Abstract
Background
Parkinson's disease (PD) patients on dopaminergic drugs may experience non-motor fluctuations (NMFs) which are often heterogeneous and respond variably to treatments.
Objective
We evaluated if personality was associated to NMFs and could modulate the NMFs responsiveness to dopaminergic medication and deep brain stimulation of the sub-thalamic nucleus (STN-DBS).
Methods
From the PREDISTIM cohort, personality dimensions of 235 PD patients were assessed by the Temperament and Character Inventory (TCI) before STN-DBS (V0). NMFs were evaluated using the NMFs Severity Scale at V0 and one year after STN-DBS (V1). Linear regression models were performed between TCI dimensions and NMFs at V0; and logistic regression models were done between TCI dimensions and 1) groups of dopa-sensitive patients (responders to ON medication at V0) versus non-dopa-sensitive ones, and 2) responders versus non-responders to STN-DBS at V1. Odds ratios (OR) were also calculated.
Results
Significant associations were found between two TCI personality dimensions (“Harm Avoidance” and “Self-Directedness”) and severity of NMFs in OFF medication at V0: PD patients with higher Harm Avoidance and lower Self-Directedness scores having more NMFs. TCI personality dimensions were not associated with the dopa-sensitivity while Novelty Seeking was significantly associated with the STN-DBS-responder group for the psychiatric (OR = 1.09 [1.02–1.17]) and for the dysautonomic NMFs (OR = 1.11 [1.04–1.18]).
Conclusions
Certain personality dimensions (Harm Avoidance and Self-Directedness) are associated with NMFs severity at baseline, and PD patients with high Novelty Seeking seem to be better candidates for NMFs improvement after STN-DBS.
Plain Language Summary
Non-motor fluctuations are highly invalidating for patients with Parkinson's disease and their response to treatments is variable. This new research examined personality dimensions impact on non-motor fluctuations and their responsiveness to dopaminergic medication and to deep brain stimulation. Two hundred and thirty-five patients with Parkinson's disease participated in the study and filled out several questionnaires to score their personality dimensions and their non-motor fluctuations severity before and after one year of deep brain stimulation. Comparisons were done between different groups of patients according to their response to dopaminergic treatments and to deep brain stimulation. As results, patients with personality traits of high “Harm Avoidance” (more anxious individuals) and low “Self-Directedness” (less goal-oriented individuals) had globally more non-motor fluctuations, and the personality dimension of “Novelty Seeking” (impulsive individuals) was related to a better non-motor fluctuations responsiveness to deep brain stimulation. Hence, non-motor fluctuations are associated with some specific personality dimensions on top of early onset of Parkinson's disease, long disease-duration, high doses of levodopa and female gender. Moreover, Parkinson's disease patients with impulsive-like personality seem to be better candidates for non-motor fluctuations improvement after deep brain stimulation.
Introduction
Patients with Parkinson's disease (PD) may experience disabling motor and non-motor fluctuations (NMFs). 1 NMFs are often heterogeneous, including psychiatric, cognitive and dysautonomic features, with variable dopaminergic responses.1–4 NMFs prevalence is not well known and varies from 17% to 100% in patients with motor fluctuations, depending on the assessment tools and on the population. 5 Classically, the occurrence of NMFs is associated with an early onset of PD, a longer disease-duration, higher doses of levodopa and female gender.1,2,4,6,7
Personality dimensions could also influence NMFs. To our knowledge, no study has yet evaluated this link between personality and NMFs, but some have highlighted some relationships between personality (using various measurement tools) and non-motor symptoms. Indeed, a recent study has shown that PD patients with a type D personality (negative emotions and low self-expression) had higher non-motor symptoms. 8 Another study, reported that personality dimensions from the Temperament and Character Inventory (TCI), such as Harm Avoidance, Self-Directedness and Novelty Seeking, were also associated with non-motor symptoms. 9
Concerning the response of NMFs after treatment, one study of 33 PD patients with NMFs found that only one out of three PD patients with NMFs had an improvement of NMFs after dopaminergic adjustment. 1 With the exception of a recent study showing that deep brain stimulation of the subthalamic nucleus (STN-DBS) does reduce levodopa-induced neuropsychiatric fluctuations in a similar way to levodopa, 10 others have reported some variability in NMF response to STN-DBS.4,5,11–17 Impulse Control Disorders (ICDs) – linked to neuropsychiatric fluctuations 18 – also seem to respond well to STN-DBS, 19 even if some studies have reported new onset of ICDs after STN-DBS. 19 In view of these observations, no predictive factor for levodopa responsiveness or improvement after STN-DBS of NMFs has yet been identified. Since in our previous study, we showed that personality dimensions were predictive of improvement in quality of life (QoL) after STN-DBS, 20 we hypothesized that they might also predict improvement of NMFs. Indeed, the personality dimensions measured by the TCI are related to different neurotransmitters systems such as the dopaminergic, serotoninergic and noradrenergic ones. 21 For example, the personality dimension of Novelty Seeking is thought to be associated with the dopaminergic system21–23; and some NMFs, such as apathy or impulsivity, are related to a hypo- or hyper-dopaminergic state.4,24 Hence, there could be a relationship between NMFs and TCI personality dimensions through these monoaminergic systems, especially the dopaminergic one.
Thus, our aim was to investigate the associations between personality dimensions and NMFs and to determine whether personality dimensions could modulate NMF response after dopaminergic treatment at baseline and/or STN-DBS one year after implantation.
Methods
Study design
This study is an ancillary analysis using data collected from the PREDISTIM study, an ongoing observational and prospective multicentric cohort (Protocol 2013-A00193–42; clinicalTrial.gov n°NCT02360683) sponsored by the University Hospital of Lille, conducted in 17 PD expert centers from the clinical research network in France (NS-Park/F-Crin), and approved from the CPP Nord Ouest-IV Ethical Committee.
Patients undergoing bilateral STN-DBS were included (n = 235). Inclusion and exclusion criteria and data collection were defined in a previous publication. 20 In short, patients having a diagnosis of PD (UKPDSBB), a disease duration ≥ 5 years, between 18 and 75 years old and undergoing STN-DBS in one of the participating centers were included in the study. Patients presenting atypical Parkinsonism, severe cognitive impairment (Montreal Cognitive Assessment score < 23) or severe psychiatric disorders were excluded. Data was collected at baseline (V0) and 1-year post-STN-DBS (V1).
Patients gave written informed consent, the study was conducted according to good clinical practice, local regulations, and data collection was compliant with GDPR rules.
Outcome measures
The NMF Severity Scale (NMF2S) is a newly validated scale to evaluate NMFs severity in PD, using 11 numeric scales rated from 0 (no fluctuations) to 10 (maximum intensity) evaluating 11 non-motor symptoms that may be fluctuating.7,25 Total NMF2S score ranges from 0 to 110 and is the sum of three sub-scores: psychiatric (anxiety, sadness and irritability), cognitive (apathy, fatigue and concentration difficulties) and dysautonomic (pain, dyspnea, restless leg syndrome, urinary trouble and excessive sweat). In the end, NMF2S total score and sub-scores are divided by their corresponding number of items to obtain a score between 0 and 10. The NMF2S was collected at V0 and V1. At baseline, NMF2S was evaluated in OFF medication (OFFmed) (an overnight withdrawal of all dopaminergic treatment, at least 8 h without dopaminergic treatment) and in ON medication (ONmed) (after taking a 150% dose of the first morning Levodopa Equivalent Dose (LED)). One-year after STN-DBS, NMF2S was evaluated in OFFmed-OFFstim and OFFmed-ONstim conditions (OFFstim was evaluated one hour after switching OFF DBS).
The TCI is a self-questionnaire evaluating seven independent personality dimensions through 226 True or False questions. 21 It was already validated for the PD population. 9 These seven TCI dimensions consist of four temperaments (Novelty Seeking, Harm Avoidance, Reward Dependence and Persistence) and three characters (Self-Directedness, Cooperativeness and Self-Transcendence). 26 Each dimension is independently scored, reflecting personality traits as a spectrum: Novelty Seeking being representative of “exploratory excitability”, “impulsiveness”, “extravagance” and “disorderliness”; Harm Avoidance of “anticipatory worry”, “fear of uncertainty”, “shyness” and “fatigability”; Reward Dependence of “sentimentality”, “attachment” and “dependence”; Persistence of perseverant and ambitious temperament; Self-Directedness of “responsibility”, “purposeful”, “resourcefulness”, “self-acceptance” and “congruent second nature”; Cooperativeness of “social acceptance”, “empathy”, “helpfulness”, “compassion” and “pure-hearted”; and Self-Transcendence of “self-forgetful”, “transpersonal identification”, “spiritual acceptance”. The TCI (personality assessment) was only collected at V0 in ONmed.
We evaluated motor and non-motor symptoms using the MDS-UPDRS (Movement Disorders Society-Unified Parkinson's Disease Rating Scale) at V0 and V1; impulsivity and impulse control disorders using the ECMP (Evaluation Comportementale dans la Maladie de Parkinson = behavior assessment in Parkinson's disease) and the UPPS-P (Urgency-Premeditation-Perseverance-Sensation Seeking-Positive Urgency impulsive behavior scale) at V0. Total LED and agonist LED were calculated at V0 and V1.
Statistical analyses
TCI dimensions and NMFs at V0
We evaluated the associations between each TCI dimension scores (explicative variables) and NMF2S total score and sub-scores at V0 (response variables), in ONmed and OFFmed using linear regression models.
TCI dimensions and NMFs response to dopaminergic drugs at V0
Changes of NMF2S total score and sub-scores between ONmed and OFFmed at V0 were assessed (paired Wilcoxon tests for the non-parametric distribution of the data, verified by Shapiro tests).
Then, two groups were created according to NMFs response to dopaminergic drugs: the “dopa-sensitive” (percentage of NMF2S total score and sub-scores improvement between ON and OFF medication at V0 ≥ 30%) and the “non-dopa-sensitive” groups. Currently, there is no minimal clinically important difference in the literature concerning this new NMF2S scale and there is no satisfactory definition of a positive response to levodopa. We therefore chose a threshold of 30% as clinically relevant for NMFs improvement based on the motor improvement obtained after levodopa (Albanese et al., 2001). 27
Logistic regressions were done between TCI personality dimensions (explicative variables) and dopa-sensitivity groups (response variable), and OR with their confidence intervals at 95% (CI95) were calculated.
TCI dimensions and NMFs response to STN-DBS at V1
One year after STN-DBS, changes of NMF2S total score and sub-scores between OFFmed-ONstim and OFFmed-OFFstim states were evaluated (paired Wilcoxon tests).
Then, two groups were created according to NMFs response to STN-DBS: the “responder” (percentage of NMF2S total score and sub-scores improvement between OFFmed-ONstim and OFFmed-OFFstim at V1 ≥ 30%) and the “non-responder” groups.
Logistic regressions were done between TCI personality dimensions (explicative variables) and DBS-responding groups (response variable), and OR with their CI95 were calculated.
All analyses were conducted on R Studio Software Version 2024.04.2, and a threshold of bilateral statistical significance of 0.05 was used after FDR corrections being applied. Interpretations were made carefully according to results strength.
Results
235 PD patients were included in the study (78 women (33%) and 157 men (67%)) with a mean age of 59.9 ± 7.4 years old, a mean disease duration of 10.8 ± 4 years and a mean total LED of 1450.2 ± 1227.8 mg/day at V0 (Table 1) which diminishes to 666.7 ± 431.9 mg/day at V1. Mean total NMF2S score at V0 was of 3.6 ± 2 in OFFmed (Table 2). Because of some missing data, only n = 214 PD patients were used for the analyses after STN-DBS at V1.
Description of the population (n = 235).

Results are given in mean ± SD (Standard Deviation); LED: Levodopa Equivalent Dose; HAMD: Hamilton Depression Scale; HAMA: Hamilton Anxiety Scale; UPPS-P: Urgency-Premeditation-Perseverance-Sensation Seeking-Positive Urgency impulsive behavior scale; ECMP: Evaluation Comportementale dans la Maladie de Parkinson (=behavior assessment in Parkinson's disease).
Changes of the NMFs and Parkinson's disease symptoms at V0 and V1.
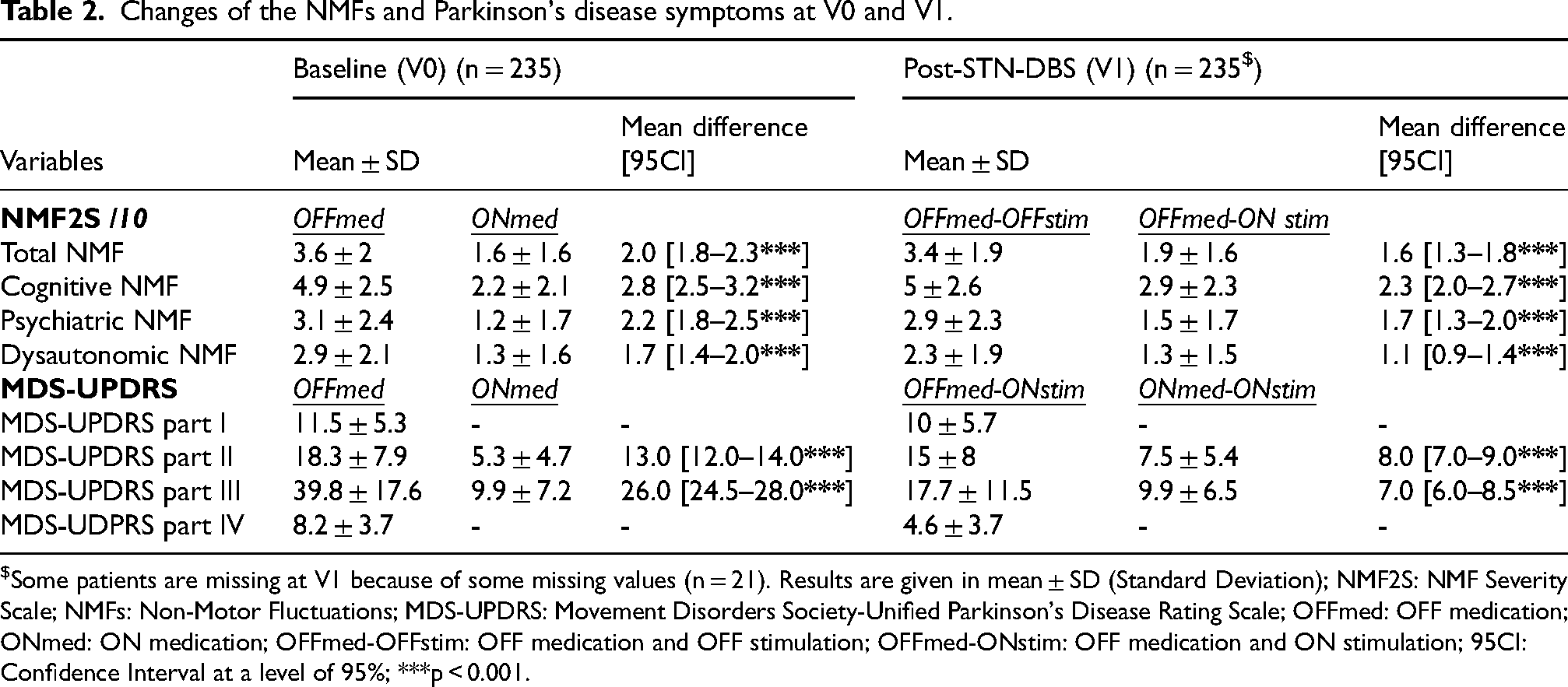
Some patients are missing at V1 because of some missing values (n = 21). Results are given in mean ± SD (Standard Deviation); NMF2S: NMF Severity Scale; NMFs: Non-Motor Fluctuations; MDS-UPDRS: Movement Disorders Society-Unified Parkinson's Disease Rating Scale; OFFmed: OFF medication; ONmed: ON medication; OFFmed-OFFstim: OFF medication and OFF stimulation; OFFmed-ONstim: OFF medication and ON stimulation; 95CI: Confidence Interval at a level of 95%; ***p < 0.001.
TCI dimensions and NMFs at V0
At baseline, significant associations were only found in OFFmed. Positive associations were found between Harm Avoidance and NMF2S total, cognitive and psychiatric scores (FDR-p.values = 0.02): PD patients with greater Harm Avoidance had higher severity of NMFs (Table 3). Self-Directedness was negatively and significantly associated with NMF2S psychiatric scores (FDR-p.value = 0.02): PD patients with greater Self-Directedness had lower severity of psychiatric NMFs (Table 3).
Linear regression models between NMF2S scores at V0 and TCI dimensions.

NMF2S: NMF Severity Scale; NMFs: Non-Motor Fluctuations; Medication corresponds to the dopaminergic state at V0; TCI: Temperament and Character Inventory; NS: Novelty Seeking; HA: Harm Avoidance; RD: Reward Dependence; P: Persistence; SD: Self-Directedness; C: Cooperativeness; ST: Self-Transcendence; *FDR-p value < 0.05 (corrected for each multiple comparison) in bold.
TCI dimensions and NMFs response to dopaminergic drugs at V0
NMF2S scores were significantly improved in ONmed versus OFFmed in NMF2S total score and each sub-score (Table 2).
No significant associations were found between NMFs dopa-sensitivity groups and TCI dimensions, (Figure 1).
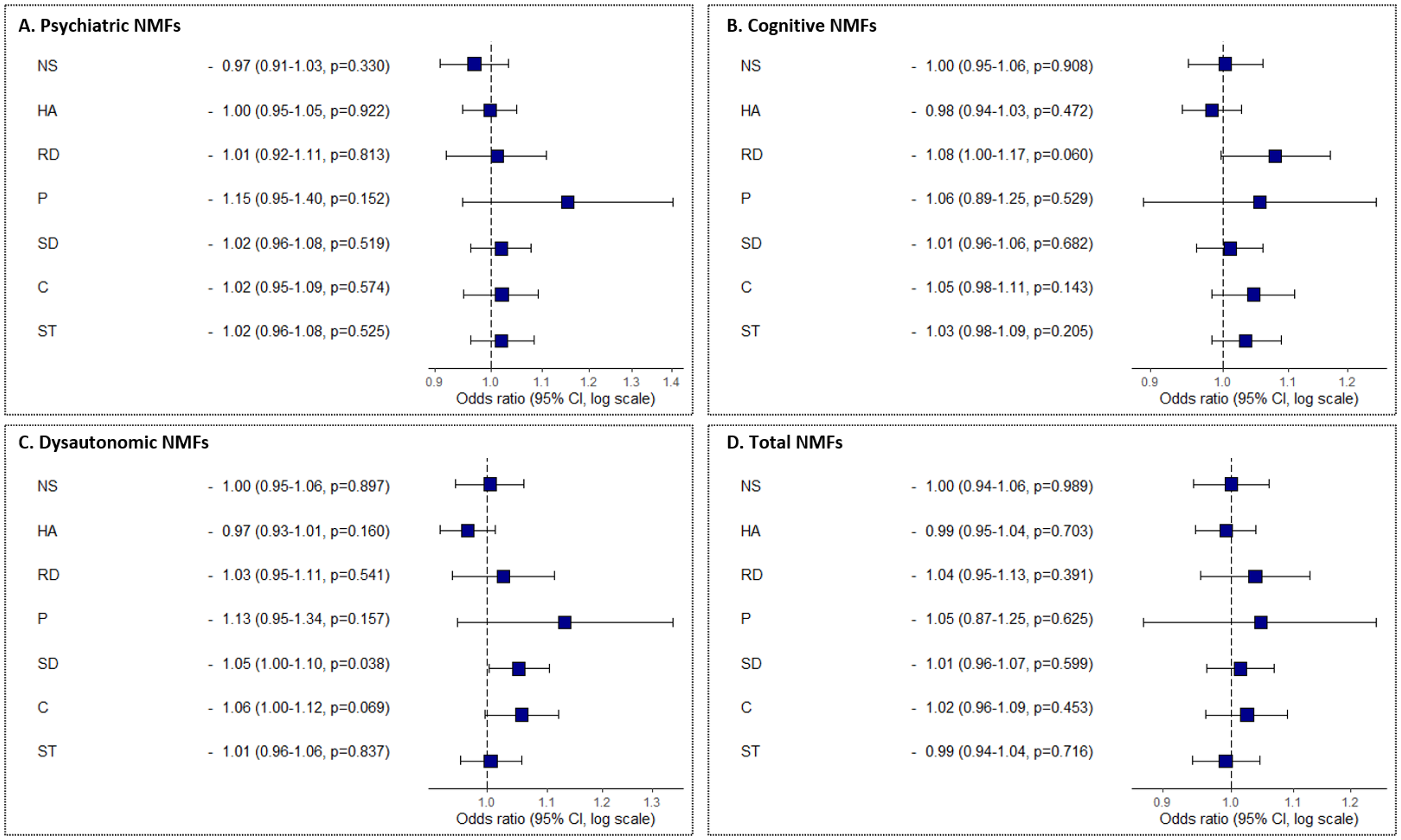
Representations of the Odds Ratio (OR) of each TCI personality dimensions concerning NMFs response to dopaminergic treatments.
TCI dimensions and NMFs response to STN-DBS at V1
At one-year after switching ON STN-DBS, NMFs were significantly improved in the total score and each sub-score of NMF2S (Table 2 – OFFmed-OFFstim vs OFFmed-ONstim scores at V1).
After STN-DBS, two significant associations were found between NMFs responsiveness to STN-DBS and a TCI dimension: Novelty Seeking was significantly associated with the DBS-responder group for the psychiatric NMFs (OR = 1.09 [1.02–1.17], p.value = 0.009) and for the dysautonomic NMFs (OR = 1.11 [1.04–1.18], p.value = 0.003) (Figure 2). PD patients in the DBS-responder group concerning psychiatric and dysautonomic NMFs had higher Novelty Seeking scores.
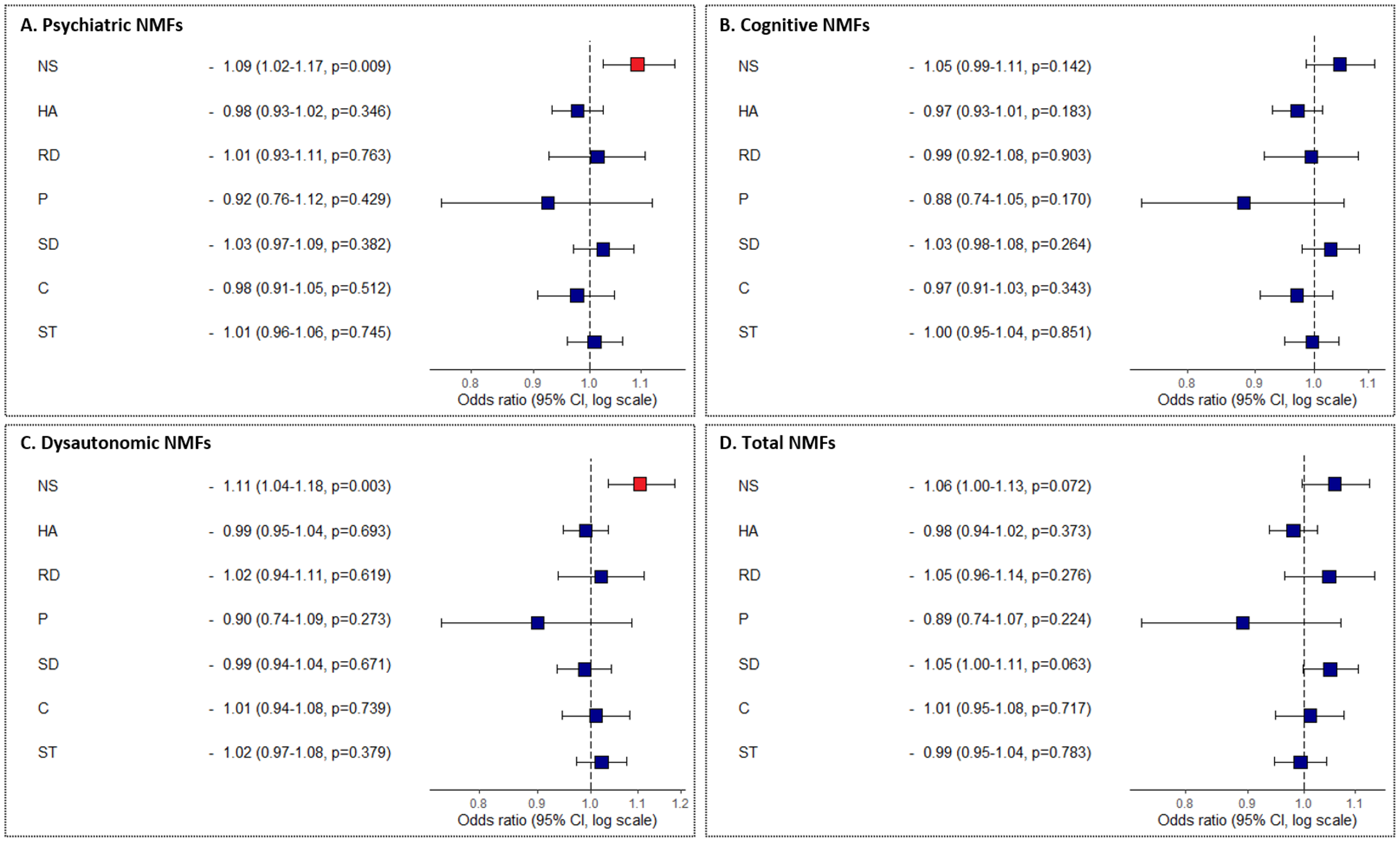
Representations of the Odds Ratio (OR) of each TCI personality dimensions concerning NMFs response to STN-DBS.
Discussion
We have found significant associations between some TCI personality dimensions and OFF-medication NMFs at baseline. Lower Harm Avoidance and higher Self-Directedness scores were associated with lower severity of NMFs, especially psychiatric NMFs. While no TCI personality dimensions were associated with NMFs dopa-sensitivity, Novelty Seeking was related to NMFs responsiveness to STN-DBS: the best improvement in psychiatric and dysautonomic NMFs after STN-DBS was found in PD patients with higher Novelty Seeking scores.
PD symptoms are part of a multifactorial system and are influenced by many factors such as genetic, sociodemographic and socio-cultural factors as well as personality (Figure 3A). In this multifactorial system, NMFs may be related to disease severity, behavioral and psychological factors, and some personality dimensions (Figure 3A).9,20,28 PD patients with high Harm Avoidance and low Self-Directedness have more severe NMFs, especially psychiatric ones in OFF medication. This could be the result of their anxiety-depressive state, which is one of the psychological factors influencing NMFs.
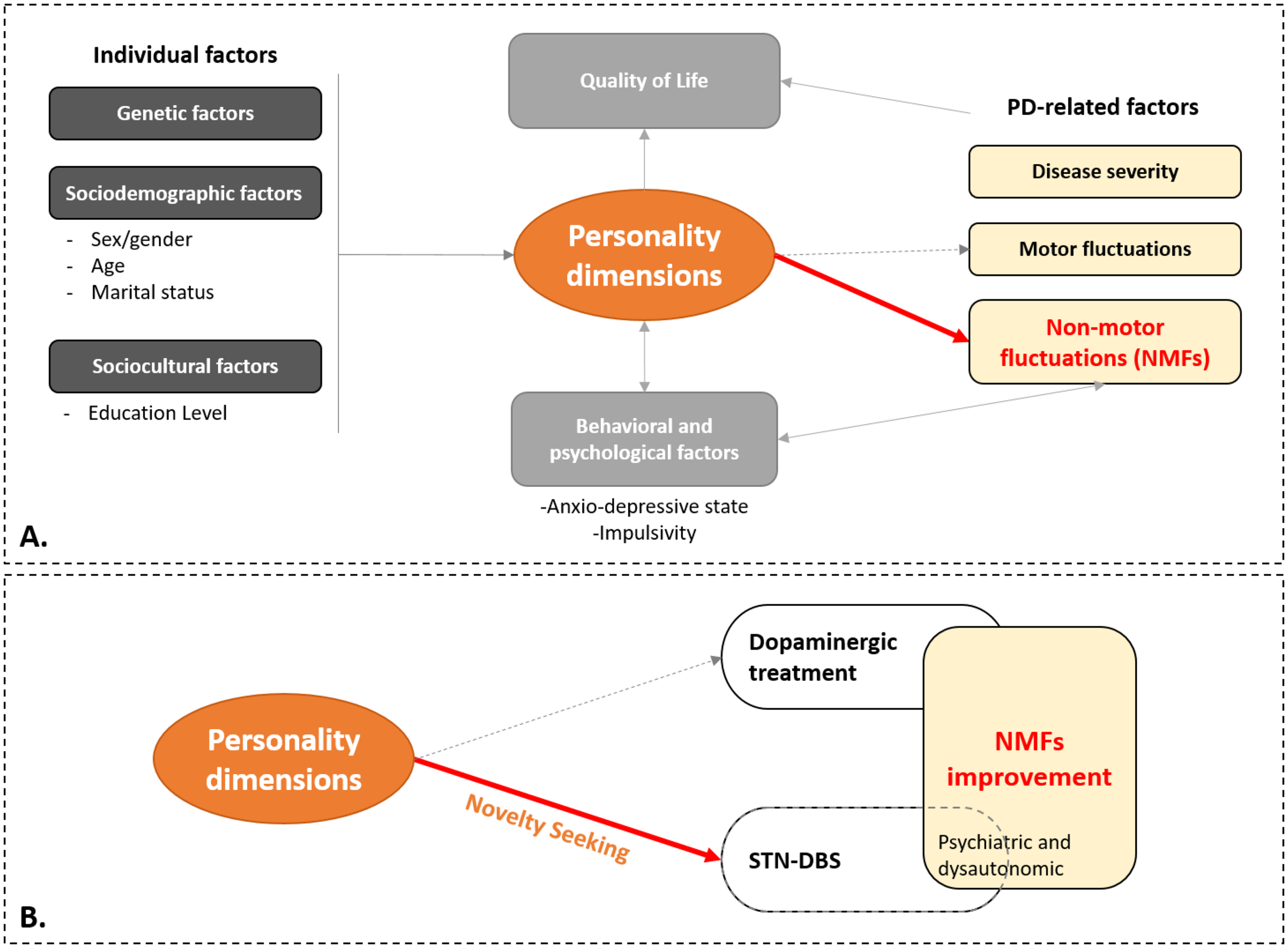
Directed acyclic graph showing representation of causal assumptions.
Harm Avoidance is a temperament related to “anticipatory worry”, “fear of uncertainty”, and “fatigability”, as described through its sub-dimensions. 21 It was often associated with depressive behavior in the literature.9,29–32 Hence, the worse global, cognitive and psychiatric NMFs in OFF-medication in PD patients with high Harm Avoidance is certainly related to this general profile of anxiety and worrying, and could be further accentuated by dopaminergic fluctuations. Self-Directedness, a character associated with “purposeful”, “resourcefulness”, and “self-acceptance”, represents efficient ways to react to diverse events. 21 Low scores in Self-Directedness have been associated with depressive features.9,29,30,33,34 Therefore, their association with higher psychiatric NMFs (anxiety, sadness and irritability) in OFF-medication is likely to be related to higher anxio-depressive state. Moreover, these associations between personality and severe NMFs could explain why QoL in PD patients is also associated with these same personality dimensions 28 : Harm Avoidance and Self-Directedness are linked to NMFs which in turn have an impact on QoL. 35
We hypothesized that NMFs response to therapies (dopaminergic treatment and/or STN-DBS) may also be related to different factors including personality dimensions (Figure 3B). Nonetheless, personality did not have a major impact on NMFs dopa-sensitivity, since no significant associations were found. This is congruent with a previous study that showed no association between personality and dopa-sensitivity on motor symptoms. 9
Regarding NMFs responsiveness to STN-DBS, only higher Novelty Seeking scores were associated with a better psychiatric and dysautonomic NMFs response (Figure 3B). Novelty Seeking is a character related to “exploratory excitability” and “impulsiveness”. 21 Hence, “Novelty Seekers” individuals tend to respond positively to new stimuli.26,36 They may be more sensitive to chronic administration of dopaminergic drugs and more prone to develop some hyperdopaminergic behaviors such as ICDs.37–39 Novelty seeking is a major predictor of impulsive and addictive behaviors. 40 Indeed, Novelty Seeking scores correlated positively with the UPPS Impulsive Behavior scale (namely the Urgency, Positive Urgency, lack of Premeditation, and Sensation Seeking sub-scores – Supplemental Table 1). Hence, after STN-DBS, the significant reduction in oral dopaminergic drugs could lead to a significant decrease in their NMFs, especially psychiatric and dysautonomic ones 41 as well as ICD as previously shown in the PREDI-STIM cohort. 19 In fact, Novelty Seeking scores correlated positively with the dopaminergic agonists LED and with its reduction at V1 (Supplemental Table 1).
Finally, all these results enhance our hypothesis of a multidimensional relationship between some TCI personality dimensions, NMFs, QoL, and behavior in PD (Figure 3).
One of the limitations of this study is the low variance explained by our models and the weak odd ratio between Novelty Seeking and response to STN-DBS, suggesting that personality should only be considered as one of multiple factors associated with NMFs. We need to investigate other aspects that are also part of personality, such as social representations and beliefs, medication and surgical treatment, which may also have an impact on NMFs and their response. TCI was only assessed before STN-DBS, and it cannot be excluded that personality dimensions could change after STN-DBS which might affect the results.42–47 We are currently running a study evaluating TCI after STN-DBS to look for changes in personality dimensions. In our analyses, we chose not to include covariates because personality, anxiety-depressive state and impulsivity are too interrelated and imbricated altogether to be used in the same models, as this would lead to redundancy. The other limitations were the lack of randomization to counterbalance medication states and post-DBS conditions, the lack of a third temporal evaluation of NMFs at a later date to confirm our results, and the fact that NMF2S evaluation was yet only used and validated in the PREDISTIM cohort. 7
In conclusion, our study suggests an association between some personality dimensions, NMFs severity and NMFs response to STN-DBS. PD patients with higher Harm Avoidance and lower Self-Directedness scores had more severe NMFs and NMFs improvement after STN-DBS was associated to higher Novelty Seeking scores. In PD patients, several factors (linked to genetics, sociodemographic and sociocultural determinants), through personality dimensions, could interact with each other and influence patients’ symptoms, behavior, quality of life and treatment responses.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251354932 - Supplemental material for Exploring the link between personality dimensions and non-motor fluctuations in Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251354932 for Exploring the link between personality dimensions and non-motor fluctuations in Parkinson's disease by Mathilde Boussac, Florent Faggianelli, Estelle Harroch, Alexandre Eusebio, Margherita Fabbri, Fabienne Ory-Magne, Emeline Descamps, Olivier Rascol, Chloé Laurencin, Ana-Raquel Marques, Mathieu Anheim, Bruno Giordana, Lucie Hopes, Caroline Moreau, Anne-Sophie Rolland, David Devos, David Maltête, Solène Ansquer, Elodie Hainque, Sophie Drapier, Jean-Philippe Brandel, Tiphaine Rouaud, Dominique Guehl, Bechir Jarraya, Mélissa Tir, Tatiana Witjas, Jean-Philippe Azulay, Christine Brefel-Courbon and in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We thank all participants for their cooperation.
We are thankful for the funding of this study by the France Parkinson charity and French Ministry of Health (PHRC national 2012), and for the participation of AXA Assurances VIE Mutuelle (France).
We are also grateful for support from the French clinical research network NS-Park/F-Crin and the Fédération de la Recherche Clinique du CHU de Lille (with Anne-Sophie Rolland, Alain Duhamel, Maeva Kheng, Julien Labreuch, Dominique Deplanque, Edouard Millois, Nolwen Dautrevaux, Victor Laugeais, Maxime Caillier, Aymen Aouni, Pauline Guyon, Francine Niset, Valérie Santraine, Marie Pleuvret, Julie Moutarde and Laetitia Thibault).
We also thank all the members of the PREDI-STIM study group: Dr Caroline Moreau, Pr Luc Defebvre, Dr Nicolas Carriere, Dr Guillaume Grolez, Dr Guillaume Baille, Dr Kreisler, Pr Jean-Pierre Pruvo, Pr Leclerc, Dr Renaud Lopes, Dr Romain Viard, Dr Gregory Kuchcinski, Mr Julien Dumont, Pr Kathy Dujardin, Mme M Delliaux, Mrs M Brion, Dr Gustavo Touzet, Pr Nicolas Reyns, Pr Arnaud Delval, Mrs Valerie Santraine, Mrs Marie Pleuvret, Mrs Nolwen Dautrevaux, Mr Victor Laugeais, Thavarak Ouk, Camille Potey, Celine Leclercq and Elise Gers (for Lille University Hospital); Jean-Christophe Corvol, Marie-Vidailhet, Elodie Hainque, Marie-Laure Welter, Lucette Lacomblez, David Grabli, Emmanuel Roze, Yulia Worbe, Cécile Delorme, Hana You, Jonas Ihle, Raquel Guimeraes-Costa, Florence Cormier-Dequaire, Aurélie Méneret, Andréas Hartmann, Louise-Laure Mariani, Stéphane Lehericy, Virginie Czernecki, Fanny Pineau, Frédérique Bozon, Camille Huiban, Eve Benchetrit, Carine Karachi, Soledad Navarro, Philippe Cornu, Arlette Welaratne, Carole Dongmo-Kenfack, Lise Mantisi, Nathalie Jarry, Sophie Aix and Carine Lefort (for the AP-HP, Paris); Dr Tiphaine Rouaud, Pr Philippe Damier, Pr Pascal Derkinderen, Dr Anne-Gaelle Corbille, Dr Elisabeth Calvier-Auffray, Mrs Laetitia Rocher, Mrs Anne-Laure Deruet, Dr Raoul Sylvie, Dr Roualdes Vincent and Mrs Le Dily Séverine (for Nantes University Hospital); Dr Ana Marques, Dr Berangere Debilly, Pr Franck Durif, Dr Philippe Derost, Dr Charlotte Beal, Carine Chassain, Laure Delaby, Tiphaine Vidal, Pr Jean Jacques Lemaire, Isabelle Rieu and Elodie Durand (for Clermont-Ferrand University Hospital); Pr Alexandre Eusebio, Pr Jean-Philippe Azulay, Dr Tatiana Witjas, Dr Frédérique Fluchère, Dr Stephan Grimaldi, Pr Nadine Girard, Eve Benchetrit, Marie Delfini, Dr Romain Carron, Pr Jean Regis, Dr Giorgio Spatola and Camille Magnaudet (for the AP-HM, Marseille); Dr Ansquer Solène, Dr Benatru Isabelle, Dr Colin Olivier, Pr Houeto JL, Pr Guillevin Remy, Mrs Fradet Anne, Mrs Anziza Manssouri, Mrs Blondeau Sophie, Dr Richard Philippe, Dr Cam Philippe, Dr Page Philippe, Pr Bataille Benoit, Mrs Rabois Emilie and Mrs Guillemain Annie (for Poitiers University Hospital); Dr Drapier Sophie, Dr Frédérique Leh, Dr Alexandre Bonnet, Pr Marc Vérin, Dr Jean-Christophe Ferré, Mr Jean François Houvenaghel, Pr Claire Haegelen, Mrs Francoise Kestens and Mrs Solenn Ory (for Rennes University Hospital); Pr Pierre Burbaud, Dr Nathalie Damon-Perriere, Pr Wassilios Meissner, Pr Francois Tison, Dr Stéphanie Bannier, Dr Elsa Krim, Pr Dominique Guehl, Sandrine Molinier-Blossier, Morgan Ollivier, Marion Lacoste, Nicolas Auzou, Marie Bonnet, Pr Emmanuel Cuny, Dr Julien Engelhardt, Olivier Branchard, Clotilde Huet and Julie Blanchard (for Bordeaux University Hospital); Pr Rascol Olivier, Dr Christine Brefel Courbon, Dr Fabienne Ory Magne, Dr Marion Simonetta Moreau, Pr Christophe Arbus, Pr Fabrice Bonneville, Dr Jean Albert Lotterie, Marion Sarrail, Charlotte Scotto d’Apollonia, Pr Patrick Chaynes, Pr François Caire and Estelle Harroch (for Toulouse University Hospital); Pr David Maltete, Dr Romain Lefaucheur, Dr Damien Fetter, Dr Nicolas Magne, Mrs Sandrine Bioux, Mrs Maud Loubeyre, Mrs Evangéline Bliaux, Mrs Dorothée Pouliquen, Pr Stéphane Derrey, Mrs Linda Vernon and Dr Frédéric Ziegler (for Rouen University Hospital); Mathieu Anheim, Ouhaid Lagha-Boukbiza, Christine Tranchant, Odile Gebus, Solveig Montaut, S Kremer, Nadine Longato, Clélie Phillips, Jimmy Voirin, Marie des Neiges Santin, Dominique Chaussemy and Dr Amaury Mengin (for Strasbourg University Hospital); Dr Caroline Giordana, Dr Claire Marsé, Lydiane Mondot, Bruno Giordana, Robin Kardous, Bernadette Bailet, Héloise Joly, Denys Fontaine, Dr Aurélie Leplus, Amélie Faustini and Vanessa Ferrier (for Nice University Hospital); Pr Pierre Krystkowiak, Dr Mélissa Tir, Pr Jean-Marc Constans, Sandrine Wannepain, Audrey Seling, Dr Michel Lefranc, Stéphanie Blin and Béatrice Schuler (for Amiens University Hospital); Pr Stephane Thobois, Dr Teodor Danaila, Dr Chloe Laurencin, Pr Yves Berthezene, Dr Roxana Ameli, Helene Klinger, Dr Gustavo Polo, Patrick Mertens, A Nunes and Elise Metereau (for Lyon University Hospital); Dr Lucie Hopes, Dr Solène Frismand, Dr Emmanuelle Schmitt, Mrs Mylène Meyer, Mrs Céline Dillier, Pr Sophie Colnat and Mrs Anne Chatelain (for Nancy University Hospital); Dr Jean- Philippe Brandel, Dr Cécile Hubsch, Dr Patte Karsenti, Dr Marie Lebouteux, Dr Marc Ziegler, Dr Christine Delmaire, Dr Julien Savatowky, Mrs Juliette Vrillac, Mrs Claire Nakache, Dr Vincent D’Hardemare and Mr Lhaouas Belamri (for the Rotschild fondation of Paris); Dr Valérie Mesnage, Dr Cécilia Bonnet, Dr Jarbas Correa Lino Jr., Dr Camille Decrocq, Dr Anne Boulin, Mrs Inès Barre, Mrs Jordane Manouvrier, Dr Bérénice Gardel, Pr Béchir Jarraya, Mrs Catherine Ziz, Mrs Lydie Prette and Mr Hassen Douzane (for the Foch Hospital of Paris-Saclay University); David Gay, Robin Bonicel, Fouzia El Mountassir, Clara Fischer, Jean-François Mangin, Marie Chupin and Yann Cointepas (for CATI (MRI acquisition management, preprocessing and data management)); Bertrand Accart, Patrick Gelé, Florine Fievet, Matthieu Chabel, Virginie Derenaucourt, Loïc Facon, Yanick Tchantchou Njosse and Dominique Deplanque (for CRB of Lille (Center of Biological Resources)); and Alain Duhamel, Lynda Djemmane and Florence Duflot (for Data management of Lille).
ORCID iDs
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the France Parkinson charity and French Ministry of Health (PHRC national 2012) with the support of the Mutuelles AXA health sponsorship program. This is an ancillary study to Protocol ID: 2013-A00193-42; ClinicalTrials.gov: NCT02360683.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used in this study is not our individual propriety. It comes from the cohort of a multicentric study. Therefore, the data can only be available on request to Research and Innovation unit of Lille University Hospital Center (David Devos: David. DEVOS@chru-lille.fr), once all the data has been analyzed.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
