Abstract
Background:
Urinary dysfunction is relatively common, however, is often underestimated and diagnosed by subjective questionnaires in patients with Parkinsonism.
Objective:
To determine the correlation between subjective urinary dysfunction and post-void residual urine volume in patients with Parkinson’s disease (PD), multiple system atrophy (MSA) and progressive supranuclear palsy (PSP), and to assess the relationship between clinical characteristics and subjective or objective urinary dysfunction.
Methods:
A total of 196 patients with de novo Parkinsonism without urological and gynecological disorders were included. For all subjects, data were collected on sonographic post-void residual urine volume and urinary symptoms. All patients were also assessed on motor symptom severity, cognitive and affective measurements, other nonmotor symptoms, 39-item Parkinson’s Disease Questionnaire, and renal functions.
Results:
Seventeen out of 196 parkinsonian patients showed abnormal post-void residual urine volumes, with ≥100 ml. The MSA group showed significantly higher post-void residual urine volume compared to the PD and PSP groups. Symptoms of the sense of incomplete emptying, weak urine stream, and nocturia were strongly correlated with post-void residual urine volume. Post-void residual urine volume was positively related to autonomic symptoms, other non-motor symptoms, patient activities of daily living, and negatively related to renal function.
Conclusion:
Increased post-void residual urine volumes were observed in small portion of early drug-naïve Parkinsonian patients, especially in MSA. Post-void residual urine volume was significantly related to subjective urinary and other autonomic dysfunctions. Voiding dysfunction is associated with not only end organ damage, but also other nonmotor dysfunctions and patient activities of daily living.
Keywords
INTRODUCTION
In clinical practice urinary dysfunction is generally assessed by subjective urinary dysfunction questionnaires and sonographic post-void residual urine volumes. However, subjective urinary dysfunctions are weakly correlated with objective tests [1].
Urinary dysfunction is present in approximately 30%–65% of patients with Parkinson’s disease (PD), which is significantly higher than healthy controls, according to prevalence studies [2, 3]. In a recent longitudinal study involving a large cohort of PD patients, urinary dysfunction was not only correlated with quality of life but was also regarded as an early marker of higher motor and non-motor disability [4].
More severe urinary dysfunctions and laboratory findings are expected to be present in atypical PD than those with PD [5]. The aims of this study were to determine the correlation between subjective urinary dysfunction and post-void residual urine volume in PD and atypical PD patients, and to assess the relationship between clinical characteristics and subjective or objective urinary dysfunction.
METHODS
Subjects
The institutional review board at Seoul St. Mary’s Hospital approved the study protocol. Each subject provided informed consent for participation.
A total of 196 de novo patients with PD [6], multiple system atrophy (MSA) [7], and progressive supranuclear palsy (PSP) [8] without urological and gynecological disorders were enrolled using each clinical diagnostic criterion. Basic characteristics were collected including age, sex, disease duration, presence of hypertension or diabetes mellitus (DM), and smoking. For all subjects, data were collected on sonographic post-void residual urine volumes and urinary symptoms using the Scale for Outcomes in Parkinson’s Disease-Autonomic (SCOPA-AUT) questionnaire [9]. All patients were also assessed with the Unified Parkinson Disease Rating Scale (UPDRS), Hoehn and Yahr (H&Y) stage, the Korean version of the Mini-Mental State Examination (MMSE), Clinical Dementia Rating (CDR), Global Deteriorating Scale (GDS), estimated Glomerular Filtration Rate (eGFR), Barthel Activities of Daily Living (B-ADL), Montgomery-Asberg depression rating scale (MADRS) [10], Non-motor Symptoms Scale (NMSS) [11], 39-item Parkinson’s Disease Questionnaire (PDQ-39) for quality of life [12], and the SCOPA-AUT questionnaire [9]. Estimated glomerular filtration rate (eGFR) was assessed by individual blood test and calculated by the equation, 186×(Creatinine/88.4)–1.154×(Age)–0.203×(0.742 if female).
Patients who had a history with urological or gynecological disorders were excluded. Patients who had abnormal post-residual urine volume were examined for structural abnormalities in the genitourinary system by urology or gynecology specialists and were also excluded in a retrospective manner.
Residual urine volume
Post-void residual urine volume was defined on portable ultrasound device using CUBEscan™ BioCon-500 (Mcube Technology, Seoul, Korea). BioCon-500 transmits ultrasound signals into the abdomen of a patient and receives the echoed signals. Using the echoed signals, the system determines the bladder’s outline and calculates bladder volume based on the outline. Bladder volume range is from 0 to 999 mL, with an accuracy of±20% or±20 mL. Abnormal post-void residual urine volume is defined as more than 100 milliliters.
Data analysis and statistics
Statistical analyses were performed using IBM SPSS statistics software version 24.0. Differences in clinical characteristics were analyzed by one-way analysis of variance (ANOVA) or analysis of covariance (ANCOVA) with Bonferroni post-hoc comparison. Pearson’s χ2 test was performed to evaluate the association between patient group and post-void residual urine volume. Correlations between objective post-void residual urine volume and individual urinary dysfunction questionnaire items measured by the SCOPA-AUT scale were tested using Spearman correlation analysis. The data were also evaluated separately by sex. Correlations were performed for post-void residual urine volume, sum of urinary dysfunction score, and other clinical characteristics which included age, disease duration, UPDRS, MMSE, eGFR, SCOPA-AUT, NMSS and PDQ 39 scores. A p-value <0.05 was considered significant.
RESULTS
This study was comprised of 163 PD (83.2%), 14 MSA (7.1%), and 19 PSP (9.7%) patients. Basic characteristics for all Parkinsonism patients are shown in Table 1. The PSP group was older, and had a higher prevalence of hypertension than the PD and MSA groups (Table 2).
Basic characteristics of patients
Values represent mean with standard deviation or numbers of patients (percentage). PD, Parkinson’s disease; MSA, multiple system atrophy; PSP, progressive supranuclear palsy; UPDRS, Unified Parkinson Disease Rating Scale; MMSE, Mini-Mental State Examination; CDR, Clinical Dementia Rating; GDS, Global Deteriorating Scale; ADL, Activities of Daily Living; NMSS, Non-motor Symptoms Scale; SCOPA-AUT, Scale for Outcomes in Parkinson’s Disease-Autonomic; PDQ39, 39-item Parkinson’s Disease Questionnaire; eGFR, estimated glomerular filtration rate.
Comparison of clinical characteristics of each group
Values represent mean with standard deviation or numbers of patients (percentage). PD, Parkinson’s disease; MSA, multiple system atrophy; PSP, progressive supranuclear palsy; UPDRS, Unified Parkinson Disease Rating Scale; MMSE, Mini-Mental State Examination; NMSS, Non-motor Symptoms Scale; SCOPA-AUT, Scale for Outcomes in Parkinson’s Disease-Autonomic; eGFR, estimated glomerular filtration rate. Analyses were performed by one-way analysis of variance test, and the χ2 test.
Abnormal post-void residual urine volume was found in 10 out of 163 PD patients, 6 out of 14 MSA patients, and 1 out of 19 PSP patients; and the MSA group had significantly higher rates of abnormal post-void residual urine volume compared to the PD and PSP groups (Fig. 1).
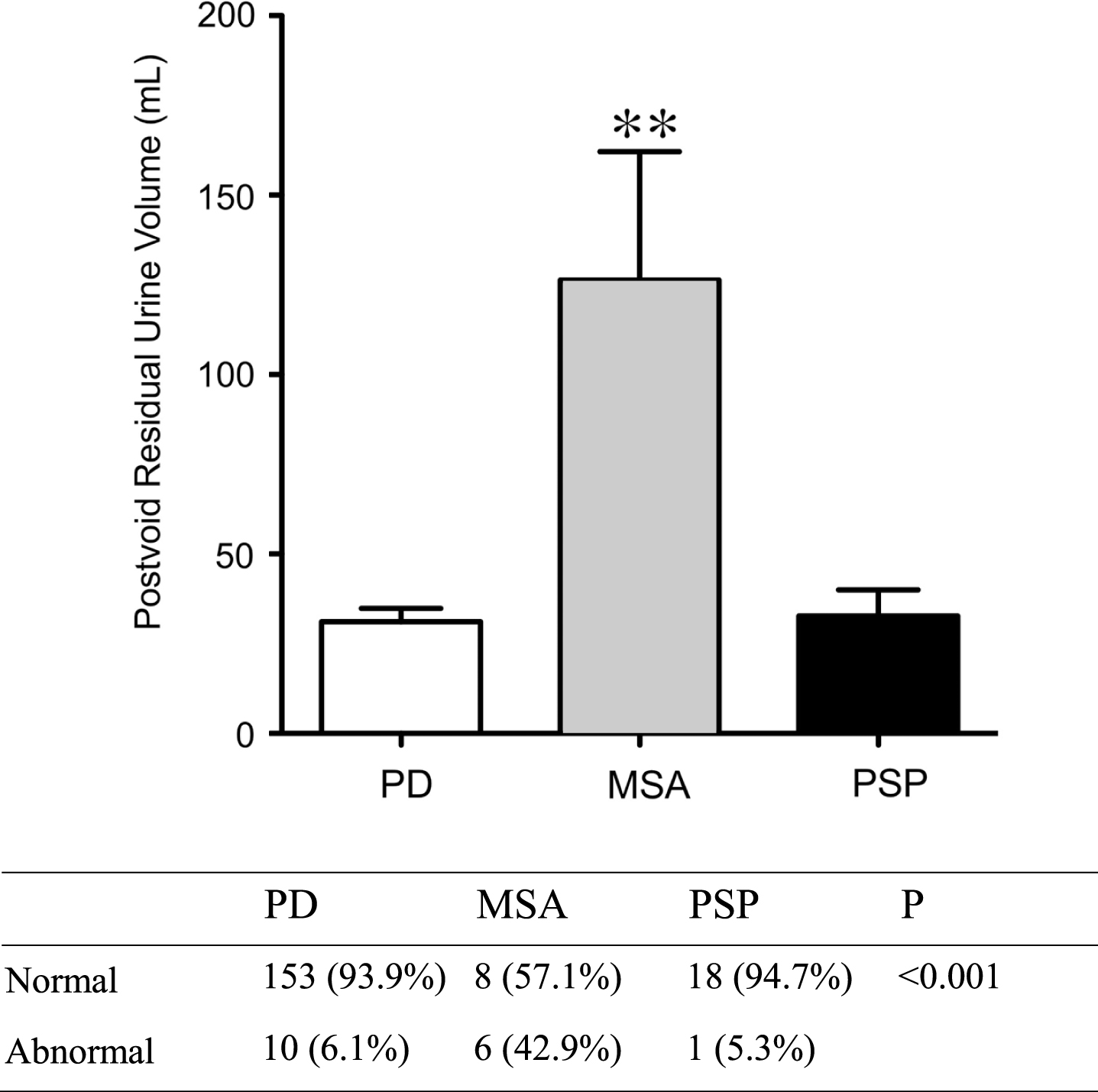
Postvoid residual urine volume of patients. Values represent the mean with standard error of the mean (SEM) or numbers of patients (percentage). PD, Parkinson’s disease; MSA, multiple system atrophy; PSP, progressive supranuclear palsy. Analyses were performed by the χ2 test and analysis of covariance (ANCOVA) controlling with age, sex, hypertension and diabetes mellitus. **<0.01.
Among subjective urinary symptoms, a sense of incomplete emptying after voiding, weak urine stream, and nocturia were significantly correlated with increased post-void residual urine volume in parkinsonian patients, especially in men (Tables 3 and 4). In females, all six urinary symptoms and the sum of the urinary dysfunction scale were not significantly associated with post-void residual urine volumes (Table 5).
Correlation analysis results among postvoid residual urine volume and urinary symptoms measured by SCOPA-AUT urinary dysfunction semi questionnaire in all Parkinsonian syndrome patients
Analyses were performed using the Spearman rank correlation coefficients.
Correlation analysis results among postvoid residual urine volume and urinary symptoms measured by SCOPA-AUT urinary dysfunction semi questionnaire in men with Parkinsonism
Analyses were performed using the Spearman rank correlation coefficients.
Correlation analysis results among postvoid residual urine volume and urinary symptoms measured by SCOPA-AUT urinary dysfunction semi questionnaire in women with Parkinsonism
Analyses were performed using the Spearman rank correlation coefficients.
Subjective urinary dysfunction score was positively associated with increasing age, UPDRS, SCOPA-AUT, NMSS, and PDQ39, and negatively related to decreasing MMSE and eGFR (Table 6). Post-void residual urine volume was related to age, eGFR, SCOPA-AUT, NMSS, and PDQ39. More urinary symptoms were correlated with the eGFR and MMSE score. Parkinsonism disease duration was not significantly related to the sum of the urinary dysfunction score or post-void residual urine volume. Urinary dysfunctions directly affected patient quality of life measured with the PDQ39.
Correlation analysis results among postvoid residual urine volume, urinary dysfunction score and other clinical characteristics
Analyses were performed using the Spearman rank correlation coefficients. UPDRS, Unified Parkinson Disease Rating Scale; MMSE, Mini-Mental State Examination; eGFR, estimated glomerular filtration rate; SCOPA-AUT, Scale for Outcomes in Parkinson’s Disease-Autonomic; NMSS, Non-motor Symptoms Scale; PDQ39, 39-item Parkinson’s Disease Questionnaire.
DISCUSSION
In the present study, post-void residual urine volume was significantly correlated with the sum of the SCOPA-AUT urinary dysfunction subscale, especially in men. This result suggests that post-void residual urine volume may be a noninvasive and objective method to evaluate urinary dysfunction in all parkinsonian patients.
Subjective urinary dysfunctions were not different among the PD, MSA, and PSP groups, however, post-void residual urine volume was significantly increased in MSA patients compared to PD and PSP patients. This may result in impaired bladder contractility in MSA patients compared to PD and PSP patients, due to the higher prevalence of weak detrusor contractility in MSA patients. This result is consistent with previous studies showing that post-void residual urine volume might be an appropriate method to differentiate MSA from PD [13, 14]. Subjects with MSA may manifest with more frequent urinary incontinence and retention than those with PD, probably due to vesicosphincteral dysfunction probably resulting from degeneration of intermediolateral cell columns and Onuf’s nucleus in the spinal cord [15, 16]. In contrast, PD is primarily induced by degeneration of dopaminergic neurons in the substantia nigra pars compacta (SNPC), which reduces the activity of gamma-aminobutyric acid (GABA)-releasing neurons projecting from the striatum to the midbrain periaqueductal grey (PAG), thus, inhibiting the micturition reflex. The loss of dopaminergic neurons also induces motor dysfunction and detrusor overactivity [5]. This is relevant to the results of several studies which report more complaints of overactive bladder symptoms such as frequency, nocturia, and urge with a minimal post-voiding residual volume in PD patients [17, 18].
Especially in male parkinsonian patients, we found that subjective urinary dysfunction score was significantly correlated with increasing post-void residual urine volume. Conversely, in female patients, SCOPA-AUT scale was not significantly associated with post-void residual urine volume. Detrusor instability and bladder outlet obstruction are more common in men than women with symptoms of urinary dysfunction [19, 20]. This result might be due to the high prevalence of benign prostate hypertrophy in elderly male patients, although we excluded patients with prostate disease and urological disorders.
Subjective urinary dysfunction score was not significantly correlated with disease duration, however, was well correlated with disease severity. This result may be due to the study’s inclusion criteria which included de-novo drug naïve parkinsonian patients and those with immobility or poor manual dexterity which could complicate urinary incontinence and retention. In this study, since we did not enroll advanced cases and not include follow-up data on severity of urinary dysfunction, future follow-up studies need to confirm that urinary dysfunction occurs in early stage PD and is an early marker of disease severity in de-novo PD patients, as reported in previous studies [4, 22].
Cognitive impairment and renal dysfunction are well-known markers of microvascular end-organ damage. In this study, high urinary dysfunction score was negatively associated with lower MMSE score. Decreased glomerular filtration rate (GFR) was significantly correlated with increased urinary dysfunction symptoms and post-void residual urine volume. Urinary dysfunction was highly prevalent in PD dementia and dementia with Lewy bodies [23], demonstrating a possible correlation between urinary dysfunction, cognitive impairment and the spreading of cortical alpha-synuclein. It has been demonstrated that different cortical regions and parts of the frontal lobe are involved in controlling cognition and emotions, which are also involved in modulating processes of bladder control [24]. However, we should consider that neurogenic bladder with increased post-void residual urine volume itself can increase the risk of chronic kidney disease and autonomic dysfunction is a common and important complication of chronic kidney disease [25].
Urinary dysfunction in PD significantly impaired quality of life as in a previous report [20], even more than motor symptoms. Therefore, effective conservative management is necessary including behavioral modification with bladder training, pelvic floor education, and biofeedback training that takes patient living facilities, mobility, and dexterity into consideration [14].
Some limitations need to be addressed. First, this study did not include an age-matched control group. Second, there was significant discrepancy in age, hypertension, and H &Y stage between the PD, MSA, and PSP groups. Our study predominantly included PD patients compared to MSA and PSP patients. We enrolled consecutive Parkinsonism patients who visited our movement disorder clinic during the same period. Third, the enrolled subjects in this study can have other medical conditions, such as diabetic autonomic neuropathy and lumbar spondylosis, that might influence urinary dysfunctions. In this study, there were no differences between DM and no DM groups in terms of subjective urinary dysfunction score (9.4±4.3 vs. 8.2±4.5, p = 0.138) and post-void residual urine volume (42.9±58.6 ml vs. 37.1±61.8 ml, p = 0.612). The prevalence of abnormal post-void residual urine volume in PD group was unrelated to the presence of DM (DM vs. no DM: 6.7% vs. 6.0%, χ2 =0.013, p = 0.893). Nonetheless, relative higher comorbidity of DM in PD group than other groups might bias to study results. Fourth, our study is not a prospective study. Therefore, further research is needed to investigate the correlation between objective and subjective urinary dysfunction in advanced PD patients.
In conclusion, significantly increased sonographic post-void residual urine volumes were observed in a small portion of early de-novo drug naïve Parkinsonian patients, especially in MSA patients. Objectively measured post-void residual urine volume was significantly related to subjective urinary dysfunctions and other autonomic dysfunctions. Voiding dysfunction is associated not only with target organ damage represented by cognitive dysfunction and renal damage, but also other nonmotor dysfunctions and patient ADL. Therefore, voiding problems due to urinary dysfunction requires attention and investigation of urinary dysfunction in PD patients; further research on mechanisms is also required.
DISCLOSURE OF CONFLICTS OF INTEREST
The authors declare no financial or other conflicts of interest.
Footnotes
ACKNOWLEDGMENTS
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2017R1D1A1B06028086).
