Abstract
Background. Fatigue is a common, persistent complaint following traumatic brain injury (TBI). Effective treatment is not well established. Objective. The current study aimed to investigate the efficacy of 4 weeks of light therapy for fatigue in patients with TBI. Methods. We undertook a randomized, placebo-controlled study of 4-week, 45 min/morning, home-based treatment with short wavelength (blue) light therapy (λmax = 465 nm, 84.8 µW/cm2, 39.5 lux, 1.74 × 1014 photons/cm2/s) compared with yellow light therapy (λmax = 574 nm, 18.5 µW/cm2, 68 lux, 1.21 × 1012 photons/cm2/s) containing less photons in the short wavelength range and a no treatment control group (n = 10 per group) in patients with TBI who self-reported fatigue and/or sleep disturbance. Assessments of fatigue and secondary outcomes (self-reported daytime sleepiness, depression, sleep quality, and sustained attention) were conducted over 10 weeks at baseline (week −2), midway through and at the end of light therapy (weeks 2 and 4), and 4 weeks following cessation of light therapy (week 8). Results. After controlling age, gender, and baseline depression, treatment with high-intensity blue light therapy resulted in reduced fatigue and daytime sleepiness during the treatment phase, with evidence of a trend toward baseline levels 4 weeks after treatment cessation. These changes were not observed with lower-intensity yellow light therapy or no treatment control conditions. There was also no significant treatment effect observed for self-reported depression or psychomotor vigilance performance. Conclusions. Blue light therapy appears to be effective in alleviating fatigue and daytime sleepiness following TBI and may offer a noninvasive, safe, and nonpharmacological alternative to current treatments.
Keywords
Introduction
Traumatic brain injury (TBI) is a leading cause of disability, with increasing prevalence among war veterans. 1 Fatigue is a common, persistent, and disabling consequence of TBI, reported by more than 60% of patients with TBI. 2 Its etiology is not well understood, although both primary (ie, brain pathology) 3 and secondary factors (depression, anxiety, and sleep disturbances)4-7 are implicated. Reported sleep disturbances include hypersomnia, insomnia, and other changes to the sleep–wake cycle, 8 although excessive daytime sleepiness (EDS) is one of the most commonly reported sleep complaints. 9
Effective treatments for fatigue and associated symptoms following TBI are limited. Pharmacological approaches include stimulant use to improve daytime symptoms and/or hypnotics to improve sleep 3 ; however, their use in patients with TBI is not well investigated and may be associated with adverse effects. Recent findings using the wake promoting agent modafinil in patients with TBI have been mixed.10,11 Given the interrelatedness of fatigue with daytime sleepiness and lowered mood following TBI, 6 a treatment approach addressing all these symptoms seems optimal.
Ocular light exposure can be both a stimulant and an antidepressant, eliciting circadian, neuroendocrine, and neurobehavioral responses. These “nonvisual” responses include resetting the circadian pacemaker, 12 acute alerting effects,13,14 and mood enhancement. 15 These effects are intensity-dependent 16 and are likely mediated primarily by a novel nonrod, noncone photoreceptor system. A subset of intrinsically photosensitive retinal ganglion cells (ipRGCs) express the blue light–sensitive photopigment melanopsin (λmax ~480 nm) and project directly to brain areas involved in nonvisual light responses. 17 In laboratory settings using controlled photon exposures, greatest improvements in alertness and depressed mood have been observed with exposure to blue or blue-enriched light.13,18 Similarly, daily home-based blue light therapy has been shown to improve depressive symptoms in seasonal affective disorder.19,20 Subjective improvements in fatigue have also been noted in patients with cancer with increasing exposure to light,21,22 as well as in healthy office workers exposed to blue-enriched white light.23,24
The primary aim of current the study was to investigate the efficacy of blue light therapy in reducing fatigue in patients with TBI.
Methods
Participants
Participants were aged 18 to 65 years, had sustained a TBI at least 3 months earlier, and self-reported significant fatigue (Fatigue Severity Scale [FSS] ≥ 4) 25 and/or sleep disturbance (Epworth Sleepiness Scale [ESS] ≥ 10 and/or Pittsburgh Sleep Quality Index [PSQI] > 5)26,27 (Figure 1). They were recruited from the TBI rehabilitation program at Epworth Hospital, Melbourne, Australia, and screened by telephone. Exclusion criteria included presence of other medical illness accounting for fatigue, including other neurological disorders, psychiatric illness requiring hospitalization, preinjury sleep disorders, or chronic fatigue syndrome; obesity or estimated high risk of Obstructive Sleep Apnea on the Berlin Questionnaire 28 ; transmeridian travel or night shift work in the preceding 6 weeks; and current use of sleep medications (prescribed or over-the-counter). Treatment with antidepressants was permitted, provided participants maintained a stable dose during study participation.
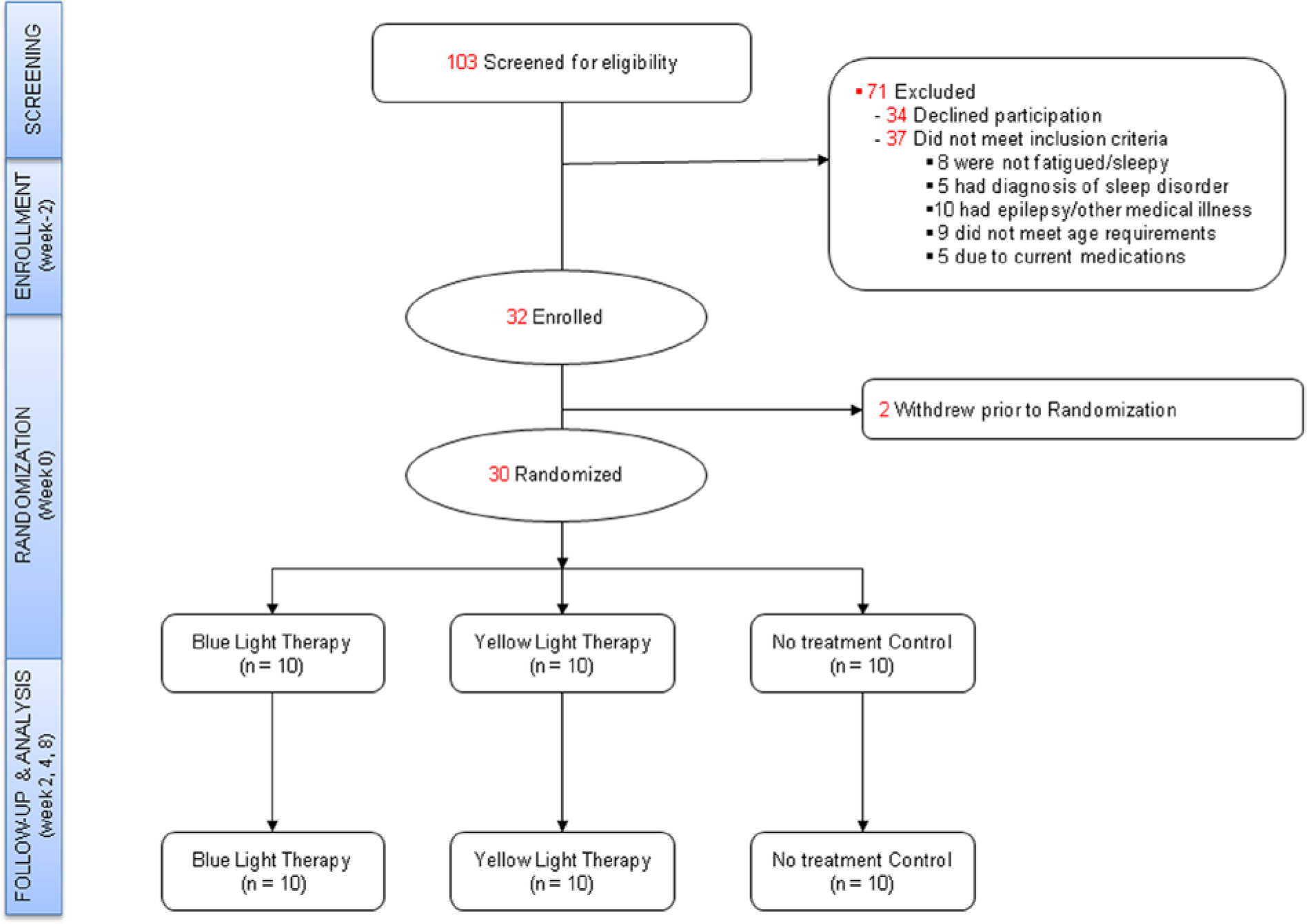
Flow of participants through the screening, recruitment, and follow-up phases of the study (CONSORT).
Procedures
The study had a randomized, placebo-controlled design, investigating 4-week, 45 min/day home-based treatment with blue light therapy (goLITE, Philips Consumer Lifestyle, American Fork, UT) compared with yellow light therapy or no treatment controls. The study was conducted between February 2009 and September 2011.
The protocol lasted 10 weeks and was divided into 3 phases: baseline (2 weeks), treatment (4 weeks), and follow-up (4 weeks; see Supplemental data for further detail; available online at http://NNR.sagepub.com/supplemental). It involved 5 home-based study assessments scheduled approximately 2 weeks apart (±3 days) and at approximately the same time of day for each participant in order to minimize the impact of time-of-day effects on assessments. Following baseline assessment (week −2), participants were randomized (week 0) to 1 of the 3 treatment conditions for the 4-week treatment phase (weeks 0-4). Randomization into 1 of the 3 treatment conditions was conducted using a concealed online-generated randomization plan (http://www.randomization.com), with 1:1:1 allocation. Due to the nature of the intervention, participants and research staff were not masked to treatment allocation. Participants were not informed of the expected efficacy of either light therapy condition and were told that the study aimed to investigate the effectiveness of both wavelengths on fatigue. Prior to treatment, participants rated their expectation of efficacy from 0 to 5 (0 = not helpful; 5 = complete remission of fatigue/sleep symptoms). Those randomized to 1 of the 2 light therapy conditions were instructed to use the device for 45 minutes each morning, within 2 hours of waking, throughout the 4-week treatment phase. During light exposure, participants were instructed to sit in front of the light panel with its centre approximately 50 cm in front of the eyes and to look into the light source for a few seconds at least every few minutes. Those randomized to the no treatment control group, as with all participants, were permitted to continue other medical, rehabilitative, or psychological interventions provided that the treatment regimen/dose did not change throughout the study treatment period. Concurrent therapies were documented.
Outcome variables were assessed during home-based visits at baseline (week −2), midway through treatment phase (week 2), at end of treatment (week 4), and again 4 weeks later (week 8; ±3 days). All participants were reimbursed $50 for study-related expenses. The study was approved by the human research ethics committees at participating institutions, and all participants provided written informed consent.
Light Treatment Devices
Both light conditions used the Philips goLITE M2 light therapy device (Apollo Health, American Fork, UT). Each 15 cm × 15 cm device contained 66 light-emitting diodes mounted 1 cm apart in an 11 × 6 array. The yellow goLITE M2 was designed as a placebo for light therapy with comparable visual brightness (illuminance). Spectral properties are displayed in Figure 2. The intensity of the blue and yellow LED devices used in the current study were below exposure limits defined by international standards for retinal blue light hazard risk, to protect against retinal photochemical injury, and both conditions had lower irradiance and photon density levels than previously used.19,20
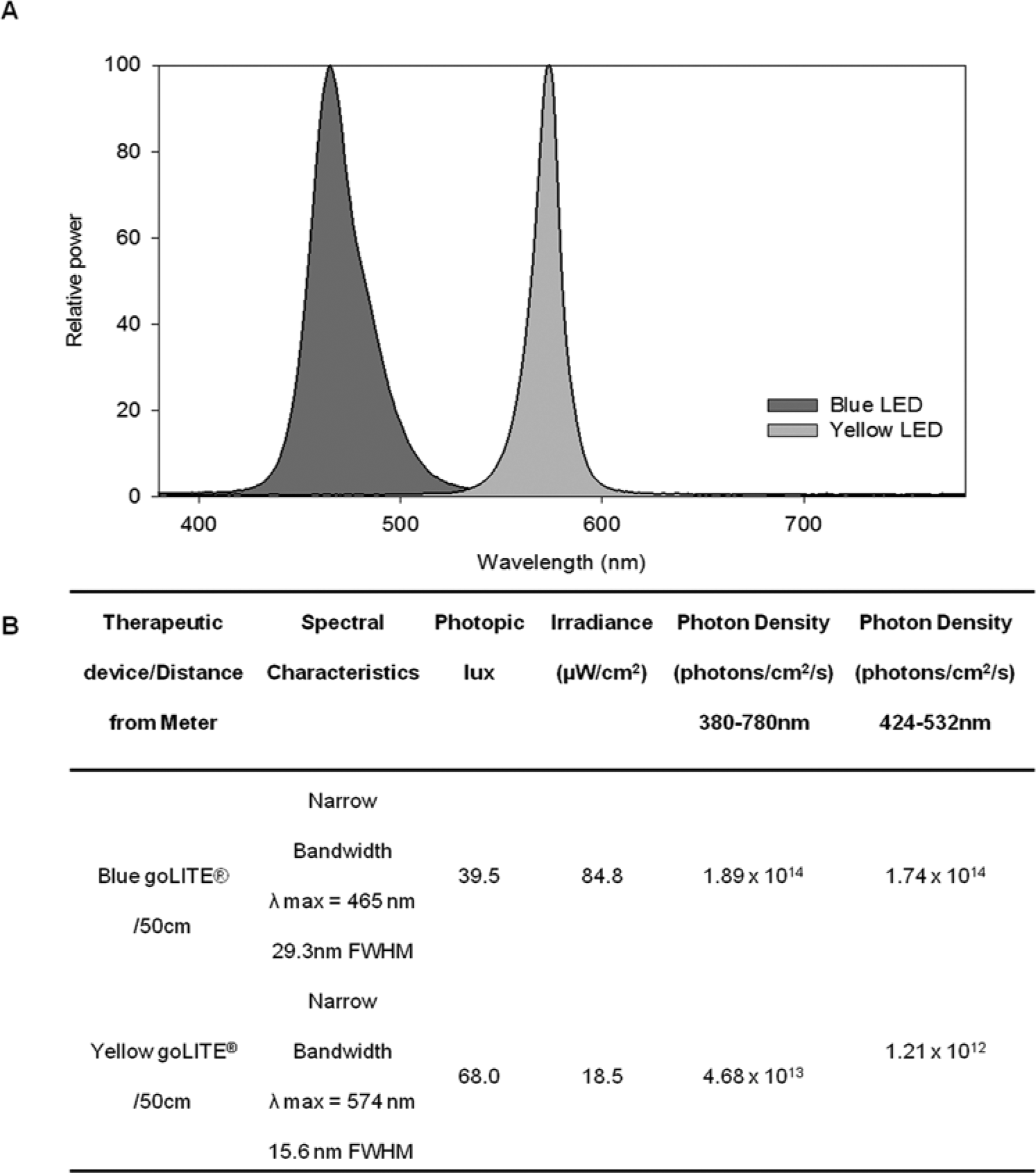
Spectral power distribution, photometric and radiometric comparisons of blue and yellow goLITE M2 devices.
Outcomes and Assessments
Fatigue, assessed using the FSS, 25 was the primary outcome measure. The FSS is a widely used measure of day-to-day fatigue assessing severity based on its impact on functioning and behavior. It comprised 9 statements rated on a scale from 1 (strong disagreement) to 7 (strong agreement). Statements included “fatigue causes frequent problems for me,” “fatigue interferes with my sustained physical functioning,” “fatigue is among my 3 most disabling symptom,” and so on. The final FSS score is the average of the 9 item responses (range = 0-7). Higher scores indicate higher subjective fatigue. In noninjured populations, FSS scores have been shown to range from 2.3 to 3.4,25,29,30 with a score of ≥4 considered indicative of clinically significant fatigue. 25 The FSS has demonstrated good reliability (Chronbach’s α = .88) and validity,25,29 and is considered sensitive to fatigue in the TBI population. 29 Secondary outcome measures were daytime sleepiness, sleep quality, and depression, assessed using the ESS, 26 PSQI modified for 2-week assessments, 27 and the Beck Depression Inventory (BDI-II), 31 respectively. The auditory version 18 of the Psychomotor Vigilance Task (PVT) 32 was used to measure sustained attention and has previously demonstrated sensitivity to short wavelength light. 18 During the 10-minute PVT testing trial, participants were required to respond to stimuli as fast as possible by pressing a response button as soon as the stimulus was presented. It provided a measure of reaction time (RT) and attentional lapses (RT > 400 ms). The mean time of day tested for each participant across the study was calculated (decimal time).
Demographic and injury-related information was collected from clinical interview and medical records. Cognitive functioning was assessed at baseline to examine possible influence on treatment outcome. Cognitive measures included the North American Adult Reading Test (NAART) 33 to estimate premorbid intellectual ability and the California Verbal Learning Test–Second Edition (CVLT-II) to assess learning ability. 34
Participants were questioned about adverse events during home-based visits at weeks 2, 4, and 8. Compliance with light therapy was recorded using a HOBO Pendant Data Logger, UA-002-08 (Onset Computer Corporation, Cape Cod, MA), attached to each light device. The logger provided a time-stamped measure of light intensity levels (lux). Participants were deemed compliant if the device was switched on at least 5 days per week on at least 3 of the 4 treatment weeks. In addition, participants were asked to record the timing and duration of light therapy use in a daily sleep diary.
Data Analysis
Differences between groups at baseline were examined using univariate analysis of variance (ANOVA) or nonparametric equivalent (Kruskall–Wallis) for continuous data and χ2 tests for categorical variables. As per previous research using the PVT,35-37 mean RT on the PVT was reciprocally transformed and lapses (x) were transformed by (√x + √(x + 1)) prior to analysis, in order to normalize distributions.
Random effects regression (with no interpolation for missing data), conducted with Stata Statistical Software, Version 11 (College Station, TX), 38 was used to compare group differences in the change in the primary outcome measure—FSS—and secondary outcome measures—ESS, PSQI, BDI-II, and PVT RT and number of lapses—across the study protocol (ie, time × treatment group interaction).39,40 The change in outcome measures over time within each treatment group was modeled as a parabolic (quadratic) function, controlling for age, gender, and level of depression (BDI-II) at baseline. For the 2 PVT outcomes, mean time tested across the study protocol was also controlled. Parabolic or quadratic models were assumed to account for the hypothesized pattern of improvement across time in the outcome measures within the light therapy intervention phase (weeks 2 and 4), in contrast to baseline (week −2) and follow-up (week 8) assessments, when there was no light exposure.
The association between participant characteristics and treatment response (ie, magnitude of change in outcome variables from baseline at week 4) was investigated using Spearman’s ρ. All available data were included in analysis. Mean ± SD are reported unless stated otherwise. A P value less than .05 was considered significant.
Results
Participant Characteristics
Of 103 individuals screened for the study, 32 were enrolled and 30 randomized (Figure 1). At inclusion, 97% reported clinically significant fatigue (FSS ≥ 4), 50% reported EDS (ESS ≥ 10), and 67% indicated poor sleep quality (PSQI > 5). Time since injury ranged from 96 days to 13.35 years (1106 ± 993.27 days; Table 1). Causes of injury included motor-vehicle accidents (60%), falls (23%), and pedestrian or pushbike accidents involving motor vehicles (17%). Duration of posttraumatic amnesia (PTA) was on average 20 days (SD = 25 days). Based on PTA, most participants had a severe injury (PTA > 1 week, 50%), followed by moderate (PTA 1-7 days, 27%), and mild (PTA < 24 hours, 23%) 35 . Following randomization, the groups did not differ significantly on demographic or injury characteristics (PTA, time since injury, age at injury), or baseline outcome measures (P > .05). The proportion of participants receiving antidepressant treatment or concurrent rehabilitative therapies (Supplementary Table S1) did not differ significantly between study groups (P > .05). No participants reported diagnosis of a DSM-IV-TR Axis I disorder other than depressive illness.
Demographic and Clinical Characteristics by Treatment Condition at Baseline a .
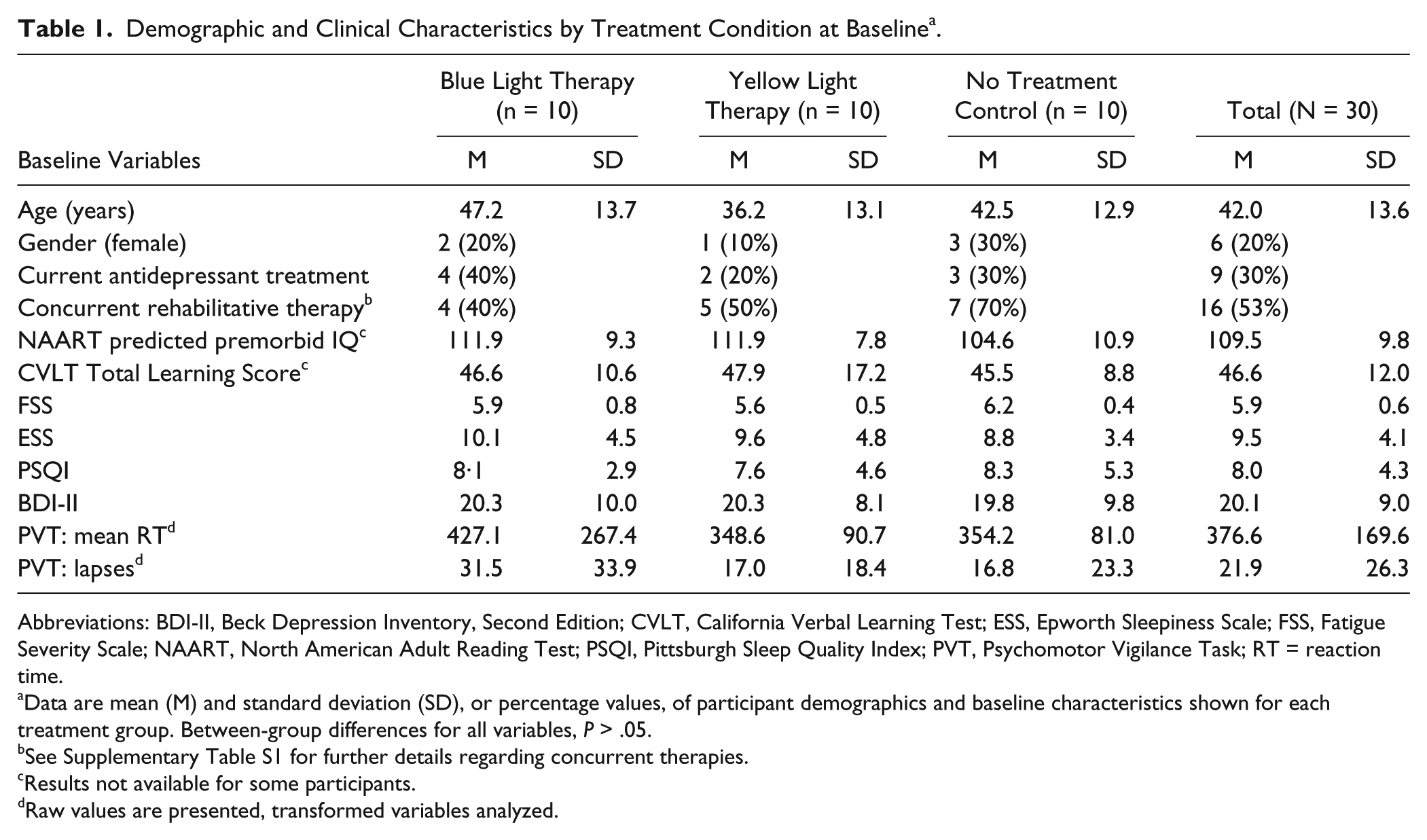
Abbreviations: BDI-II, Beck Depression Inventory, Second Edition; CVLT, California Verbal Learning Test; ESS, Epworth Sleepiness Scale; FSS, Fatigue Severity Scale; NAART, North American Adult Reading Test; PSQI, Pittsburgh Sleep Quality Index; PVT, Psychomotor Vigilance Task; RT = reaction time.
Data are mean (M) and standard deviation (SD), or percentage values, of participant demographics and baseline characteristics shown for each treatment group. Between-group differences for all variables, P > .05.
See Supplementary Table S1 for further details regarding concurrent therapies.
Results not available for some participants.
Raw values are presented, transformed variables analyzed.
Outcome Measures
Figures 3 and 4 show the change in symptom severity for primary and secondary outcome measures from baseline for each treatment group (also see Supplementary Table S2). A reduction in fatigue, daytime sleepiness, depression, and improvements in psychomotor RT and attentional lapse frequency can be seen during the treatment phase, particularly for those who received blue light therapy. Improvements in these measures did not appear to persist following cessation of the treatment, at week 8.
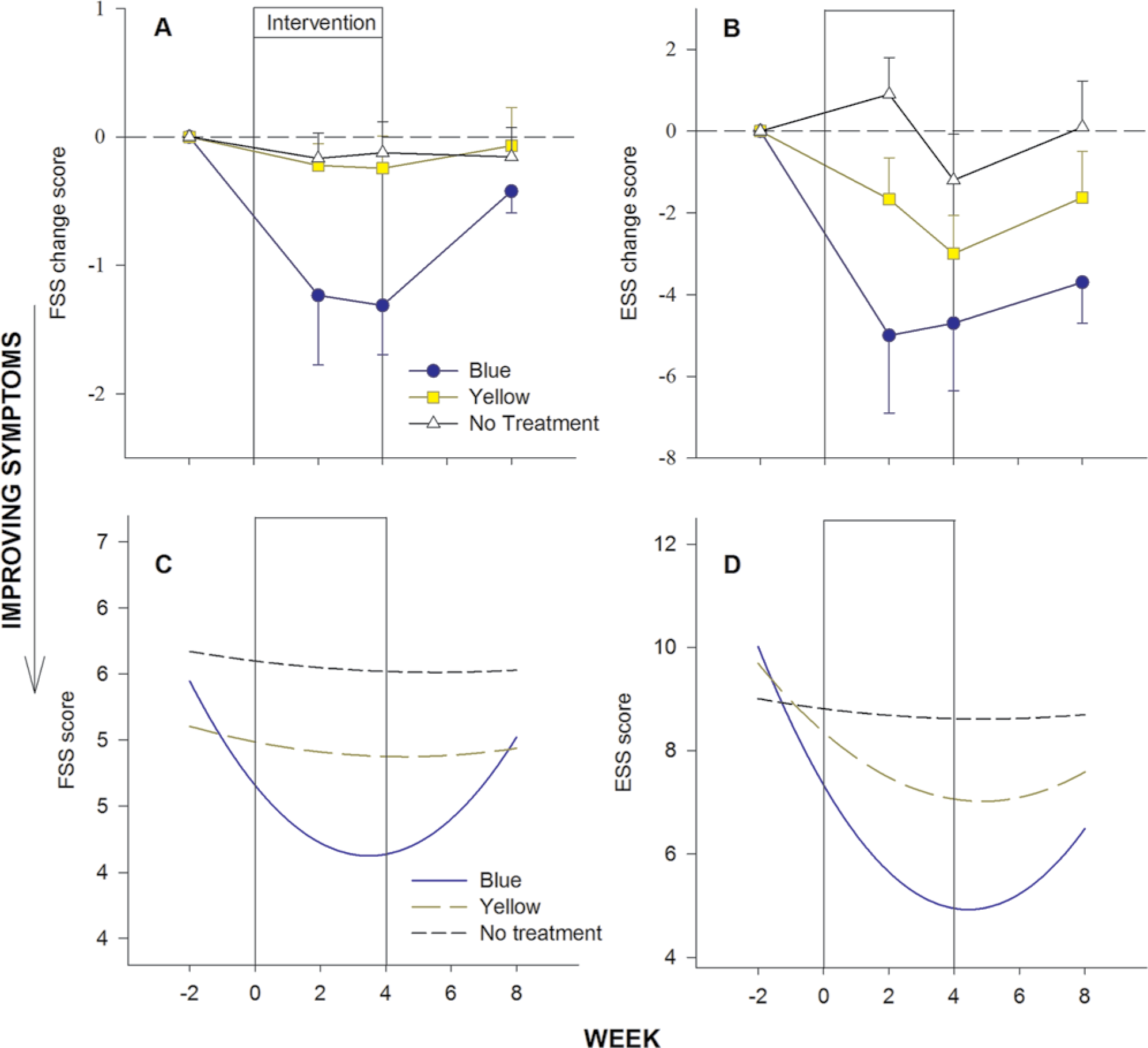
Mean change from baseline (A, B) across the study protocol, shown with the fitted parabolic curves for each group (C, D), on measures of fatigue (A, C) and daytime sleepiness (B, D).
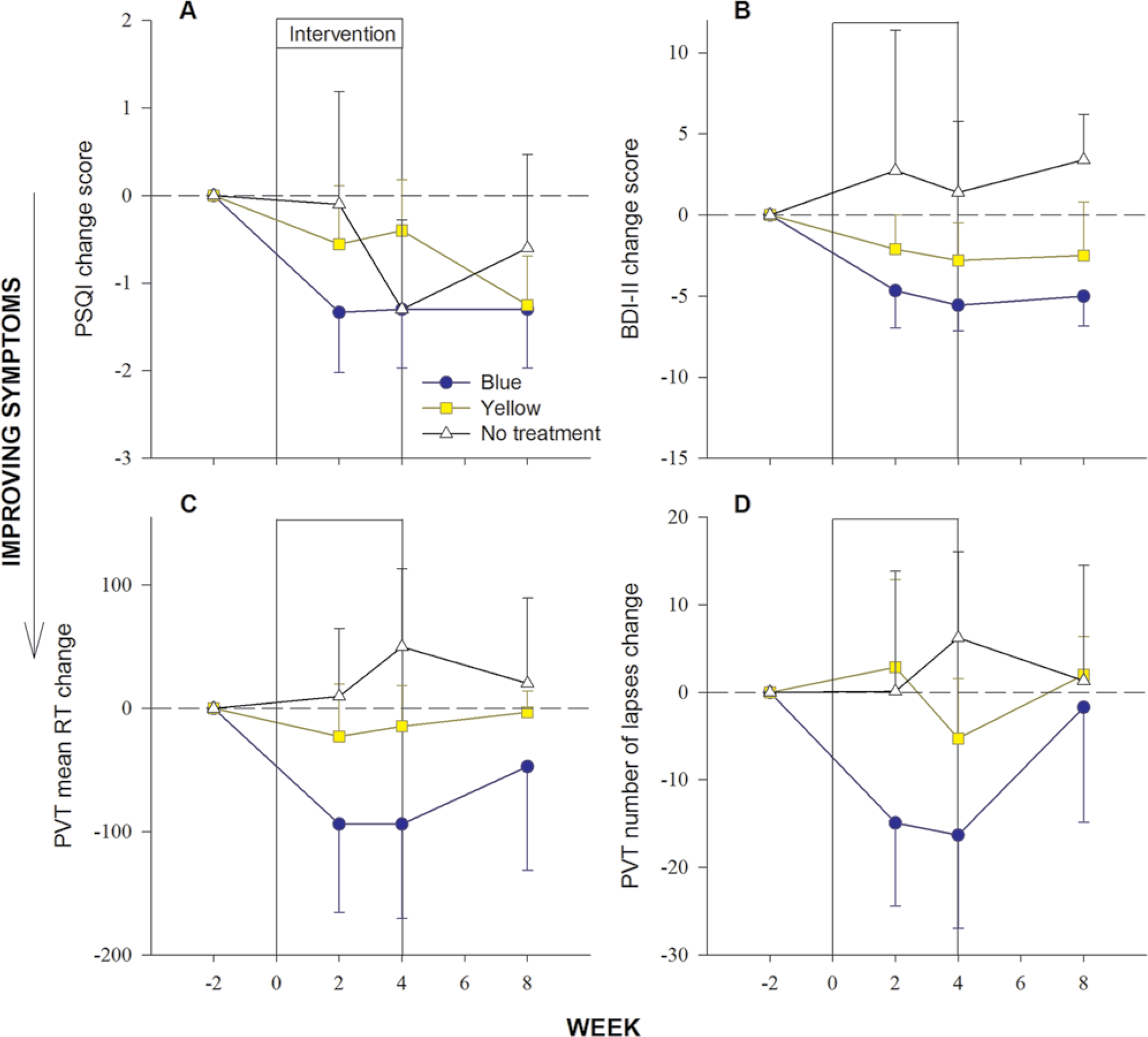
Mean change from baseline in sleep quality (A), depression severity (B), and reaction time (C) and number of lapses (D) on the Psychomotor Vigilance Task.
With regard to the primary outcome measure, the random effects regression revealed a significant reduction (both linear and quadratic) in fatigue across the study protocol in those who received blue light therapy (Table 2), with large effect size (Supplementary Table S2). Specifically, those in the blue light therapy group experienced a greater overall (linear) decline in fatigue (by .44 units per week [P < .001]) relative to controls. The significant departure from linearity reflected in the parabolic function (Table 2, Figure 3) additionally highlights that the improvement in fatigue occurred during use of blue light therapy (ie, during the intervention phase: weeks 2 and 4). Compared to no treatment controls, the blue light group showed a significant improvement in fatigue (difference from no treatment control in quadratic time coefficient 0.04, P < .001). These improvements in fatigue (linear and parabolic) were not observed in the yellow light therapy group relative to no treatment controls, and there was no significant deviation in fatigue over time in the no treatment control group.
Random Effects Regression of FSS and ESS a .
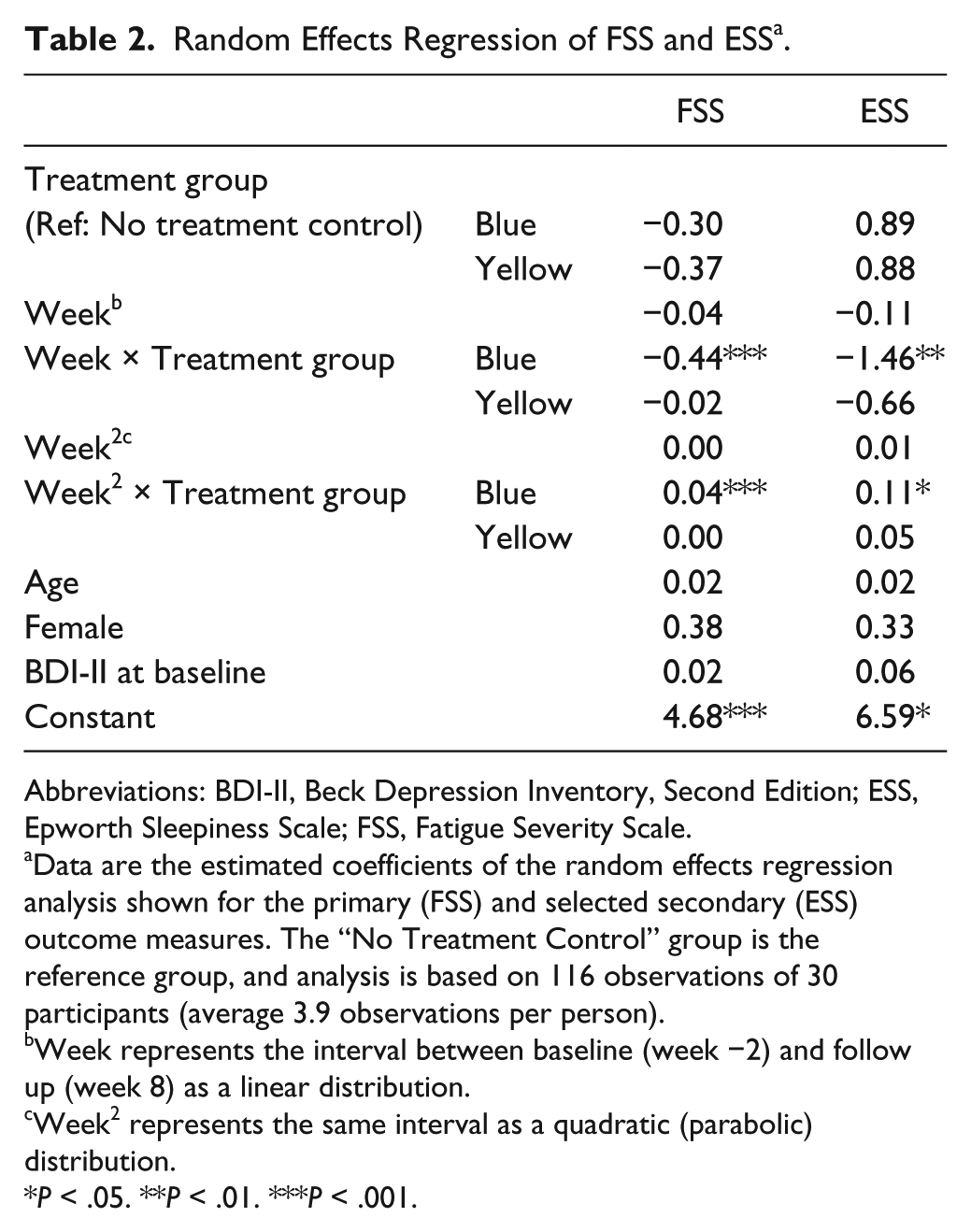
Abbreviations: BDI-II, Beck Depression Inventory, Second Edition; ESS, Epworth Sleepiness Scale; FSS, Fatigue Severity Scale.
Data are the estimated coefficients of the random effects regression analysis shown for the primary (FSS) and selected secondary (ESS) outcome measures. The “No Treatment Control” group is the reference group, and analysis is based on 116 observations of 30 participants (average 3.9 observations per person).
Week represents the interval between baseline (week −2) and follow up (week 8) as a linear distribution.
Week2 represents the same interval as a quadratic (parabolic) distribution.
P < .05. **P < .01. ***P < .001.
A significant reduction (both linear and quadratic) in the secondary outcome measure of daytime sleepiness (ESS) was also observed in the blue light therapy group (Table 2, Figure 3), with medium effect size (Supplementary Table S2). Compared with the no treatment controls, a significantly greater overall (linear) decline in daytime sleepiness (by 1.46 units per week [P < .01]) was observed over time in the blue light group. The significant parabolic model for the blue light group as compared with the no treatment control group (difference from control in quadratic time coefficient 0.11, P < .01) also indicated that this improvement in daytime sleepiness occurred during the intervention phase (Table 2, Figure 3). Significant deviations in daytime sleepiness over time were not observed in the yellow light therapy and no treatment control groups. For other secondary outcome variables (PSQI, BDI-II, PVT-RT, and PVT-lapses), there was no evidence of significant changes (linear or quadratic) in symptom severity across time that were unique to any treatment condition (P > .05).
Treatment Expectation, Compliance, and Adverse Events
Participants randomized to blue or yellow light therapy groups did not differ in their expectation of treatment efficacy (blue light therapy: mean = 3.0, SD = 1.3 units; yellow light therapy: mean = 1.9, SD = 1.0 units, P > .05). There was no significant difference in number of days the light therapy was switched on, between blue (24.0 ± 4.7, n = 6) and yellow (22.6 ± 2.7, n = 8) groups (P > .05). Sleep diary reports also indicated comparable compliance (eg, percentage of recorded days with “compliant” light therapy use defined as 45 ± 10 minutes, within 2 hours of waking) between the blue (81.9 ± 17.5%, n = 9) and yellow (74.0 ± 12.51%, n = 9) groups (P > .05). On average, participants in both groups reported using the light therapy in the morning but outside of the 2-hour window on an additional 8% of days. Adverse events were reported as follows: blue light therapy: headache (1 day, patient A), diarrhea (14 days, patient B, considered unrelated to intervention); yellow light therapy: headache, light glare, or “brightness” (2-3 days, patient C), soreness around the eyes (2-3 days, patient D). All adverse events resolved spontaneously and did not result in discontinuation of therapy. There was no significant association between demographic or injury characteristics, or cognitive function, and the average magnitude of change (over the 4-week treatment period) in symptom severity of fatigue or daytime sleepiness with blue light treatment (P > .05).
Discussion
We observed a significant reduction in fatigue in patients with TBI who received 4 weeks of 45 min/day home-based treatment with high-intensity blue light therapy. This effect was not observed in individuals within the yellow light therapy and no treatment control groups. This reduction in fatigue was accompanied by significant reductions in the secondary outcome measure of daytime sleepiness, but was not associated with demographic, injury, or cognitive characteristics of the participants. To our knowledge, this is the first reported use of light therapy in the TBI population.
Our primary finding of reduced fatigue with use of blue light therapy is consistent with earlier reports in healthy individuals exposed to blue-enriched white light in the workplace.23,24 The additional observation of improvement in daytime sleepiness seen with blue light therapy also supports previous research in healthy individuals13,18,41 and is consistent with the reported interrelatedness of fatigue and daytime sleepiness in individuals with TBI. 6
The (quadratic) pattern of change seen in fatigue and daytime sleepiness across the 10-week protocol suggests an alleviation of these symptoms during the 4-week treatment phase, with evidence of trend toward previous levels 4 weeks after treatment cessation, implying that the effects of blue light exposure do not persist after treatment cessation. These patterns of change in fatigue and daytime sleepiness were not replicated by the yellow light therapy, suggesting that the alleviating effects were unique to the blue light therapy group. Moreover, the effects of yellow light therapy on fatigue and daytime sleepiness were indistinguishable from those of the no treatment controls. The magnitude of reduction in fatigue and daytime sleepiness we observed after 4 weeks of treatment with blue light therapy was greater than that reported following 6 weeks of daily treatment with 100 to 200 mg modafinil in a comparable TBI sample. 11 Worth noting, however, is the evident interindividual variability of fatigue and daytime sleepiness (Supplementary Table S2) within each treatment group and across the duration of the study. This variability present at baseline, in particular for the ESS (secondary outcome), may reflect the known interindividual or person-specific differences in fatigue and sleepiness often seen in healthy individuals under conditions of sleep loss.42,43 Other possible explanations for this degree of interindividual variability in outcomes include the varying influence of associated secondary symptoms (eg, sleep disturbance, pain, mood) 6 and/or limitations of self-report given the possibility of insight and recall difficulties within this clinical population. Finally, although currently not well understood, individual differences in response to light exposure may also play a role.
Despite significant reductions in fatigue and daytime sleepiness seen in those using blue light therapy, significant changes in other secondary outcomes were not observed in any treatment condition, although there was evidence for a nonsignificant trend toward improved depression and sustained attention (Figure 4). In contrast to prior research, which has demonstrated reduced depressive symptoms when using a similar blue light intervention and application regimen,19,20 our findings did not demonstrate statistically significant reduction in depression with use of blue light therapy during the treatment phase. Similarly, improvements in self-reported fatigue and daytime sleepiness during blue light therapy were not reflected in assessment of sustained attention (PVT). In view of the small effect sizes observed for the BDI and PVT, we appear to be inadequately powered to detect significant improvements with blue light. However, given that improvement of PVT performance has been demonstrated in a smaller sample of healthy, sleep-deprived individuals,18,25 the result may also be attributed to increased variability due to differences in the testing environment (ie, home-based vs laboratory) and/or patient population. Also, despite improvements in self-reported daytime symptoms, our clinical sample of patients with TBI did not report significant improvement in sleep quality. Sleep disturbances in patients with TBI are likely to be heterogeneous, attributable to varying mechanisms. The possibility that light therapy may be effective in a subgroup that show symptoms of circadian rhythm sleep disorder 44 should be investigated.
Proposed mechanisms for the alerting effects of blue light involve activation of melanopsin-containing intrinsically (short wavelength/blue-) photosensitive retinal ganglion cells. 45 Although we found blue light to be superior to yellow light in alleviating fatigue and daytime sleepiness, it is difficult to interpret the findings in this context for several reasons. Our study did not compare light wavelengths of equivalent photon density, and our blue light therapy contained approximately 4 times more total photons overall than the yellow light therapy. As such, it is difficult to determine if the observed improvements were specifically wavelength-dependent as opposed to a simple dose (intensity) response. We note that broad-spectrum white light might also be effective, as shown in other populations. 15 Furthermore, several domains may be involved in the demonstrated effects of this blue light therapy—including the ability of light to (a) act as an acute stimulant, (b) act as an antidepressant, and/or (c) shift the timing of circadian rhythms. Given the previously reported interrelationships between fatigue, daytime sleepiness, and mood, 6 it is plausible that some or all of these domains may be involved in this heterogeneous population; however, our study cannot distinguish between these potential mechanisms.
An inherent limitation in light intervention studies is the lack of a placebo control to light in normally sighted individuals. Attempts to develop appropriate placebo light sources have had limited success. 46 We attempted to account for this issue by using a “placebo” yellow light that contained less photons in the short wavelength range. Although treatment allocation could not be masked, ratings of treatment expectation did not differ significantly between blue and yellow light therapy groups, suggesting that our findings are not directly attributable to treatment expectancy. As previously discussed, findings from the study may also be affected by reduced statistical power, participant heterogeneity, and limitations of self-report. Research with increased sample sizes and possible inclusion of objective measures of sleepiness may be warranted.
Our findings establish the basis for large-scale randomized controlled trials of light therapy for fatigue in TBI. Future research equating blue light therapy and placebo conditions on total photon densities will assist in differentiating between wavelength versus intensity-dependent effects of light in this population. A strength of the current study was its attempt to objectively monitor light therapy compliance. We cannot, however, objectively confirm that participants remained seated in front of the light source for the entire treatment period.
In conclusion, our findings suggest that 45 min/day of home-based blue light therapy may offer a safe, effective, nonpharmacological, and inexpensive therapy for alleviation of fatigue and daytime sleepiness symptoms in patients with TBI. These findings have important implications for enhancing rehabilitation participation and long-term quality of life following TBI.
Footnotes
Acknowledgements
We thank the Monash Epworth Rehabilitation Research Centre for assistance with participant recruitment.
Authors’ Note
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
KLS and JT report no disclosures.
JLP has received royalties from Psychology Press and Guilford Press for textbooks on traumatic brain injury that do not mention light therapy, and has received an honorarium and travel and accommodation support to present a lecture on fatigue and sleep disturbance at the TBI Interagency Conference in Washington, DC, May 2011.
SWL has received consulting fees from Apollo Lighting, American Family and Wyle Integrated Science and Engineering (NASA), and holds current consulting contracts with Naturebright, Sound Oasis, and Wyle Integrated Science and Engineering (NASA). He is/was a consultant on federally funded projects at Brigham and Women’s Hospital, Thomas Jefferson University, and Warwick Medical School; and has received one-time lecture fees from Takeda Pharmaceuticals North America, I Slept Great/Euforma, LLC, and Emergency Social Services Association Conference, UK; unrestricted equipment gifts from ResMed Inc, Philips Lighting, and Bionetics Corporation; unrestricted monetary gift to support research from Swinburne University of Technology, Australia; a fellowship gift from Optalert, Pty, Melbourne, Australia; advance author payment and royalties from Oxford University Press; payment for editing a textbook section from Elsevier; honoraria from Servier Inc for writing an article for Dialogues in Clinical Neuroscience and from AMO Inc for writing an educational monograph, neither of which refer to the companies’ products; honoraria and/or travel and accommodation support for invited seminars, conference presentations, or teaching from 2nd International Symposium on the Design of Artificial Environments; 8th International Conference on Managing Fatigue; American Academy of Sleep Medicine; American Society for Photobiology; Apollo Lighting; Bar Harbor Chamber of Commerce; Bassett Research Institute; Canadian Sleep Society; Committee of Interns and Residents; Coney Island Hospital; FASEB; Harvard University; Illinois Coalition for Responsible Outdoor Lighting; International Graduate School of Neuroscience; Japan National Institute of Occupational Safety and Health; Lighting Science Group Corp; Lightfair; National Research Council Canada; New York Academy of Sciences; North East Sleep Society; Ontario Association of Fire Chiefs; Philips Lighting; Thomas Jefferson University; University of Montreal; University of Tsukuba; University of Vermont College of Medicine; Utica College; Vanda Pharmaceuticals; Velux; Warwick Medical School; Woolcock Institute of Medical Research; Wyle Integrated Science and Engineering (NASA); investigator-initiated research grants from Respironics Inc, Philips Lighting, Apollo Lighting, and Alcon Inc; service agreement with Vanda Pharmaceuticals; sponsor-initiated research contract with Vanda Pharmaceuticals; 2 investigator-initiated research grants from the ResMed Foundation. SWL has also served as a paid expert witness on behalf of 3 public bodies on arbitration panels related to sleep, circadian rhythms, and work hours. SWL holds a process patent for the use of short-wavelength light for resetting the human circadian pacemaker and improving alertness and performance, which is assigned to the Brigham and Women’s Hospital per hospital policy. He has also received revenue from a patent on the use of short-wavelength light, which is assigned to the University of Surrey.
SMWR has served as a consultant through his institution to Vanda Pharmaceuticals, Philips Respironics, EdanSafe, The Australian Workers’ Union, Rail, Bus and Tram Union, and National Transport Commission, and has through his institution received research grants and/or unrestricted educational grants from Vanda Pharmaceuticals, Takeda Pharmaceuticals North America, Philips Lighting, Philips Respironics, Cephalon, and ResMed Foundation, and reimbursements for conference travel expenses from Vanda Pharmaceuticals. His institution has received equipment donations or other support from Optalert, Compumedics, and Tyco Healthcare. He has also served as an expert witness and/or consultant to shift work organizations.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from Transport Accident Commission, Jack Brockhoff Foundation, Centre for Integrated Research and Understanding of Sleep (CIRUS), The University of Sydney, and RACV Sir Edmund Herring Memorial Scholarship. goLITE devices were donated by Apollo Health, since bought by Philips Lighting.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
