Abstract
Background:
Supervised exercise training alleviates motor symptoms in people with Parkinson’s disease (PD). However, the efficacy of exercise to improve nonmotor symptoms such as cognitive function is less well known.
Objective:
To systematically review evidence on the efficacy of different exercise modes (coordination exercise, resistance exercise, aerobic exercise) on domain-specific cognitive function in patients with PD.
Methods:
Parallel-group randomized controlled trials published before March 2018 were included. Primary outcome measures included global cognitive function and its subdomains, and the Unified Parkinson’s Disease Rating Scale was included as a secondary outcome. Methodological quality was assessed using the Physiotherapy Evidence Database scale.
Results:
The literature search yielded 2,000 articles, of which 11 met inclusion criteria. 508 patients (mean age 68±4 years) were included with a disease severity from 1 to 4 on the Hoehn & Yahr stage scale. Overall study quality was modest (mean 6±2, range 3–8/10). In 5 trials a significant between-group effect size (ES) was identified for tests of specific cognitive domains, including a positive effect of aerobic exercise on memory (ES = 2.42) and executive function (ES = 1.54), and of combined resistance and coordination exercise on global cognitive function (ES = 1.54). Two trials found a significant ES for coordination exercise (ES = 0.84–1.88), which led to improved executive function compared with that of non-exercising control subjects.
Conclusion:
All modes of exercise are associated with improved cognitive function in individuals with PD. Aerobic exercise tended to best improve memory; however, a clear effect of exercise mode was not identified.
INTRODUCTION
Parkinson’s disease (PD) is a highly prevalent age-related condition with an average prevalence of 1601 per 100,000 population in North America, Europe and Australia [1, 2]. PD has traditionally been characterized as a movement disorder, but was recently redefined as a heterogeneous multisystem disorder [3] that includes non-motor symptoms such as hyposomnia [4, 5], mood disturbances [6–8] and, especially, cognitive decline. Up to 57% of patients suffering from PD develop mild cognitive impairment (MCI) within 5 years of their initial diagnosis [9–11], and the majority will eventually develop dementia if they survive more than 10 years [3]. The underlying neurophysiological mechanisms for cognitive decline in PD are not completely understood, but an accumulation of amyloid plaques, mitochondrial dysfunction and neurotransmitter changes are all suggested to contribute [3, 12–14].
As part of efforts to develop effective treatments and strategies to prevent cognitive decline in older adults, exercise has shown promising results. Regular physical exercise promotes angiogenesis, neurogenesis and synaptic plasticity [15–17] and has the potential to protect cognitive function with ageing [18–20]. In PD, different exercise modes (e.g., aerobic training [21–23], resistance training [24–26], forced exercise training [27, 28], dance [29, 30] and balance training [31–33]) have all been shown to improve motor symptoms such as tremor, gait disturbances, postural instability, and bradykinesia. While recently published guidelines do not recommend one specific exercise mode [34], it has been previously suggested that a combination of exercise modes might be best to improve motor function in individuals with PD [35].
Less is known about the effects of exercise on cognitive function in individuals with PD, and the influence of specific modes. Previous reviews suggested that both aerobic and resistance training, or a combination of them, might influence cognitive function positively [21, 36]. However, other reviews have suggested that various forms of physical activity, including aerobic, resistance and coordination exercises such as Qigong and Tai Chi, have negligible effects on cognitive function [37, 38]. These inconsistent findings are likely influenced by the wide range of exercise modes (i.e., aerobic, resistance & balance training) that have been used. Furthermore, there are reports that the exercise-induced benefits on cognitive function might be domain specific (e.g., related to attention, processing speed, executive function, memory, or working memory) [39–41]. For this reason, it is important to consider domain-specific neurocognitive outcomes to gain a better understanding of the effects of exercise on cognitive function in PD [40–42].
This is supported by emerging human and animal evidence that each mode of exercise training has a distinct influence on brain function [43–49]. Coordination training, but not aerobic training, is associated with improved speed of processing [50]; whereas memory is improved with aerobic training and not coordination training in healthy older adults [51]. Improvements in memory were associated with an increased hippocampal volume, while the increase in speed of processing was associated with an increased volume in the basal ganglia (globus pallidus) [50, 51]. These divergent effects on brain function suggest that different exercise modes are likely to influence cognitive function differently, but this has yet to be firmly established in individuals with PD.
Therefore, the purpose of the current study was to compare the effects of different exercise modes on various measures of cognitive function in individuals with PD by systematically reviewing previous randomized controlled trials. The primary aim was to compare the effects of aerobic training, resistance training and coordination training on cognitive function. Furthermore, the secondary aim was to identify combinations of different exercise modes (e.g., aerobic and resistance training) and evaluate their effect on cognitive function. With this approach, this study aimed to determine the most effective forms of exercise to address the specific cognitive impairments associated with PD.
MATERIALS AND METHODS
This systematic review was conducted following the international guidelines established by PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) [52].
Data search
Five electronic databases were searched to identify relevant publications: PubMed, Cochrane Central Register of Controlled Trials, Scopus, LIVIVO and Web of Science. Relevant keywords from three categories – “disease”, “exercise” and “cognition” – were combined with each other, and entered in the different databases. The category
Inclusion criteria according to the PICO (population, intervention, comparator, outcome) [53] format was used to select eligible studies. Only randomized controlled trials (RCTs) were included. Study populations consisted of individuals with idiopathic PD without any restriction placed on the stage of the disease or its severity. Trials targeting secondary or acquired PD were excluded. Exercise programs lasting at least 4 weeks with at least one supervised exercise session per week were considered eligible. Exercise interventions included aerobic training, resistance training, coordination training or a combination of any of these exercise modes. Studies that evaluated the combination of an exercise intervention with other treatments (e.g., drug therapy, education programs) were excluded.
The primary outcome measure was cognitive function assessed by a valid and standardized measurement (e.g., neuropsychological tests) at baseline and after the intervention. Measures of cognitive function were further stratified into different cognitive domains, similar to that used by Smith et al. [40] and Engeroff et al. [41]. The domains were global cognitive function, attention, executive function, speed of processing, memory and “other” (see Table 1). Outcomes related to attention were subdivided into general attention and speed of processing, whereas outcomes for working memory and memory, which are often used interchangeably [54], were combined into a single category.
Domain-specific cognitive tests included in this review
The Unified Parkinson’s Disease Rating Scale (UPDRS) [55] was included as a secondary outcome as it is commonly used to assess the longitudinal course of PD. The UPDRS consists of six different parts that can be assessed on their own or as a summative score.
Study selection
One reviewer conducted the literature search (T.S.) and downloaded the results from all databases into bibliographic software (EndNote X8; Thompson Reuters, New York, USA). After eliminating duplicates, titles and abstracts were screened to identify eligible papers. Full texts of eligible studies were obtained and assessed. Quality of the eligible papers was assessed separately and in duplicate by three reviewers (T.S., R.B. & J.W.) using the Physiotherapy Evidence Database Scale for randomized controlled trials (PEDro) [56]. One reviewer (T.S.) extracted the data from included studies and two reviewers checked the accuracy of extracted data (R.B. & J.W.). Disagreements between reviewers were resolved by consensus or by a third reviewer when necessary.
Data extracted from each included trial consisted of (1) characteristics of trial participants, (2) characteristics of the exercise training intervention(s) (e.g., frequency, duration, mode), (3) methods used to assess primary and secondary outcomes, and (4) assessment of between-group comparative data before (baseline) and after intervention.
Data analysis
Trials were divided according to the different types of exercise interventions and subdivided into aerobic training, resistance training, coordination training or combined exercise modes. When trials contained two (or more) different exercise intervention groups, both were compared to the control group and to each other separately. The included studies used different neuropsychological tests to assess cognitive function (primary outcome). Due to this large variety of tests, a meta-analysis could not be conducted. Corresponding authors were contacted when data were missing or when data were not reported as mean and standard deviation (SD).
Pre-post intervention changes in mean and SD were used to calculate mean differences and between-group effect sizes (ES). Hedges’ bias-correction for small sample sizes was applied and between-group ES and 95% confidence intervals (CI) were calculated as follows: between-group ES = (
RESULTS
A total of 2,000 citations were retrieved through the search strategy. After duplicates were excluded (
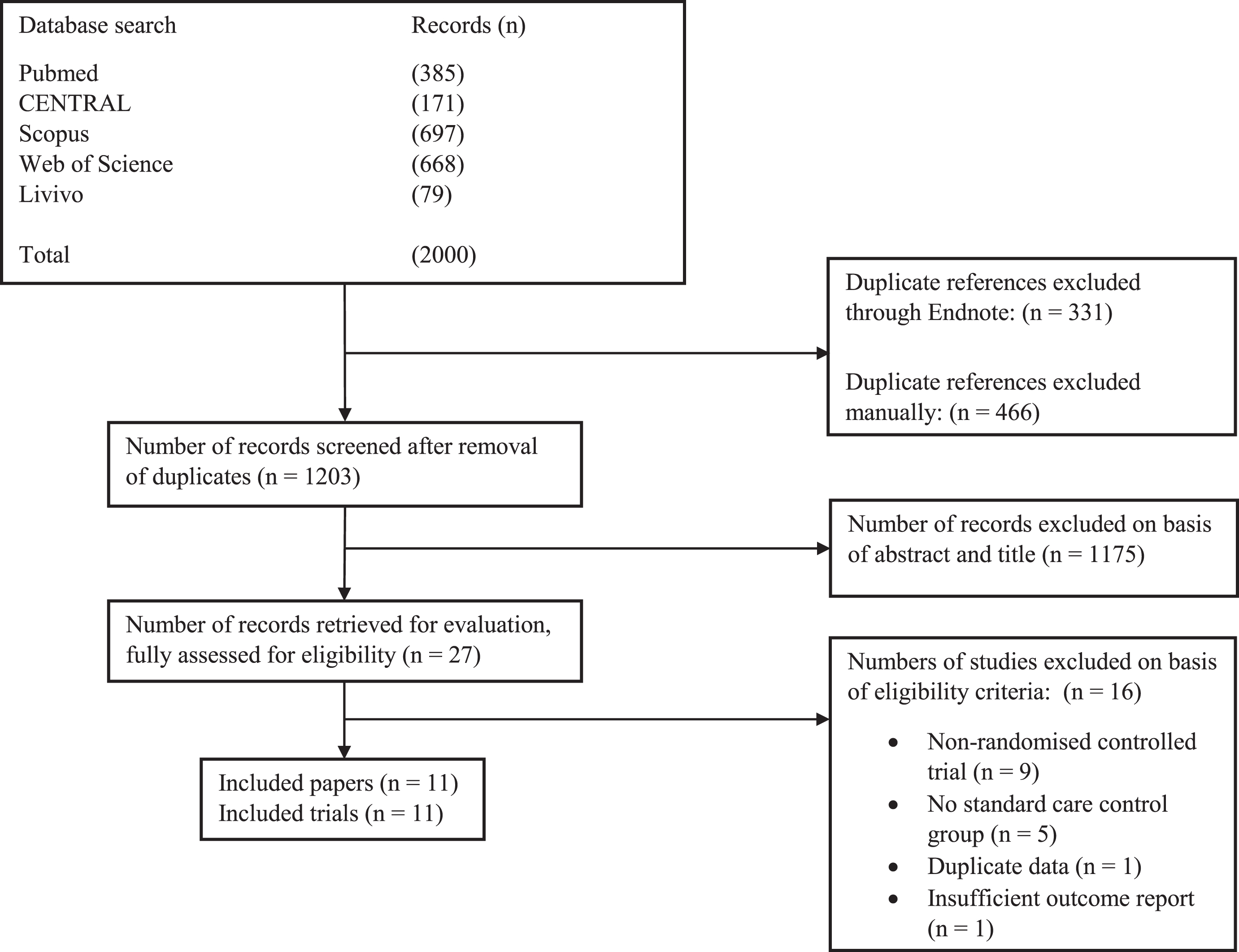
Study flow diagram (CENTRAL = Cochrane Register of Controlled Trials).
Results: Quality assessment
The included studies had a modest overall quality based on the average PEDro score (mean±SD 6±2, range 3–8/10, Table 2). The participants in all included studies were randomly allocated to their respective group and baseline characteristics only differed significantly in one study, where the control group had more years of education [85]. Eligibility criteria was reported in seven of the eleven trials [77, 82–85], but none of the included studies reported blinding of the exercise intervention therapists or participants. However, this is somewhat common and difficult to avoid in trials that evaluate the effects of an exercise intervention. Apart from two studies [83, 85], all trials used blinded assessors, but only three studies presented sufficient information about concealed allocation [77, 82]. Four studies had a greater drop-out rate than 15%, which might have had an effect on the outcomes of these studies [76, 85]. Most of the studies reported point measures and measures of variability (91%) as well as between group statistics (100%), but only three studies specifically stated that all participants received the originally allocated treatment (intention-to-treat analysis) [82, 86].
Quality assessment of the included studies (PEDro scale)
aItem does not contribute to total score; # = scores are available at PEDro database (https://www.pedro.org.au/) [54].
Study characteristics
The study and patient characteristics are presented in Table 3. In total 904 participants were screened for eligibility and 508 participants were included in the different trials. Idiopathic PD was an inclusion criterion for all trials, and all but one trial [85] excluded participants with early signs of cognitive impairment. Most of the studies used a specific cut-off score of the Mini Mental State Examination to detect cognitive impairment (cut-off scores differed between 23 and 26). Most of the studies defined any other major comorbidities (such as cardiac diseases, other neurological disorders) as an exclusion criterion. Further, the studies that did not specifically exclude participants with depression or apathy, did not include participants with severe symptoms of these disorders according to the studies’ different baseline scores [82, 85]. A mean age of 68±4 years was reported across the studies and the included participants’ PD severity ranged from 1 to 4 on the Hoehn & Yahr stage scale [87].
Study characteristics (Values are mean±SD; NR = not reported; AE = aerobic exercise; RE = resistance exercise; CE = coordination exercise; CG = control group; MoCA = Montreal Cognitive Assessment; MMSE = Mini-Mental State Examination; HY = Hoehn & Yahr)
Different exercise interventions were used by the included studies and a summary of the characteristics of the interventions is presented in Table 4. The intervention periods differed between four [82] and twenty-six [78] weeks, with an exercise frequency between once [80] and three times [76, 85] a week. Five studies analyzed the effects of aerobic exercises [76, 85], one study the effects of resistance exercises [84], and five further studies the effects of coordination exercises [76–78, 86]. In addition, three studies investigated the effects of combined resistance and coordination exercises [79, 84]. In three of the included trials the control group met regularly to control for the effect of social interaction [79, 84] and in one further study the control group was provided with information about the benefits of exercise [83]. In the other trials, the control group consisted of standard care and did not receive additional treatment [76–78, 86].
Characteristics of the exercise interventions (AE = aerobic exercise; RE = resistance exercise; CE = coordination exercise; CG = control group; NR = not reported; NA = not applicable; HR = heart rate)
Outcome measures
Executive function was the most commonly assessed (
The neuropsychological tests most often used were the Montreal Cognitive Assessment score [82–84], the Trail Making Test A + B [81, 85], the digit span forward and backwards [76, 85], and the Stroop colors task [76, 85].
Primary outcome: Global cognitive function
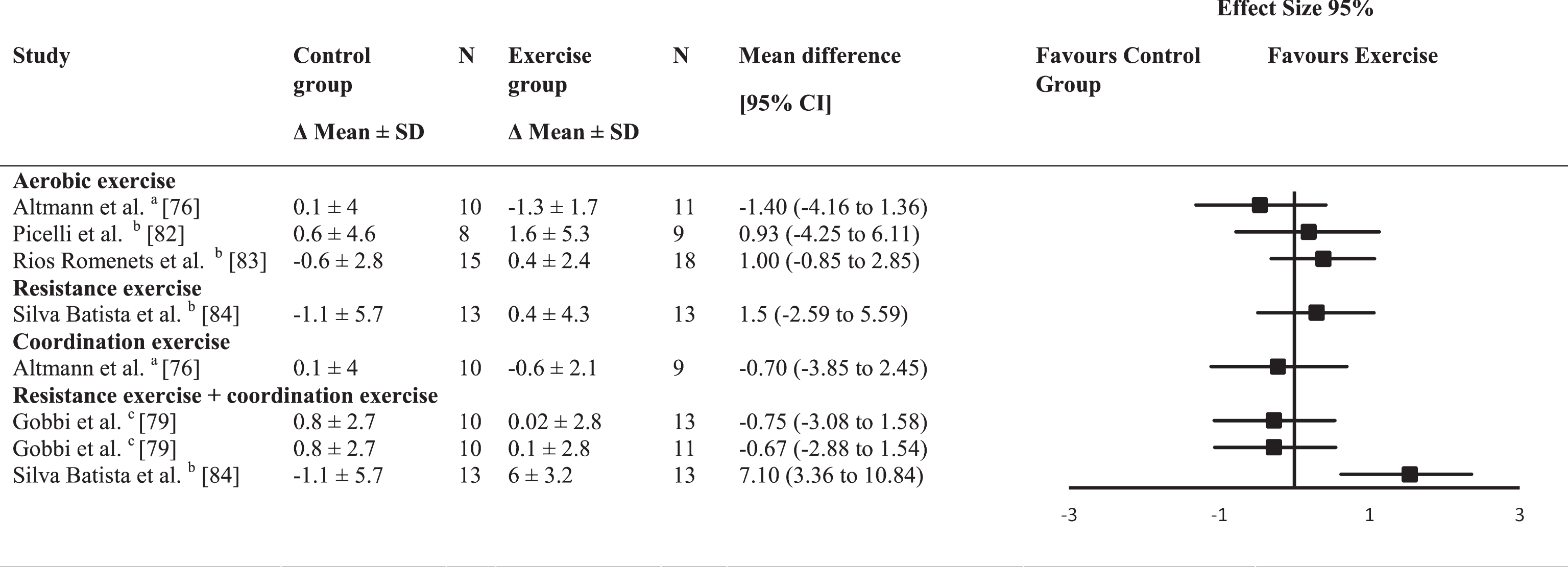
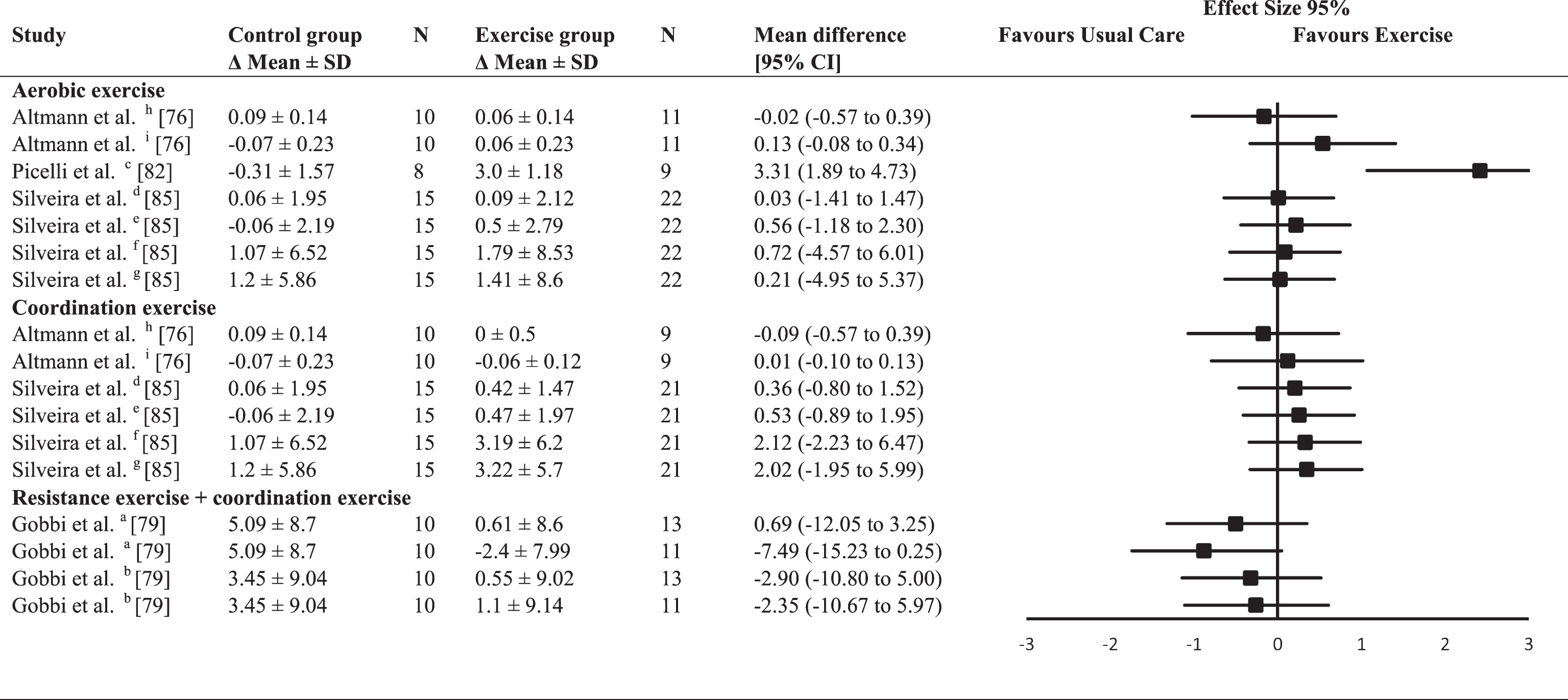
Of the five studies assessing global cognitive function (Table 5) two performed an aerobic exercise intervention [82, 83] and one other study included an aerobic exercise intervention as well as a coordination exercise intervention [76]. One intervention consisted of resistance exercise and a combination of resistance exercise and coordination exercise intervention [84]. The fifth study analyzed the effects of a combined resistance and coordination exercise intervention [79]. The between-group ES were trivial for aerobic exercises (0.04; range –0.46–0.39) and small for resistance exercises (ES = 0.30) or the combined resistance and coordination exercises (0.33; range –0.27–1.54. The average between-group ES for coordination exercises was small and negative (ES = –0.22). Only one of the estimated ES reached the level of significance and was in favor of combined resistance and coordination exercises in comparison to the control group (ES = 1.54;
Effect size calculations for global cognitive function (forest plot detailing between-group effect size and 95% confidence intervals for each outcome in each trial)
a =Mattis Dementia Rating Scale; b = Montreal Cognitive Assessment (MoCA); c = Mini-Mental State Examination (MMSE).
Primary outcome: Executive function
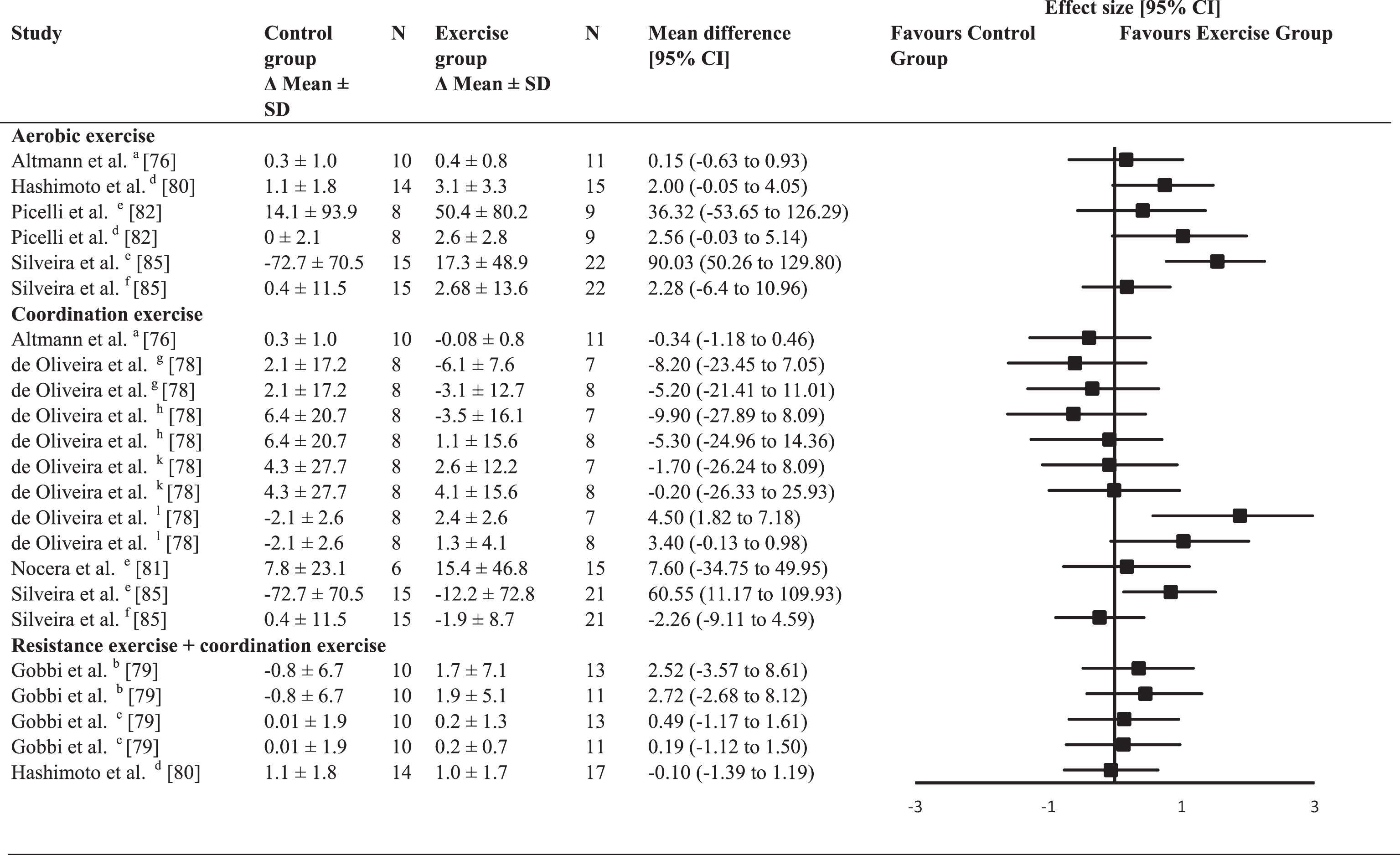
A broad variety of neuropsychological tests were used to assess executive function, which contributes to the large range of mean differences (Table 6). The average between-group ES for aerobic exercise interventions was moderate (mean ES = 0.68; range 0.17–1.54), whereas the average between-group ES for coordination exercise interventions (mean ES = 0.13; range –0.62–1.88) and for combined resistance and coordination exercise interventions was trivial (ES = 0.13; range –0.06–0.46). Significant improvements in executive function (
Effect size calculations for executive function (forest plot detailing between-group effect size and 95% confidence intervals for each outcome in each trial)
a =composite z scores; b = Wisconsin Card Sorting Test (WCST) - perseverative errors; c = WCTS categories completed; d = Frontal Assessment Battery (FAB); e = Trail Making Test (TMT) B; f = Stroop Word & Colour Interference; g = WCST – number of trials; h = WCST – correct answers; k = WCST - number of errors; l = Raven colored matrix.
Primary outcome: Attention
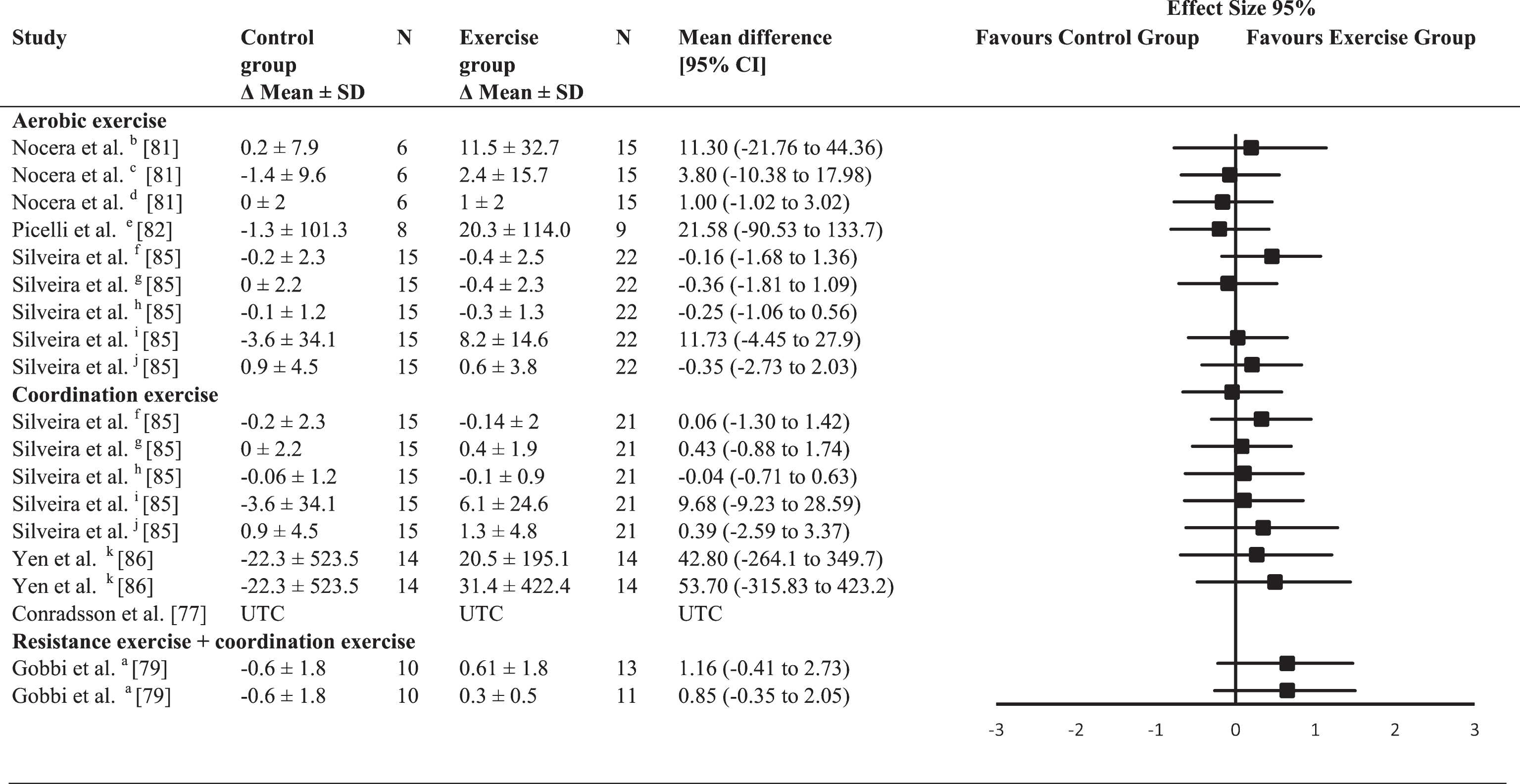
No significant ES were found for attention (Table 7). The average between-group ES were trivial for aerobic exercise interventions (mean ES = 0.02; range –0.2–0.46), small for coordination exercise interventions (mean ES = 0.20; range –0.04–0.35), and moderate for combined resistance and coordination exercise interventions (mean ES = 0.65).
Effect size calculations for attention (forest plot detailing between-group effect size and 95% confidence intervals for each outcome in each trial)
a = Wisconsin Card Sorting Test (WCST) – failure to maintain set; b = Trail Making Test (TMT) A; c = letter fluency; d = digit backward; e = TMTA; f = digit forward; g = digit backward; h = Corsi block test; i = TMT A; j = letter fluency; k = digit backward; UTC = unable to calculate.
Primary outcome: Speed of processing
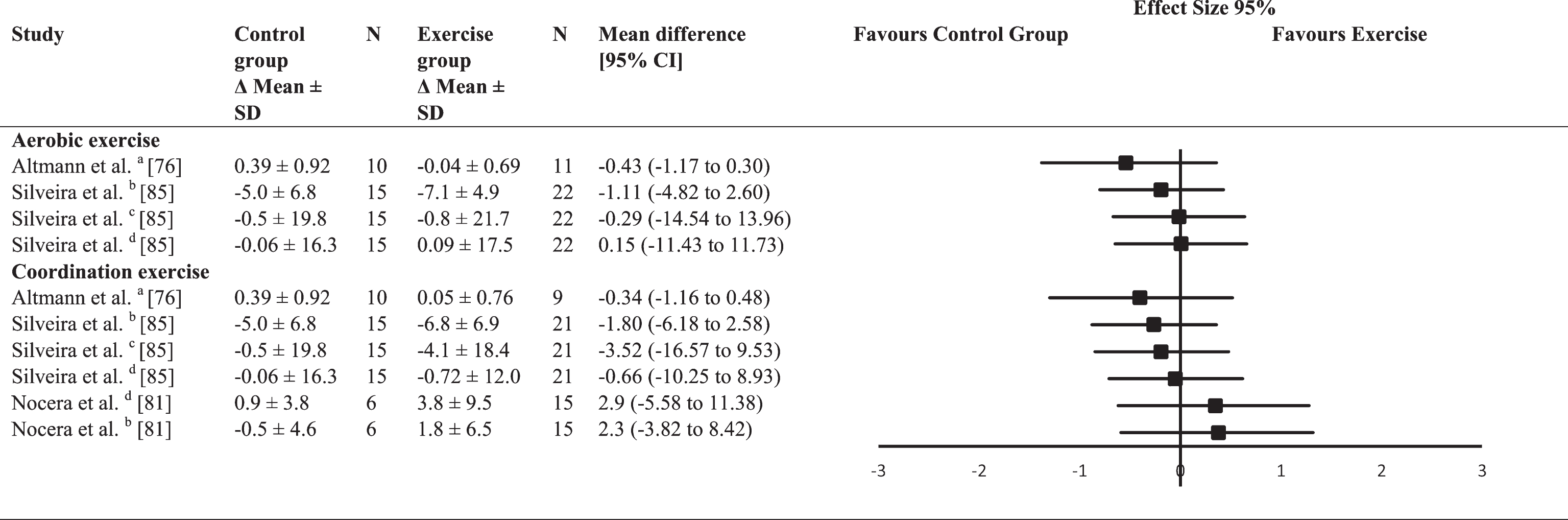
None of the included studies had a significant between-group ES for speed of processing (Table 8). The two studies [76, 85] that included aerobic exercise intervention (ES = –0.18; range –0.54–0.01) as well as the three studies with a coordination exercise intervention (ES = –0.03; range –0.40–0.38) had a trivial average between-group ES.
Effect size calculations for speed of processing (forest plot detailing between-group effect size and 95% confidence intervals for each outcome in each trial)
a = composite z scores; b = category fluency; c = Stroop Word; d = Stroop Color.
Primary outcome: Memory
Four studies used different neuropsychological tests to assess memory (Table 9). Two of them investigated the effects of aerobic and a coordination exercise interventions [85], whereas the other two investigated the effects of aerobic exercise [82], and combined resistance and coordination exercise [79]. Average between-group ES was trivial for coordination exercise interventions (ES = 0.18; range –0.17–0.35) and small for both aerobic exercise interventions (ES = 0.45; 0.01–2.42) and combined resistance and coordination exercises (ES = –0.49; range –0.88––0.26). One trial demonstrated a significant ES in favor of the aerobic exercise intervention (ES = 2.42;
Effect size calculations for memory (forest plot detailing between-group effect size and 95% confidence intervals for each outcome in each trial)
a =Wechsler Memory Scale I; b = Wechsler Memory Scale II; c = memory with interference test; d = California Verbal Learning Test (CVLT) short; e = CVLT long; f = Rey-Osterrieth Complex Figure Test (Rey-O) - short; g = Rey-O –long; h = 1-back task; i = 2-back task.
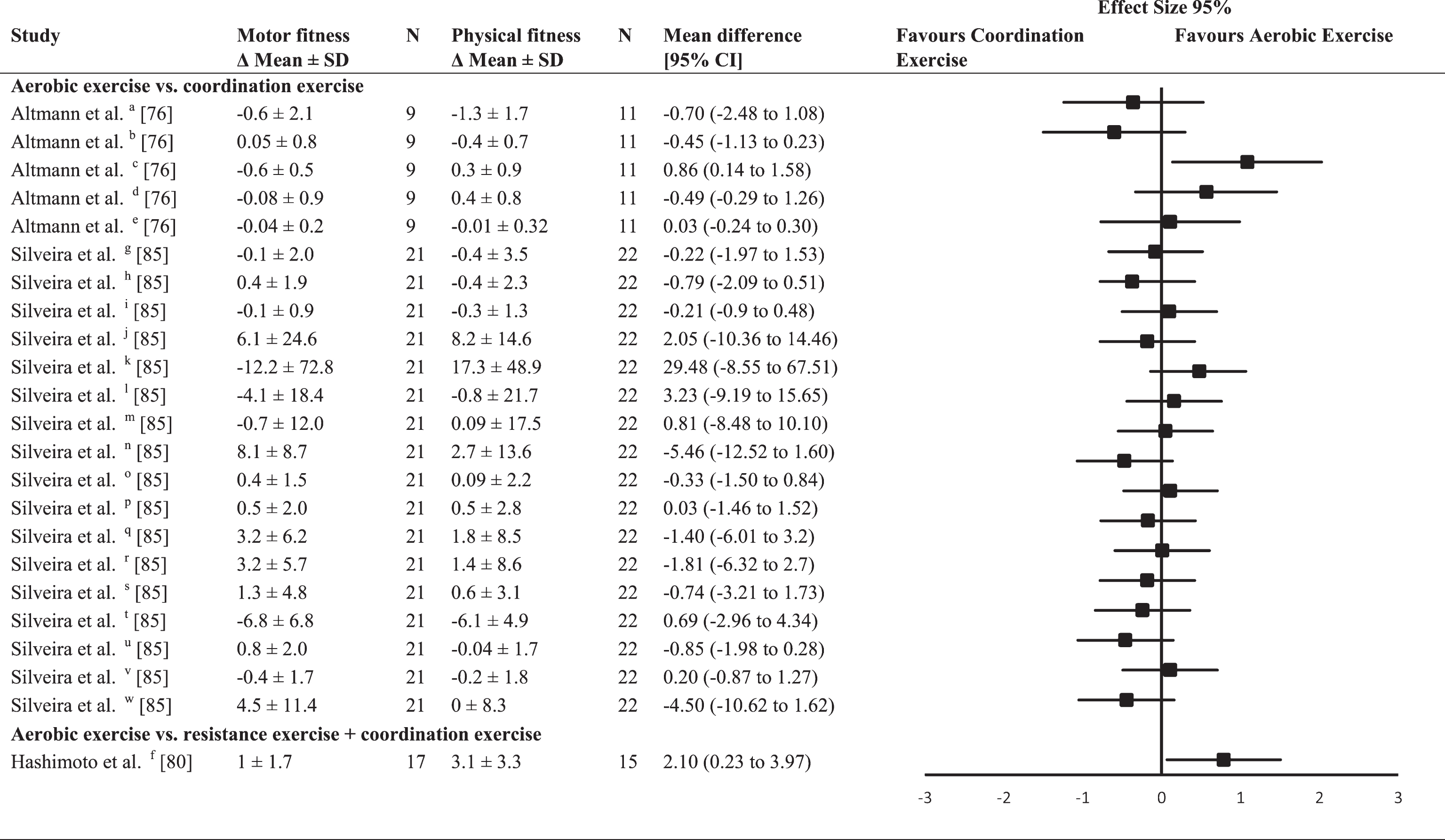
Aerobic versus coordination exercise
Besides comparing the outcomes to a control group, one study compared an aerobic exercise intervention with a combined resistance and coordination exercise intervention (ES = 0.79) [80] and two other studies compared the effects of an aerobic exercise intervention with a coordination exercise program [85]. Table 10 shows the between-group ES for these studies across several cognitive domains. The average between-group ES for the comparison of aerobic exercise and coordination exercise interventions was trivial (ES = –0.04; range –0.6–1.09). However, two trials revealed a significant ES, and both favored aerobic exercise with a large ES (1.09) for memory [76] and a moderate ES (0.79) for executive function [80], in each study respectively.
Effect size calculations between different exercise modes (forest plot detailing between-group effect size and 95% confidence intervals for each outcome in each trial
a = global cognitive function – Mattis Dementia Rating Scale; b = composite z scores - speed of processing; c = composite z scores – memory; d = composite z scores – executive function; e = composite z scores – language; f = executive function (Frontal Assessment Battery); g = attention (digit span forward); h = attention (digit span backward); i = attention (Corsi block test); j = attention (Trail Making Test A); k = executive function (Trail Making Test B); l = speed of processing (Stroop Color); m = speed of processing (Stroop Word); n = executive function (Stroop Color Word interference); o = memory (California Verbal Learning Test (CVLT) short); p = memory (CVLT long); q = memory (Rey-Osterrieth Complex Figure Test (Rey-O) – short); r = memory (Rey-O long); s = speed of processing (phonemic fluency); t = attention (verbal fluency); u = language (Boston Naming Test); v = visuospatial function (Intersecting Pentagons); w = visuospatial function (Benton Line Orientation Test).
Secondary outcome: Unified Parkinson’s Disease Rating Scale
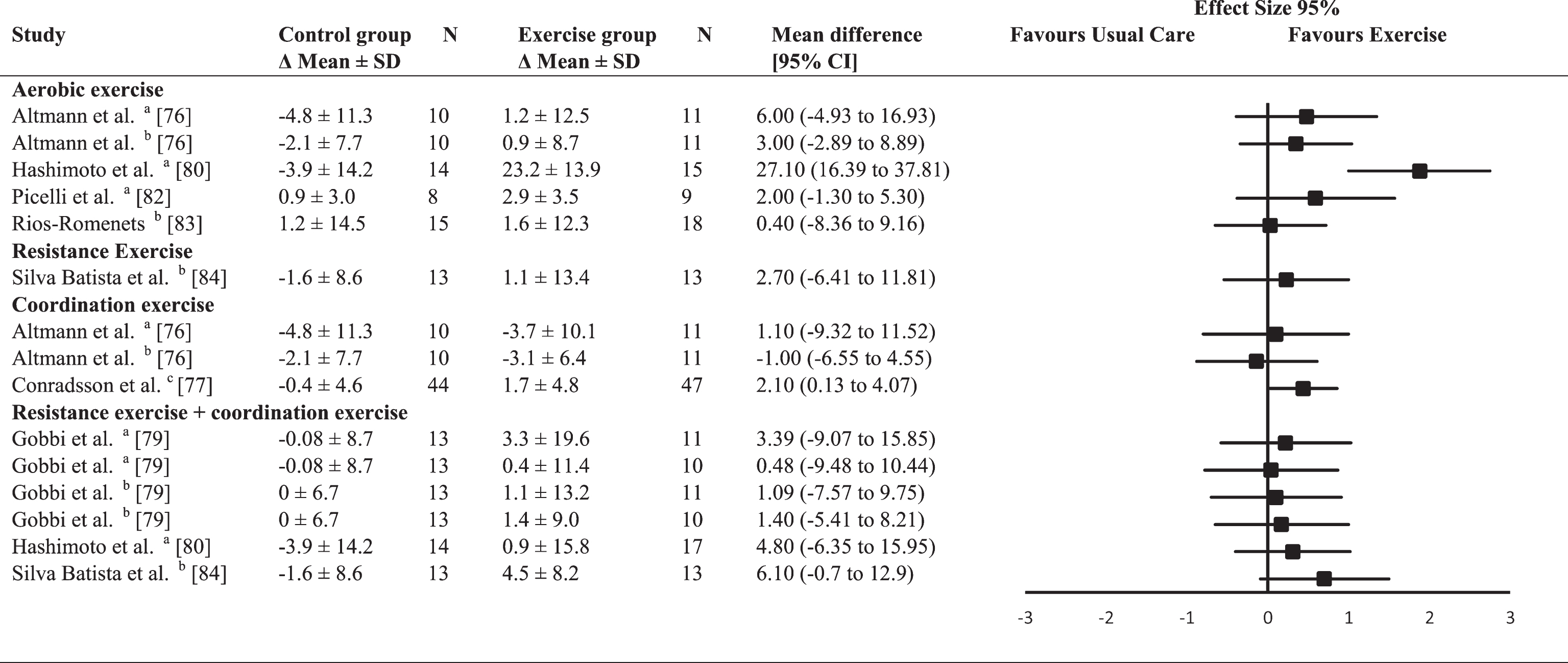
Seven of the included studies used the UPDRS to evaluate the overall health status of their participants [76, 82–84] (Table 11). Two of these studies reported the total score of the UPDRS as well as the subscore of the motor evaluation [76, 79]. Furthermore, two studies reported either the total scores [80, 82] or the scores of the motor evaluation [83, 84] and one of the included studies reported the subcategory activities of daily living [77] (Table 11). Besides one negative between-group ES in the coordination exercise interventions (ES = –0.014), all other ES tended to be in favor of the exercise interventions. The average between-group ES was trivial for coordination exercise interventions (ES = 0.13; –0.14–0.44), small for combined resistance and coordination exercise interventions (ES = 0.26; range 0.1–0.7), and moderate for aerobic exercise interventions (ES = 0.67; range 0.03–1.88). Two of the trials demonstrated significant improvements in the UPDRS with aerobic exercise (
Effect size calculations for the Unified Parkinson’s disease rating scale (forest plot detailing between-group effect size and 95% confidence intervals for each outcome in each trial)
a = Unified Parkinson’s Disease Rating Scale (UPDRS) – total score; b = UPDRS – motor score; c = UPDRS – activities of daily living.
DISCUSSION
This review included eleven RCTs that analyzed the effect of different exercise modes (aerobic exercise, resistance exercise, coordination exercise, and combined resistance and coordination exercise) on at least one cognitive domain in individuals suffering from PD. Executive function, attention and memory tended to improve after each form of exercise in comparison to these cognitive functions in the non-exercise control group. However, we did not observe this trend for speed of processing. Global cognitive function tended to improve after aerobic, resistance, and combined resistance and coordination exercises, but not after coordination exercises. The average between-mode ES ranged from small to moderate, but often lacked statistical significance, and therefore this review was unable to identify a clear effect of exercise mode on the cognitive domains. However, in five trials a significant between-group ES in favor of the exercise group over the non-exercising control group was identified for tests of specific cognitive domains. These significant between-group ES included a positive effect of aerobic exercise on memory and executive function, and of combined resistance and coordination exercise on global cognitive function. Two trials found a significant ES for coordination exercise, which led to improved executive function compared with that of non-exercising control subjects.
Global cognitive function
We identified one significant between-group ES, which favored combined resistance and coordination exercises over the non-exercise control group [84]. The other exercise modes did not lead to any significant changes, suggesting that combined resistance and coordination exercises could be best to improve global cognitive function in individuals with PD. However, another study that investigated the effect of combined resistance and coordination exercises did not replicate the above findings. These conflicting results may be due to methodological differences or differences in participant characteristics [79]. The MoCA was most commonly used (3 of 5 studies [82–84]) to assess global cognitive function, which is consistent with current recommendations for PD [88]. This instrument is possibly more sensitive for detecting early cognitive decline in individuals with PD than are the other instruments used in the studies (MMSE, Mattis-Dementia Rating Scale (MDR)) [89–92]. This sensitivity difference could explain the conflicting outcomes of these two studies, because the MoCA scores changed significantly, but not the MMSE or MDR scores.
The only significant between-group ES was found for the intervention group with the lowest reported baseline MoCA scores (mean score 20.8) [84]. Participants with a MoCA score of 20.8 probably had MCI: MCI produces MoCA scores from 19 to 25, and scores from 19 to 21 can indicate late-stage MCI [90, 93]. Accordingly, multimodal exercise training may only increase global cognitive function in individuals with both PD and MCI, which concurs with recent recommendations of the American Academy of Neurology for the treatment of MCI [94]. Future research should focus on assessments sensitive to early and subtle changes in global cognitive function. Based on the findings of our review, combined resistance and coordination exercises might be best to improve global cognitive function. However, given the aforementioned differences in participant characteristics, these findings require confirmation.
Executive function
The transition from normal cognition to MCI is often characterized by impaired executive function in individuals with PD [95–98]. Exercise has a stronger effect on executive function than on any other cognitive domain in healthy older adults [49, 99]. However, we identified only three instances of a significant ES, where either aerobic or coordination exercises were superior to control [78, 85]. A recent study in healthy older adults suggests that exercise mode does not specifically affect executive function [20]. In contrast, another study that compared aerobic exercise to combined resistance and coordination exercises found a significant effect for aerobic exercise [80].
According to previous studies, the apparent effect of exercise can vary with different neuropsychological tests and may relate to selective deficits in executive function (e.g., control of interference, cognitive flexibility, and planning). This factor may partially explain the results of our review [41, 100]: only the TMT B, which assesses cognitive flexibility, and the ‘Raven colored matrix’ task, which uses a problem-solving task associated with visuospatial dimensions, revealed significant changes [101, 102]. In the same studies, we observed no changes in other neuropsychological tests that assess planning and control of interference (e.g., Wisconsin card sorting task, Stroop test) [78, 85]. Hence, exercise may improve specific deficits in individuals with PD.
The TMT B was used by Silveira et al. [85] and by two other studies that investigated the efficacy of either aerobic or coordination exercises. These two studies observed a positive tendency for exercise [81, 82]. The lack of significance might be explained by the smaller sample sizes and the participant characteristics in these studies. Silveira et al. included participants with MCI; their sub-analysis revealed that the performance of only the cognitively impaired participants changed significantly after the exercise intervention [85]. These combined findings suggest exercise is effective in improving executive function in individuals with PD who experience MCI, which agrees with our findings for global cognitive function. Future studies should concentrate on selective deficits in executive function and use larger sample sizes in order to provide further insight into the efficacy of different exercise modes.
Attention and speed of processing
We did not identify significant effects of any exercise mode on attention or speed of processing, although many of the neuropsychological tests used in the review studies meet recent recommendations of how to assess these functions in individuals with PD [97, 104]. Our results contrast with findings of a recent systematic review in healthy older adults and in individuals with MCI and with dementia. The review identified attention and speed of processing among the cognitive domains most consistently improved by exercise [20]. However, the authors stated that at least 52 sessions of about 60 minutes may be needed to improve cognitive performance [20]. No exercise intervention in our review included this many sessions, which may explain why we found no significant effects. Additionally, simple attention tasks might not be complex enough to detect changes in cognitive performance in small sample sizes [41, 99]. Ideally, future research should use longer intervention periods with more exercise sessions and batteries of complex cognitive tests in order to detect small, but important, changes in these functions.
Memory
Individuals with PD often suffer from deficits in short-term and long-term memory, which can be present before the onset of other cognitive complaints [103]. However, we identified only four studies that assessed the effect of an exercise intervention on memory. Of these, one found a significant between-group ES favoring the aerobic exercise intervention over the control group [82]. This study used the auditory consonant trigram test, which is a short-term memory retention task [104, 105]. The findings of this study agree with those of previous research in healthy older adults and individuals with MCI that showed aerobic exercise was associated with improved memory [51, 106]. Another of the studies compared aerobic exercise and coordination exercise regimens, finding the aerobic mode superior in improving memory [76]. Therefore, we speculate that aerobic exercise has a superior effect on improving memory in individuals with PD, because no other exercise mode (coordination exercise, combined coordination and resistance exercise) led to significant changes. This would support previous findings that proposed the superior role of aerobic exercise for slowing disease progression in PD [107].
Another study in our review used aerobic exercise training but found no significant improvement in memory [85]. In the studies in which aerobic exercise did improve memory, participants exercised on a treadmill, whereas the participants in this study exercised on stationary bikes. Together, these findings suggest that treadmill training could be superior to cycling on a stationary bike for improving memory. The unique associations between memory and distinct aspects of postural control and gait in individuals with PD could explain the differences between cycling on a stationary bike and walking on a treadmill [108]. Treadmill training improves postural instability and gait disturbances, so this form of exercise might address the clinical signs of parkinsonism better than cycling [108–110]. Therefore, future research should be encouraged to directly compare these exercise modalities. As memory is likely to worsen in individuals with PD, and an impaired memory decreases quality of life, validated treatment options are urgently required [111].
Secondary outcomes: Unified Parkinson’s Disease Rating Scale (UPDRS)
The UPDRS instrument is commonly used to assess individuals with PD. Using the UPDRS results as an outcome, our finding that in general exercise tended to improve symptoms of PD agrees with that of previous reviews [30, 112]. We did not find a significant effect of mode: the two studies with significant improvements after the intervention (cf. non-exercise control group) employed either aerobic or coordination exercises [77, 80]. Overall, our findings suggest that clinicians and researchers should be encouraged to prescribe exercise in individuals with PD; however further evidence is required to confirm the optimal form of exercise.
Insights into possible physiological mechanisms
We identified that various exercise modes have the potential to improve both global cognitive function and executive function in individuals with PD, but that aerobic exercise may be best for improving memory. In addition, one study reported that aerobic exercise was superior to combined resistance and coordination exercises in improving executive function. These findings indicate that a particular exercise mode could be more effective than other modes in improving certain cognitive domains. This difference in effect could result from different mechanisms within the brain that are triggered by specific exercise modes.
In healthy older adults, aerobic and resistance exercise affect the brain in different ways. Aerobic exercise improves hippocampal volume, modulated by a higher expression of brain-derived neurotrophic factor (BDNF) [49, 114], whereas resistance exercise more likely produces higher concentrations of insulin-like growth factor 1 (IGF-1), with a smaller, or no, increase in BDNF [114, 115]. Both exercise modes increase synaptic plasticity within the hippocampus after training, which is associated with an improved memory [46, 51]. However, one study found that aerobic exercise training activated glutamatergic signaling proteins (e.g., N-methyl-D-aspartate and postsynaptic density protein) while resistance exercise training increased the expression of protein kinase C alpha, tumor necrosis factor alpha, and interleukin 1 beta [46]. Because altered glutamatergic neurotransmission and neuronal metabolic dysfunction are part of the pathophysiology in PD, we speculate that an activation of glutamatergic signaling proteins might have a greater effect in individuals with PD [116, 117]. The potential of glutamatergic signaling proteins in improving cognitive function, especially memory, has been discussed in previous research with humans and animals and may explain why aerobic exercise better improves memory, as we identified in the current review [117–120]. However, this remains speculative and needs further proof.
In addition, animal studies have revealed that aerobic exercise produces neuronal and mitochondrial protection and increases nigrostriatal neurotrophic factor levels in parkinsonian mice [121]. Mitochondrial dysfunction is seen in both PD and Alzheimer’s disease, further supporting the role of aerobic exercise in improving cognition, especially memory, in individuals with PD [122]. However, more research is needed to completely understand the physiological mechanisms behind the effects of aerobic exercise in individuals with PD.
Besides aerobic and resistance exercises, coordination exercises are suggested to increase the volume of the hippocampus and the basal ganglia (globus pallidus) in healthy older adults [45, 123], possibly via the ‘vestibular pathway’. Coordination (e.g., balance) exercises often engage the vestibular system, which not only contributes to spatial cognition but is also anatomically connected to the medial-temporal lobe (e.g., hippocampus) and to parieto-temporal cortical networks [47, 124]. This vestibular pathway may also trigger the striatum [47], which is part of the basal ganglia – the brain area affected by PD. This pathway could explain the potential of coordination exercises to improve cognitive performance and general disease severity in individuals with PD, which we found in our review [78, 85].
Evidence exists that different physiological responses to specific exercise modes accompany increased cognitive performance. However, the role of each pathway in individuals with PD is not fully understood. Assessing physiological mechanisms and cognitive function to identify the pathways that are responsible for behavioral changes is recommended to provide further insight into the efficacy of different exercise modes. Such information would help to identify efficient treatment strategies.
Social interaction
Besides changes in physiological mechanisms, another benefit of group exercises is environmental enrichment, particularly social enrichment, which might also prevent cognitive decline [125]. However, the results of our review do not support this notion. Three of the included RCTs compared the effects of group exercises to a control group, which met regularly to control for the effect of social interactions [79, 84]. Whereas significant results were found for executive function [82] and global cognitive function [84] following the exercise interventions in two studies, the control groups did not improve in any of the studies. Furthermore, one study investigated the effect of a group or an individual exercise class and only the participants of the individual exercise class improved their cognitive performance significantly [78], suggesting that exercise-induced changes are more important for improving cognitive performance than any effects of social interaction. Nevertheless, we should not underestimate the power of social interactions during exercise classes as part of a holistic treatment of PD because of the potential benefits for quality of life and mood [126–128].
Strength and limitations
To prescribe exercise interventions for maintaining or improving cognitive function, we need a better understanding of the specific effects of different exercise modes in individuals with PD. Therefore, our review systematically analyzed the effect of aerobic, resistance and coordination exercises on domain-specific cognitive function, which provides further insight into previous findings [24, 36–38]. Additionally, we strictly followed the PRISMA guidelines, only included RCTs, and calculated between-group ES using the mean difference between pre-tests and post-tests to address limitations of previous studies [36–38].
The large number of neuropsychological tests used in the studies, coupled with differences in outcome parameters and the small number of trials within each cognitive domain, prevented us undertaking a meta-analysis. We excluded conference abstracts and other non-peer-reviewed publications from the current review, although we cannot exclude the possibility of publication bias completely. Average between-group ES does not take the grade of evidence for each outcome or differences in study sample sizes into account, as does the overall (weighted) ES of a meta-analysis.
Further, the included studies used a wide range of exercises, which made it difficult to categorize and compare them. For example, we cannot exclude the possibility that different forms of exercises such as treadmill training or stationary bike training lead to different adaptations, although both are considered aerobic exercise. Nevertheless, given the current lack of RCTs, analyzing the efficacy of different exercise modes is an important first step before comparing different training forms within a mode. Also, some studies did not sufficiently report how they monitored exercise intensity during the exercise classes, particularly during coordination exercises [76, 81–86]. To provide further insight into the role of intensity, future study designs should include measuring exercise intensities in convenient ways (e.g., heart rate monitors, subjective rating of perceived exertion). Besides the range of exercises and the lack of monitoring intensity, the intervention periods in the included studies differed between 4 to 26 weeks. Further, average sessions per week as well as length of the exercise classes ranged between once to three times a week and 30 to 60 minutes per session, which marks a high heterogeneity of the different study designs. As a previous review discussed a dose-response effect of exercise in healthy older adults and recommended a minimum of 52 sessions lasting 60 minutes to improve cognition, it is possible that the different study designs may have influenced the results of this review [20]. However, we identified significant between group-effects in studies with an intervention period of 4, 12, 16, and 26 weeks, as well as an average participation per week of two to three times with exercise classes lasting between 45 and 60 minutes [76, 81–86]. Therefore, we were not able to identify a specific effect of exercise volume (intervention period, length of exercise classes, average exercise sessions per week), but recommend that future studies should aim to identify the best dose-response relationship.
Previous sample size calculations for the effects of exercise interventions on disease severity in individuals with PD (
Conclusion
This systematic review found no clear effect of exercise mode on domain-specific cognitive function in individuals with PD, but that aerobic exercise tended to best improve memory in individuals with PD. Further, exercise in general appears to be associated with improved cognitive function in individuals with PD. However, improvements might be limited to certain cognitive domains (global cognitive function, executive function or memory). Besides cognitive function, exercise, regardless of mode, tended to slow disease progression in individuals with PD. The currently limited number of high-quality RCTs makes it difficult to draw further conclusions, but emphasizes the need for more quality investigations.
Exercise has shown promising effects on cognitive function in healthy older adults, and as a treatment strategy in individuals with MCI, but the effects of exercise on cognitive function in individuals with PD require verification. In addition, in order to define optimal and specific training programs, the effects of different exercise modes on cognitive performance should be directly compared. The findings would allow the prescription of goal-oriented exercise programs to improve cognitive performance. Researchers must focus on uniform reporting of their findings (e.g., use recommended neuropsychological tests) to guarantee comparability among studies and should aim to assess underlying physiological mechanisms in order to completely understand the effect of exercise. Further, future studies should use homogeneous reporting of exercise intensity and mode and should also analyze the long-lasting effect of exercise on cognitive function in individuals with PD.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
Some aspects of the work were supported by the Universities Australia and German Academic Exchange Service (DAAD) cooperation scheme. Additionally, Tim Stuckenschneider was supported by a DAAD Scholarship. We would like to thank two unknown reviewers for their valuable feedback on a previous version of the manuscript.