Abstract
To determine if recently reported changes in sensory thresholds during migraine attacks can also be seen in cluster headache (CH), we performed quantitative sensory testing (QST) in 10 healthy subjects and in 16 patients with CH. Eight of the patients had an episodic CH and the other eight a chronic CH. The tests were performed on the right and left cheeks and on the right and left side of the back of the hands to determine the subjects' perception and pain thresholds for thermal (use of a thermode) and mechanical (vibration, pressure pain thresholds, pin prick, von Frey hairs) stimuli. Six patients were examined in the attack-free period. Three were also willing to repeat the tests a second time during an acute headache attack, which was elicited with nitroglycerin. The healthy subjects performed the experiments in the morning and evening of the same day to determine if sensory thresholds are independent of the time of day. If they were, this would allow estimation of the influence of the endogenous cortisone concentration on these thresholds. The control group showed no influence of the time of day on the thresholds. There was a significant difference in pain sensitivity between the back of the hands and the cheeks (P < 0.05): higher thresholds were found on the back of the hands. The thresholds generally exhibited little intersubject variability, indicating that QST is a reliable method. There was also a significant difference between the test areas in the patient group (P < 0.001): the cheeks were also more sensitive than the back of the hands. In comparison with reference data of healthy volunteers, the detection thresholds were increased in the patients on both test areas. These were statistically significant for warmth, thermal sensory limen (TSL), heat and pressure on the back of the hands (P < 0.04) and for the warmth and TSL thresholds on the cheeks (P < 0.05). There were no differences in the thresholds regardless of whether the patients were examined in or outside of a cluster bout. Furthermore, we found no cutaneous allodynia in the three patients tested during an attack. The increased sensory thresholds on the cheeks as well as on the back of the hands are in agreement with an increased activation of the patients' antinociceptive system. The seasonal variation and the temporal regularity of single attacks as well as the findings in imaging studies indicate that the hypothalamus is involved in the pathophysiology of CH. In view of the strong connectivity between the hypothalamus and areas involved in the antinociceptive system in the brainstem, we hypothesize that this connection is the reason for the increased sensory thresholds in CH patients found in our study.
Keywords
Introduction
The diagnosis of cluster headache (CH) is based on the typical clinical criteria as described in the International Headache Society (IHS) Classification (1). Clinical features suggest the pathophysiological involvement of the trigeminal, the parasympathetic and the sympathetic systems. The pain is typically centred in the first division of the trigeminal nerve. During acute attacks there is activation of the trigeminovascular reflex indicated by the elevation of calcitonin gene-related peptide (CGRP) serum concentrations ipsilateral to the pain (2). Some autonomic findings (lacrimation, rhinorrhoea) and elevated serum vasoactive intestinal peptide concentrations ipsilateral to the pain during attacks suggest the activation of the parasympathetic system and a dysfunction of ipsilateral sympathetic innervation (ptosis, miosis and impaired sweating) (2). The characteristic relapsing–remitting course with seasonal variation and the temporal regularity of single attacks indicate that the biological clock, namely the hypothalamus, also plays a role. The involvement of the hypothalamus is supported by hormonal findings (cortisol and melantonin concentrations) (3), the elevation of soluble interleukin-2 receptors in the serum (4), evidence of imaging studies (5, 6) and the successful stimulation of this area for treatment of CH (7).
There is also activation of the trigeminovascular system during a migraine attack, as indicated by elevated CGRP concentrations during the attacks. Burstein and coworkers were the first to demonstrate the development of an allodynia with a typical spatial and temporal distribution during migraine attacks (8, 9). Cutaneous allodynia appears within 1 h after throbbing headache begins and can last from 2 to 4 h to the end of the attack. This alteration of sensory perception is interpreted to be a sign of secondary sensitization of pain-processing second-order neurons due to the nociceptive inflow during the attack. Furthermore, cutaneous brush allodynia has been shown to occur in short-lasting unilateral neuralgiform headache with conjunctival injection and tearing (SUNCT) with no temporal delay between onset of pain and development of allodynia (10). These studies suggest that initiation and duration of cutaneous allodynia linked to central sensitization are determined by the temporal profile of the headache questioning if it can be seen in all trigeminal-driven headache disorders. Since both migraine and CH have in common this activation of the trigeminovascular reflexes, we investigated whether patients with CH develop similar changes in the sensory thresholds and if these thresholds are altered.
Methods
Subjects
Ten paid healthy volunteers (five females, five males) with no history of any chronic disease or headache disorder (mean age 29.7 years, range 22–53 years) participated in the experiment. The healthy subjects performed the tests twice within 1 day (morning and afternoon) according to the endogenous cortisone concentration for time-of-day-independent thresholds.
Sixteen CH patients participated, eight chronic (CCH) and eight episodic (ECH), diagnosed on the basis of the IHS criteria (15 men, one woman; mean age 42.8 years, range 21–65 years). The mean duration of the disease was 9.8 years (range 0.2–21 years) and the frequency of attacks ranged from one to six per day. The average severity of the headache on a visual analogue scale (VAS) from 0 to 100 (0 = not painful; 100 = maximum pain imaginable) was stated as 73 (range 50–99). At least 11 patients reported typical accompanying ipsilateral, autonomic symptoms, such as conjunctival injection, lacrimation or nasal congestion. Brain magnetic resonance imaging (MRI) studies were normal in all patients. Three patients with ECH were examined twice, in and outside of a cluster bout. The other five ECH patients out of bout and the eight CCH patients were tested only once. Three patients (two ECH, one CCH) agreed to repeat the tests during a nitroglycerin-triggered acute CH attack. To minimize the pain for those three patients due to the duration of the tests, only the thermal thresholds and the mechanical detection and pain thresholds were obtained. The tests were started as soon as the three patients reported a subjective sensation of the beginning of the attack. As soon as the tests were performed or the patient finished them because of too much pain, the patients were given sumatriptan (6 mg s.c.) or zolmitriptan (5 mg nasally). Afterwards they rated the attack on a VAS (0–100) as 70, 80 and 90, whereas they rated the general severity of the headache previously as 62, 58 and 52, respectively.
The Institutional Ethics Committee approved the study and all subjects gave their written informed consent.
Quantitative sensory testing
The pain and detection thresholds for thermal and mechanical stimuli associated with the second sensory division of the trigeminal nerve and also due to our previously collected data of a healthy control group were measured by quantitative sensory testing (QST) bilaterally on the cheeks. Furthermore, we performed QST on the back of the hands based on Burstein's observation of spatial and temporal allodynia during a migraine attack. Vibration and pressure pain were only assessed on both hands.
Thermal tests
A Modular Sensory Analyser (MSA, SOMEDIC Sales AB, Stockholm, Sweden) was used to determine the perception and pain thresholds for warm and cold stimuli. The linear heating and cooling of the contact surface of the thermode (1°C/s, 12.5 cm2) was continuously monitored by a feedback circuit (temperature limits 5 and 50°C). The reference temperature was kept constant at 32 ± 0.2°C. The thermode was attached to the dorsum of the hand with a blood pressure cuff with a constant pressure of 20 mmHg (11).
Thermal testing was performed by the method of limits (12–14). Starting from the reference temperature, warm and cold stimuli were applied (Δtemp. 1°C/s). The subjects were asked to press a button as soon as they noticed a change in temperature (perception threshold) or a painful sensation (pain threshold). By repeating this process, five infra- and five suprathreshold values were obtained. After multiplication of these 10 values, the tenth root was taken as the individual threshold (geometric mean).
Mechanical perception and pain thresholds
The mechanical detection threshold was determined by using a set of 12 calibrated von Frey hairs (VFH; Marstock, Marburg, Germany; diameter 0.4–0.65 mm, force 0.25–512 mN). Beginning with a force of 16 mN, which each subject felt, the next lower hair was applied until the subject no longer felt the stimulus. Then the next higher hair was applied until the subject noticed the stimulus. Using the method of limits, the geometrical mean was taken as the individual mechanical detection threshold.
The mechanical pain threshold was tested with a series of seven standardized pinprick probes (diameter 0.2 mm, force 8–512 mN). The lowest force (8 mN) was applied first and was followed by the next higher force until the subject signalled a pricking pain. According to the method of limits, five infra- and five suprathreshold values were obtained and the geometric mean was considered as the individual mechanical pain threshold.
Vibration sensation
A standardized tuning fork (64 Hz, 8/8 scale) was placed on the styloid process of the ulna to measure the patients’ deep sensitivity. The measurement was repeated three times and the subjects were asked to signal when they no longer felt the vibration. The geometric mean of three increasing stimulus intensities was taken as the individual vibration threshold.
Pressure pain
To test deep pain perception, a pressure algometer with a force of 200 N and a contact area of 1 cm2 was placed on the thenar muscle of both hands. The algometer was pressed on the muscle until the subject signalled that in addition to the pressure sensation there was a feeling of pain. Using three continuous ramps of increasing pressure intensities of approximately 50 kPa/s, the geometric mean was established as the pressure threshold.
Wind-up
Perceptual wind-up was tested by an initial single stimulus followed by sets of 10 punctuated stimuli (256 mN) in the test areas (1 cm2). The subject were asked to rate the perceived pain of the first stimulus as well as the following 10 stimuli in relation to the first one on a verbal rating scale (0 = not painful, 100 = maximal pain imaginable). The test was repeated five times. The geometric mean of the results was calculated to describe the relation between the pain intensity of the set of 10 stimuli and the first single stimulus. High values for this measurement indicated that the pain sensitivity of the subject had become stronger after repeated stimulation. In the wind-up test a constant of 0.1 was added to all ratings in order to avoid loss of data in case the first rating was zero (15, 16).
Statistical analysis
A ‘general linear model’ was used to analyse the effect of several repeated measurement factors on the thresholds described above. For the healthy volunteers, there were several factors with two levels: (i) time of day (morning/evening); (ii) test area (back of hands/cheeks); and (iii) side of body (right/left).
According to the analysis for the CH patients, the factors were determined as: (i) time (in bout/out of bout/during a headache attack); (ii) test area (back of hands/cheeks); and (iii) side of body (non-affected side/affected side).
For the three patients who performed the tests in bout as well as out of bout only the data of ‘out of bout’ were taken to obtain a statistical analysis (multivariate
Results
Control group
The results for the thermal and mechanical pain and perception thresholds are presented as means and standard deviations (SD) in Table 1. The homogeneity of the test results proved that QST was a reliable method for gathering qualitative and quantitative results. The univariate test of repeated measures
Means and SD of the thermal thresholds (°C) and mechanical thresholds (mN) and wind-up (N/cm2) of healthy volunteers (n = 10)

Significant difference between hands and cheeks.
TSL, Thermal sensory limen.
Cluster headache patients
Results of QST outside of an acute attack
The multivariate
Multivariate
Means and SD of the thermal thresholds (°C), the mechanical perception and pain thresholds (mN) for the cluster headache patients (n = 16), and also for the wind-up test (N/cm2) (n = 12) and the mechanical thresholds tested on the back of hands (n = 15)

Significant difference between hands and cheeks.
TSL, Thermal sensory limen.
Results of QST between in bout and out of bout
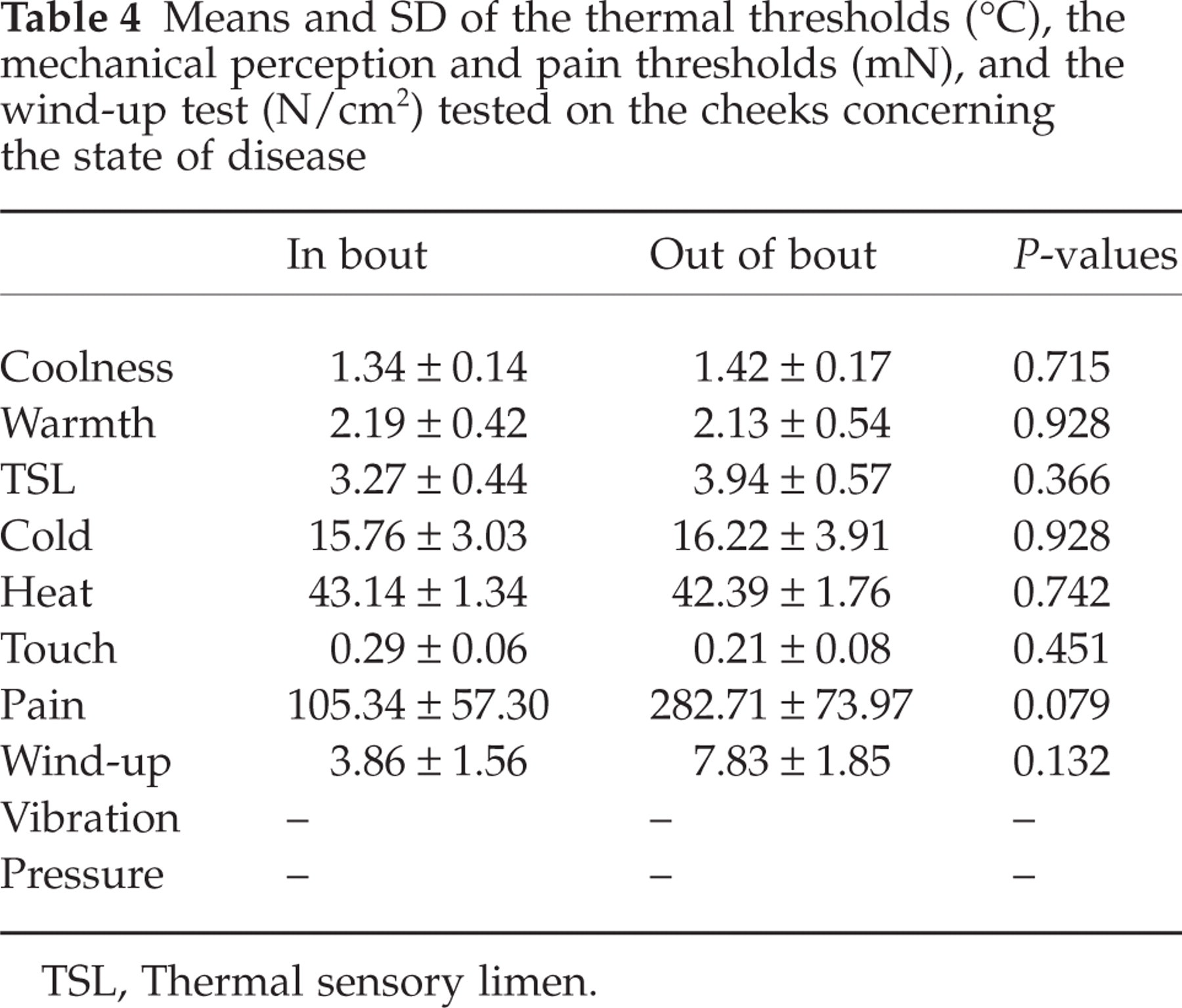
Although there was no significant difference shown in the measured sensory thresholds with regard to the state of the CH, the detailed means and SDs are shown in Tables 3 and 4 for the back of the hands and the cheeks separately. Only in the mechanical pain threshold could a significant difference between in bout and out of bout be seen on the back of the hands (P < 0.005). There was a non-significant trend for patients with CH to be more sensitive out of bout than in bout, which is the case in six out of 10 tests on the back of the hands and in five out of eight tests on the cheeks.
Means and SD of the thermal thresholds (°C), the mechanical perception and pain thresholds (mN), wind-up test (N/cm2) and the mechanical thresholds tested on the back of the hands concerning the state of disease

Significant difference between in and out of bout.
TSL, Thermal sensory limen.
Means and SD of the thermal thresholds (°C), the mechanical perception and pain thresholds (mN), and the wind-up test (N/cm2) tested on the cheeks concerning the state of disease
TSL, Thermal sensory limen.
Results of QST during an acute cluster attack
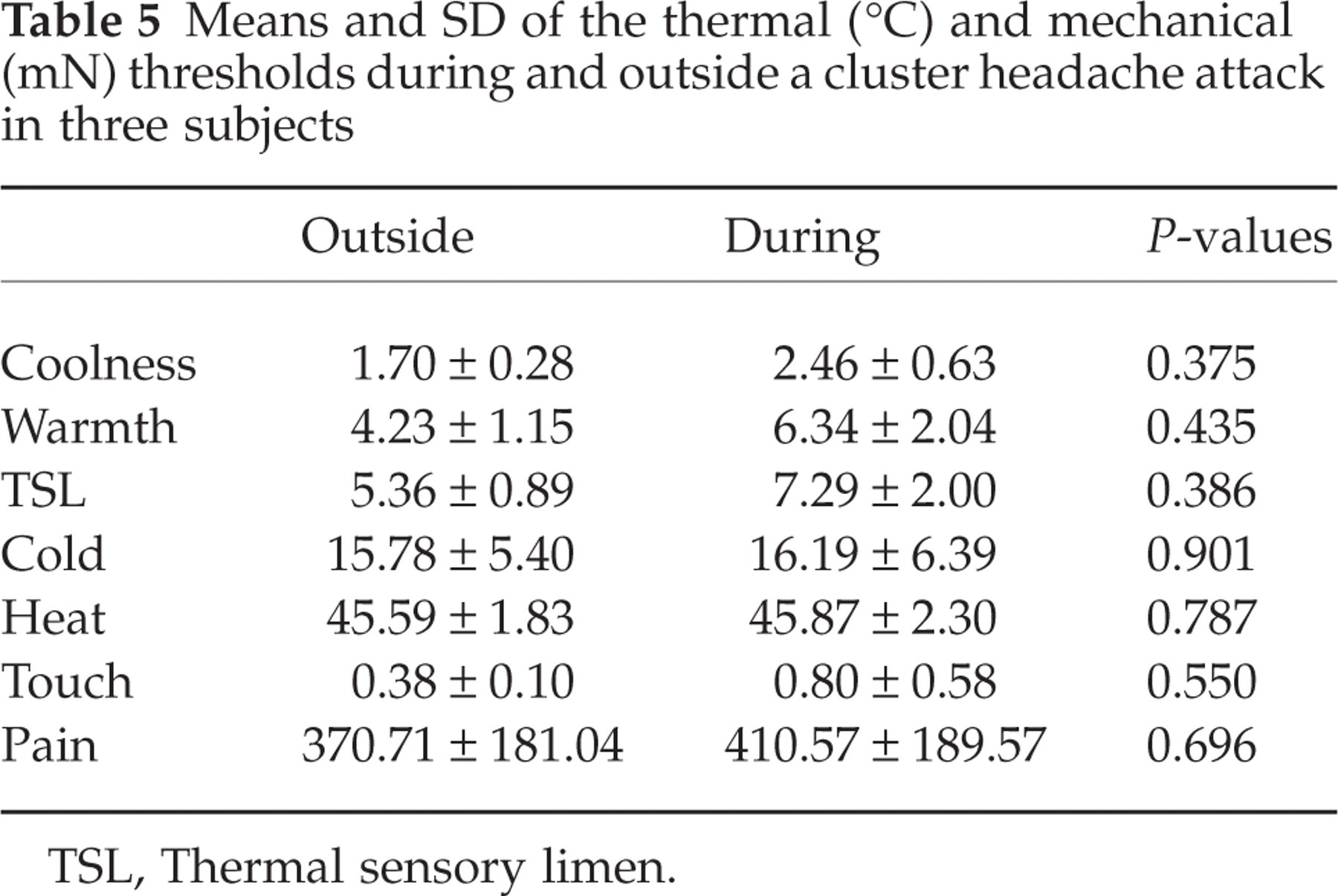
The thresholds for three patients obtained outside and during an attack were compared but no significant difference in the thresholds was found (P < 0.4) (Table 5). There was also no sign of allodynia in these three patients. However, there was a non-significant trend for the patients to be more sensitive outside an attack than during an attack in all tested sensory thresholds except in the cold pain threshold.
Means and SD of the thermal (°C) and mechanical (mN) thresholds during and outside a cluster headache attack in three subjects
TSL, Thermal sensory limen.
Comparison of volunteers/patients
The volunteers’ thresholds were used as reference data for the CH patients. Since there was no significant difference for the patients tested in bout or out of bout, the data of the patients were combined. The multivariate
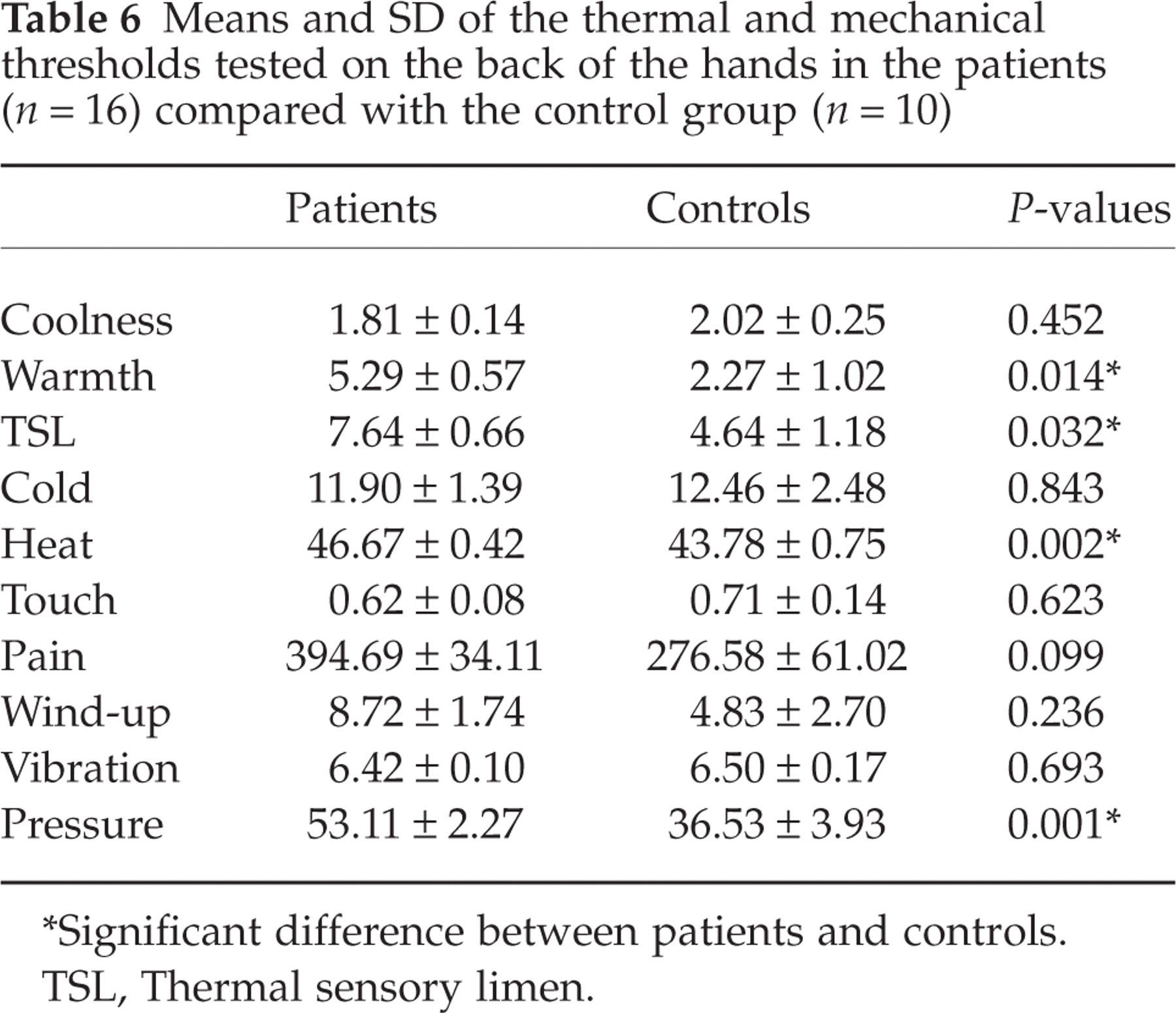
Means and SD of the thermal and mechanical thresholds tested on the back of the hands in the patients (n = 16) compared with the control group (n = 10)
Significant difference between patients and controls.
TSL, Thermal sensory limen.
Means and SD of the thermal and mechanical thresholds tested on the cheeks in the patients (n = 16) compared with the control group (n = 10)

Significant difference between patients and controls.
TSL, Thermal sensory limen.
The thresholds of patients were significantly elevated on the back of the hands in the tests for warmth (P < 0.014), thermal sensory limen (TSL) (P < 0.032), heat (P < 0.002) and pressure (P < 0.001). In the case of the cheeks, the thresholds for warmth (P < 0.047) and for TSL (P < 0.020) were significantly increased in the patients.
All in all, there was a tendency for the CH patients to be less sensitive in perceiving thermal and mechanical stimuli than healthy controls on both test areas, the back of the hands and the cheeks. We found no evidence for cutaneous allodynia either on both test areas or in or out of bout.
Discussion
The present study has demonstrated that patients with CH have bilaterally increased detection thresholds for mechanical and thermal perception and pain stimuli compared with those of a healthy control group. These increased thresholds were not restricted to the trigeminal innervation, but were also found on the back of the hands regardless of whether the patients were examined in or outside of a cluster bout.
Migraine and CH have an overlapping feature in their pathophysiology, namely the involvement of the trigeminovascular system. Otherwise, there are fundamental differences, e.g. the hypothalamus is not involved in migraine. In migraine, several studies have presented QST data of patients examined during acute migraine attacks (8, 9, 17). Approximately two-thirds of the migraineurs tested in these studies developed scalp allodynia with a characteristic spatial spread first to the ipsilateral hand and afterwards to the contralateral side of the face, depending on the duration and severity of the attack. However, migraine patients tested interictally did not differ from controls in their pain thresholds and did not develop cutaneous allodynia (18). There was some evidence of a slight interictal sensitization of the trigeminal structures, since the patients showed signs of increased wind-up. This contradicts our findings in CH. Thus, we believe that it is unlikely that central sensitization plays a dominant role in the pathophysiology of CH. This is another aspect in which CH differs from migraine.
In migraine the presence of allodynia also positively correlates to a longer disease duration (19). Scalp allodynia during a migraine attack is best attributed to a central sensitization of second-order neurons within the caudal trigeminal nucleus (8, 9). This in turn is consistent with findings of a study on migraineurs in between attacks. The authors reported increased temporal summation to painful mechanical stimuli in the referred-pain area mediated by the induction of wind-up in second-order trigeminovascular neurons (18). Cutaneous allodynia has also been described in SUNCT (10), suggesting that the time to onset and duration of central sensitization is associated with the temporal profile of the disorder, leading to the question of whether it can occur in all trigeminovascular-driven headache disorders.
Woolf and Salter have described two forms of plasticity of pain pathways (20). First, ‘activation-dependent plasticity’, in which second-order neurons in the dorsal horn of the spinal cord are activated by peripheral input, expressed as wind-up. In the second form, ‘modulation of nociceptive synaptic transmission’, dorsal horn neurons become sensitized in response to peripheral input.
Peripheral parasympathetic mechanisms probably do not play a pathophysiological role in this process, since modulation of the parasympathetic outflow by applying lidocain to the sphenopalatine ganglion reduced the migraine pain, but did not affect the development of the cutaneous allodynia (21). In contrast, early treatment of migraine headache with triptans seems to prevent the development of allodynia (22). All in all, these findings support the view that a sensitization of peripheral nociceptive fibres is not the cause of the allodynia, but rather a central sensitization of trigeminal second-order neurons in the dorsal horn of the spinal cord according to the ‘activation-dependent plasticity’ described by Woolf and Salter.
One study has reported that brush allodynia showed a weak correlation between the duration and type of disease in four of 10 tested patients diagnosed to have CH (two ECH and two CCH) (23). However, in our study brush allodynia and pressure allodynia differed in their activation of diverse nociceptors. While that study divided the patients qualitatively according to the type of CH, it also observed allodynia in one patient during an acute attack. In contrast, we pooled our data on ECH and CCH quantitatively, since we did not find any significant differences in the thermal and mechanical pain and detection thresholds that were related to the state of the disease. Nevertheless, we consistently found significantly elevated detection and pain thresholds for warm temperature (P < 0.04) as well as for pressure pain (P < 0.001) on the back of the hands and increased thresholds for warmth and TSL (P < 0.05) on the face of 16 CH patients. Since these increased detection thresholds were seen not only in the area with trigeminal innervation but also on the back of the hands, we believe this indicates that a central process is responsible for the observed threshold increase.
How do the bilaterally increased sensory thresholds fit in with the concept of a strictly unilateral headache? First, several reports have shown a side switch from bout to bout and, more rarely, also during a cluster bout. Moreover, Leone has described a patient who was successfully treated with unilateral stimulation of the posterior hypothalamus and who later developed attacks on the contralateral side (7). Second, since the intensity dependence of cortical auditory-evoked potentials, a function of the central raphe-hypothalamic serotonergic system, differs on both sides in CH patients compared with healthy controls, this is further support for the view that there is a bilateral disturbance of the hypothalamic pain control system (24).
In summary, two main conclusions can be drawn from the present study. First, there was no evidence for cutaneous allodynia in CH in or outside of the attack and therefore we exclude peripheral as well as central sensitization in this headache. This feature seems to be a major difference between CH and migraine. Second, our results point to changes in the central pain control system which were most probably due to a bilateral disturbance of the hypothalamic-raphe system. This is also based on the anatomical neighbourhood of the posterior hypothalamus, which belongs to the supraspinal pain control system, and on positron emission tomography imaging studies showing increased activity in the posterior hypothalamus as well as changes in cell density of the pixel-based morphometry on the MRIs (5, 6).
Acknowledgements
We thank Mrs J. Benson for her help in editing the manuscript. The study was partially supported by a grant of the BMBF ‘chronischer Kopfschmerz’, project D1.
