDVR) using a single 18Fluorodopa PET scan combined with a tissue reference model to avoid arterial puncture for plasma 18Fluorodopa determination [8]. The potential biases of these two aspects are largely disease-dependent and increases with disease severity and are thus less relevant in our study with its homogenous, well-matched early PD cohort (for detailed discussion on PET technology, please refer to [9, 10]).
Together, relative elevation of putaminal dopamine turnover as part of compensatory mechanisms in early PD comprises an important disease-intrinsic factor for the development not only of later motor complications, but also of neuropsychiatric fluctuations as the most frequent type of non-motor fluctuations in late-stage PD [28, 29]. Since we have previously demonstrated that dopamine turnover increases after the initiation of levodopa treatment [8], our results moreover imply that an additional increase of intrinsic dopamine turnover by levodopa could play a pathophysiological role for the induction of levodopa-related non-motor complications. On the other hand, striatal dopamine turnover is not predictive for later occurrence of other non-motor fluctuations or other major health outcomes. These results should be treated as hypothesis generating and require confirmation.
CONFLICTS OF INTERESTS
Alexander Storch and Heinz Reichmann have received unrestricted research grants, speaker honoraria and consultancy fees from Pfizer Pharma GmbH, Berlin, Germany. The other authors have no other conflict of interest to report related to this article.
M. Löhle has been funded by a research grant of the Center for Regenerative Therapies Dresden, and received honoraria for presentations from Boehringer Ingelheim, GlaxoSmithKline, MEDA Pharma and UCB Pharma.
Dr. W. Hermann reports grants from Teva GmbH, Berlin, Germany, grants from UCB Pharma, Germany, outside the submitted work.
Dr. Wolz reports personal fees from Boehringer Ingelheim, Zambon, Valeant, Desitin, TEVA, UCB Pharma, Abbvie, Licher, and Daiichi Sankyo, outside the submitted work.
B. Beuthien-Baumann has received honoraria for presentations/lectures from Pfizer, Henning Berlin, Sanofi and Lilly.
H. Reichmann was acting on advisory boards and gave lectures and received research grants from Bayer Health Care, Boehringer/Ingelheim, Cephalon, Desitin, GlaxoSmithKline, Merck-Serono, Novartis, Orion, Pfizer, Solvay, TEVA/Lundbeck, UCB Pharma, and Valeant.
Dr. A. Hermann has received funding from the Federal Ministry of Education and Research (BMBF), the Deutsche Gesellschaft für Muskelkranke, the Roland-Ernst-Stiftung Saxony and the Helmholtz-Association. He served on the editorial board of BMC Neurology.
Dr. Storch has received funding from the Deutsche Forschungsgemeinschaft (DFG) and the Helmholtz-Association. He received unrestricted research grants from GKC Melbourne, Pfizer, and TEVA Pharma, honoraria for presentations/lectures/consultancies or advisory boards from AbbVie, Bayer Healthcare, Bial, GKC, Grünenthal, UCB, Zambon, Ca, AbbVie, TEVA, Lundbeck, and UCB Pharma, outside the submitted work. He has served on the editorial boards of Stem Cells and Stem Cells International.
J. Mende, Dr. Oehme, Dr. van den Hoff, and Dr. Kotzerke report no disclosures.
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank all patients for their willingness to participate in our study. Furthermore, we are grateful for help of the members of our movement disorders outpatient clinic at Technische Universität Dresden, the PET division of the Helmholtz-Zentrum Dresden-Rossendorf and the Coordination Center for Clinical trials at Technische Universität Dresden (particularly Uta Schwanebeck and Xina Grählert).
The initial randomized trial was supported by an unrestricted grant from Pfizer Pharma, Berlin, Germany. The financial sponsors of the study had no role in the study design, data collection, data analysis, data interpretation or writing of the report. The corresponding author had the final responsibility for the decision to submit for publication.
