Abstract
Here, we describe a five year old girl with congenital HIV who had a six-week onset of rapidly deteriorating mobility and progressive proximal muscle weakness, associated with a raised Creatine Kinase (CK) level of 4330 U/L [25–200 U/L], subsequently diagnosed with an inflammatory myositis. Potential causes were investigated by paediatric neurology and immunology teams. Her viral load had been undetectable over the preceding two years, excluding a primary HIV myositis. While MRI scanning did not show evidence of definite myositis, a muscle biopsy showed evidence of an inflammatory process, comprising a moderate endomysial, perimysial and perivascular mononuclear (CD8 + T cell) infiltrate with increased MHC expression. No particular features of dermatomyositis or immune-mediated necrotising myopathy were identified and there were no features of an inclusion body myositis.
Given the absence of active HIV infection, the role of anti-retroviral medications was considered. She had had a recent switch in medication, from twice daily Raltegravir (an Integrase Strand Transfer Inhibitor, INSTI) to once daily Dolutegravir (an INSTI) while continuing on an established daily protocol of Abacavir and Lamivudine (Nucleoside Reverse Transcriptase Inhibitors). Changing the Dolutegravir back to Raltegravir, in combination with continuing Lamivudine and Abacavir for two months made no difference to her weakness or CK levels. Moreover, this drug regimen had been well-tolerated over the preceding 19 month period. Changing the anti-retroviral regime completely to a single drug class (Protease Inhibitors) of Ritonavir and Darunavir, resulted in a dramatic improvement in her symptomatology. Within ten days she regained the ability to stand and walk, with a reduction in her CK from 1700 U/L at time of switch to 403 U/L [25–200]. This case highlights the potential risk of developing inflammatory myositis from anti-retrovirals even 19 months into treatment.
INTRODUCTION
Inflammatory myopathies are a heterogeneous group of conditions that are the most common form of acquired muscle disease. There are many categories and aetiologies to inflammatory myopathies; idiopathic, genetic, immunological, iatrogenic, metabolic and endocrine. An inflammatory myopathy may also be referred to as a myositis, which is the term that will be used in the rest of the article. A myositis secondary to a viral illness (often Influenza virus) is a frequent cause of weakness and limping in children. It is often referred to as Benign Acute Childhood Myositis. This condition typically presents as refusal to walk and tenderness of the calf muscles bilaterally with pain on passive or active dorsiflexion [1–3]. The symptoms of myositis last for approximately 1-2 weeks in duration [2, 3]. HIV is also known to cause myositis [4, 5] while medications used to treat the condition have also been reported to result in myositis, including steroids and anti-retroviral agents such as Zidovudine [4].
Myositis as a consequence of anti-retroviral treatment usually presents with evidence of muscle inflammation and breakdown, leading to raised CK levels, myoglobinuria and ultimately to muscle weakness. Zidovudine, a Nucleotide Reverse Transcriptase Inhibitor (NRTI), has been well established as a drug capable of causing mitochondrial toxicity [6]. Zidovudine’s mechanism of action involves competing with natural nucleoside substrates of HIV reverse transcriptase and consequently may disrupt human mitochondrial DNA expression by inhibition of mitochondrial γ-DNA polymerase. Myopathy was reported in 17%of patients treated with Zidovudine monotherapy for over 9 months [7]. Zidovudine is not alone as an anti-retroviral capable of causing myositis; Raltegravir (an INSTI) which is normally well tolerated, has been cited in case reports with CK elevations, rhabdomyolysis and myopathy in post-marketing surveillance [8–10].
Without therapy, HIV itself can cause a myositis and in such cases a muscle biopsy can be useful to distinguish the aetiology. Ragged red fibers on modified Gomori trichrome stain and COX-negative fibers are evident with Zidovudine, in contrast to the inflammatory cell infiltrates that can be seen in HIV-associated myositis [11]. Almost all case reports of anti-retroviral therapy associated myositis have been from the adult population with only very limited case reports in children. We present a case of a child with HIV who was well established and compliant with their antiretroviral combination therapy for 19 months but subsequently developed features of myositis as evidenced via laboratory testing and muscle biopsy.
METHODOLOGY
Case description and clinical features
Our patient had an uneventful birth and infancy with no medical interventions noted until the age of 2 years when they received a diagnosis of Autism. At this time, the patient was noted to have minimal communication, with preservation of gross motor development. At the age of 4 years the patient was identified through contact tracing, to have contracted HIV through vertical transmission following maternal seroconversion in late pregnancy. In the initial clinical assessment, it was found that the patient had a high viral load of 9037 copies/ml and a CD4 count of 0.51×109. Her weight was 16 kg and height 101 cm (both 25–50th centile). She had not, until this age, had any hospital admissions, however she had received some infrequent courses of oral antibiotics from their GP and additionally, the mother had noted that she took longer than other children to clear viral infections.
At the age of 4 years and 4 months she was started on a triple combination of anti-retrovirals comprising Raltegravir 100 mg BD (an INSTI), Lamivudine 150 mg OD (an NRTI) and Abacavir 300 mg OD (also an NRTI) and was followed up under the Infectious Diseases and Immunology. Her viral load and CD4 counts were monitored and it was noted that she achieved prompt viral suppression to undetectable levels, indicating compliance with treatment. Her gross motor development was noted to be progressing appropriately during a clinic review aged 4 years and 6 months. Her communication had also developed as she acquired new skills. She still required verbal prompting for tasks, though demonstrated good fine motor control.
At the age of 5 years and 10 months she had remained well with no hospitalisations. She had an uncomplicated flu-like illness at this time but had not required inpatient care. When reviewed by the Immunology team one month later, her medications were adjusted in order to facilitate a simpler, once daily, dosing for her therapy. In doing so, twice-daily Raltegravir was switched for Dolutegravir, a once-daily medication of the same drug class.
At 6 years of age, the patient developed walking difficulties and was assessed by her developmental team and physiotherapists. A repeat assessment within 24 hours found an increased tendency to fall, and marked instability. Within a further 24 hours, she had developed a waddling gait and refused to walk. She required a buggy for mobility. She demonstrated a regression in her fine motor skills. She required support when standing due to proximal muscle weakness. A blood test at this time revealed a CK of 4339 U/L [range 25–200 U/L] and an ALT of 163 U/L [10–40 U/L] (Fig. 1). Her raised CK and ALT persisted for over 6 weeks, as did her symptoms.
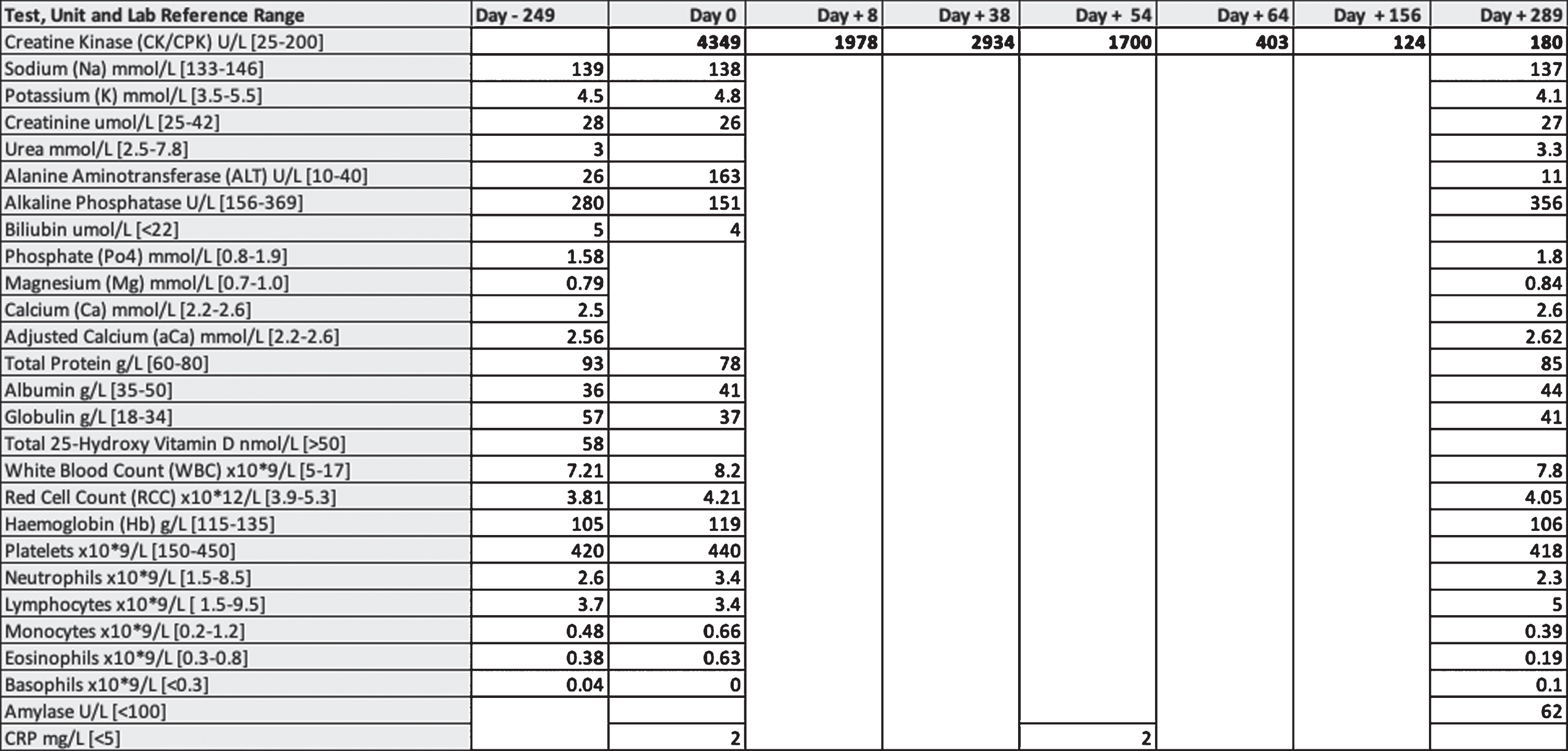
Laboratory test results. This table presents the blood test results performed before, at presentation (Day 0), during medication changes and for 289 days after presentation.
During her inpatient admission for investigations it was found that she could sit without support but not stand. Power was 3/5 bilaterally in the lower limbs and 5/5 bilaterally in the upper limbs. She was found to have hypopigmented patches on the left leg and right side of the chest, which had been present since birth. There was no evidence of any spinal abnormality. An MRI spine reported as normal and an MRI of her lower limbs showed no definite evidence of myositis.
She was reviewed by her HIV team during admission and the HIV viral load was confirmed to be well controlled, excluding a primary HIV myositis. The possibility of a reaction to her medication was considered. The recently commenced Dolutegravir was switched back to Raltegravir and Lamivudine and Abacavir continued. No improvement was noted clinically after nearly two months and therefore a complete class switch to Protease Inhibitors was undertaken wherein Lamivudine, Abacavir and Dolutegravir were discontinued and she was commenced on daily Ritonavir (100mg) and Darunavir (600mg). She received at this time a buggy due to her poor mobility.
Within 2.5 weeks of stopping her previous medications and switching to Ritonavir and Darunavir, her CK fell from 2934 U/L, to 403 U/L. It was at this time that the patient showed an interest in walking independently again. Her strength and mobility improved and she was observed to stand with a wide base of support and to walk several metres independently. The trend in CK in relation to clinical symptoms is demonstrated in Fig. 2.
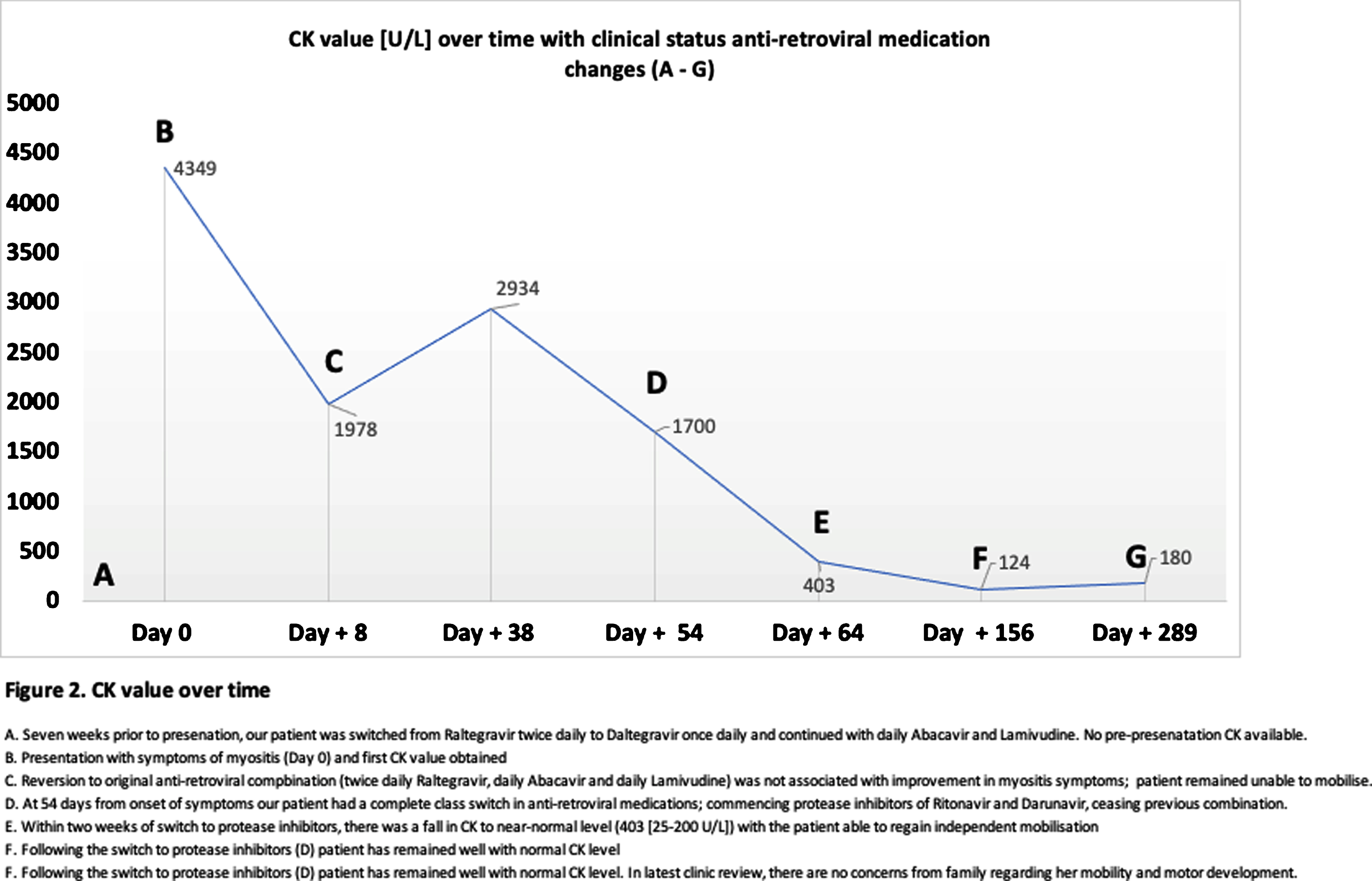
CK value [U/L] over time with clinical status anti-retroviral medication changes (A - G). This line graph shows the raised CK value on initial presentation of 4339 U/L, falling to 1978 U/L within a month during which the time the patient became immobile. The CK remained raised, in association with immobility, until a complete switch of the anti-retroviral combination therapy was undertaken, after which there is a fall in this value and trend of rapid clinical improvement noted. Changes in clinical status and anti-retroviral medications are annotated A to G.
When followed up 8 months after presentation, her muscle function had fully normalised and her CK and ALT had returned to within the normal range; CK 180 U/L [25–200 U/L], ALT 11 U/L [10–40 U/L] (Fig. 1). She has remained symptom-free with no myalgia and has shown normal progression of her gross and fine motor development.
Investigations
A number of modalities of investigations were performed as part of her initial workup including blood tests, spine and muscle MRI imaging, nerve conduction studies, echocardiography, muscle biopsy and genetic testing. Blood test investigations, as summarised in Fig. 1, were unremarkable, with the exception of CK and ALT values (please refer to Fig. 2 for trend of CK over time). Auto-immune causes were excluded by negative antibody screens for Rheumatoid factor, anti-ds DNA, anti-ENA and Hep2000/Hep 2000 type. MRI of the lower limbs (T1, STIR and TSE sequences) at 6 weeks into the development of her symptoms showed no definite focal signal abnormality within the muscles of the lower legs. There was no sign of fatty atrophy and the lower leg muscles had a symmetric, normal morphology and volume. This imaging did not provide radiological evidence of myositis. At 8 weeks into the development of symptoms and 1 week after the complete change of anti-retroviral medication, she also underwent an MRI whole spine (T2 TSE and T1 Flair). This was equally reported as a normal study.
At 2 months from development of symptoms and 2 weeks after the discontinuation of Dolutegravir, Lamivudine and Abacavir, a nerve conduction study and electromyography were performed. The nerve conduction study found no evidence of a large fibre peripheral neuropathy. Needle EMG of the right deltoid muscle did not show abnormal spontaneous activity. There were however, highly polyphasic low amplitude and short duration motor units which recruited briskly to a full interference pattern. These findings suggested a myopathy but were not aetiology specific. There was no spontaneous activity to suggest muscle fibre necrosis or inflammation though it was noted that this could not be excluded.
A biopsy of the right quadriceps muscle was performed during her admission. This skeletal muscle biopsy demonstrated features of an inflammatory myopathy (see Fig. 3). There were patchy areas of a mild to modest endomysial mononuclear infiltrate. In addition, foci of perivascular inflammation were observed, particularly within the perimysial septae. Invasion of intact fibres was not seen. No active segmental muscle fibre necrosis was identified and no rimmed vacuoles were identified. A background of mild myopathic features was also noted, with mild variation in myofibre diameters and scattered modestly atrophic fibres. A slight increase in internal nuclei was noted but no increase in interstitial connective tissue was observed nor were structural abnormalities characteristic of classic congenital myopathies observed.
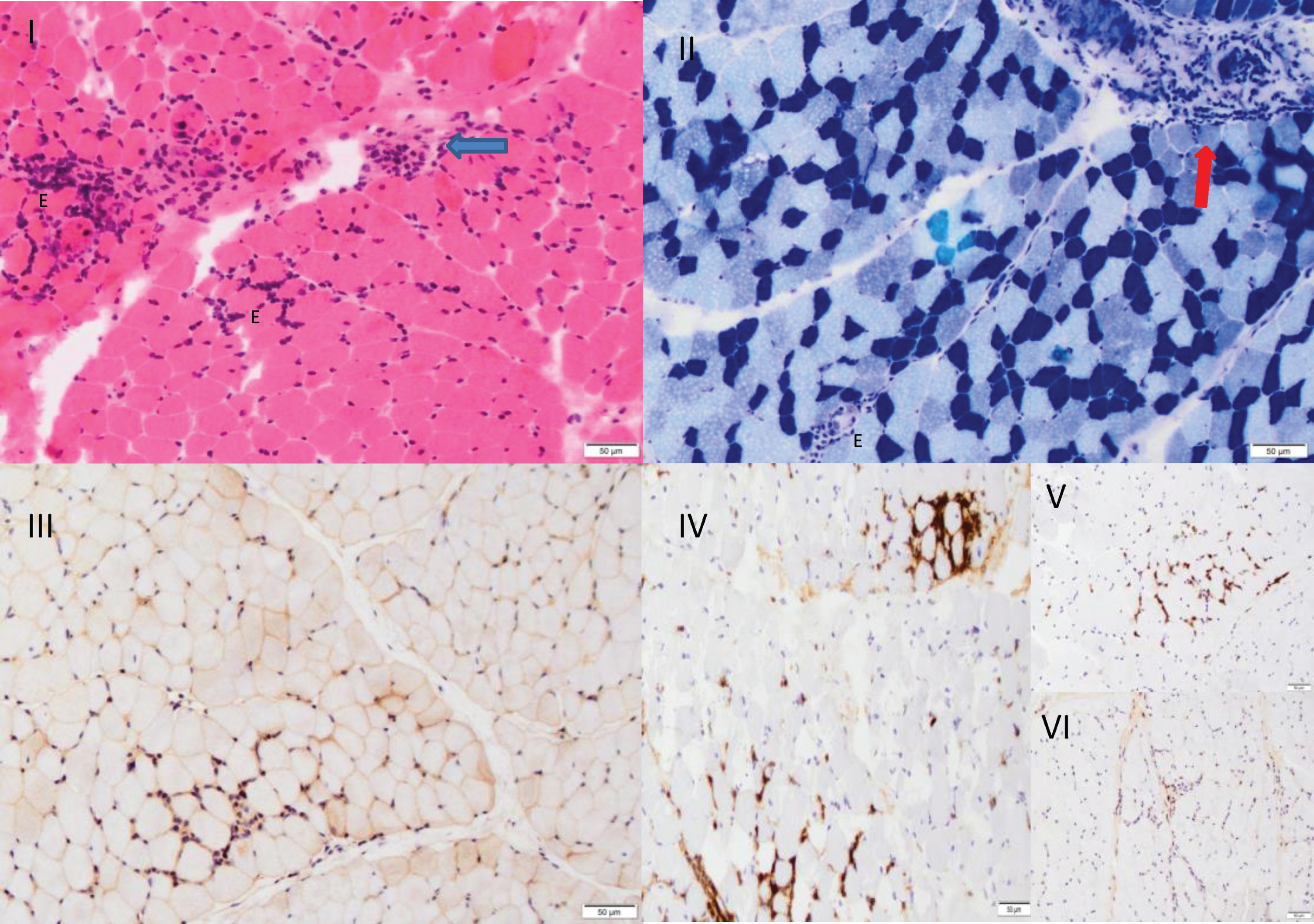
Muscle biopsy. I. H&E 20x showing patchy areas of a mild to modest endomysial mononuclear inflammatory infiltrate (E) and perimysial perivascular inflammation (arrow) on a background of variability in myofibre diameters. II. MATPase 10x showing a type II fibre predominance with a relative retention of the normal chequerboard architecture. Atrophy of type I fibres is noted. Focal endomysial (E) and perimysial perivascular inflammation (arrow) are also observed. III. B2 microglobulin 20x highlighting generalised increased sarcolemmal expression around myofibres in keeping with an inflammatory myopathy. IV. CD45 20x demonstrating the presence of endomysial lymphocytes which comprise predominantly CD8 + lymphocytes. V. (V) with only a rare CD4 + T lymphocyte identified (VI).
The panel of histochemical and immunohistochemical stains did not demonstrate any features of a pre-existent metabolic myopathy or congenital dystrophy, and no features of a classical congenital myopathy were identified. Immunostaining showed upregulation of MHC class I on muscle fibres and immunostaining for lymphocyte markers confirmed the presence of a moderate endomysial, perimysial and perivascular mononuclear CD8 + T cell infiltrate. There were no specific features of dermatomyositis or immune-mediated necrotising myopathy nor where there rimmed vacuoles or p62 positive inclusions to suggest inclusion body myositis. The features did not indicate a specific aetiology but were consistent with an inflammatory myositis particularly given the setting of a known HIV infection.
To investigate for cardiac involvement, an echocardiogram was performed during the inpatient admission which showed no significant changes and a structurally normal heart. CK isoenzymes did show a raised level of CK-MB isoenzyme, 129 U/L, [5–25 U/L] making up 18%of total [0–5%], suggestive of cardiac muscle inflammation, however an ECG was performed which was normal with no ischaemic changes. There was no evidence of CK-BB.
Genetic findings
A molecular genetic analysis for suspected mitochondrial disease was undertaken. The analysis identified no pathogenic or likely pathogenic mtDNA variants in the patient’s muscle DNA.
DISCUSSION
We present a child with sudden and profound deterioration in mobility and proximal muscle weakness believed to have developed secondary to anti-retroviral therapy with Lamivudine, with a possible contribution from Raltegravir. Lamivudine and Abacavir have not been frequently cited in the literature as a potential cause of inflammatory myositis. In one retrospective case control study, Lamivudine was implicated in myositis with a documented CK rise observed in 3 patients prescribed Lamivudine for treatment of Hepatitis B [12]. Abacavir is not reported as a cause of myopathy or myalgia, although is listed in the British National Formulary for Children as potentially causing lethargy.
Considering the INSTI drug class as a potential cause of myositis in this patient, there is no supporting evidence for Dolutegravir causing myositis, however rare cases from different reports exist of Raltegravir induced myositis. Dolutegravir as a cause of myositis was studied in the adult population; there was no evidence of an increased risk of grade 3 or 4 CK level rise (defined as a x5 or x10 ULN rise, respectively) [13]. In listed anti-retroviral toxicities, myositis is not listed among its adverse effects [10, 11]. The newer agent, Raltegravir, of the same drug class, has been reported as causing myalgia due to underlying mitochondrial enzyme restriction [11]. In one randomised controlled trial [9] performed in the adult population, Raltegravir-based therapy was associated with a higher prevalence of symptomatic skeletal muscle toxicity (reported myalgia 19%versus 3%p < 0.001 and myopathy 4%versus 0%p < 0.03), which did not seem to be concentration or time dependent, nor however associated with elevated CK (14%versus 16%p < 0.639). Proximal myopathy was considered an uncommon but significant side effect of Raltegravir exposure. The findings of the trial by Lee et al., were similar to a prospective study [8] for patients receiving Raltegravir where 5 out of 342 patients (1.5%of cohort) had a CK rise three times the upper limit of normal and of these, four were asymptomatic for muscle symptoms and one was symptomatic (0.3%of cohort). Patients on concurrent Atazanavir, a Protease Inhibitor, were more likely to experience muscle symptoms. As in the Lee et al., study, no cases of rhabdomyolysis were observed. These reports suggest a possible role of Raltegravir in causing myositis in our patient.
Secondary mitochondrial failure or deficiency is a previously described mechanism of action resulting in myotoxicity [14] but this is not supported in our patient’s case by the muscle biopsy or genetic findings. The possibility of a prolonged viral myositis cannot be entirely excluded, however, as mentioned earlier, the commonest virus known to cause this, Influenza, is associated with a shorter duration of symptoms, typically less than 3 weeks duration and is associated with myalgia.
As for practical considerations, it is not known if there were manual difficulties in the preparation, storage or administration of this patient’s medications, which replaced a twice daily alternative anti-retroviral. It is also possible that the combination therapy used may have induced liver toxicity. The drug levels were not available at the time of symptom development to indicate possible impairment of drug metabolism, although a raised ALT seen in this patient’s course may support a co-existing liver inflammation.
Based on the available literature on myositis induced by anti-retroviral therapy use, we suggest that this child’s presentation was secondary to Lamivudine therapy possibly with a contribution from Raltegravir, a rarer cause of myositis in the adult population. Stopping these medications and switching to a different class of anti-retrovirals induced a rapid improvement both clinically and biochemically and this well-defined temporal relationship suggests that medication changes were the instrument in this patient’s recovery.
Footnotes
ACKNOWLEDGMENTS
We would like to thank the parents for their consent for this case report and for allowing the publication of the results and comments on this case. The authors would like to thank the support of the contributing departments in the diagnosis and management of this rare presentation.
