Abstract
Background:
Myotonic dystrophy type 1 (DM1) is a multisystemic neuromuscular disorder causing a plea of impairments, of which fatigue and apathy are some of the most frequent non-muscular symptoms. No curative treatment exists to date, and patients only have access to limited effective care, which are intended to decrease the burden of specific symptoms in daily life.
Objective:
This study aimed to assess whether a 12-week strength training program has an impact on fatigue/daytime sleepiness, apathy, and disease bruden in men with DM1.
Methods:
Eleven participants completed the Fatigue and Daytime Sleepiness Scale (FDSS) and the Myotonic Dystrophy Health Index (MDHI) at baseline, at 6 and 12 weeks, and at 6 and 9 months. Also, the Apathy Evaluation Scale (AES) was filled out at baseline, at 12 weeks, and at 6 and 9 months.
Results:
Results show significant effects of the training program both on apathy and fatigue/daytime sleepiness, effects that are respectively greater at three and six months after the end of the program than at its very end. However, no difference was observed regarding the overall disease burden.
Conclusion:
These findings are promising for patients with DM1 considering that few non-pharmacological treatments are available.
ABBREVIATIONS
Clinician version of the Apathy Evaluation Scale
Cytosine, thymine and guanine
Myotonic dystrophy type 1
Excessive daytime sleepiness
Fatigue and Daytime Sleepiness Scale
Hospital and Anxiety Depression Scale
Myotonic Dystrophy Health Index
Nonparametric longitudinal data test
Relative treatment effect
INTRODUCTION
Myotonic dystrophy type 1 (DM1), the most prevalent adult-onset neuromuscular disease [1, 2], is a progressive genetic condition caused by abnormal cytosine, thymine and guanine (CTG) repetitions on the Dystrophy Myotonic Protein Kinase gene [3]. DM1 may be divided into five different phenotypes according to the age at onset of symptoms and severity: congenital, infantile, juvenile, adult, and late-onset [4]. DM1 is a multisystemic disease that affects namely the cardiac, endocrine, digestive, musculoskeletal and central nervous systems [2]. As for behavioral symptoms, they may consist of avoidant personality disorder [5], apathy [6–8], daytime sleepiness and fatigue [9, 10]. Daytime sleepiness and fatigue are frequently referred to as the most frequent non-muscular symptoms of the condition, being present respectively in 88% and 91% [11]. These latter symptoms are associated with higher depressive levels, lower quality of life, and disrupted social participation in individuals with DM1 [12–14]. Apathy, defined as a reduction of initiative, interest, and emotion related to goal-directed behaviors, is also associated with decreased quality of life [15] and is a significant predictor of restriction of social participation in DM1 [7]. In addition, higher levels of fatigue and daytime sleepiness were associated with sleep-related complaints in persons affected by DM1 [7, 10].
Without curative treatment, care aiming to reduce the patient-reported burden of symptoms is highly relevant. There is strong research evidence for the efficacy of exercise as a non-pharmacologic treatment option for disturbed sleep [16]. Moreover, exercise training, and notably strength (or “resistance”) training, has been shown to have positive effects on self-rated levels of fatigue [17–23], daytime sleepiness [24, 25], and sleep quality [26] in various medical conditions. Some authors advanced that exercise leads to better sleep quality which in turn results in lower fatigue and daytime sleepiness levels, and higher levels of daily activities and motivation [19, 20]. A recent 10-month cognitive behavioral therapy (CBT) with optional graded exercise therapy in severely fatigued adult DM1 patients [27] found increased levels of physical activity and exercise capacity, higher self-perceived capacity for activity and social participation, and a decrease in experienced fatigue. However, no evidence of improved self-reported quality of life or decreased apathy was observed.
We conducted a 12-week supervised strength training program that aimed to maximize muscle function in men with DM1, for which results related to muscle adaptations (strength, functional capacity, and change in fiber type and size) were previously published [28].
The objective of this study was to evaluate the immediate and medium-term effects of a 12-week lower limb strength training program on symptoms of fatigue/daytime sleepiness, apathy, and patient-reported health.
MATERIALS AND METHODS
Recruitment
Participants were recruited among DM1 patients followed at the neuromuscular clinic of Saguenay, Québec, Canada. Inclusion criteria were being aged between 30 to 65 years, having a DM1 diagnosis confirmed by genetic analysis, and being able to walk without assistance and give informed consent. The exclusion criteria were to present the congenital phenotype of DM1 and to have a contraindication for maximal strength testing and training or muscle biopsy. Further details on the procedures for DM1 patients’ selection can be found elsewhere [28] (Appendix 1 for the complete list of inclusion/exclusion criteria).
The study was approved by the Ethics Review Board of the Centre intégré universitaire de santé et de services sociaux of Saguenay–Lac-Saint-Jean (Québec, Canada) and all participants gave a written informed consent.
Training program
This study is part of a larger project on the effects of a 12-week lower limb strength training program on muscle strength, vastus lateralis fiber size, and function in men with DM1 [28]. All participants completed a 12-week training program composed of two weekly sessions. Evaluation sessions occurred at baseline (T1), at week 6 (T2), and at week 12 (T3) as well as at six months (T4) and nine months (T5) after the beginning of the program. All 21 sessions of training were supervised by either trained physiotherapy student or physiotherapists (M-PR or ED). At each session, participants performed five lower limb training exercises (leg extension, leg press, hip abduction, squat, and plantar flexion) and completed 3 series of a maximum of 6–8 repetitions of each, with a 3-minute rest between each series. Load for each exercise was determined by individual trials carried out in the first session and adjusted for each participant throughout the program to ensure that they can complete a maximum of 6–8 repetitions of movement in the full range of motion of the targeted exercise before failure.
Clinical measurements
All evaluations and questionnaires were conducted or supervised by an experienced psychologist (BG). Figure 1 shows the study and assessment timeframe.
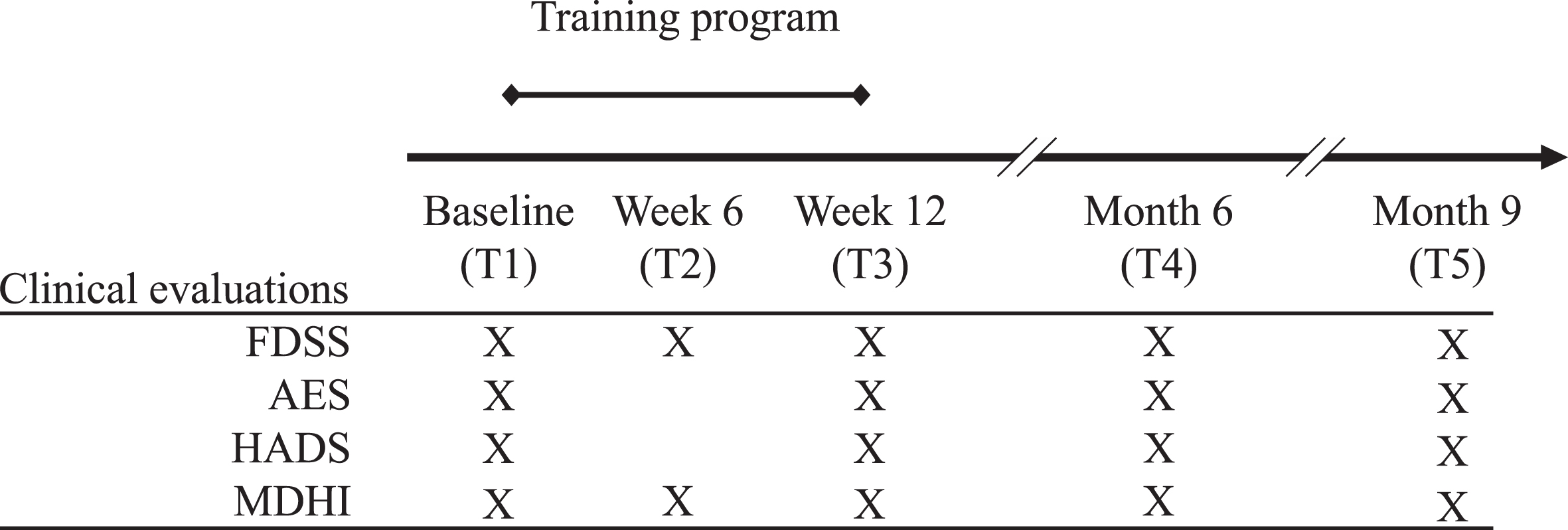
Protocol evaluation session’s timeline and studied variables. The different evaluation session times are marked on the timeline. An “X” marks the corresponding evaluation period in which tests and evaluations took place. The training program was completed twice a week between the baseline and week 12. Fatigue and Daytime Sleepiness Scale: FDSS; Apathy Evaluation Scale: AES; Hospital Anxiety and Depression Scale: HADS; Myotonic Dystrophy Health Index: MDHI.
Fatigue and excessive daytime sleepiness
Fatigue and daytime sleepiness were assessed using the 12-item Fatigue and Daytime Sleepiness Scale (FDSS) [29], a scale specifically devised for DM1. It provides interval measures of fatigue and daytime sleepiness on a single continuum addressing the same health construct. In its validation study, the FDSS fulfilled all Rasch model expectations. Moreover, an additional study confirmed the use of the FDSS as a reliable and valid instrument to measure fatigue and daytime sleepiness in patients with DM1 for either clinical or research purposes [30]. The raw sum score varies from 0 to 24 (24 indicating higher levels of fatigue and daytime sleepiness).
Apathy
Apathy was assessed using the clinician version of the Apathy Evaluation Scale (AES-C) [31], a commonly used scale that has been identified as a promising tool to assess apathy in DM1 [32]. The AES-C contains 18 items based on the subject’s thoughts, emotions, and actions over the last month. Specifically, clinicians have to rate participants’ level of apathy based on a semi-structured interview including questions on their interests, activities and daily routines. The total score varies between 18 and 72 points, a higher score indicating a higher level of apathy. The AES-C presents good internal consistency when used in DM1 (Cronbach’s Alpha = 0.87) [33].
Disease burden and impact of key symptoms
The Myotonic Dystrophy Health Index (MDHI) [34] was used to assess patients’ overall disease burden and the impact of key symptomatic themes. The MDHI is a disease-specific patient-reported outcome measure composed of 114 questions. It has an excellent test-retest reliability (ICC = 0.95) and can differentiate cohorts of DM1 patients having a low or high disease burden. Three subscales were selected for their theoretical proximity to the main variables of the present study, namely Ability to do activities (14 items), Fatigue (4 items), and Sleep (4 items).
Anxiety and depression
Anxiety and depression levels were assessed using the Hospital Anxiety and Depression Scale (HADS) [35]. This self-administered scale contains 14 items divided into two subscales: anxiety (7 items) and depression (7 items). One advantage of this scale is that it contains no item that assesses somatic symptoms which could be confounded with symptoms of DM1 and increase artificially depression/anxiety scores.
Data analysis
Effects of the training program on symptoms of fatigue/daytime sleepiness, apathy, disease burden, and anxiety/depression were tested with nonparametric repeated measures ANOVA especially suited for longitudinal factorial designs with small samples (R 3.5.0, package nparLD version 2.1) [36]. This type of rank-based ANOVA has no assumption about distributions and is robust to outliers and changes in distribution shapes across time. No assumption is made about the covariance matrix. The nparLD posthoc tests were performed with Wilcoxon’s signed-rank test for the intra-group comparisons. Post-hoc analyses comprised four evaluation times (T1 –baseline, T3 –week 12, T4 –month 6 and T5 –month 9), and five time-points comparisons: the effect of the whole training program assessed at the end of the training program (T1–T3: significant positive changes in scale scores are expected); the effect of the whole training program assessed three months after the end of the training program (T1–T4: significant positive changes in scale scores are expected); the effect of the whole training program assessed six months after the end of the training program (T1–T5: significant positive changes in scale scores are expected); the maintained effects of the training program until month 6 (T3–T4: no change or positive changes in scale scores are expected); the maintained effects of the training program until month 9 (T3–T5: no change or positive changes in scale scores are expected).
Differences were considered significant if p-value <0.05. Results are presented as raw scores at baseline (for clinical consideration), then as Relative treatment effect (RTE) values. RTE values vary between 0 and 1, with 0.5 as the null hypothesis value. When RTE is above 0.5, values within this condition are generally higher than random values drawn from any other conditions. The medium effect size requires values either above 0.64 or below 0.36 [37]. Effect size is considered high when RTE scores are outside the range [0.29–0.71] [37]. In this condition, no additional correction for multiple comparisons had to be performed.
RESULTS
Participants
Of the 32 eligible patients, 18 refused to participate, two could not be reached, and one was under investigation in cardiology. Eleven men with DM1 completed the 12-week training program (see Table 1 for participants’ characteristics and all baseline scores). There were no dropouts during the training program. No patient missed more than 2 sessions out of 21 (giving a minimum of 90.5% training attendance and a mean attendance of 97% for the whole group).
Participant characteristics and baseline scores for fatigue/sleepiness, apathy, anxiety, depression and the patient perception of their health
All participants were men with DM1. Abbreviations: AES-C: clinician version of the Apathy Evaluation Scale; CTG: cytosine-thymine-guanine repeats; FDSS: Fatigue and Daytime Sleepiness Scale; HADS: Hospital and Depression Scale; MDHI: Myotonic Dystrophy Health Index.
Fatigue/daytime sleepiness
A significant overall training program effect on fatigue/daytime sleepiness was observed (p < 0.01) (Table 2). More particularly, FDSS scores significantly decreased between T1 and T5 (p = 0.024) and tended to decrease between T1 and T3 (p = 0.057) (Table 3). No change was observed between T1 and T4 (p = 0.121), between T3 and T4 (p = 0.84), and between T3 and T5 (p = 0.28) (Table 3). However, the position of the FDSS RTE scores from T1 to T5 suggests a medium treatment effect (Fig. 2A).
Assessment times relative treatment effect and overall training effect
Overall training effects were estimated with nonparametric repeated-measures ANOVA. *p < 0.05. **p < 0.01. Abbreviations: FDSS: Fatigue and Daytime Sleepiness Scale; AES: Apathy Evaluation Scale; HADS: Hospital Anxiety and Depression Scale; MDHI: Myotonic Dystrophy Health Index.
Between assessment times’ pairwise analyses
Abbreviations: FDSS: Fatigue and Daytime Sleepiness Scale; AES: Apathy Evaluation Scale; HADS: Hospital Anxiety and Depression Scale; MDHI: Myotonic Dystrophy Health Index.
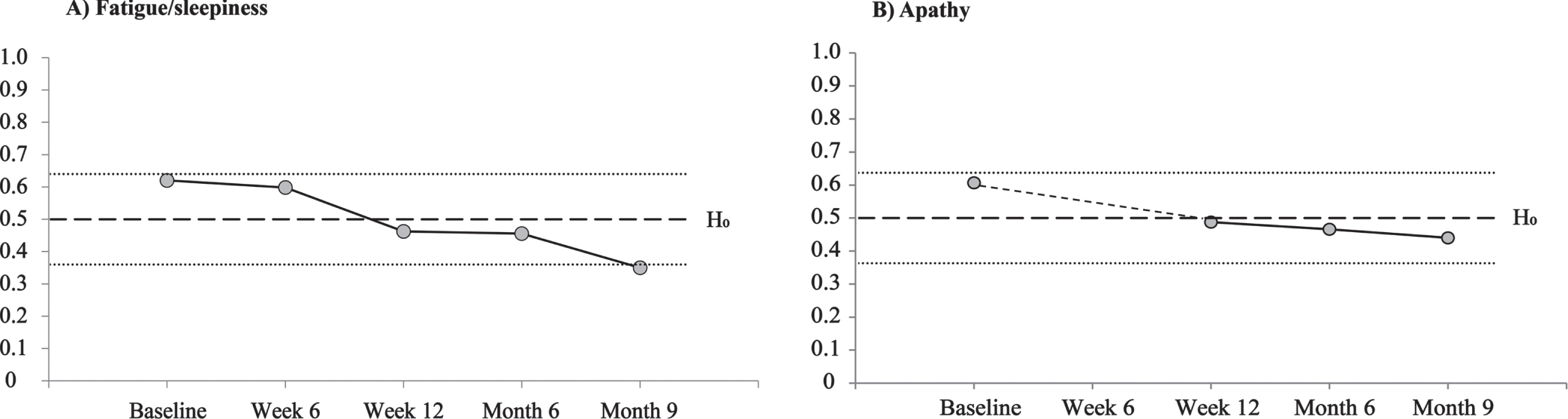
Evolution over the five evaluation periods of A) fatigue/sleepiness and B) apathy, presented in Relative treatment effect values. The two small dotted lines up and down the null hypothesis (H0; Relative treatment effect = 0.5) represent the medium effect size (Relative treatment effect = 0.36 and 0.64).
Apathy
Regarding the AES-C, a significant overall training program effect on apathy was observed (p < 0.05) (Table 2). More precisely, AES-C total score significantly decreased between T1 and T3 (p = 0.018) and between T1 and T4 (p = 0.025) and tended to decrease between T1 and T5 (p = 0.055). No change was observed between T3 and T4 (p = 0.47) nor between T3 and T5 (p = 0.84) (Table 3). Effect size cannot be considered medium, but RTE scores were over the 0.5 line at baseline and were below this line at week 12 (Fig. 2B).
Overall disease burden and impact of key symptoms
No significant overall training program effect was observed for MDHI total score as well as for scores of Ability to do activities, Fatigue, and Sleep subscales (Table 2).
Anxiety and depression
No significant overall training program effect was observed for anxiety and depression levels (Table 2).
DISCUSSION
This is the first study, to our knowledge, to show that a 12-week strength training program can reduce debilitating neurobehavioral symptoms known to exact a heavy toll on DM1 patients. More particularly, results indicated that the effects of the training program on apathy and fatigue/daytime sleepiness levels were greater three and six months after the end of the program, respectively. In line with the recent review by Kline and colleagues (2021) [16], the present results support the view that physical activity improves sleep outcomes. Indeed, clinical, neurophysiological, and histopathologic findings support the hypothesis that hypersomnia-like symptoms of DM1 patients may represent a manifestation of a primary central disturbance of sleep regulation. It must be borne in mind that DM1 is reportedly a progeroid disease that is characterized by restricted social participation and that less working outside of the home and social isolation can contribute to sleep issues. Hence, there is a distinct possibility that the 12-week strength training program improved social participation in addition to activity levels per se, thus contributing to lesser fatigue and daytime sleepiness symptoms. More generally, one can assume that a supervised strength-training program can induce significant strength and functional gains in men with DM1, gains that may in turn increase feelings of energy, motivation, confidence, and interest in activities.
Our results are in line with the results of the only study to date showing a positive effect on fatigue/daytime sleepiness using a non-pharmacological treatment in DM1, namely a CBT and standard care plus optional graded exercise in a large randomly selected sample of patients with severe fatigue [27]. Since exercise was optional in this latter study, its specific effect on fatigue compared to those of CBT cannot though be specified. Considering that the strength training program used herein relied on a highly standardized protocol and that positive effects were found for both fatigue/daytime sleepiness and apathy, such a training program could advantageously be used in future multicenter design studies or randomized clinical trials to diminish disabling neurobehavioral symptoms.
In addition, results showed mounting effects of exercise on DM1 patients’ apathy levels over 6 months after the end of the program. In the study by Okkersen and colleagues (2018) [27], the prospective 16-month follow-up did not show a significant change in apathy levels [27] despite increased perceived capacity for activity and social participation. As previously advanced [38], a treatment solely based on thoughts and emotions such as CBT may not be sufficient to reduce apathy in patients with DM1, while a more concrete anchorage such as perceived physical improvement might be required.
On the other hand, the 12-week strength training program did not affect self-reported anxiety and depression levels. These symptoms were primarily assessed as control variables to evaluate potential emotional adverse effects of the program. Fortunately, no such impact occurred in the present study. On the contrary, 3 out of 11 participants reported “better mood” as the most self-reported changes related to the program [28]. It is possible that, for participants, perceiving themselves as stronger and in a better physical shape had a positive impact on their mood.
According to the participants’ perception of Sleep, Fatigue, and Ability to do activities assessed with the MDHI, the absence of a significant effect may be surprising considering that AES and FDSS revealed an effect, and both share conceptual proximity with these MDHI subscales. However, a closer look both at the instructions to the respondents and at the nature of items may bring some answers. Regarding first the instructions, the MDHI asks patients “How much are the following things currently affecting your life?” One can observe that it does not aim to assess the severity of a given symptom. Indeed, the MDHI was specifically devised to estimate the overall disease burden and impact of key symptomatic themes in DM1 patients [34]. In addition, the Sleep subscale includes one item on daytime sleepiness, one item on excessive need for sleep, and one item on difficulty falling asleep at night. Hence, these items address distinct sleep phenotypes of DM1 and it should also be stressed that several DM1 patients with EDS do not exhibit prolonged nocturnal sleep and/or long sleep latencies [13]. While it was demonstrated that the FDSS measures a single factor [29], the factorial and convergent validity of the MDHI Fatigue and Sleep subscale remains, to our knowledge, to be demonstrated.
Study limitations
One important drawback of this study is the small number of participants. Another limitation is the absence of a control group that would lend greater credence that significant outcome improvements stem from the strength training program per se. Also, the control group should ideally be exposed to low levels of muscle stimulation (e.g., an aerobic program) while controlling for potential positive effects (e.g. participating in a training program, being part of a study, being stimulated by therapists) that can account for variations in perceived symptoms of fatigue, sleepiness, and motivation. DM1 is a very slowly progressive disease and both fatigue and daytime sleepiness are not expected to change significantly over short periods in the absence of a given intervention. Moreover, our long-term natural history study revealed an increase in symptoms rather than a decrease [39]. In addition, the FDSS does not allow to distinguish fatigue symptoms from daytime sleepiness symptoms. Forthcoming evaluations and studies which perform a strength training program could use distinct standardized fatigue scales and daytime sleepiness scales in DM1 patients with severe fatigue and daytime sleepiness. Finally, sleep-wake patterns were not assessed by either a sleep diary or actigraphy.
CONCLUSIONS
This study revealed that a 12-week strength training program can significantly reduce fatigue/daytime sleepiness and apathy levels over 6 months in men affected by DM1. Since these latter symptoms are known to impact negatively on social participation and quality of life, it may bring major benefits for DM1 patients and their relatives to engage in a strength training program or other interventions designed to augment activity levels, including lasting functional gains [28].
Further studies should assess the additional contribution of CBT on strength training programs on fatigue, daytime sleepiness, and apathy of DM1 patients using namely focus group interviews. Indeed, occupational therapists and psychologists may help participants in raising awareness of the growing confidence in their strength, energy, and capacities and therefore support them to transfer it into increased actual activities and daily-life participation.
Despite limitations, the present findings are promising and remind us that when strategies for physical and functional rehabilitation are tested, it is highly relevant to also consider the assessment of possible secondary benefits on more “subjective” symptoms. Finally, the positive and persistent effects found 6 months after the end of the program are quite encouraging given the paucity of non-pharmacologic treatments available for DM1 patients and the heavy toll on the quality of life of these debilitating symptoms.
DECLARATION OF COMPETING INTEREST
All authors report having no conflicts of interest to disclose.
Footnotes
ACKNOWLEDGMENTS
The authors of this study would like to thank all the participants who graciously gave their time and were invested in the completion of this highly demanding training program. We would like to thank Dr Cynthia Gagnon for the gracious support of her research team, the Groupe de recherche interdisciplinaire sur les maladies neuromusculaires. We would also like to thank Dr. Jean Leblond for his support with the statistical analyses. We will also thank the Université du Québec à Chicoutimi, more specifically to the staff of the sports pavilion for the access to the equipment and rooms for training and evaluations.
FUNDING
This project was funded by the Fondation du Grand Défi Pierre Lavoie. Dr. Elise Duchesne is supported by a Chercheur-boursier Junior 1 salary award from the Fonds de recherche du Québec-santé (FRQS-311186). Marie-Pier Roussel holds a Ph.D. study grant from the Fonds de recherche du Québec-santé.
APPENDIX 1: INCLUSION AND EXCLUSION CRITERIA
Inclusion criteria DM1 diagnosis must be confirmed by genetic analysis; Male gender, aged between 30 and 65 years old; Be able to walk without assistance; Consent of the neurologist must be given to participating in this study; Must reside in the Saguenay-Lac-St-Jean region Subjects must be able to give their consent freely and voluntarily. Exclusion criteria Patients with any other form of muscular dystrophy are excluded; Finally, patients with any contraindications listed in the table below were excluded (as presented in their medical files):
