Abstract
Purpose:
Chilaiditi’s sign (CS), hepatodiaphragmatic interposition of the intestine, was caused by morphological abnormalities such as diaphragmatic atrophy, intestinal dilation, and liver atrophy. The sign is potentially important due to associations with clinically recurrent abdominal pain or even colonic volvulus. Late-onset Pompe disease (LOPD) could have the high prevalence of CS because of widened hepatodiaphragmatic space, following diaphragmatic atrophy, and the abnormal dilation of intestine caused by glycogen accumulation in smooth muscle of intestine. Our aim was to investigate the prevalence of CS in LOPD, and to identify the risk factors of CS in LOPD patients.
Methods:
Medical records of genetically confirmed patients of Pompe disease at the National Center Hospital, National Center of Neurology and Psychiatry were retrospectively reviewed. We evaluated CS using chest X-ray (CXR) and abdominal CT and assessed the prevalence of CS in LOPD patients. We also divided the patients into two groups, CS and non-CS group, and evaluated the factor associated with CS compared to clinical variables between groups.
Results:
Three of seven (43%) were detected in CS. CS group (P5-7) and non-CS group (P1-4) were obtained. In comparison of clinical variables, the severity of atrophy in right diaphragms was significantly higher in CS than non-CS groups (p = 0.029). Also, the frequency of abnormal position of right diaphragm and liver, and abnormally dilated bowel was seen in all of CS patients, but none of non-CS patient (p = 0.029, each).
Conclusion:
In LOPD patients, the prevalence of CS was much higher of 43%, compared to healthy groups, or even in similarly respiratory muscle impaired neuromuscular diseases. The anatomically abnormal position of diaphragm and liver, atrophy and fat infiltration of diaphragms, and abnormally dilated bowel were significantly associated with CS in LOPD. We should pay more attention to CXR or abdominal CT as follow up in LOPD patients.
Keywords
INTRODUCTION
Late-onset Pompe disease (LOPD) is an autosomal recessive, lysosomal storage disorder caused by partial deficiency of acid α-glucosidase (GAA) (OMIN#232300) [1]. The residual GAA activity causes gradual accumulation of glycogen in skeletal muscles, leading to progressive myopathy and respiratory failure.
Although evidence has been growing for the involvement of smooth muscle in LOPD patients, with reports of vascular morphological abnormalities such as basilar artery dilation or aortic aneurysms [2, 3], manifestations in the gastrointestinal tract (GIT), which shows close functional and anatomical associations with smooth muscle, remain poorly understood. In addition, autopsy data from patients with LOPD have shown accumulation of glycogen in the GIT and dysfunction of smooth muscle in the small intestine [4–7].
Chilaiditi’s sign (CS), defined as hepatodiaphragmatic interposition of the intestine, is a rare anatomical abnormality [8]. The sign is often not associated with symptoms, but is potentially important due to associations with clinically recurrent abdominal pain or even colonic volvulus, following compression of the gastric outlet by the abnormally placed colon in CS [9]. The cause of CS has been considered to involve morphological abnormality of the right diaphragm, liver, and intestine, such as diaphragmatic weakness, liver cirrhosis, and abnormal gas accumulation due to aerophagia [10].
Patients with neuromuscular disease (NMD) can easily develop this sign because of diaphragmatic weakness. In particular, patients with LOPD may have a tendency to develop CS because of the presence of two main factors: diaphragmatic factors and bowel factors. No previous reports have described CS in LOPD.
Our aim was to investigate the prevalence of CS in LOPD, and to identify risk factors for CS in LOPD patients to compare clinical variables between CS and non-CS groups. We discussed the importance of adding CS to the list of intestinal complications in LOPD, and suggested using chest X-ray (CXR) and abdominal CT to screen patients with LOPD for CS.
METHODS
Patients
Medical records of patients with genetically confirmed Pompe disease at the National Center Hospital, National Center of Neurology and Psychiatry were retrospectively reviewed. Seven patients with LOPD (2 males; median age, 58 years) were enlisted. All study protocols were approved by the National Center Hospital Ethics Committee (approval no. A2018-024).
Patients 1, 2, 4, 6 and 7 have been previously reported [11–14].
We collected information including age at clinical onset, diagnosis, enzyme replacement therapy started, mechanical ventilation started, phenotype of gene mutation, and cause of death, if the patient died, from medical records. We also collected whether subjects needed mechanical ventilatory support, and whether invasive or non-invasive ventilation was applied.
CS evaluation
All patients repeatedly underwent chest and abdominal CT to evaluate respiratory muscles, including the diaphragm. CT examinations were performed using a Somatom Definition AS scanner (Siemens Medical Solutions, Forchheim, Germany).
CS was defined as interposition of the small intestine or colon into the hepatodiaphragmatic space on abdominal CT, regardless of whether the case was symptomatic or asymptomatic in the present study [8].
Findings from abdominal CT and CXR were also compared (Fig. 1).
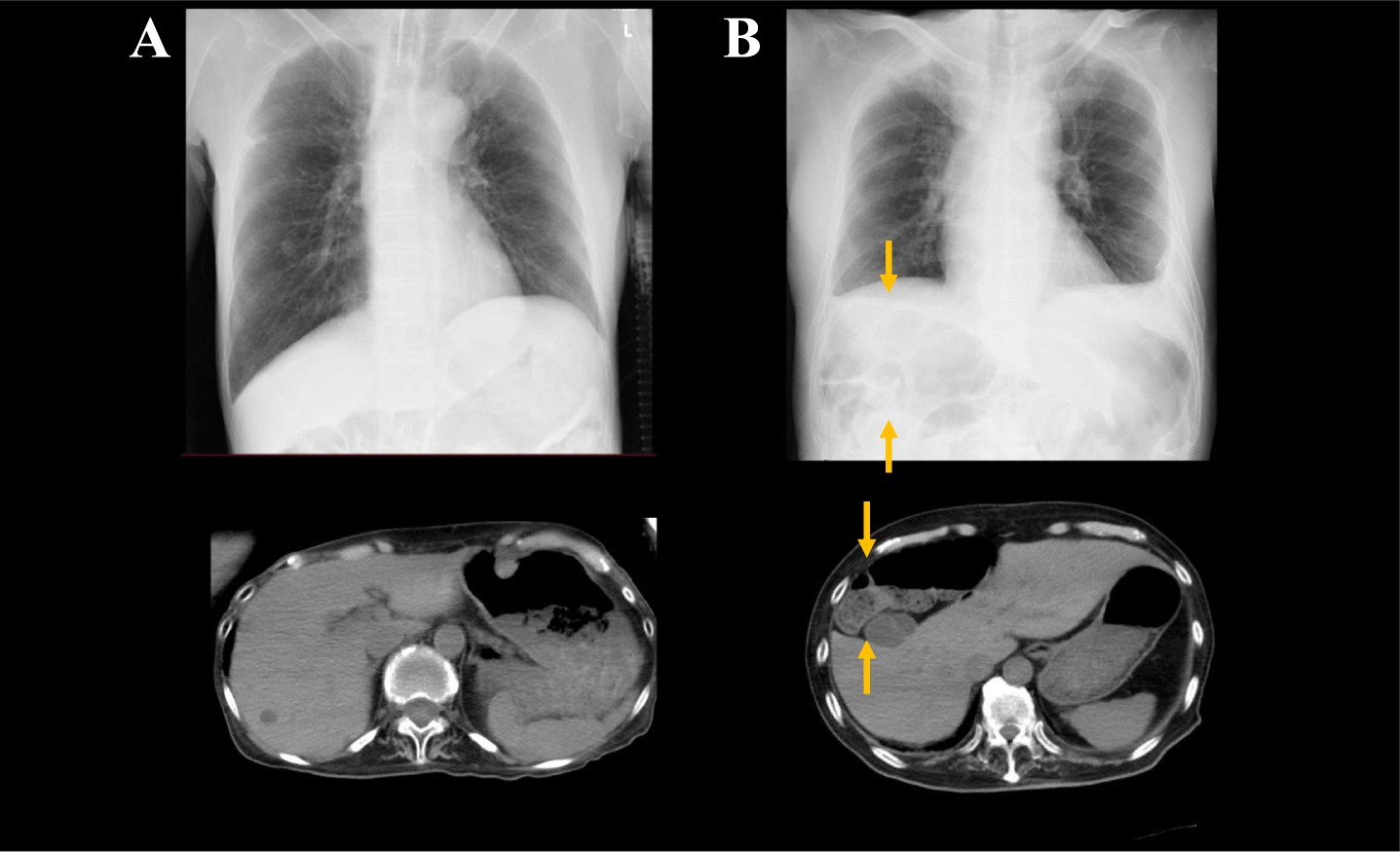
The findings on CXR and abdominal CT in non-CS and CS patients.
Diaphragm factor
The right diaphragm was evaluated regarding the severity of atrophy and fat infiltration on abdominal CT and height on CXR. The severity of diaphragm atrophy was evaluated according to the Mercuri score [15, 16], as the grading of muscle appearance in NMD on CT. This score shows the severity of muscle atrophy and fat infiltration, as follows; score 0: right and left diaphragm pillars are visible and do not show fatty replacement; score 3: unilateral pillars are visible or even partially infiltrated, whereas the other is absent; and score 4: diaphragm pillars are fully replaced on both sides (Fig. 2).
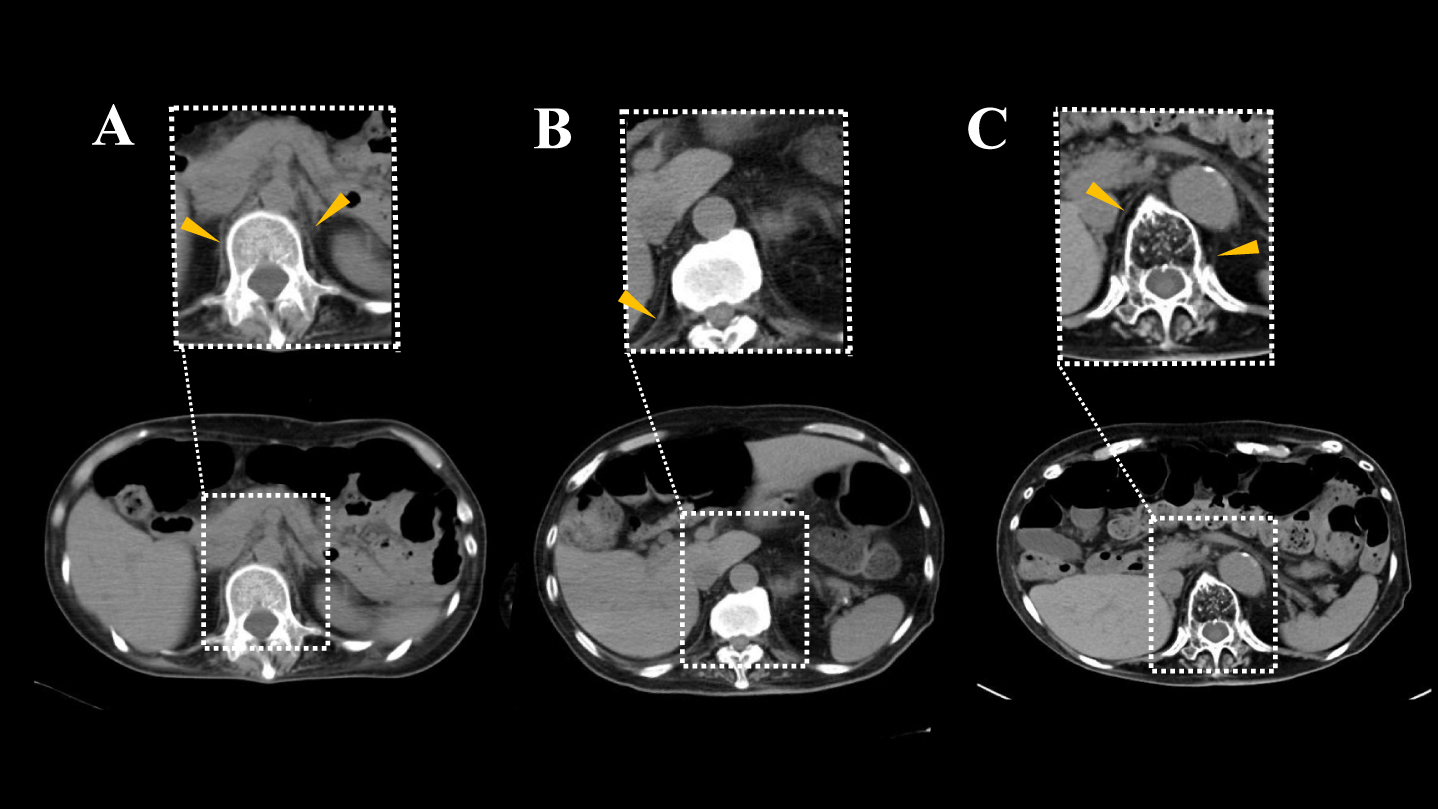
Evaluation of diaphragm muscle based on Mercuri score. In
The position of the right diaphragm was considered abnormal when the height of the right diaphragm was more than 2 cm higher compared to the height of the left diaphragm [17], according to findings of hemi-diaphragms differing even within one rib intercostal space height (approximately -2 cm) of each other on frontal inspiratory CXR [18].
Liver factor
The size and shape of the liver was evaluated. Normally, the liver was higher than the left diaphragm on CXR. When the upper border of the liver was detected under the left diaphragm, the position of liver was considered abnormally higher [19].
Bowel factor
Dysphagia was defined as difficulty with swallowing solids or fluids, and a need to devise food modifications. All patients had been evaluated for swallowing function by videofluorography or videoendoscopy, and adequate food modifications (including gastrostomy) had been provided before the study.
Aerophagia was defined as diffuse gastric, small bowel and colonic distension without signs of obstruction with uncomfortable gastrointestinal symptoms, such as abdominal distension, bloating, belching, and flatulence [20]. This finding could be caused by excessive and repetitive air swallowing, particularly during non-invasive positive pressure ventilation (NPPV), by imbalances in supportive inspiratory and spontaneous expiratory phases [21].
We also detected the maximum caliber of dilated bowel on chest or abdominal X-ray. The small bowel was defined as abnormally dilated if the caliber was more than 30 mm [22], and the large bowel was defined as dilated if the caliber was more than 60 mm [23].
Body component measurement
All patients underwent body composition measurement including lean body mass (LBM), total fat weight (TFW), and body mass index (BMI) using dual-energy X-ray absorptiometry (Discovery Bone Densitometer; Hologic, Bedford, MA) [24].
The duration between body component measurements and evaluation of CS was ≤2 weeks.
Statistical analysis
We divided subjects into a CS group and a non-CS group, and compared clinical variables between groups to extract factors associated with CS.
Baseline characteristics and radiological findings were compared between CS and non-CS groups. Numerical values are reported as medians and interquartile ranges. Data were statistically analyzed using the chi-square test for categorical variables, and the Mann-Whitney U test for nonparametric analyses. Two-sided p-values less than 0.05 were considered significant. All data were analyzed using R statistical software (version 4.1.0).
RESULTS
Case presentation (Table 1)
We have previously reported the clinical presentations of
Clinical and molecular data of 7 LOPD patients
Age is expressed in years. ERT: enzyme replacement therapy.
As proximal limb weakness gradually progressed, she visited us at 30 years old, at which time a slight elevation of serum creatine kinase of 256 IU/l (normal range: 45-163 IU/l) was detected. Muscle biopsy and reduced GAA activity led to a diagnosis of LOPD.
Although the patient could walk without any support, progressive ventilatory defects were already apparent on spirometry. She developed sudden respiratory distress at 35 years old, necessitating mechanical ventilatory support with tracheostomy during day and night. Enzyme replacement therapy (ERT) was initiated at 56 years old.
ERT was initiated at 76 years old, requiring admission to our hospital every 2 weeks for administration.
We have previously reported the clinical course of
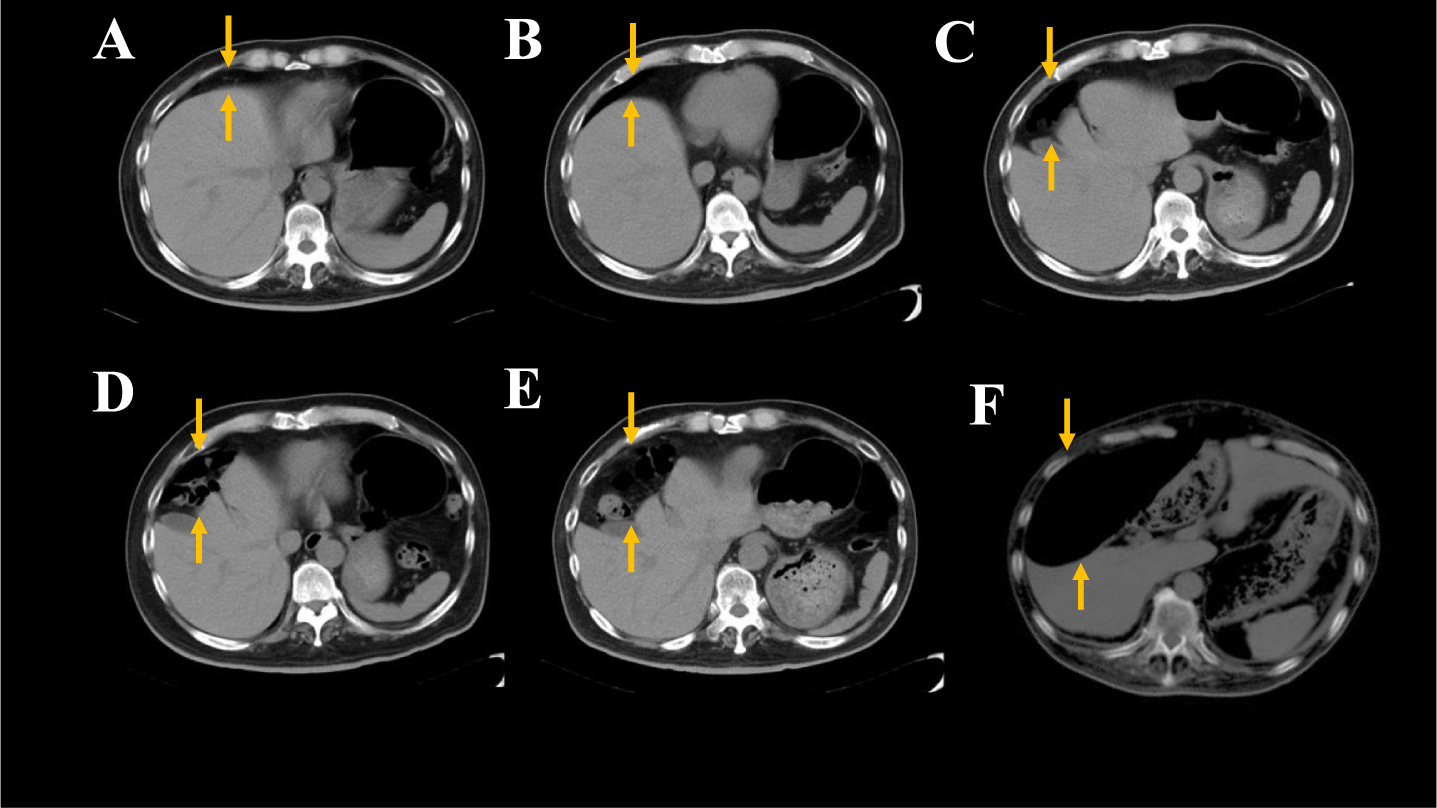
Temporal changes of abdominal CT findings in CS patient. In
At 74 years old, the patient suddenly died of bowel obstruction without any evident risk factors such as history of surgery or intestinal cancer, suggesting that CS may have been associated with the cause of his death, given previous reports of CS as an uncovered cause of intestinal volvulus or obstruction [25]. Other two patients in CS group have not developed intestinal volvulus.
Baseline characteristics of all patients (Tables 1, 2)
Median age at disease onset, considered as the time of first symptom onset, was 25 years (range, 22–36 years). Median age at diagnosis was 39 years (range, 26–46 years). ERT and mechanical ventilation were started at median ages of 54 years (range, 40–61 years) and 36 years (range, 34–54 years), respectively. All our cases required mechanical ventilation, five on NPPV alone during the night, and two on tracheostomy positive-pressure ventilation (TPPV) throughout the day and night. The phenotype of genetic mutation in all cases was reported as a variant of the GAA gene [26].
Three cases were dead as of the start of the present study.
Comparison of clinical variables between CS and non-CS groups
Three of seven patients (43%) showed CS. Characteristics of the CS group (Patients 5–7) and non-CS group (Patients 1–4) are shown in Table 2.
Demographics and clinical characteristics by Chiliditi and non-Chiliditi symptoms groups
CS: Chiliditi sign, ERT: enzyme replacement therapy.
In a comparison of clinical variables, sex, age at clinical onset, diagnosis, at CT evaluations of respiratory muscles, time of starting ERT, and time of starting mechanical ventilation did not differ significantly between groups. Similarly, no significant differences were seen between groups in duration from clinical onset to start of ERT, of ERT, or mechanical ventilation support. It was notable that all patients in the CS group showed recurrent abdominal pain, but only one in the non-CS groups.
In terms of body component measurements, LBM, TFW, and BMI all tended to be higher in the CS group than in the non-CS group, but these differences were not statistically significant. It was notable that all in CS group showed recurrent abdominal pain, but only one in non-CS group (Table 2).
In the evaluation of well-known risk factors of CS, severity of atrophy and fat infiltration in the right diaphragms were significantly higher in the CS group than in the non-CS group (CS vs. non-CS: median grade 3, range 3–4 vs. median grade 0, range 0–3, p = 0.029) (Table 3).
Comparison of risk factor of Chiliditi syndome between groups
Values are presented as n (%), CS: Chiliditi sign, NPPV: Non-invasive positive pressure ventilation.
In addition, abnormal position of the right diaphragm, liver under the left diaphragm, and abnormally dilated bowel were seen in all CS patients, but none of non-CS patients (CS vs. non-CS, 100% vs. 0%, p = 0.029, each).
Other well-established risk factors such as the frequency of using NPPV, dysphagia, gastrostomy, aerophagia, and liver atrophy did not differ significantly between groups.
DISCUSSION
In 1990, Chilaiditi first reported the abnormal sign of the transverse colon interposed between the liver and right diaphragm using early roentgenology, and this phenomenon was therefore named eponymously [8]. In the present study, CS was seen in three of the seven cases of LOPD (43%), much higher than the frequency of approximately 0.25–0.28% in the general population [27].
Although diaphragm weakness, one of the major risk factors of CS, was usually detected in NMD, especially in the advanced phase, few studies have evaluated the prevalence of CS in NMD. Our search only revealed investigations of CS in Duchenne muscular dystrophy (DMD), with a prevalence of 6.2% [28], and myotonic dystrophy type 1 (DM1), with a prevalence of 12.5% [29]. The prevalence of NPPV use was not mentioned in either of those studies.
The reason for the prevalence of CS being much higher in LOPD than in healthy groups, or even in NMD with similarly impaired respiratory muscles, remains unclear because we did not compare CS between LOPD and other NMDs. One possible reason is that the intestine in patients with LOPD may be more easily interposed into the hepatodiaphragmatic space because of the increased elasticity of the intestines, following a gradual accumulation of glycogen in the smooth muscles of the intestines [4–6]. Also, considering the diaphragm was usually more severely affected in DMD than in LOPD, the high prevalence of CS in LOPD in the present study may be associated with the higher severity of the enrolled patients.
Causes of CS are mainly classified as follows [30, 31]:
CS can thus be caused by abnormalities of the liver, colon or right hemidiaphragm.
In comparing risk factors between CS and non-CS groups of LOPD patients, the severity of atrophy and fat infiltration in the right diaphragm, and the frequencies of an abnormal position of the right diaphragm, liver under the left diaphragm, and abnormally dilated bowel were all significantly higher in patients with CS. Those findings may indicate an elevated position of the right diaphragm and lowered position of the liver could widen the hepatodiaphragmatic space, and the dilated intestinal bowel could be more easily interposed into this space.
On the other hand, LBM, TFW, and BMI tended to be higher in the CS group than in the non-CS group, although the differences were not significant. Accumulation of visceral fat around the liver widens the hepatodiaphragmatic space, and may allow easier interposition of the intestine [32]. In the present study, all subjects showed advanced-stage disease and visceral fat accumulation in each group may have been similar, contributing to the lack of significant difference between groups.
Interestingly, NPPV resulting in aerophagia did not differ significantly between groups, despite being a known risk factor for CS [10]. In terms of bowel dilation as a bowel factor for CS, NPPV was estimated to more easily result in development of CS compared to TPPV, because of the continuous entry of air into the stomach [21]. In the present study,
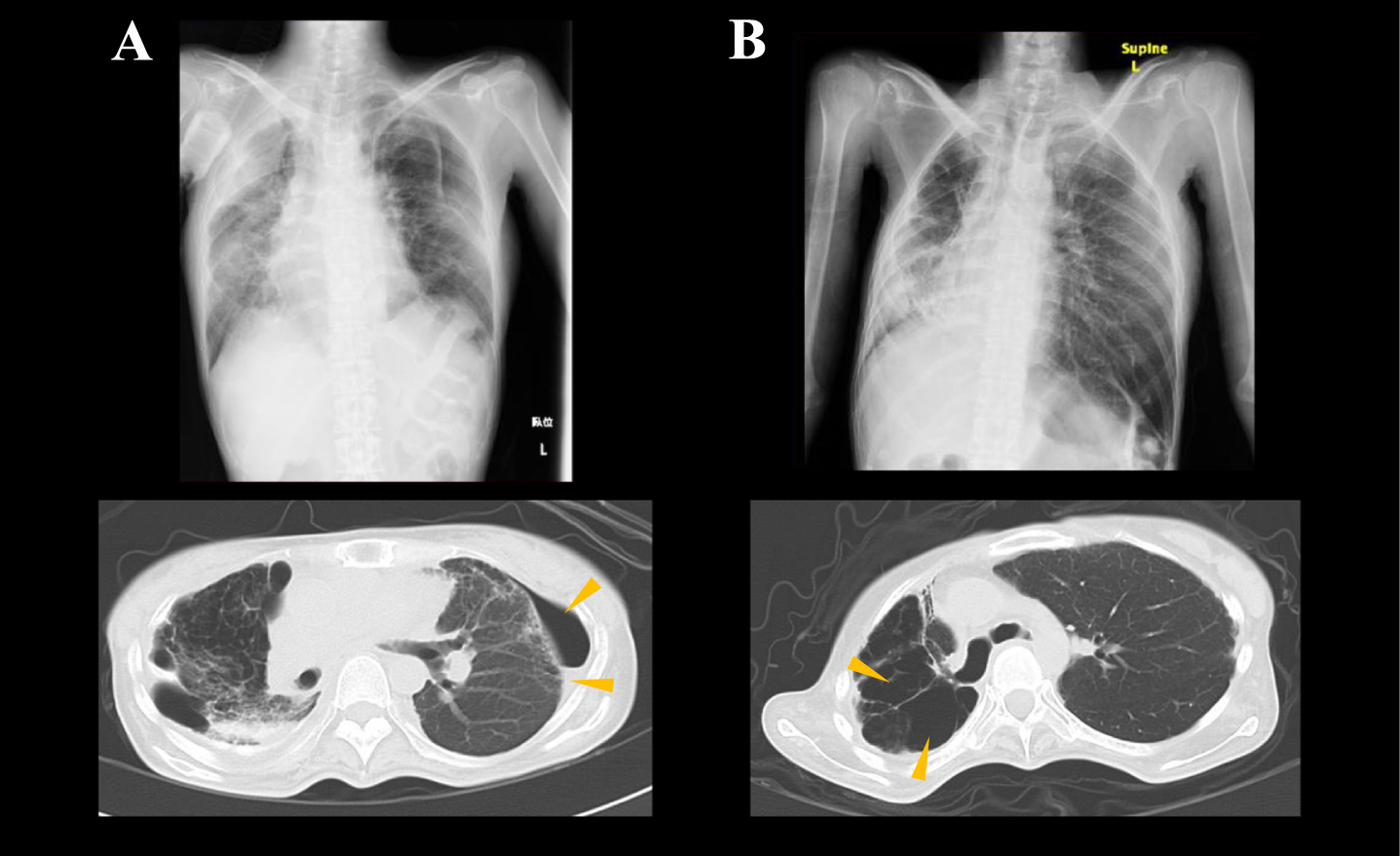
Marked right-side shift of the mediastinum on chest CT in two non-CS patients with NPPV.
A number of limitations to this study should be considered. First, the number of subjects was quite small, because of the low prevalence of LOPD. Second, we evaluated diaphragmatic atrophy by using abdominal CT. It was well-known that CT-based quantification of fatty muscle atrophy has limitations of detection. However, in advanced-phase late-onset Pompe disease, MRI cannot be easily performed because most of them required continuous positive airway pressure. Thus, in real-world clinical setting, CT examinations may also play an important role in evaluating Chilaiditi’s sign in advanced-phase late-onset Pompe disease.
Third, CS was not compared with other NMDs in the present study, and the high prevalence of CS in LOPD compared to other NMDs could have resulted from simple differences in clinical stage. Also, the high number of mechanical ventilated patients in the present study could contribute to the high prevalence of Chilaiditi’s sign. In addition, lack of detail of lung functional parameters, such as percent age of vital capacity or peak cough flow, was also a limitation. However, in intermittent end-tidal carbon dioxide monitoring, the values were stable and within normal range.
Further investigation to clarify the prevalence of CS in each NMD and compare risk factors including mechanical ventilation among diseases is warranted. Finally, though accumulation of glycogen in the smooth muscles of the intestines was reported in LOPD [4–6], association between glycogen accumulation and development of CS reveal unclear. Three of seven (Patient 1 and 2; non-CS, Patient 6; CS) had been dead when the study was started, and Patient 1 was the only patient we conducted the post-mortem examination. Thus, further histological studies is needed to confirm the evidence of intestinal abnormalities in LOPD with CS.
The routine evaluation of CXR should also play an important role in ruling out CS in patients with LOPD. If CS is suspected, further delineation of the liver and diaphragm by CT and referral to a gastroenterologist for further management and follow-up may be worth recommending. Preventing the risks associated with CS requires physician awareness and screening using at least CXR and CT to detect patients with CS.
CONCLUSION
The prevalence of CS was much higher in LOPD patients (43%) than in healthy groups or even groups with NMD resulting in similar respiratory muscle impairment, such as DMD and DM1.
Anatomically abnormal positions of the diaphragm and liver, atrophy and fat infiltration of diaphragms, and abnormally dilated bowel were all significantly associated with CS in LOPD.
Since CS can cause intestinal symptoms or even contribute to sudden death due to intestinal volvulus, CXR or abdominal CT require more attention as follow-up examinations in LOPD patients.
DECLARATIONS ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was conducted in accordance with the Declaration of Helsinki, and was approved by the National Center Hospital Ethics Committee (A2018-024).
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
Please contact author for data requests.
FUNDING
This work was supported by Research on rare and Intractable Diseases of Health and Labor Sciences Research Grants and an Intramural Research Grant (2–4, 2–6) for Neurological and Psychiatric Disorders from the NCNP.
COMPETING INTERESTS
The authors declare that they have no competing interests.
AUTHOR’S CONTRIBUTIONS
JT and MM analyzed the patient data and were major contributors in writing the manuscript. IN performed the histological and genetical examination in patients, and contributed to the diagnosis. HA, NS, YT helped to draft the manuscript. All authors read and approved the final manuscript.
Footnotes
ACKNOWLEDGMENTS
Not applicable.
DISCLOSURES
Financial Disclosures:
Junichiro Takahashi - Reports no disclosures
Madoka Mori-Yoshimura - Reports no disclosures
Hajime Ariga - Reports no disclosures
Noriko Sato - Reports no disclosures
Ichizo Nishino - Reports no disclosures
Yuji Takahashi - Reports no disclosures
