Abstract
Background and Objective:
Duchenne muscular dystrophy (DMD) is a degenerative X-linked muscle disease. Death frequently results from complications in cardiopulmonary systems. Preclinical/early diagnosis of cardiac autonomic abnormalities may aid initiate cardioprotective therapy and enhance prognosis.
Methods:
A cross sectional, prospective study of 38 DMD boys compared with 37 age-matched healthy controls was conducted. Lead II electrocardiography and beat-to-beat blood pressure were recorded to assess heart rate variability (HRV), blood pressure variability (BPV), and baroreceptor sensitivity (BRS) in a standardized environment. Data were analysed and correlated with disease severity and genotype.
Results:
In the DMD group, the median age at assessment was 8 years [IQR 7–9 years], the median age at disease onset was 3 years [IQR, 2–6 years], and the mean duration of illness was 4 years [IQR, 2.5–5]. DNA sequencing showed deletions in 34/38 (89.5 %) and duplications in 4/38 (10.5%) patients. The median heart rate in DMD children was significantly higher [101.19 (Range, 94.71–108.49)] /min compared to controls [81 (Range, 76.2–92.76)] /min (
Conclusion:
This study demonstrates a distinct early impairment of neuro-cardio-autonomic regulation in DMD. Simple yet effective non-invasive techniques such as HRV, BPV, and BRS may help identify cardiac dysfunction in a pre-clinical state, paving the way for early cardio-protective therapies and limiting disease progression in DMD patients.
Keywords
INTRODUCTION
Duchenne muscular dystrophy (DMD) is an X-linked recessive genetic disorder that affects approximately 1 in 3500–5000 live male births worldwide [1]. DMD is diagnosed using multiplex polymerase chain reaction (mPCR) or multiplex ligation-dependent probe amplification (MLPA) techniques to look for mutations in the DMD gene [2]. Typically, most patients are diagnosed around age 5 and, subsequently, lose ambulation around age 10 [3]. In the later stages of disease progression, muscles governing vital functions, such as respiration and cardiac movement, are frequently affected, leading to death [2, 4]. Currently, 30% of global DMD patients survive beyond 30 years of age due to nocturnal ventilation and spinal surgery [3].
One of the most distinguishing features of DMD is the progressive degeneration and necrosis of the cardiomyocytes [5]. The use of traditional diagnostic measures to avoid heart failure and mortality in people with DMD is limited. However, DMD patients frequently exhibit irregular circadian rhythm and reduced heart rate variability (HRV), leading to severe congestive heart failure [6]. Consequently, evaluating the electrophysiology of the heart can lead to identifying patterns of dysfunction that can foresee the progression of decline in cardiac function [7]. Accordingly, resting heart rate is a simple measurement with substantial prognostic significance [8].
Additionally, HRV offers a precise, non-invasive way to indirectly and quantitatively evaluate the autonomic nervous system (ANS) that regulates heart physiology. The method is based on variations in RR intervals, or the time between successive heartbeats [9]. It has been shown that changes in HRV can predict adverse cardiovascular outcomes.
Since 1995, a number of studies have been conducted to investigate HRV in patients diagnosed with DMD [10, 11]. Reviewing the role of HRV in DMD reveals that it possesses significant importance in determining the common influences of the sympathetic and parasympathetic branches of the autonomic nervous system (ANS), pulmonary diseases, age and/or stage of the disease, the impact of medication on enhancing autonomic modulation, and the usefulness of HRV as a predictor of sudden cardiac death [12]. Since heart failure in DMD has increased drastically, assessments based solely on HRV may not provide sufficient information on ANS activity due to HRV’s methodological equivocation [13]. Hence additional tools along with HRV in assessing autonomic-cardiac modulation are mandated.
The term “blood pressure variability” refers to the changes in blood pressure has a predictive role in both the morbidity and mortality associated with cardiovascular disease [14]. The baroreflex mechanism is responsible for the short-term maintenance of blood pressure stability. This mechanism’s effectiveness can be evaluated using the cardiovagal baroreflex sensitivity (BRS) index, which examines the degree to which the RR interval adjusts reflexively in response to a shift in systolic blood pressure [13].
In the present study, we have, for the first time, assessed the beat-to-beat blood pressure variability (BPV) and baroreflex receptor sensitivity (BRS) along with standard HRV evaluation to obtain a composite picture of autonomic modulation in the early stages of DMD. Further, the HRV, BPV, and BRS data were correlated with disease severity to predict cardiac dysfunction in a pre-clinical stage and pave the way for early cardio-protective therapies in children with DMD.
MATERIALS AND METHODS
This prospective observational study included 38 children with genetically confirmed DMD and 37 normal boys of similar ages and socioeconomic statuses who had no prior history of neuromuscular symptoms or heart disease. The study was approved by the institution’s ethical committee, and all participants (controls/patients) or their legal guardians provided written informed consent. The children with DMD were recruited from the neuromuscular clinic of a tertiary care university hospital in India. Genetic analysis was performed using the Multiplex ligation-dependent probe amplification (MLPA) method to screen for all 79 exons of the dystrophin gene using the SALSA MLPA probe sets P034 and P035 (MRC-Holland, Amsterdam, the Netherlands), as per the manufacturer’s instructions [15]. Patients with DMD who had deletions or duplications confirmed by genetic testing were considered for inclusion. DMD Boys with cardiac comorbidities and / or use of medications such as ACE inhibitors, beta-blockers, and other drugs with potential chronotropic effects were excluded from the study. The assessment protocol consisted of heart rate variability (HRV), Blood pressure variability (BPV) and Baroreflex sensitivity (BRS).
The autonomic function tests were performed under standardised settings in the Autonomic laboratory, Department of Neurophysiology, NIMHANS [16, 17]. In brief, children were advised to have breakfast two hours before the tests. Further, they were instructed to empty their bowels and bladder before the tests. All tests were conducted between 9 am to 11 am in a quiet ambience maintained with a constant temperature. The children were acclimated to the laboratory environment and briefed about the tests to alleviate any anxiety that may impede the recordings.
Continuous blood pressure (BP) and electrocardiogram (ECG) were monitored for 20 minutes after the patient rested for 15 minutes. A five-minute, artefact-free segment from ECG and BP recordings was used for the analysis of HRV, BPV, and BRS [18].
Heart rate variability assessed from electrocardiogram
All individuals underwent artifact-free lead II electrocardiogram (ECG) recording in the supine resting position. Signals were conveyed through an analogue to digital converter (Power Lab, 16 channels data acquisition equipment, AD Instruments, Australia) with a sampling rate of 1024 Hz. To facilitate analysis, the raw ECG was converted into a series of consecutive RR intervals. An automatic programme (Lab chart pro version 8, AD instruments, Australia) that enables visual verification of the unprocessed ECG and breathing signals was used to capture and analyse the data offline. Lab Chart Pro and ECG Analysis Software Version 2.3.2, which employ the Hamilton-Tompkins QRS detection method for ECG detection, were used to calculate time and frequency domain parameters for an error-free 5 minute ECG segment for analysis [19] (Fig. 1).
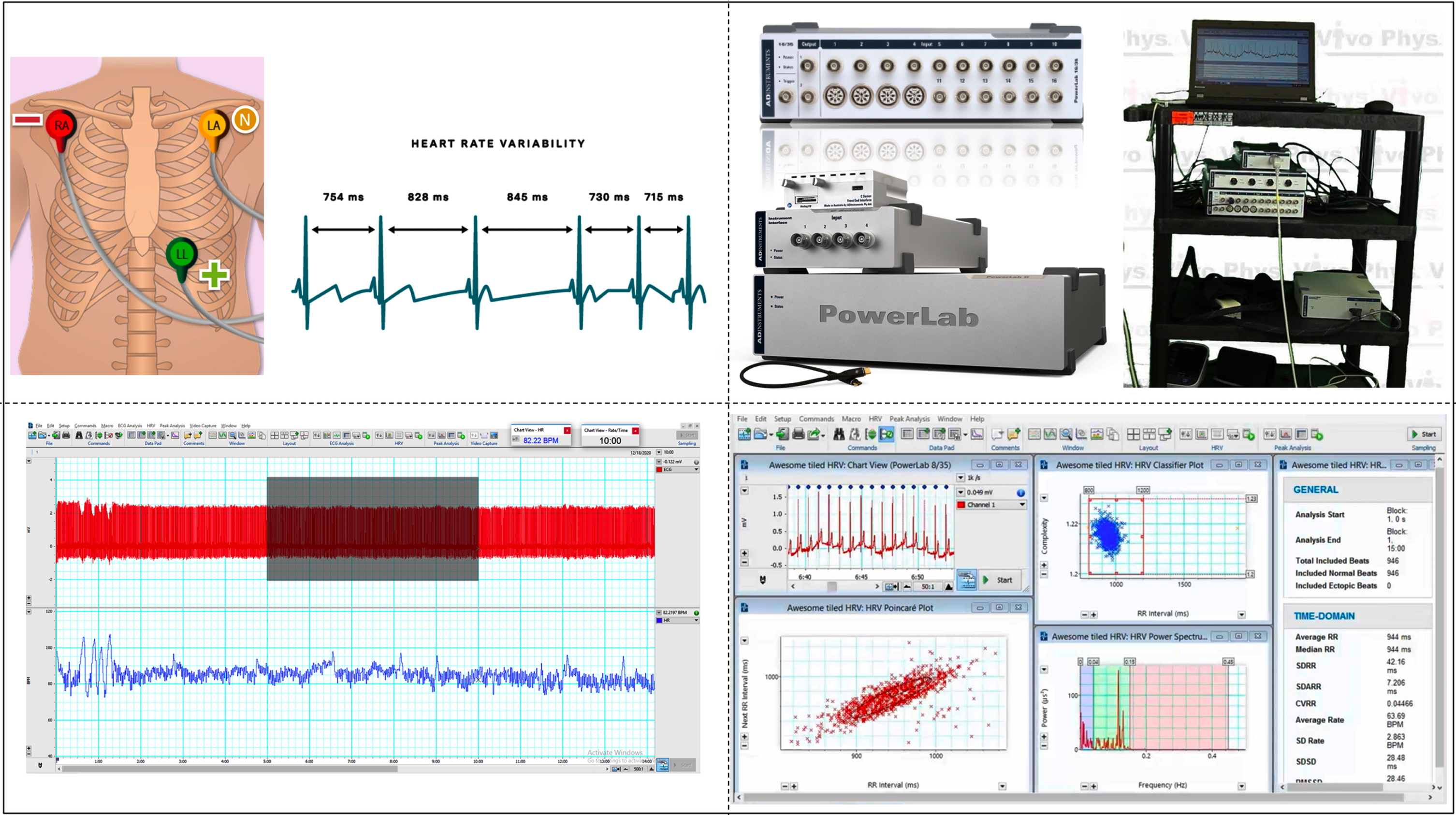
Demonstrates an individual recording and analysis of HRV.
Time-domain analysis
The analysis and comparison of R-R intervals were performed using descriptive statistical methods. Heart rate fluctuations were measured using (a) standard deviation of RR intervals sensitive to all sources of variation (SDNN); (b) root mean square successive difference of RR intervals (RMSSD); the number of NN intervals differing by >50 ms from the preceding interval (NN 50) and the percentage of intervals >50 ms different from the preceding interval (pNN50).
Frequency-domain analysis
For frequency-domain parameters, the non parametric Fast Fourier Transform (FFT) was used. The different FFT components and their corresponding frequency ranges were as follows: (a) total power (TP) (0–0.4 Hz) - reflecting sympathetic and parasympathetic tone; (b) high frequency (HF) (0.15–0.4 Hz) - indicative of parasympathetic/vagal tone and respiration; (c) low frequency (LF) (0.04–0.15 Hz) indicating sympathetic as well as parasympathetic tone. These are then used to determine the LF normalised unit (LF nu) and HF normalised unit (HF nu), which demonstrate sympathovagal balance and represent the proportional value of each component in proportion to the total power minus the VLF component [19].
Blood Pressure Variability (BPV) recording and analysis
The Finometer (Finapres Medical Systems, The Netherlands), a non-invasive beat-to-beat BP monitor attached to the middle finger, was employed to keep track of blood pressure for 15 minutes. The procedure has been explained in detail previously [20, 21]. The Finometer detects and measures cardiovascular parameters using the Model flow method. For off-line analysis, Nevrokard cardiovascular parameter analysis (CVPA) software (version 2.1.0) was used (Fig. 2). The time domain parameters (systolic blood pressure –SBP, diastolic blood pressure –DBP, mean blood pressure –MBP, coefficient of variation –COV) and other cardiovascular parameters like Pulse Pressure (PP), Cardiac Output (CO), Stroke Volume (SV), Ejection Time (ET) and Peripheral Resistance (PR) along with Aortic compliance (AC) and Aortic Impedance (AI) were calculated [22].
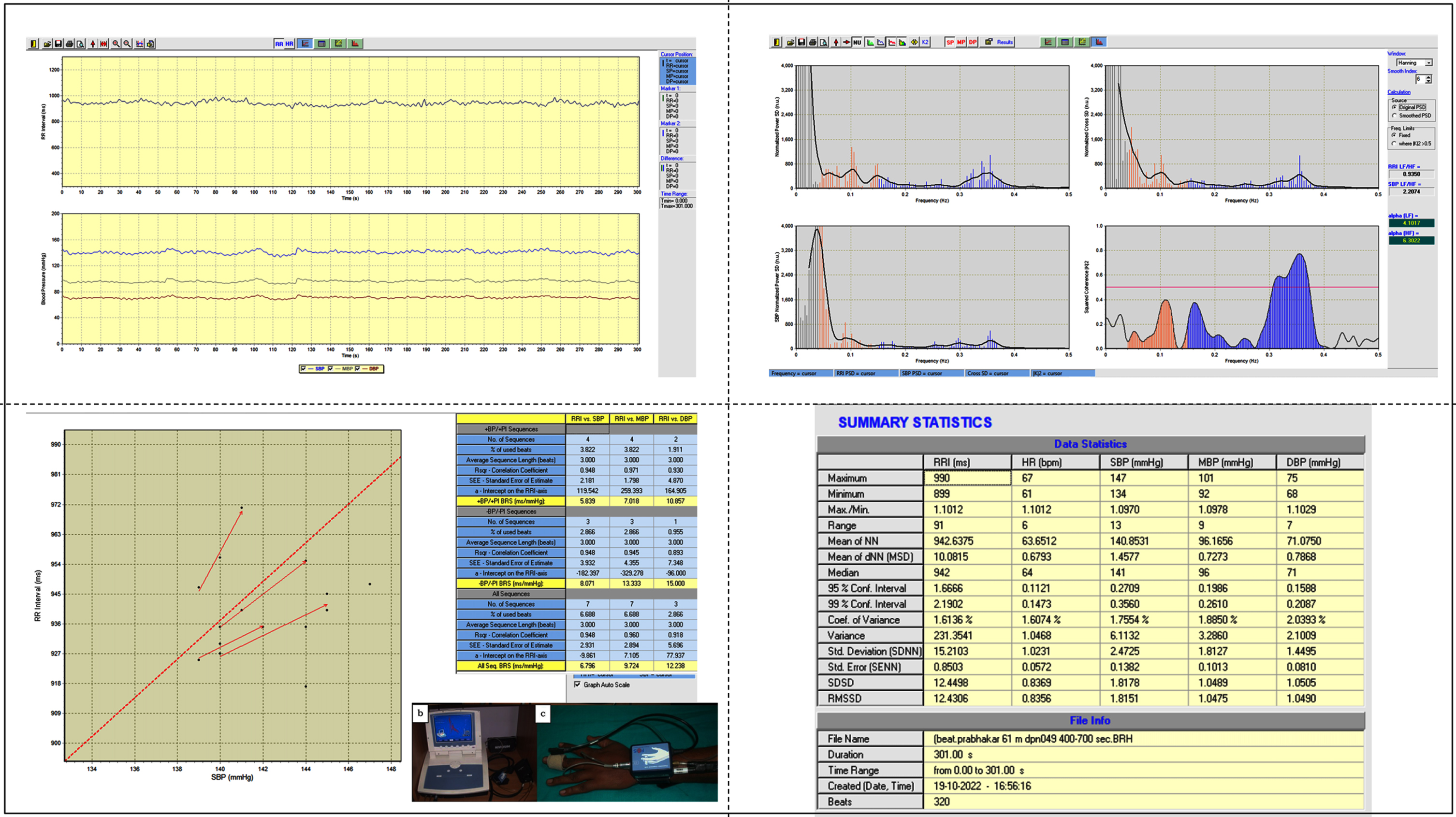
Demonstrates an individual recording and analysis of BPV and BRS.
Baroreflex sensitivity (BRS) recording and analysis
Measuring changes in R-R interval with blood pressure changes gives the BRS that can be calculated using sequence method and/or spectral methods. In the sequence method of BRS measurement, spontaneous beat-to-beat variations in heart rate are measured as a function of the beat-to-beat variations in blood pressure [23]. The sequence approach is based on the recognition of three or more beats in a row in which a progressive rise or fall in systolic blood pressure is followed by a progressive lengthening or shortening of the R-R interval. The threshold settings for considering beat-to-beat systolic blood pressure and R-R interval changes as sequence were 1 mm Hg and 6 ms, respectively. The sensitivity of the reflex was calculated by estimating the slope of the regression line linking changes in systolic pressure to changes in R-R interval. All computed slopes were finally averaged to obtain the BRS (up, down and total BRS) [23]. BRS was determined by measurement of the alpha-index using the spectral method: the transfer function of variability in the systolic pressure signal to the variability in the RR interval. For the spectral BRS method, autoregressive spectral analysis of SBP and R-R intervals was obtained. As a result, the cross-coherence between the low-frequency power of SBP and R-R interval (α-LF) and the cross-coherence between the high-frequency power of SBP and R-R interval (α-HF) was obtained [24].
Statistical analysis
All data were analyzed using IBM SPSS (IBM Corp. Released 2013. IBM SPSS Statistics for Windows, Version 22.0. Armonk, NY: IBM Corp, USA). Shapiro–Wilk test was performed to check the normality of the data. Results of continuous measurements of HRV, BPV and BRS, are presented as Median and interquartile range. ‘Mann- Whitney U’ test was used to assess significant differences in the study parameters between patients and controls. The level of significance was kept at
RESULTS
Phenotypic characteristics of the study cohort
In the DMD cohort, the median age at assessment was 8 years [interquartile range 7–9 years], median age at onset of illness was 3 years [range, 2–6 years] and mean duration of illness was 4 years [range, 2.5–5] (Table 1a). The median height and weight were 119 (Range, 115.75–124) cm and 22.5 (Range, 18.55–26.48) kg respectively. The height was significantly lower compared to the age and weight-matched boys in the control group. All children suffering from DMD had lower limb symptoms and associated truncal weakness. Delayed motor milestones with late age of onset of independent walking were observed in children with DMD (20/38 (52.6%)). Frequent falls with buckling were reported in 28/38 (73.7%) at the time of presentation. Toe walking was seen in 16/38 (42.1%), and none had lost ambulation at the time of examination.
Demographic profile of the study population
Values are expressed as median (Interquartile range: Q1–Q3) and
Further, calf hypertrophy was present in all DMD patients. Kyphoscoliosis was seen in 9/38 (23.6 %). The mean creatine kinase level was 14269±9140.3 IU/L. The majority of the children were on oral steroids: 33/38 were on regular oral prednisolone at 0.75 mg/kg/day, 3/38 were on daily oral deflazacort at 0.9 mg/kg/day, and two patients from the study group were drug naïve at the time of evaluation. No significant difference in socio-economic status was observed between the two groups statistically. The demographic details and the clinical features of the patient and control group are summarized in Table 1a and 1b.
Genotypic characteristics of the DMD children
MLPA results showed exon deletions in 34 and duplications in 4 cases. Family history was positive with a definite affected member in 6/38 cases and consanguineous marriage was present in 6/38 cases. Disruption of the reading frame was present in 33/38 (86.8%), and the in-frame mutations were not in concordance with the reading frame theory in 5 cases. Mutations distal to exon 44 were present in 29 cases, and proximal mutations occurred in 9 cases. Most mutations occurred in the distal hotspot region (Exon 45–55). Single exon deletions were present in 7 patients, and the most common type was exon 45 (2 cases). Among the multi-exon mutations, exon 45–50 deletion was the most common and was present in 4 cases.
The heart rate variability parameters
The median heart rate was significantly higher in DMD patients compared to controls [101.2 (Range, 94.7–108.5)] /min vs [81 (Range, 76.2–92.8)] /min. The median SDNN, RMSSD and all other frequency domain parameters were significantly reduced in DMD children (Table 2).
Heart Rate Variability (HRV) in DMD and healthy children
Values are expressed as median (Interquartile range: Q1–Q3); HR –heart rate; NN –R-R interval (ms); HR –heart rate (bpm-beats per minute); SDNN –Standard deviation of NN intervals (ms); RMSSD- Square root of the mean of the sum of squares of differences between adjacent NN intervals (ms); TP- Total power; LF- Low frequency; HF- high frequency; nu- normalized units.
Blood pressure variability and baroreflex sensitivity parameters
We recorded continuous beat to beat finger arterial BP from 34 DMD and 24 healthy children because the rest of the cohort was restless during recording and uncooperative.
The mean blood pressure and diastolic blood pressure in DMD children were statistically different from the ones in healthy volunteers (Table 3a). The stroke volume was significantly reduced in DMD. The pulse pressure and the cardiac output were also significantly affected in patients along with ejection time. In the DMD patients, the aortic impedance was also significantly increased with reduction in aortic compliance. However, there was no statistical significant change in the coefficient of variation of systolic blood pressure among patients with DMD.
Blood pressure variability (BPV) in DMD and healthy children
Values are expressed as median (Interquartile range: Q1–Q3);SBP –Systolic blood pressure, MBP –Mean blood pressure, DBP –Diastolic BP, MU (medical unit - mmHg.s/mL); lpm- litres per minute; mMU –milli-mmHg.s/mL. Values expressed as median (Interquartile range).
For BRS the values were recorded from 35 DMD and 24 healthy children and the results were compiled in Table 3b.
All BRS parameters (up, down and total BRS) were significantly reduced in children with DMD conveying disturbance in controlling short-term fluctuation of blood pressure. Though the alpha-LF was not significantly different between the two groups, indicating no change in sympathetic tone, there was reduced alpha-HF indicating low baseline parasympathetic tone.
Baroreflex sensitivity (BRS) in DMD and healthy children
Values expressed as median (Interquartile range: Q1–Q3); LF- Low frequency; HF- High Frequency.
Correlation analysis
The age at onset and duration of illness showed a correlation with alpha-HF in HRV (
DISCUSSION
To our knowledge, this is the first study in the literature assessing both short-term HRV as well as BPV and BRS parameters in children with genetically confirmed DMD. This was a prospective hospital-based case-control study with a multimodal evaluation of 38 patients with DMD and 37 healthy volunteers showing a definite early impairment of neuro-autonomic control in DMD before the onset of clinical symptoms.
In our DMD cohort, the motor developmental delay was observed in 52.6 % of cases. They all showed predominantly lower limb symptoms. Dominant features were calf hypertrophy, proximal limb weakness and toe walking. These findings are comparable to earlier studies from India and other countries [25, 26]. The mutational patterns in the DMD gene observed in our cohort are also comparable to large studies in different population groups [27–29]. The concordance to reading frame theory was observed in more than 85% of cases in the present cohort. We therefore conclude that our patient group is an appropriate sample.
The height in the DMD group was significantly lower compared to the age-matched healthy children. We speculate that either chronic steroid usage during the growth spurt or the progressive dystrophic process itself may be the causes [30].
Though the usefulness of HRV in stratifying patients at the highest risk for Sudden cardiac death (SCD) is still controversial [31], the extent of cardiac damage in DMD children is challenging to assess clinically. This is mainly due to their limited physical activity and usually sedentary lifestyle. In addition, the primary cause of death in DMD in developing countries is inadequate supportive care and sudden cardiorespiratory complications [32]. So, by evaluating neuro-autonomic-cardiovascular control using HRV and blood pressure variability, we may obtain cost-effective, non-invasive clinical results that can serve as detectors or early predictors of cardiac dysfunction, thereby helping initiate appropriate pharmacological treatments.
The study results showed a significant reduction in the RR (SDNN) interval and an increased HR. The rise in the HR demonstrates the ineffectiveness of the DMD heart in pumping blood into the peripheral vascular system forcefully. These findings were confirmed by a significant reduction in BPV parameters like stroke volume (SV), cardiac output (CO), ejection time (ET), and aortic compliance. In addition, peripheral resistance and aortic Impedance were significantly increased in DMD compared to healthy children. These findings point to the failure of sympathetic nervous system control over cardiac activity in DMD. However, to our knowledge, none of the earlier studies has demonstrated this pattern of autonomic behaviour in patients with DMD. Therefore, further cardiac evaluation of these patients could provide evidence for any cardiac impairment.
Similar to a previous study [6], a significant difference in the modulation of HRV was observed in DMD children compared to controls, even in the early stages of the disease, before the loss of ambulation. Typically, cardiac complications generally set in after loss of ambulation in DMD [32]. Though findings show low pulse pressure levels in DMD, this did not reflect parasympathetic overplay in HRV. There was also a substantial reduction in BRS parameters. Up, down and total BRS were significantly reduced in DMD, indicating failure of peripheral baroreceptor mechanisms to sustain short-term blood pressure fluctuation.
Literature states that in DMD, beyond the age of 10, boys usually begin to develop cardiomyopathy that is initially asymptomatic with regional wall motion abnormalities that increase with age [33, 34]. It is hypothesized that the mechanism of cardiac muscle involvement is like that of skeletal muscle with dystrophin deficiency. This deficiency leads to loss of sarcolemma integrity, fiber necrosis, and finally, replacement of myocardium with connective tissue or fat [35].
Further, from our records, 12 out of 38 had undergone echocardiography, but only one showed mild Mitral regurgitation (MR) and Tricuspid regurgitation (TR). This indicates that electrical changes in the heart and autonomic dysregulation occur very early compared to structural changes. This is in line with the literature. Dhargave et al. showed sympatho-vagal imbalance in DMD children compared to healthy controls, with the duration of illness ranging between 1 and 8 years [6]. Nigro et al. have noted that heart involvement may emerge at any stage of the disease, and there may be no correlation between cardiac damage and the degree of skeletal muscle damage [36]. The same authors have also investigated the progress of cardiac involvement in DMD, and a variety of asymptomatic ECG changes were found in children under the age of ten [37]. Yotsukura et al. initially described the impairment of the autonomic nervous system in DMD and reported a significant increase in sympathetic activity and a decrease in parasympathetic activity [38]. A study by Lanza et al. and Katliorienė confirmed these observations in DMD patients. The authors reported a marked impairment of the parasympathetic branch [12, 39] and concluded that significantly lower HRV parameter values were noted in patients with DMD. In another study, authors identified early autonomic involvement in ambulatory DMD patients, which was reflected by SDNN changes [12]. Thomas and his colleagues showed that DMD patients had reduced HRV and autonomic dysfunction prior to the onset of heart failure, which is associated with myocardial fibrosis [40]. Thus, our study emphasises the value of monitoring short-term continuous beat-to-beat blood pressure and baroreflex sensitivity parameters in patients with DMD. This method has the advantage of being able to be used in children with DMD in a single setting of BP cuff placement for 15–20 minutes. Thus, as demonstrated in the current study, an early cardiac evaluation may help quickly identify cardiac dysfunction and the subsequent increased risk for SCD in the absence of clinical signs, as well as suggest an appropriate time point for pharmacological intervention. However, the benefit of this approach needs to be validated in a clinical trial.
Moreover, DMD has a strong association between diminished HRV and myocardial fibrosis. These patterns are evident in patients with early-stage DMD and become more prominent as disease severity and age increase. Further, autonomic testing in DMD shows consistent alterations in circadian rhythm and HR variability. It has been proposed that the early increase in HR is due to altered parasympathetic activity, whereas the latter increase is due to increased sympathetic tone. It has been suggested that the persistent activation of the sympathetic nervous system in DMD can be a driving force in the pathological formation of myocardial fibrosis [11].
However, the mechanisms that mediate this autonomic dysfunction remain largely unidentified. Stimulating the exercise pressor reflex, an autonomic reflex originating in skeletal muscle, contributes considerably to the modulation of sympathetic and parasympathetic activity. Since muscular dystrophy causes skeletal myopathy, the function of this reflex may become aberrant as the disease progresses. As such, it may contribute to or aggravate the resulting autonomic dysfunction [41].
The autonomic nervous system is integrally engaged in regulating blood flow to skeletal muscle through modulating cardiac output, blood pressure, and vascular resistance [42]. Therefore, changes in the ability of the autonomic nervous system to control these hemodynamic variables could drastically reduce blood flow to peripheral skeletal muscle. This could initiate and/or exacerbate the primary degenerative processes of the disease.
In cardiac muscle cells, dystrophin has an vital role, identically to skeletal muscle, i.e., the lack of dystrophin can cause an increase in the structural vulnerability of cardiac and vascular smooth muscles, membrane instability, interruption of Ca2+ balance, enlarged production of reactive oxygen species, and mitochondrial dysfunction. Peterson JM et al. [43] demonstrated that the NF-κB of cardiomyocytes impairs the cardiac response to β-adrenergic stress, hence providing the first indication that the classic NF-κB signalling derived from cardiomyocytes plays an active role in the promotion of dystrophic cardiac dysfunction with an interruption in calcium homeostasis is considered the cause for the pathology of dystrophic hearts. The study also demonstrated that pharmacological inhibition of NF-κB in mdx mice for only one month can reverse existing stress-induced cardiac muscle dysfunction and that inhibition of NF-κB systemically and specifically in cardiomyocytes reverses the pathological disability of cardiac muscle preparations for responding to β-adrenergic stimulation.
In the clinical setting, pharmacological inhibition of NF-κB via corticosteroid therapies has been shown to improve adrenergic responses and cardiovascular autonomic response, as evidenced by an increase in HRV in people with DMD [44]. Thus NF-κB plays a critical role in the pathophysiology of cardiac autonomic dysfunction in DMD.
Further, the context in which cardiac autonomic dysfunction can be taken as indicative of underlying cardiac disease is based on the bidirectional relationship between the inflammation/immune system and the central nervous system [45, 46]. It has been postulated that the central nervous system can regulate immune system function through two plausible mechanisms: receptor-specific pro- or anti-inflammatory effects of noradrenaline (norepinephrine) released by sympathetic postganglionic fibers on primary and/or secondary lymphoid organs [46]; or the direct anti-inflammatory effects of acetylcholine on splenic macrophages (the source of acetylcholine in the spleen is still unclear), which suppress the release of tumor necrosis factor (TNF) and other cytokines (Tracey, 2002). Conversely, the immune system can also modulate autonomic activity via the activation of afferent neural inputs from the lymphoid organs to the brain centers responsible for autonomic regulation [45, 46], through the direct effects of inflammatory markers accessing autonomic brain centers devoid of a blood–brain barrier [45], or via the direct effects of cytokines produced locally in the brain by glia [45]. This reflex arc, which has been termed the ‘inflammatory reflex’ by Tracey et al. (2002), is an integrative response that aims to offer a rapid, balanced, and reversible defensive response to the presence of pathogens, injury, and disease. However, under conditions of altered homeostasis, the impairment of one axis of this reflex arc may seriously compromise the other, and it is plausible that this neural–immune imbalance will occur in a number of inflammatory conditions, resulting in the development and progression of cardiovascular disease [47]. This mechanism pertaining to inflammation and cardiovascular autonomic dysfunction was explained earlier in disease conditions pertaining to rheumatoid arthritis (RA) and diabetes mellitus by Tiago Peçanha et al. [47]; Francesca Ingegnoli et al. [48]; Gaetano Antono Lanza et al. [49]. It has been shown that serum C-reactive protein levels are significantly associated with depressed HRV. Cardiovascular involvement, in terms of reduced baroreflex sensitivity and heart rate variability-derived indices, is a common comorbidity in patients with RA and has been linked to autonomic nervous system dysfunction, which in turn is influenced by increased levels of circulating pro-inflammatory cytokines. The majority of common cardiac and mental health conditions associated with RA comorbidities had potential links to systemic and joint inflammation. Similar evidence might exist in DMD that the circulating inflammatory activators and by-products of inflammation, most of which originate in skeletal muscle, might lead to the development and progression of cardiovascular disease in children with DMD.
We acknowledge the limitations in this study. The lack of echocardiogram in a majority of the study population is not available for correlation studies. Similarly, the number of children with proximal mutations in DMD gene is low and might limit the inference of the correlation analysis between mutation sub-types. Further, the study needs to be tested in a clinical trial design.
CONCLUSION
In this study, we showed alterations in autonomic tone in DMD. DMD children observed a loss of vagal tone and an increase in sympathetic tone, appearing to have a multifactorial origin in the absence of significant structural changes of the heart. This confirms the utility of these measures as predictors of adverse cardiac outcomes in DMD. Further, the age at onset and duration of illness showed a significant correlation with ANS activity. This suggests that HRV, BPV and BRS can be used as a preclinical markers of cardiovascular complications in DMD patients, offering therapeutic intervention to prevent SCD.
CREDIT AUTHORSHIP CONTRIBUTION STATEMENT
All authors approved the final manuscript as submitted and agree to be obliged for all aspects of the work.
DATA AVAILABILITY
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
