Abstract
Background:
Pathological gambling (PG) in Parkinson’s disease (PD) manifests as a persistent and uncontrollable gambling behavior, characterized by dysfunctional decision-making and emotional impairment related to high-risk decisions.
Objective:
The aim of this study was to explore the relationship between personality traits and prefrontal cortex activity in PD patients with or without PG.
Methods:
Thus, hemodynamic cortical activity measured by functional near-infrared spectroscopy (fNIRS) and Iowa Gambling Task (IGT) performance were recorded in forty-six PD patients, divided into three groups according to their gambling status: PD patients with active gambling behavior (PDG); PD patients who remitted from PG (PDNG); and a control group (CG) composed by patients with PD only.
Results:
Results indicates that gambling behavior in PD patients is strongly predictive of dysfunctional cognitive strategy; affecting anomalous cortical response with a left hemispheric unbalance in dorsal areas; and it is related to more reward sensitivity than impulsivity personality components.
Conclusions:
PDG patients differed from PDNG and CG from both behavioral and brain response to decision-making. Overall, these effects confirm a pathological condition related to cognitive and emotional aspects which makes the patients with PGD victims of their dysfunctional behavior.
INTRODUCTION
In association to the most obvious movement disorder characterizing Parkinson’s disease (PD), several studies showed that PD patients may often exhibit some clinical non-motor symptoms, such as Impulse Control Disorders (ICDs). ICDs include behaviors like pathological gambling, hypersexuality, compulsive shopping and binge eating, that were found to be a significant source of morbidity for patients with PD receiving dopamine replacement therapy [1, 2]. Among these, pathological gambling (PG) manifests as a persistent and uncontrollable gambling behavior, despite financial losses and social problems, characterized by a diminished control over the problematic behavior and with a tension or an appetitive urge state prior to engagement in the behavior [3]. This definition of PG is in line with the conceptualization of ICDs as “behavioral addictions” or “disinhibitory psychopatologies” [2–5].
Recent studies investigated the etiology of pathological gambling in PD and suggested that therapies with dopamine agonists are the medication with the strongest association with the development of PG [1, 7]. However, some risk factors, such as male gender, young age at PD onset, genetic predisposition and personality traits such as impulsivity and high novelty seeking have been found to predispose vulnerable individuals to this disorder [1, 6]. Within this frame, PG in PD can be considered as a side effect of the dopaminergic treatment in a relatively vulnerable clinical population. According to the “dopamine overdose hypothesis” postulated by Poletti and colleagues [7], PD patients with ICD strongly process immediate positive rewards or pleasure, because exogenous levodopa administration increases dopaminergic levels and facilitates an excitatory activity in the direct pathway of the cortico-striato-thalamo cortical loops but, on the other hand, it prevents decreases in dopaminergic systems, altering the inhibitory activity of the indirect pathway in the same cortical loop, which translates to a reduced ability to control maladaptive behaviors and to learn from negative rewards, despite significant impairment in social and occupational functioning [7, 8]. Ventral regions and dorsal striatal circuits in PD might be adversely affected by this imbalance in dopaminergic systems and might lead to dysfunctional decision making and cognitive control processes that, indeed, were found consistently across studies in PD with ICD [1, 8–15].
To get more in detail, not only the cognitive functioning seems to be impaired in these clinical population, but also emotional components have been identified to have a role when reward signals turn in favor of immediate outcomes in the case of decision-making [16]. Indeed, dysfunctional decision processes are related to an hyperactivity in the emotional system, mediated by frontal and medial structures, such as the PFC, and more specifically orbitofrontal cortex (OFC), anterior cingulate cortex (ACC) and amygdala, which exaggerate the rewarding impact of external reinforces; and an hypoactivity in the prefrontal cortex (such as left ventromedial areas, vmPFC, and mainly the dorsolateral prefrontal cortex, DLPFC), which predicts the long-term consequences of a given action and that is a critical component for working memory and executive processes (planning, inhibition, attention shifting, cognitive flexibility, and metacognition) [17, 18].
Regarding the emotional motivational system, recent studies based on Gray’s BIS/BAS model [19] suggested that behavioral motivational responses related to personality traits are crucial to the generation of emotions, and those were found to be relevant to approach (reward) and withdrawal (inhibition) in the decisional process [16, 17]. Concerning reward mechanisms, Behavioral Inhibition System (BIS) and Behavioral Activation System (BAS) measures represent a useful tool for testing a potentially anomalous reward sensitivity in neuropsychiatric populations, such as addictions, compared to healthy participants [16, 20]. BAS was conceptualized as a motivational system that is sensitive to signals of reward and non-punishment, engaging behavior toward a payoff and away from a loss. Thus, in general reward becomes a positive reinforcement for action (approach behavior) and a normal level of BAS functionally affects positive emotional attitudes, while extreme levels of reward sensitivity have been linked to impulsivity disorders [21]. On the other hand, punishment promotes negative reinforcement of avoidance (withdrawal), and extreme levels of BIS induce to anxiety-related disorders [22].
A central aspect is that BIS/BAS system owns its cortical correlate on the PFC: whereas the left PFC activity was shown to be implicated in approach-related motivations (appetitive) and positive emotions (reward processing), the right PFC activity was found to be involved in withdrawal-related motivations (aversive) and negative emotions (punishment) [23–26]. Previous research found that people with substance use disorders, gambling disorder or high-level of BAS sensitivity to reward displayed significantly riskier decision making, choosing a larger potential reward even at a greater risk of penalty. Moreover, their electroencephalographic activity revealed a frontal hemispheric activation asymmetry in favor of the left PFC (DLPFC and ACC), reflecting an increased responsiveness to more risky choices [16, 17].
Both emotional and cognitive impairments characterizing PD patients with PG were often investigated using a well-validated decision-making under uncertainty task, the IOWA Gambling Task (IGT) [27, 28]. In this task, patients were asked to select 100 cards from four decks identical in appearance but different in reward feedback. They need to choose between these four decks without knowing that two of them are disadvantageous (more risky, producing immediate large rewards but also significant money loss) or advantageous choices (characterized by small and frequent rewards but less loss) and their goal is to maximize their profit and avoid losing money as far as possible [29]. The IGT is supposed to test emotional-based learning and to be associated to PFC functioning [30, 31]. Evidences from IGT have showed that pathological gamblers with PD behave as patients with vMPFC damage or addictions, showing dysfunctional decision-making [7, 33].
So far both clinical and neuroimaging studies (fMRI and PET) identified the neural networks involved in the decision-making processes associated with IGT execution in PD patients with PG; but special attention was given to frontal areas (vMPFC, ACC, OFC and DLPFC), that seem to be crucial for successful decision-making and IGT performance [34–37].
However, diversely from PET and fMRI techniques, different advantages seem to characterize the application of a relatively new imaging measure such as functional near infrared spectroscopy (fNIRS) for monitoring brain activity and for exploring the contribution of specific PFC areas in response to IGT [38]. This technique is safe and non-invasive, portable, closer to real-life situations, not as susceptible as fMRI to movement artifacts (so it is suitable for certain population like PD patients with movement disorders), not very expensive and not producing any loud noise. Moreover, it is particularly well-suited for evaluating the PFC activity, which is among the regions involved in the emotional and decision-making processes [39].
To our knowledge, only one pilot study on healthy participants focused on the role of DLPFC during IGT performance using NIRS technique [40]. During the first part of the IGT task (1st–50th choice), Bembich and colleagues [40] found a DLPFC activation (slightly lateralized on the right side): in the first period (1st–25th choice) this area was active for low-risk options and was hypothesized to reflect attention shifting; while in the second period (26th–50th choice) DLPFC was active for high-risk options (i.e., more disadvantageous choices), perhaps reflecting the functions of response inhibition and behavioral control. It should be noted that although this study pointed out interesting aspects of DLPFC role in IGT, it presented some limitations: such as the small sample of young healthy participants, the differences in the study design (subjects performed physically a modified version of the IGT task) and the lack of information on the personality traits and mood state characterizing participants (variables proved to influence IGT performance [41].
Otherwise, our present study is the first using an optical imaging technique (fNIRS) to investigate decision-making and concomitantly reward processes (BIS/BAS component) during an IGT task in a clinical sample like PD patients with PG. In addition, we directly compared a control group (CG) of PD patients with PD people who actually show PG (PD Gamblers, PDG) on one side, and PD patients with previous history of PG, that do not show PG anymore (PD Non-Gamblers, PDNG). By studying a pure Parkinson’s group, we deleted the possible confusing effect related to the pathology per se (as in the case of using only and healthy control group). Our first hypothesis was that PD patients with PG would display alterations in PFC hemodynamic activity during the decision-making process for disadvantageous options, showing an excessive reward sensitivity and a lack of inhibition skills. Therefore, we expected that these effects should be marked by an increase of oxy-Hb responses, mainly in the left PFC regions which were found to be related to excessive reward attitudes (increased positive values attributed to potential rewarding cues). In addition, reward trait (revealed by BIS/BAS measure) [20, 42], and impulse control disease (measured by Barratt Impulsiveness Scale, BIS-11, [43]) should mark this impairment mainly in PDG than PDNG and CG. In contrast, a more similar profile is expected for PDNG and CG, who may show a more functional response to immediate reward (more risky options) with a more balanced activity by the left and right PFC.
METHOD
Subjects
A total of forty-six patients with PD were included in the study (37 males and 9 females; age range = 45–75, Mage = 62.93, SD = 7.76). They were recruited from outpatients who were diagnosed and regularly treated by an experienced neurologist (NM) at Centro Parkinson ASST G. Pini-CTO of Milan, Italy. The recruitment criteria include the patient’s competency in understanding the nature of this study and their ability to provide informed consent on his/her own behalf. Exclusion criteria were a) main hearing or vision impairment; b) a previous history of other neurological and psychiatric diseases; c) major depression disorder (according to DSM IV criteria and BDI-II; [44]); d) diagnosis of Dementia and/or Mini-Mental State Examination (MMSE) below 26 out of 30, according to the working criteria proposed by the Movement Disorders Society [45]; e) presence of mild cognitive impairment (MCI).
Patients underwent a clinical and neuropsychological evaluation composed by MMSE [46], the Frontal Assessment Battery (FAB; [47] and the Clock Drawing Test (CDT; [48]). Subjects gambling behavior was assessed using a clinical interview (DSM IV criteria) and QUIP-RS (Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease; [49]) administered to the patient and then checked with the caregiver. Tests were administered by a trained neuropsychologist (CS) in the morning and in the on phase (Table 1).
Demographic and clinical characteristics, and neuropsychological assessment of included patients with Parkinson’s disease.
*age and education corrected scores
Patients were assigned to three different groups, on the base of their gambling status: (1) PD patients with active pathological gambling behavior (N = 15; PDG), (2) PD patients with a previous history of PG (N = 14; PDNG), (3) the control group composed by patients with PD only (N = 17; CG). All patients were treated with levodopa and/or dopamine agonists; daily dosage was recorded and controlled. Disease severity was evaluated with the Unified Parkinson Disease Rating Scale (UPDRS) and correspond on average to the Hoehn & Yahr stage II [50].
All subjects were Italian and were fully informed about the research aims, including that no compensation was provided for their participation. Written informed consents were obtained from all participants and the research protocol was approved by the Ethics Committee of the institution where the work was carried out (Centro Parkinson ASST G. Pini-CTO of Milan, Italy). The study was conformed to the principles of the Declaration of Helsinki.
Personality characteristics measures
BAS score
The Behavioral Activation Scale (BAS) from the Italian version [51] of the 20-item “Behavioral Inhibition/BAS” [20] scale was used to assess sensitivity to reward, impulsive sensation seeking personality characteristics and a propensity for setting more approach goals (goals of moving toward something). Globally, this self-report questionnaire includes 24 items (20 score items and 4 fillers, each measured on five-point Likert scale), and two total scores for BIS (range = 7–28; items 7) and BAS (range = 13–52; items 13). BAS also includes three subscales: a) Reward Responsiveness, that correspond to the tendency to respond to rewarding outcomes with energy and enthusiasm, 5 items (possible scores range from 9 to 20), b) Drive, that captures motivation to pursue goals, and c) Fun Seeking, that reflects the tendency to pursue positive experiences without regard to potential threats or costs, both 4 items (possible scores from 4 to 16). Based on these measures, two total scores (BIS and BAS total) and three BAS subscales were calculated for each group. Only BAS total score was used for the successive statistical analysis (Cronbach’s alpha coefficient = 0.511).
BIS-11
The Barratt impulsiveness scale, version 11 (BIS-11) [43, 52] was used to assess the personality/behavioral construct of impulsivity (Cronbach’s alpha coefficient = 0.546). The BIS-11 scale consists of thirty items that provide a total score for general impulsivity, and scores for three other subscales. The total score was considered in the present study.
Behavioral measure
Iowa Gambling Task
Patients completed a computerized version of the IGT task (adapted for an online Italian version, [27]) in which they had to select a card from among four decks identical in appearance but varying in their amounts of monetary reward and punishment. The task object is to maximize the profit (starting with a fictive loan of € 2000 play money) by drawing a total of 100 trials and avoid losses. This task is a sensitive measure of decision-making that simulates real-word decision making situation demanding the evaluation of the magnitude and timing of rewards and punishments under uncertain conditions. All participants received standard instructions for the IGT: they were told that the goal of the game was to win as much as possible, otherwise, to avoid losing money by selecting a card, one at a time, from the four decks. They were informed that each card drawn would indicate how much they had won and whether there was a penalty as well. They were also warned that some decks were more advantageous than others, and that they were free to switch from one deck to another at any time and as frequently as they liked. They were not informed of the schedule of wins and losses in each deck or given advice on how to draw the cards, nor were they told how many cards they would be allowed to draw yet. To obtain the IGT goal, subjects must find out the most advantageous deck and prevalently pick cards out from that deck. Gain and losses are different for each card selected from the four decks: decks A and B are more risky and disadvantageous (“Disadvantageous Decks”, DD), producing immediate large rewards but they are accompanied by significant money loss (extreme punishments), so they cost more in the long term; decks C and D are advantageous (“Advantageous Decks”, AD), because the penalty amounts are lower in these low-paying decks, resulting in an overall gain (reward) entailing better log-term strategies, despite the initial reduced short-term gain. In summary, high-risk options implied that there is the chance of a great reward, but there is also high risk for a loss. By contrast, low-risk options are often characterized by lower reward, but also low risk for loss.
For this task the quantitative parameter consisted in a behavioral measure computed for the whole 100 card selections. We counted the number of selections from DD (A and B) and the number of selections from AD (C and D) and then derived the net score for the game [(C + D)-(A + B)] (IGT index); a positive score in the IGT score indicates the presence of a positive, advantageous long-term strategy [17, 53].
Procedure
During the experimental phase patients sat on a chair in a darkened and sound isolated room, facing a computer monitor (1280×1024 pixels). The pc was placed approximately 80 cm from the subject, with a visual horizontal angle of 6° and a vertical angle of 8°. Participants used a mouse to choose the card and they had no restriction of time to respond, although they were required to make a decision in a short time interval. They perform the IGT task during NIRS measures recording and the result of their choice (gain or loss) was shown on the screen for a duration of 6 seconds, with an inter-stimulus interval of 12 s before the following IGT choice. At the beginning of the experiment before the task onset, we registered 120 seconds resting period as baseline. The entire experimental procedure was completed in about 1 hour. After completing the experimental phase, BIS-11 and BIS/BAS questionnaires were administered to patients.
Cerebral oxygenation measure
fNIRS data acquisition
fNIRS measurements were conducted with NIRScout System (NIRx Medical Technologies, LLC. Los Angeles, California) using an 8-channel array of optodes (4 light sources/emitters and 4 detectors) covering the prefrontal area (respectively the DLPFC and OFC). Emitters were placed on the following positions: FC3-FC4 and F1-F2 for DLPFC; AF3-AF4 and Fpz for OFC. While detectors were placed on: FC1-FC2 and F3-F4 for DLPFC; Fp1 and Fp2 for OFC (Fig. 1). Emitter-detector distance was kept at 30 mm for contiguous optodes and near-infrared light of two wavelengths (760 and 850 nm) was used. NIRS optodes were positioned on the subject’s head using a NIRS cap according to the international 10/5 system. Resulting channels are reported: Ch 2 (FC3-FC1) and Ch 5 (F1-F3), Ch 4 (FC4-FC2) and Ch 7 (F2-F4), corresponding respectively to the left (Ch2 and Ch5) and right (Ch4 and Ch7) DLPFC (Brodmann Area 9); also, Ch 9 (AF3-Fp1) and Ch11 (Fpz-Fp1), Ch 10 (AF4-Fp2) and Ch 12 (Fpz-Fp2) correspondingly to the left (Ch9 and Ch11) and right (C10 and C12) OFC (Brodmann area 11) [54].
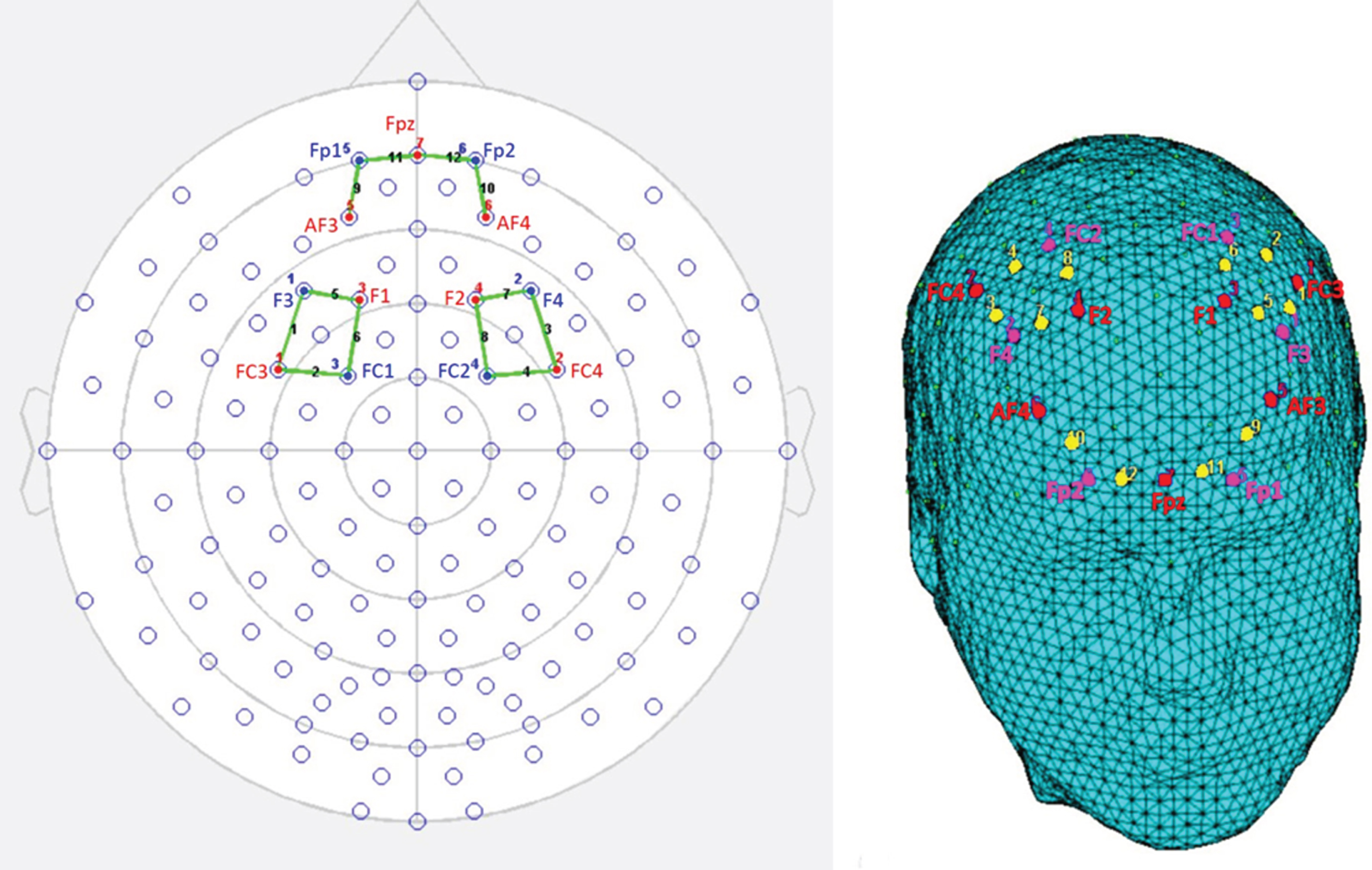
fNIRS montage. A) Schematic representation of channels fNIRS positioning on a 2d head, which includes 10–20 electroencephalography system positions. B) The location of fNIRS channels on DLPFC and OFC regions illustrated on a 3d rendered head. The emitters (red) were placed on positions FC3-FC4 and F1-F2 for DLPFC; AF3-AF4 and Fpz for OFC, while detectors (fuchsia) were placed on FC1-FC2 and F3-F4 for DLPFC; Fp1 and FP2 for OFC. Resulting channels (yellow) were as follows: Ch 2 and Ch 5 correspond to the left DLPFC, while Ch 4 and Ch 7 correspond to right DLPFC. Ch 9 and Ch 11 correspond to the left OFC; Ch 10 and Ch 12 correspond to right OFC.
With NIRStar Acquisition Software, changes in the concentration of oxygenated (O2Hb) and deoxygenated hemoglobin (HHb) were recorded continuously throughout the task, starting from a 120 s resting baseline. Signals obtained from the 8 NIRS channels were acquired with a sampling rate of 6.25 Hz and analyzed and transformed with nirsLAB software (v2014.05; NIRx Medical Technologies LLC, 15Cherry Lane, Glen Head, NY, USA), according to their wavelength and location, resulting in values for the changes in the concentration of oxy and deoxygenated hemoglobin for each channel, scaled in mmol*mm.
The raw O2Hb and HHb data from each channel were digitally band-pass filtered at 0.01–0.3 Hz. Then, the mean concentration of each channel was calculated by averaging data across the trials, an average value for each condition (advantageous decks C and C, called AD and disadvantageous decks, A and B, called DD) was calculated starting from the onset (the decisional process from the appearing of the deck) for the following 6 s. The mean concentration value of 6 s before the trial was used as event-related baseline. According to the mean concentrations in the time series, the effect size in every condition was calculated for each channel and subject. The effect sizes (Cohen’s d) were calculated as the difference of the means of the baseline and trial divided by the standard deviation (sd) of the baseline: D = (m1-m2)/s, with m1 and m2 being the mean concentration values during baseline and trial, respectively, and s the SD of the baseline. Then, the effect sizes obtained from the 8 channels were averaged in order to increase the signal-to-noise ratio. Although NIRS raw data were originally relative values and could not be directly averaged across subjects or channels, effect sizes normalized data could be averaged regardless of the unit since the effect size is not affected by differential pathlength factor (DPF). Finally, the event-related responses to stimuli with respect to each baseline signs have been inverted [55–57].
RESULTS
Data analysis
Three sets of analyses were performed with respect to behavioral (IGT index), hemodynamic (NIRS) and trait (BAS; BIS-11) measures.
Firstly, a repeated measure ANOVA with independent factor Group (CG, PDNG, PDG,) was applied to IGT measure. Secondly, a set of ANOVA models was applied to NIRS measures, including advantageous/disadvantageous choices AD/DD (2, AD, DD); Group (3, PDG, PDNG; CG); Lateralization (2, Left channels, Right channels); Localization (2, DLPFC – Ch2, Ch4; Ch5; Ch7; OFC – Ch9, Ch10, Ch11, Ch12) as fixed factors. For all the ANOVA tests, the degrees of freedom were corrected using Greenhouse–Geisser epsilon where appropriate. Post-hoc comparisons (contrast analyses) were applied to the data. Bonferroni test was applied for multiple comparisons. In addition, the normality of the data distribution was preliminary tested (kurtosis and asymmetry tests). The normality assumption of the distribution was supported by these preliminary tests.
Finally, correlation analyses between BAS and Group variable; BIS-11 and Group variable (r Spearman coefficients); BAS scales and IGT variable; BIS-11 and IGT variable (Pearson coefficients) were performed. Bonferroni test was applied for multiple comparisons.
IGT score
Group factor showed significant main effect (F [2, 50] = 8.54, p≤0.001, η2 = 0.41) at ANOVA. Indeed post-hoc paired comparisons revealed more negative IGT values (more DD than AD choices) for PDG than PDNG (F [1, 50] = 7.06, p≤0.001, η2 = 0.38) and than CG (F [1, 50] = 9.51, p≤0.001, η2 = 0.44). No other effect was statistically significant (Fig. 2).
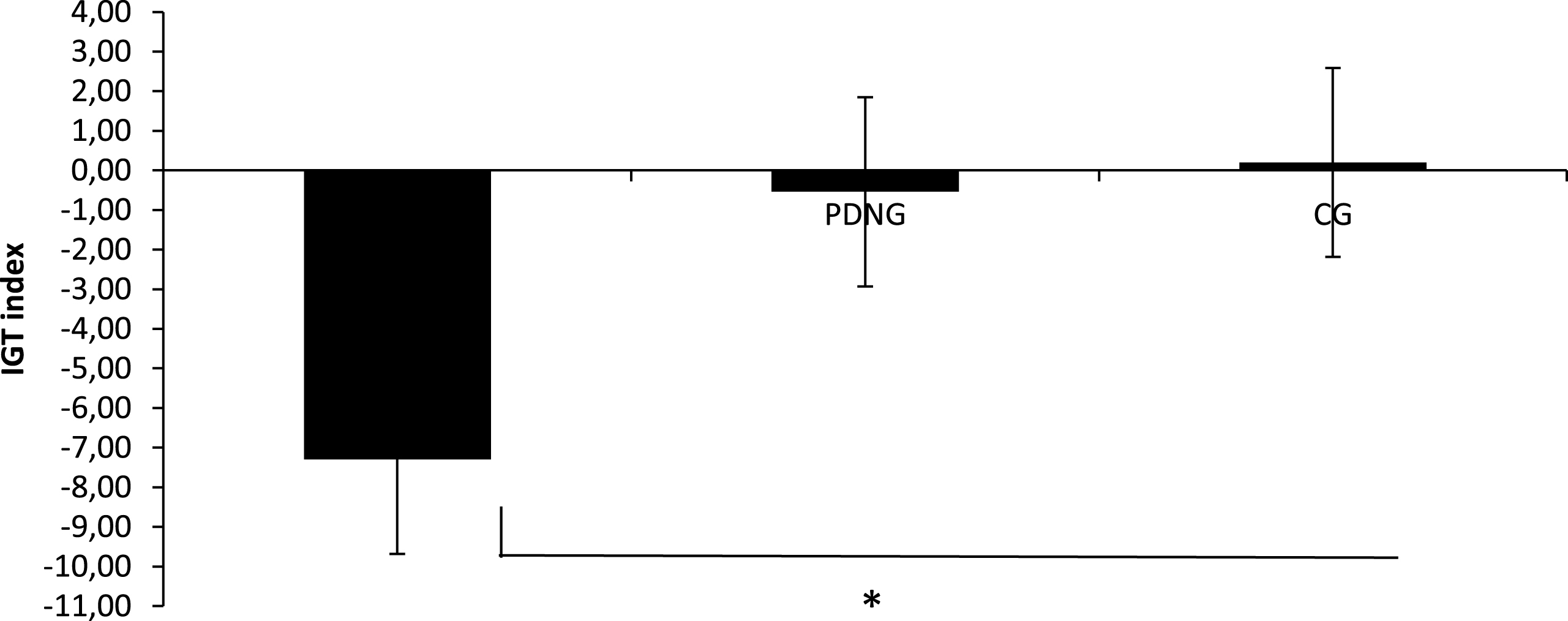
IGT Index. IGT index as a function of group (PDG; PDNG; CG).
fNIRS
The statistical analyses were applied to D dependent measure for O2Hb and HHb-concentration. The analysis on HHb did not reveal significant effects, and for this reason we reported only results for O2Hb-values.
As shown by ANOVA Group×AD/DD (F [2, 51] = 9.32, p≤0.001, η2 = 0.44) and Group×AD/DD×Localization X Lateralization (F [6, 51] = 11.34, p≤0.001, η2 = 0.47) effects were statistically significant. As for the simple effects for the significant interaction Group×AD/DD, higher cortical response to DD was found for PDG than PDNG (F [1, 50] = 7.45, p≤0.001, η2 = 0.39) and CG (F [1, 50] = 9.13, p≤0.001, η2 = 0.43). In addition about the second interaction effect, PDG showed higher cortical response to DD within the left than the right DLPFC (F [1, 50] = 9.11, p≤0.001, η2 = 0.43). In addition PDG revealed increased brain activity than PDNG (F [1, 50] = 8.77, p≤0.001, η2 = 0.40) and CG (F [1, 50] = 10.09, p≤0.001, η2 = 0.45) in response to DD within the left DLPFC. No other comparison was statistically significant (Fig. 3).
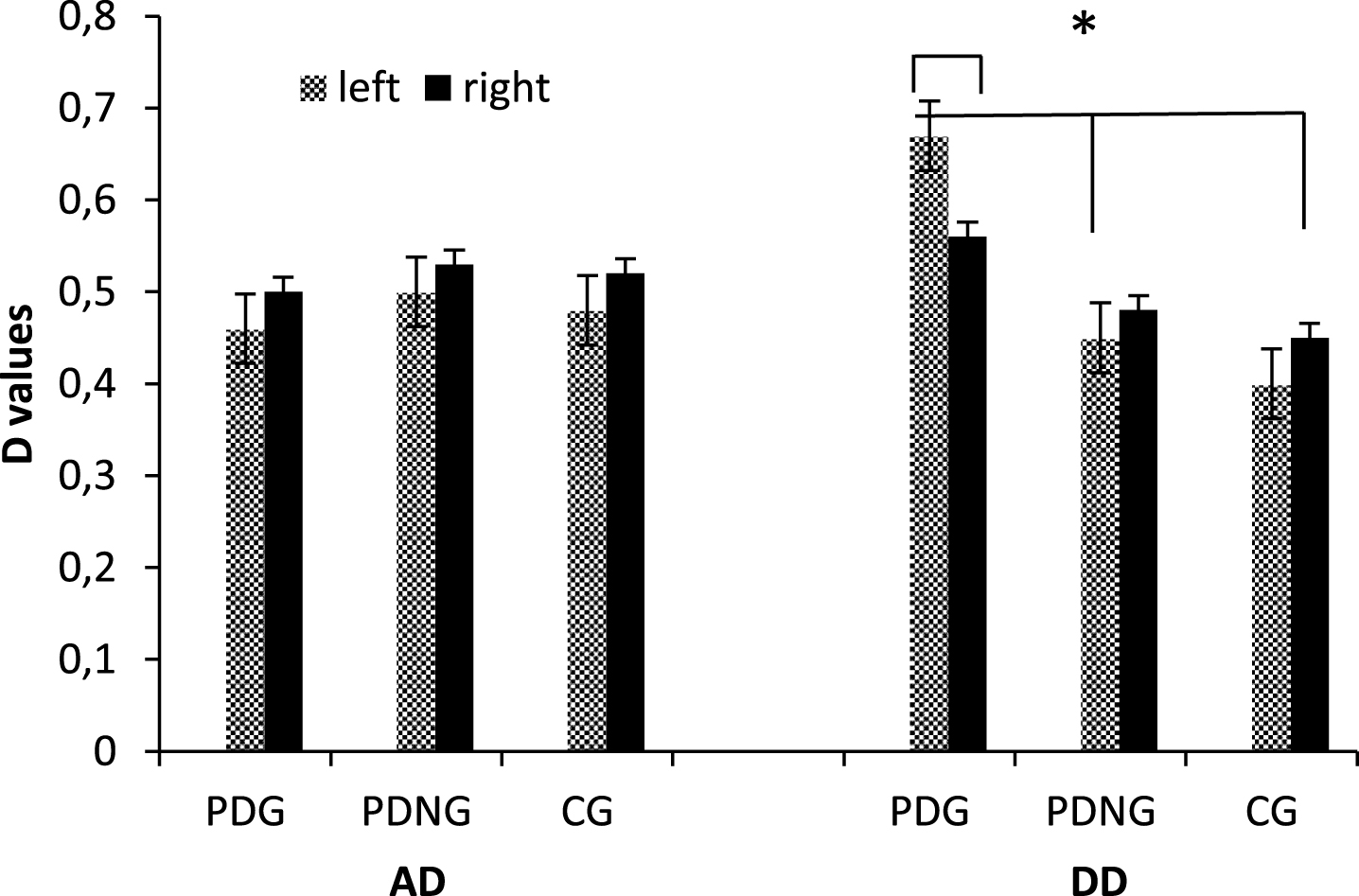
Hemodynamic activity as a function of group (PDG; PDNG; CG), AD/DD, and Lateralization.
Correlation analysis
Correlation analyses were applied firstly to BAS and Group variable; BIS-11 and Group variable (r Spearman coefficients) from one hand; BAS scales and IGT variable; BIS-11 and IGT. As shown by Spearman correlation coefficients, group was not significantly associated to BIS-11 scale (r = 0.341, p = 0.13). In contrast, BAS measures showed significant correlational coefficients, with positive association between group and BAS values (r = 0.566, p≤0.001): higher BAS values were mainly related to PDG.
About IGT measure, it was negatively associated respectively to BIS-11 r = –0.518, p≤0.001 and BAS r = –0.553, p≤0.001 scale: more negative IGT (more DD) was correlated to higher BIS-11 and BAS and values (Fig. 4).
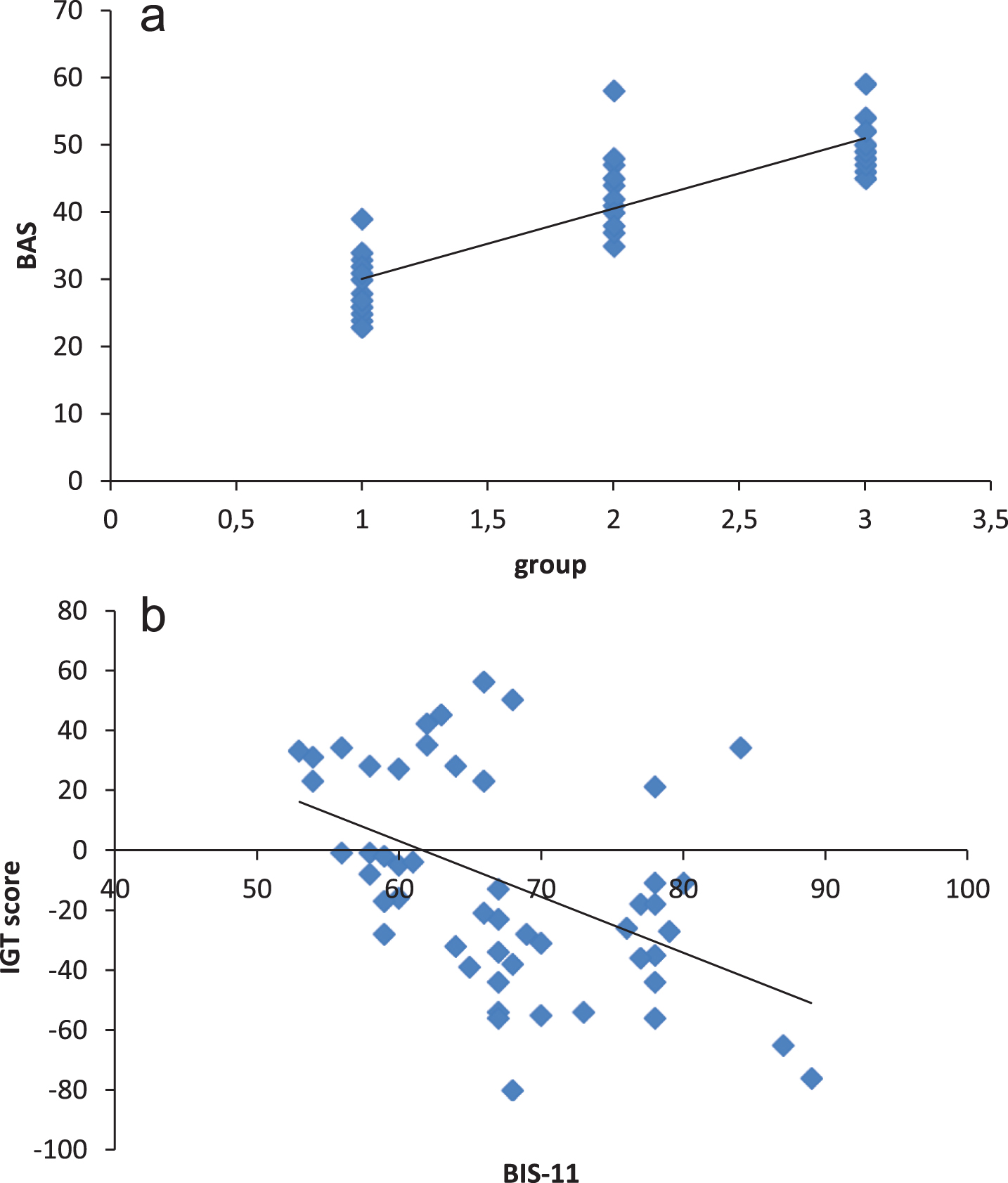
Correlational values for: (a) group and BAS; (b) BIS-11 and IGT scores; (c) BAS and IGT score.
DISCUSSION
The present research exploited fNIRS technique to study the differences of cerebral hemodynamic activity in DLPFC and OFC of a sample of patients with PD, divided in three different groups according to their pathological gambling behavior, during IGT task execution, a well-validated decision-making under ambiguous situations task. Some main results are reported below. First, the behavioral performance, measured using the IGT task, revealed significant differences across the three groups: it was found an impairment in PDG group when facing functional options, indeed these patients showed a relevant prevalence of choices from DD. This result could be considered as a demonstration of a dysfunctional decision-making behavior in PD patients with PG, that was already highlighted by previous research using the same task [13, 58]. Moreover, this effect of poor performance on the IGT associated to impulsive choices and to the preference of small, immediate rewards over large delayed rewards, has been already described in other clinical populations with addictions, such as stimulant abusers [59], pathological gamblers [60], polysubstance dependent patients [61] and cocaine abusers [62, 63]. The evidence that in the PDG group consistently increased the response to immediately rewarding events as compared to the PD and PDNG reinforces the assumption that a motivational impairment is observable in the case this population, as for addictions. A sort of “reward sensitivity bias” seems to be the most relevant variable inducing the subject to overestimate the immediate (unreal) reward at a detriment to the delayed (real) reward [62]. In contrast both PDNG and CG showed a functional decision-making behavior, with the ability to opt for the most advantageous options, although in absence of immediate large reward. These groups were able to develop a positive strategy over time in order to obtain a real gain. To our knowledge, we were the first considering PD patients with a previous history of PG. This result was highly relevant to underline the significance of remitted gambling condition. Indeed, it is the first study which explored in detail this point, compering PDG with PDNG and we may state that the gambling behavior in PD is strongly related to contingent conditions. Both emotional and impulse control impairments characterized PD patients with PG, in contrast to PDNG profile. This effect was supported by hemodynamic data, which underlined the functional, more than structural, impact of prefrontal activity in relationship with PG.
Secondly regarding hemodynamic activity, significant effects were found as a function of groups in the PFC: PDG showed a significant increased DLPFC response to high-risk and potentially more rewarding options (DD). On the one hand, this result may suggest the difficulty to actively control their impulse to achieve an immediate reward when a fictitious winning condition is present. This is hypothesized to be related to a frontal lobe cognitive dysfunction in monitoring impulsivity, delayed discounting and in managing inhibitory control mechanisms in PDG [62, 64]. Recent fMRI studies highlighted the specificity of DLPFC reduced volume abnormalities as a predictor of restricted behavioral repertoires in alcohol-dependent patients [65] and in cocaine-dependent polysubstance abusers [66]. On the other hand, DLPFC activity was found to increase when a subject is involved in a variety of executive functions, such as action selection in decision-making processes, reversal learning, reward prediction error and behavior adjustments [67]. In this case, PD patients with PG seems to engage a significant greater quantity of neural resources when facing disadvantageous options, if compared to PDNG and PD groups, perhaps because of the need of a major effort to use cognitive strategy to actively regulate reward processing [68]. This could be supported by a fMRI study on patients with DLPFC damage that were found to be unable to understand and succeed in IGT reversal learning [69]. Altogether, these evidences lead to the hypothesis that impaired dorsal striatal connectivity in PDG could disrupt the propagation of value information to DLPFC and contribute to the observed “addictive behavioral impairment”. Although their effective inadequate behavior, PDG are willing to go on with the dysfunctional options to obtain an (apparent) rewarding choices. That is because PDG appeared more sensitive to risky cues (potentially rewarding), being emotionally involved in the reward processing [16].
In general, our results may suggest a major involvement of the frontal regions in Parkinson’s disease patients with gambling behavior for both cognitive and emotional processes. Specifically, the present results underlined the role of the DLPFC to modulate the rewarding effect in the case of dysfunctional decision-making. We already know from previous research that DLPFC is involved in predicting the long-term consequences of a given action and that is a critical component for working memory and executive processes (planning, inhibition, attention shifting, cognitive flexibility, and metacognition) [17, 18]. Moreover, studies targeting DLFPC with transcranial direct current stimulation (tDCS) and repetitive TMS have shown that modulation of activity in DLPFC significantly reduces not only risk-taking behavior [70], but also craving for alcohol [71], cocaine [72] and food [73].
In contrast, the absence of differences among the groups with regard of the OFC, which did not appear to directly support the behavioural choices, can be considered as a relevant result. Indeed, no different activation as a function of deck types was found for OFC area, that together with the ACC was found to be involved in emotional processing, exaggerating the reward impact of external reinforcers [18]. Taken together these results may suggest a relevant role by the DLPFC more than other PFC components.
However, interestingly, a significant effect was also revealed as a function of the cortical lateralization. Indeed, the DLPFC sensitivity to rewarding (and dysfunctional) conditions was mainly supported by the dorsal left side activation more than the right side. This result may confirm the PFC unbalance effect (with specific lateralization within the left hemisphere) which was found in response to more rewarding cues also in the case of not dysfunctional choices. As previously observed, the left PFC activation that has been considered in general as a factor able to explain the tendency to prefer more reward-related conditions (in terms of positive emotions and approach behavior) and was found to be a critical marker of dysfunctional decisional behavior in Substance Use Disorders (SUD) populations [62, 74]. Indeed, this left hemispheric hyperactivation known as “hemispheric unbalance effect” was observed to reflect a cognitive bias in conditions considered as more appetitive and rewarding and to be associated also to high-BAS healthy subjects, that are characterized by the inability to evaluate and to reconstruct a realistic thought about the cognitive strategy they adopted, in terms of efficacy, functional planning, flexibility, and self-knowledge, with generally more limited in their ability to formulate a metacognitive knowledge of their own behavior [17].
Thirdly, these behavioral and neurophysiological trends were partially confirmed by correlational analyses. Indeed, whereas impulsivity trait (BIS-11) was not directly related to group variable, BAS profile was largely related to group profile, that is higher BAS was more related to PDG than PDNG and CG group. In addition, the behavioral inability to opt for the functional strategy (as elucidated by IGT index) was highly related to impulsivity (BIS) and reward (BAS). Based on these results we may suppose that both these personality traits, more reward sensitivity than impulsivity, may be intrinsically related to gambling behavior in Parkinson’s disease. Whereas previous models on gambling addiction underlined the possible role of both rewarding effect and impulsivity trait [6], based on the present data, we may suggest a more crucial role for the reward sensitivity bias more than the impulsivity component. A possible explanation may suggest that the significant relationship between BAS scoring and PG behavior supports the direct impact of rewarding construct in explaining the difference between remitted or not remitted gambling behavior. Indeed higher level of BAS (and reward-related mechanism) may be considered predictive of future pathological gambling behavior, whereas low level of BAS may predict a higher ability to contrast reward bias, as shown by PDG group. Future research may more directly explore the relationship between these two personality variables.
To summarize, based on previous results, we may suppose that impulsivity and reward sensitivity personality components are related to anomalous cortical response and that this is predictive of dysfunctional cognitive strategy and gambling behavior in PD patients. All these effects, and mainly the role of BAS measure, may reinforce the pathological condition which makes the PDG victims of their dysfunctional behavior. In contrast, when reward bias is limited, as shown in PDNG, the impulse control is favored, and gambling behavior may be contrasted with more adequate strategies. Future research could better consider this important factor of reward-related mechanisms to develop a usable clinical measure predictive of a possible remission behavior or not in PG.
To conclude, two emerging limitations of the present study are the reduced number of participants per group and the absence of a direct comparison with a control group composed by healthy subjects only. As future steps, it would be necessary to integrate these results with the previous limitations and with some other important constructs that were found to have a role within the frame of pathological gambling, such as impaired self-awareness [60] and metacognition.
CONFLICT OF INTEREST
The authors have no conflict to report.
Footnotes
ACKNOWLEDGMENTS
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors report no conflict of interests. Acknowledgement to Fondazione Grigioni for the support.
