Abstract
Objective
To investigate the value of myositis-specific autoantibodies in the diagnosis of idiopathic inflammatory myopathy and tumor risk prediction in patients with clinically suspected idiopathic inflammatory myopathy.
Methods
This retrospective study analyzed the myositis-specific autoantibodies and clinical characteristics of 357 patients with clinically suspected idiopathic inflammatory myopathy. Statistical analyses were conducted to assess the associations between myositis-specific autoantibodies and clinical symptoms.
Results
Among the 357 patients with suspected idiopathic inflammatory myopathy, the co-occurrence rate of anti-synthetase and anti-Ro52 was 52.63%. Univariate analysis demonstrated significantly higher diagnostic rates of idiopathic inflammatory myopathy in patients positive for anti-synthetase and anti-Ro52 than in those who were seronegative. Patients who tested positive for anti-signal recognition particle exhibited distinct serological profiles and diagnostic outcomes compared with those who tested negative. Significant associations were observed between myositis-specific autoantibodies and tumor biomarkers. Multivariate logistic regression analysis identified serum creatinine and anti-synthetase antibody positivity as independent diagnostic predictors of idiopathic inflammatory myopathy.
Discussion
Anti-synthetase antibodies were independently associated with idiopathic inflammatory myopathy. Co-positivity for anti-synthetase and anti-Ro52 was closely related to the diagnosis of idiopathic inflammatory myopathy. Myositis antibodies were significantly correlated with tumor biomarkers, highlighting their potential utility in early cancer screening. Elevated serum creatinine was also independently associated with idiopathic inflammatory myopathy, exhibiting diagnostic significance.
Conclusion
Myositis-specific autoantibodies have potential clinical value for early tumor screening and the diagnosis of idiopathic inflammatory myopathy.
Keywords
Introduction
Idiopathic inflammatory myopathy (IIM) comprises a heterogeneous group of autoimmune disorders characterized by a subacute onset and progressive proximal muscle weakness, often resulting in significant functional impairment. The disease spectrum classically includes five major subtypes, namely, dermatomyositis (DM), anti-synthetase syndrome (ASS), immune-mediated necrotizing myopathy (IMNM), polymyositis (PM), and inclusion body myositis (IBM). 1 Notably, patients diagnosed with PM in early pathological observations were reclassified as having IMNM or ASS in many cases, 2 leading to a significant reduction in the number of patients in the PM subgroup. 3 IBM was excluded because it is nonamenable to treatment. 4 First-line treatments typically involve systemic glucocorticoids (e.g. prednisone), often combined with immunosuppressive agents in refractory cases. Notably, extramuscular involvement frequently accompanies IIM, occasionally preceding or overshadowing myopathic symptoms. Key systemic features include cutaneous eruptions, interstitial lung disease (ILD), overlapping connective tissue disorders, and cardiac complications. 1 Clinical heterogeneity in IIM is particularly pronounced at disease onset, with substantial variability in symptom presentation, severity, and organ-specific involvement.
Multiple autoantibodies have been classified as myositis-specific autoantibodies (MSAs) or myositis-associated autoantibodies (MAAs). 5 MSAs serve as disease-specific biomarkers for diagnosing IIM. In clinical scenarios where muscle biopsy is contraindicated or atypical symptoms are present, MSAs demonstrate considerable diagnostic utility. 6 Beyond diagnosis, these autoantibodies carry prognostic implications and may inform therapeutic decisions. 7 Emerging evidence indicates that MSA profiling enables precise clinical subclassification in IIM patients. 6 Considering these clinical implications, this study systematically investigated the correlations between MSA profiles, diagnostic categories, and serological parameters, aiming to provide evidence-based insights to optimize diagnostic and therapeutic strategies for patients with suspected IIM.
Classical MSAs include anti-synthetase, anti-Mi2, and anti-signal recognition particle (SRP) antibodies. Anti-Mi2 antibodies are associated with classic DM, correlating with a favorable prognosis, response to corticosteroids, and low malignancy rates. 8 Anti-SRP antibodies are linked to IMNM, typically characterized by a severe and acute onset, although chronic insidious presentations have also been documented. 9 Anti-Jo-1 was the first anti-synthetase antibody identified. In addition to anti-Jo-1, seven anti-synthetase antibodies have been reported, generally with epidemiological prevalence rates between 1% and 5% or lower. In this study, we focused on four common anti-synthetase antibodies, namely, anti-Jo-1, anti-PL-12, anti-PL-7, and anti-EJ. With rare exceptions, each patient typically exhibits only one of these autoantibodies. However, patients with ASS share similar clinical manifestations, such as myositis, ILD, polyarthritis, fever, Raynaud’s phenomenon, and mechanic’s hands, collectively defining ASS. 10
Research indicates that the risk of malignancy significantly increases within the 3 years before IIM onset. 11 Autoantibody seroconversion frequently precedes clinical symptom onset in IIM. This study focused on patients with suspected IIM who exhibit clinical features that do not meet the current classification criteria for a definitive diagnosis. The study objectives were to characterize the prevalence and patterns of MSAs in this pre-diagnostic population and evaluate potential correlations between autoantibody profiles, progression to definitive IIM diagnosis, and tumor marker expression. The findings may offer clinical utility in early intervention strategies and oncological surveillance in these patients.
Methods
This retrospective study analyzed the myositis autoantibody profiles and clinical characteristics of 357 patients with suspected IIM treated between 1 January 2021 and 30 September 2023. 12 Clinical data were extracted retrospectively from electronic medical records and included sex, age, alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatine kinase (CK), lactate dehydrogenase (LDH), α-hydroxybutyric dehydrogenase (αHBDH), albumin (ALB), creatinine (CRE), neuron-specific enolase (NSE), carcinoma embryonic antigen (CEA), carbohydrate antigen 153 (CA153), CYFRA21-1 (CYF), carbohydrate antigen 724 (CA724), carbohydrate antigen 199 (CA199), alpha-fetoprotein (AFP), carbohydrate antigen 125 (CA125), anti-tyrosine (anti-Jo-1), anti-alanine (anti-PL-12), anti-threonine (anti-PL-7), anti-glycine (anti-EJ), anti-Mi2, anti-SRP, and anti-Ro52. The diagnosis of IIM was established according to the 2017 EULAR/ACR classification criteria. 1
The inclusion and exclusion criteria were established using Walter’s framework. 4 The inclusion criteria were as follows: (a) symmetrical proximal muscle weakness necessitating high-dose glucocorticoid therapy; (b) symptom duration ≤24 months; and (c) patients with a connective tissue disorder and/or cancer. The exclusion criteria were as follows: (a) alternative etiologies for muscle weakness (medication-induced myotoxicity and inherited neuromuscular disorders); (b) neurological evaluations indicative of neurogenic pathology (distal weakness greater than proximal weakness, asymmetric presentation, and distal muscle atrophy or fasciculations).
Final enrollment decisions were made by board-certified rheumatologists following comprehensive diagnostic evaluations.
The study was conducted in accordance with the Declaration of Helsinki (1975, as revised in 2013). Patient data were de-identified to protect confidentiality; no personally identifiable information was retained. The reporting of this study conformed to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 12
Antibody detection
Myositis autoantibodies were detected using a line immunoblot assay according to the manufacturer’s instruction (EUROLINE: Autoimmune Inflammatory Myopathies 16 Ag, EUROIMMUN, Lübeck, Germany). Autoantibody results were categorized as negative, weakly positive, moderately positive, or strongly positive; only moderate or strong positive results were considered.
Serological parameter detection
Serum ALT, AST, CK, LDH, αHBDH, ALB, and CRE levels were measured using Hitachi 7600 Automatic Analyzer. Serum NSE, CEA, CA153, CYFRA21-1, CA724, CA199, AFP, and CA125 were measured using Roche Cobas 8000 Automatic Analyzer. Calibrators and quality-control materials matching the reagent standards were used, and quality control assessments were performed at high, medium, and low levels.
Statistical analysis
Statistical analyses were performed using SPSS software (version 25; SPSS Inc., Chicago, IL, USA). Quantitative data with a non-normal distribution were presented as medians with interquartile ranges (M (P25, P75)), and the Mann–Whitney U test was employed for intergroup comparisons. Qualitative data were compared between the two groups using the chi-squared test. Multiple logistic regression with a stepwise approach was conducted to identify independent risk factors. Receiver operating characteristic (ROC) curves were constructed to evaluate diagnostic performance. Statistical significance was set at P < 0.05.
Results
Distribution of MSAs in 357 patients with suspected IIM
Among 357 patients with suspected IIM, 40 tested positive for MSAs, of whom 45% showed positivity for anti-synthetase antibodies. Table S1 shows the general characteristics of the sample. The positivity rates for anti-Jo-1, anti-PL-12, anti-PL-7, and anti-EJ antibodies in the anti-synthetase group are presented in Figure 1. One patient was positive for both anti-Mi2 and anti-synthetase antibodies. This patient exhibited co-positivity for anti-Mi2 and anti-PL12.

Distribution of MSAs in 357 patients with suspected IIM. N (%). MSAs: myositis-specific autoantibodies; IIM: idiopathic inflammatory myopathy.
Co-positivity for MSA with anti-Ro52
The number and frequency of patients with MSA co-positivity with anti-Ro52 are presented in Table 1. Among 357 patients, 50 were positive for anti-Ro52 antibodies. The frequency of co-positivity for anti-Ro52 with anti-synthetase antibodies was 52.63%, whereas that for anti-Ro52 with anti-SRP antibodies was 41.67%.
The frequency and number of patients with MSA co-positivity with anti-Ro52.

MSA: myositis-specific autoantibody; IIM: idiopathic inflammatory myopathy; SRP: signal recognition particle.
Clinical characteristics of patients positive for anti-synthetase antibodies
Table 2 summarizes the clinical characteristics of patients who were positive versus those who were negative for anti-synthetase antibodies. Among the 357 patients, 338 were negative for anti-synthetase antibodies. To minimize potential bias from untested antibodies, patients who were negative for anti-synthetase antibodies, rather than all who were negative for antibodies, were employed as controls. Patients who were positive for anti-synthetase antibodies exhibited significantly higher IIM diagnostic rates than those who were negative. The diagnosis rate of IIM in patients who were negative for anti-synthetase antibodies was 21.01%. Serological parameters, including CK, LDH, αHBDH, ALB, and CK/CRE, differed significantly between the two groups.
Clinical characteristics of patients positive for anti-synthetase antibodies.

IQR: interquartile range; ALT: alanine aminotransferase; AST: aspartate aminotransferase; CK: creatine kinase; LDH: lactate dehydrogenase; αHBDH: α-hydroxybutyric dehydrogenase; ALB: albumin; CRE: creatinine; IIM: idiopathic inflammatory myopathy.
Clinical characteristics of patients positive for anti-Mi2 antibodies
Table 3 displays the clinical characteristics of patients who were positive versus those who were negative for anti-Mi2 antibodies. The analysis revealed no significant differences in the characteristics between the two groups, except for CK/CRE ratios.
Clinical characteristics of patients positive for anti-Mi2 antibodies.

IQR: interquartile range; ALT: alanine aminotransferase; AST: aspartate aminotransferase; CK: creatine kinase; LDH: lactate dehydrogenase; αHBDH: α-hydroxybutyric dehydrogenase; ALB: albumin; CRE: creatinine; IIM: idiopathic inflammatory myopathy.
Clinical characteristics of patients positive for anti-SRP antibodies
Significant differences were observed in the serum ALT, AST, CK, LDH, αHBDH, and CRE levels; CK/CRE ratios; and IIM diagnostic rates between patients who were positive versus those who were negative for anti-SRP antibodies (Table 4).
Clinical characteristics of patients positive for anti-SRP antibodies.

SRP: signal recognition particle; IQR: interquartile range; ALT: alanine aminotransferase; AST: aspartate aminotransferase; CK: creatine kinase; LDH: lactate dehydrogenase; αHBDH: α-hydroxybutyric dehydrogenase; ALB: albumin; CRE: creatinine; IIM: idiopathic inflammatory myopathy.
Clinical characteristics of patients positive for anti-Ro52 antibodies
Significant differences were observed in the AST, LDH, αHBDH, ALB, and CRE levels; CK/CRE ratio; sex distribution; and IIM diagnostic rates between patients who were positive versus those who were negative for anti-Ro52 antibodies (Table 5).
Clinical characteristics of patients positive for anti-Ro52 antibodies.
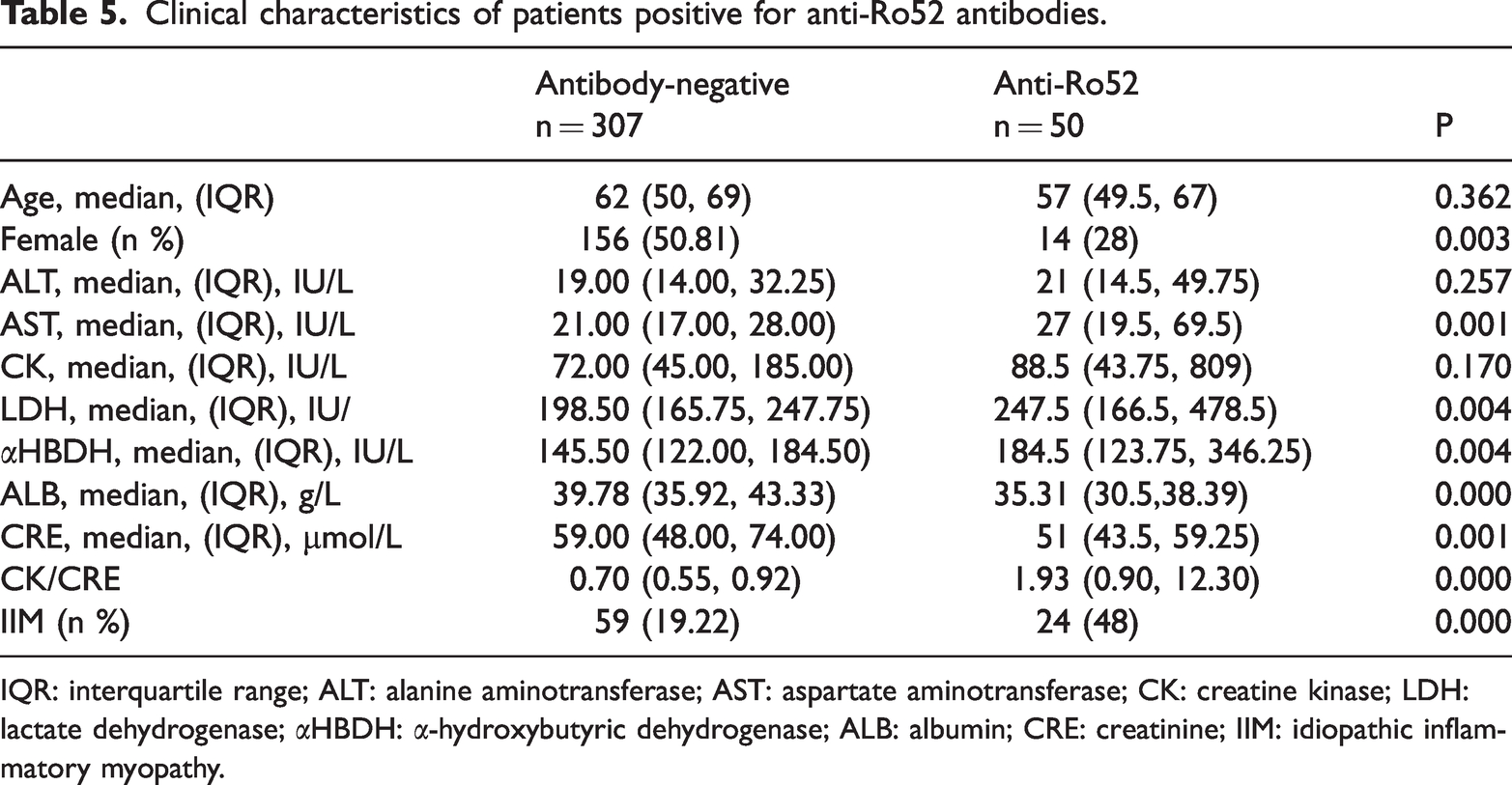
IQR: interquartile range; ALT: alanine aminotransferase; AST: aspartate aminotransferase; CK: creatine kinase; LDH: lactate dehydrogenase; αHBDH: α-hydroxybutyric dehydrogenase; ALB: albumin; CRE: creatinine; IIM: idiopathic inflammatory myopathy.
Relationship between tumor markers and myositis antibodies
Patients who were positive for anti-synthetase antibodies exhibited significantly higher CA199 levels than those who were negative (Figure 2). Patients who were positive for anti-SRP antibodies showed significantly higher NSE levels than those who were negative. Patients positive for anti-Ro52 antibodies demonstrated significantly higher levels of NSE, CA153, and CA125 than those who were negative. In contrast, no significant elevation was observed in the tumor biomarkers between patients who were positive versus those who were negative for anti-Mi2 antibodies.

Relationship between tumor markers and myositis antibodies. N: negative, P: positive. *P < 0.05.
Multiple logistic regression analysis of IIM
Table 6 presents a comparative analysis of serological parameters between patients with and without IIM. Variables including age; ALT, AST, CK, LDH, αHBDH, ALB, CRE, NSE, and CEA levels; and CK/CRE ratio showed statistically significant differences between the two groups.
Univariate analysis of serological parameters between the IIM and non-IIM groups.
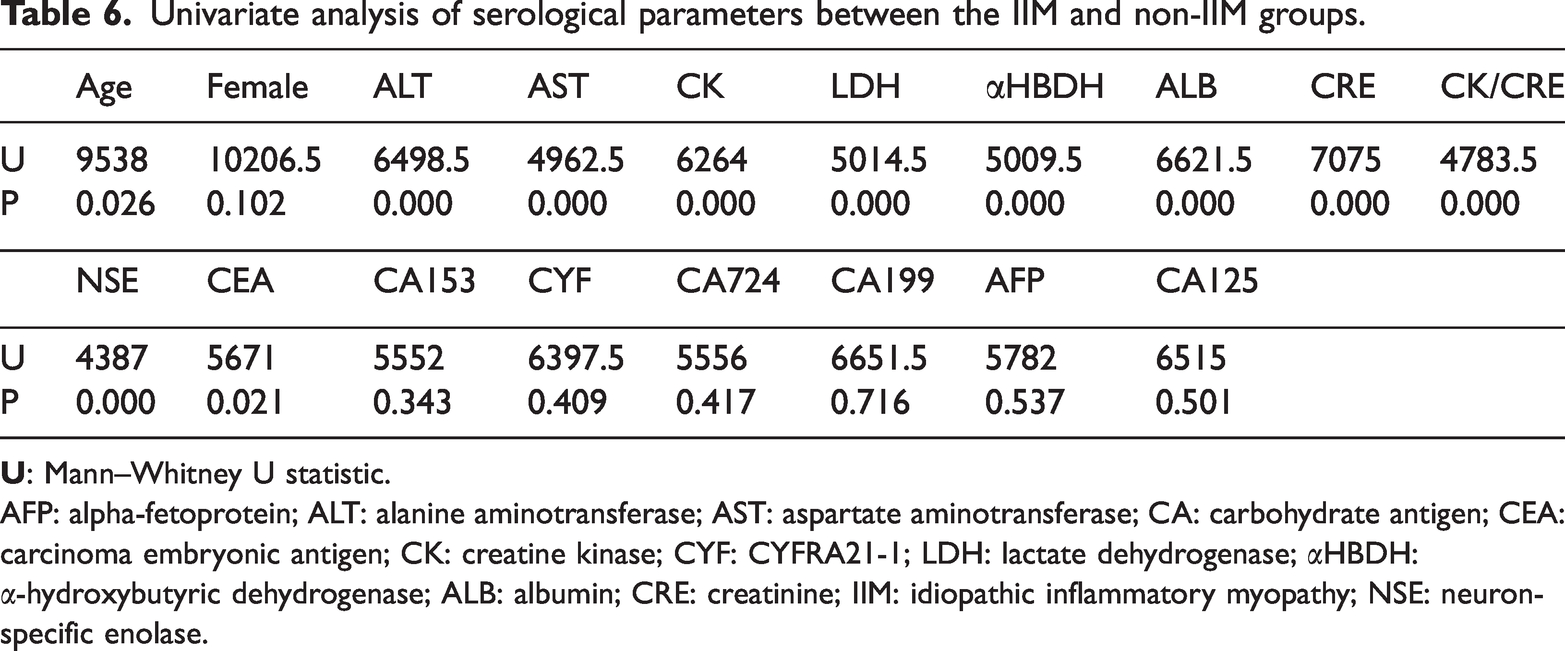
AFP: alpha-fetoprotein; ALT: alanine aminotransferase; AST: aspartate aminotransferase; CA: carbohydrate antigen; CEA: carcinoma embryonic antigen; CK: creatine kinase; CYF: CYFRA21-1; LDH: lactate dehydrogenase; αHBDH: α-hydroxybutyric dehydrogenase; ALB: albumin; CRE: creatinine; IIM: idiopathic inflammatory myopathy; NSE: neuron-specific enolase.
Using IIM diagnosis as the dependent variable, multiple logistic regression analysis incorporating significant variables from the univariate analysis identified CRE and anti-synthetase antibody positivity as independent factors influencing IIM (Table 7).
Multiple logistic regression analysis of IIM.

ALT: alanine aminotransferase; AST: aspartate aminotransferase; CEA: carcinoma embryonic antigen; CK: creatine kinase; LDH: lactate dehydrogenase; αHBDH: α-hydroxybutyric dehydrogenase; ALB: albumin; CRE: creatinine; IIM: idiopathic inflammatory myopathy; NSE: neuron-specific enolase; OR: odds ratio; CI: confidence interval.
ROC analysis for predictors of IIM
As CRE was identified as an independent predictor of IIM, ROC curves were plotted for CRE and CK/CRE (Figure 3). The ROC analyses showed statistically significant results (Table 8).

ROC analysis for predictors of IIM. ROC: receiver operating characteristic; IIM: idiopathic inflammatory myopathy.
ROC analysis for predictors of IIM.

AUC: area under the curve; CK: creatine kinase; CRE: creatinine; IIM: idiopathic inflammatory myopathy; CI: confidence interval.
Discussion
In patients with suspected IIM, the co-occurrence rate of anti-synthetase and anti-Ro52 antibodies was high (52.63%), while the co-positivity rate for anti-SRP and anti-Ro52 antibodies reached 41.67%. The underlying mechanisms of Ro-52 antibody co-occurrence with other myositis antibodies remain unclear. Anti-Ro52 antibodies were most frequently co-positive with anti-synthetase antibodies, suggesting their common involvement in specific immune response pathways. A previous report indicated that co-positivity rates of anti-Ro52 with other anti-synthetase antibodies, such as anti-EJ, anti-OJ, anti-PL-7, and anti-PL-12, range from 33% to 100%. 13 In our study, 80% of patients co-positive for anti-Ro52 and anti-synthetase antibodies were diagnosed with IIM. Logistic regression analysis identified anti-synthetase antibody positivity as an independent factor associated with IIM diagnosis. Additionally, patients who were double-positive for anti-Jo-1 and anti-Ro52 antibodies reportedly have poorer prognoses. 14 Antibody co-positivity impacts disease progression and prognosis. Clinical attention should be paid to the co-positivity of anti-Ro52 with anti-synthetase antibodies.
In our study, 92% of anti-SRP antibody-positive patients were diagnosed with IMNM, indicating that anti-SRP antibodies have very high specificity for diagnosing IIM. Studies on IMNM pathogenesis have demonstrated that anti-SRP antibodies may induce muscle weakness and fiber necrosis in mice, 15 suggesting that anti-SRP antibodies are not only diagnostic biomarkers but may also play a crucial role in IMNM pathogenesis. It is established that serum CK levels in IMNM are higher than those in other IIM subtypes.16,17 Our findings showed significantly elevated levels of serum indicators such as ALT, LDH, and αHBDH in patients with anti-SRP positivity and decreased CRE levels. These serological markers closely reflect the pathological mechanisms of IMNM, characterized by severe, rapidly progressive muscle weakness and muscle fiber necrosis, with resultant muscle mass reduction leading to decreased CRE levels.
Anti-Mi2 antibodies target the nucleosome remodeling deacetylase complex. Previous clinical studies have reported the association of anti-Mi2 positivity with DM-specific clinical features, including Gottron’s papules, heliotrope rash, shawl sign, and V-sign. 18 Patients positive for anti-Mi2 typically respond favorably to steroid treatment and have a better prognosis. 19 Prior studies have shown significantly elevated CK levels in DM patients positive for anti-Mi than those who were negative. 20 In contrast, our study showed no statistically significant differences in CK and LDH levels or other indicators between anti-Mi2-positive and -negative patients. However, the CK/CRE ratio was significantly higher in anti-Mi2 antibody-positive patients than in those who were negative, suggesting that the CK/CRE ratio serves as a more sensitive marker for muscle injury. Only 30% of anti-Mi2 antibody-positive patients met the diagnostic criteria for IIM, indicating low diagnostic specificity of anti-Mi2 for IIM. Previous research has shown that patients positive for anti-Mi2 do not tend to manifest symptoms of multi-system involvement, such as pulmonary involvement or polyarthritis. 21 Furthermore, the serological markers and tumor markers showed no significant differences between anti-Mi2 antibody-positive and -negative groups, suggesting that clinical manifestations in anti-Mi2 antibody-positive patients were relatively mild.
The risk of cancer increases within 3 years before and after the onset of IIM. 22 Up to 25% of IIM patients are diagnosed with cancer within 3 years of disease onset, 23 with the most common malignancies including lung cancer, ovarian cancer, colorectal cancer, lymphoma, breast cancer, and nasopharyngeal carcinoma. 24 Cancer remains the leading cause of death in adults with IIM, partly due to delayed diagnosis of cancer. 25 The majority of IIM-associated cancers are diagnosed at an advanced stage. 26 Therefore, we investigated the relationship between myositis antibodies and tumor markers to identify suitable risk factors for early cancer screening. The International Guideline for IIM-Associated Cancer Screening recommends anti-Ro52, anti-SRP, anti-synthetase, and anti-Mi2 antibodies as indicators for cancer screening. 22 Our study found significantly elevated levels of CA199 in anti-synthetase antibody-positive patients; significantly elevated levels of NSE in anti-SRP antibody-positive patients; and significantly elevated levels of NSE, CA153, and CA125 in anti-Ro52 antibody-positive patients. However, no significant elevations in tumor markers were observed among anti-Mi2 antibody-positive patients. In our cohort, only 30% of anti-Mi2 antibody-positive patients were diagnosed with IIM. Additionally, a previous study reported an anti-Mi2 antibody positivity rate of 19.2% in patients with a definitive diagnosis of IIM. 6 Therefore, the lack of elevated tumor markers in anti-Mi2 antibody-positive patients may be attributable to the relatively small number of patients with a confirmed diagnosis of IIM in this subgroup.
We found that 49% of patients positive for myositis antibodies were not diagnosed with IIM. International guidelines indicate an increased cancer risk within 3 years before IIM onset, and myositis antibodies typically appear before the clinical manifestation of IIM. Therefore, understanding the relationship between myositis antibodies and cancer is crucial for early cancer screening. Our research demonstrated significant associations between specific myositis antibodies and elevated tumor markers. Anti-synthetase antibodies were correlated with increased levels of CA199, an important marker commonly elevated in pancreatic, colorectal, liver, and gastric cancers. Anti-SRP antibodies were correlated with NSE, a recognized marker for small cell lung cancer. Anti-Ro52 antibodies were correlated with elevated levels of NSE, CA153, and CA125, which are markers associated with lung and liver cancers. Tumor markers exhibit certain limitations in terms of both sensitivity and specificity; thus, they should not be used as standalone diagnostic indicators. However, positive tumor marker results indicate a cancer risk in patients. 26 In our study, we specifically investigated the correlation between MSA and tumor markers, with the findings providing evidence for the potential association between myositis antibodies and tumors.
We also collected preliminary data on malignancies within our study population, identifying two cases of malignancy among anti-synthetase antibody-positive patients, two cases among anti-SRP antibody-positive patients, and five cases among anti-Ro52 antibody-positive patients. Our findings suggest that tumor biomarker profiles vary according to different MSA subsets. However, the absence of long-term follow-up represents a limitation. Future studies incorporating 3–5 years of follow-up data to evaluate cancer incidence in patients with distinct antibody profiles are warranted.
Logistic regression analysis identified serum CRE as an independent predictor of IIM. Creatine in the muscle tissue is converted into CRE, released into the circulation, and cleared by the kidneys. 27 Reduced muscle mass decreases the amount of creatine available for its conversion into CRE. Although CK is commonly employed as a diagnostic biomarker for myositis, variations in serum CK levels due to membrane pathology and residual muscle mass may limit its utility. The rate of conversion from creatine to CRE is more directly related to muscle mass reserve. 28 Table S2 shows statistically significant differences in the serum CK and CRE levels as well as the CK/CRE ratio among the three antibody groups compared with other indicators. Thus, we included the CK/CRE ratio as a more sensitive indicator. A previous study reported that the level of serum CRE decreases in various neuromuscular diseases. 29 Studies examining CRE levels in IIM patients have shown significantly lower levels during the active disease phase compared with those during the inactive phases; 30 moreover, in juvenile DM, the urinary CRE/choline ratio was correlated with disease severity. 31 Our results similarly indicated that decreased serum CRE level was independently associated with IIM occurrence. Reduced muscle mass represents a critical pathological change in IIM. The ROC curve analyses of CRE and CK/CRE yielded area under the curve values of 0.662 and 0.738, respectively, underscoring the potential diagnostic significance of the CK/CRE ratio in IIM.
Conclusion
In summary, anti-synthetase antibodies are independent predictors of IIM. Co-positivity for anti-synthetase and anti-Ro52 antibodies is closely related to IIM diagnosis and warrants clinical attention. Myositis antibodies are significantly correlated with tumor markers and thus have considerable utility in early cancer screening. Decreased CRE level is an independent factor associated with IIM, and the CK/CRE ratio has notable diagnostic value.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251362969 - Supplemental material for The value of myositis-specific autoantibodies in the diagnosis of idiopathic inflammatory myopathy and tumor risk prediction
Supplemental material, sj-pdf-1-imr-10.1177_03000605251362969 for The value of myositis-specific autoantibodies in the diagnosis of idiopathic inflammatory myopathy and tumor risk prediction by Lili Shi, Rong Min, Yanyan Wu, Jiahui Liu, Xia Liu and Peichang Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251362969 - Supplemental material for The value of myositis-specific autoantibodies in the diagnosis of idiopathic inflammatory myopathy and tumor risk prediction
Supplemental material, sj-pdf-2-imr-10.1177_03000605251362969 for The value of myositis-specific autoantibodies in the diagnosis of idiopathic inflammatory myopathy and tumor risk prediction by Lili Shi, Rong Min, Yanyan Wu, Jiahui Liu, Xia Liu and Peichang Wang in Journal of International Medical Research
Footnotes
Acknowledgements
The authors would like to acknowledge the support by the HUIZHI Talent Leadership Development Program of Xuanwu Hospital as well as Beijing Sail Plan for Talents Development.
Author contributions
PW and LS put forward research ideas and set the overall research goals. YW organized and analyzed the data. LS was responsible for writing the manuscript. The remaining authors assisted in collecting the data and writing the manuscript. All authors contributed to the article and approved the submitted version.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Consent for publication
None.
Ethics approval
This was an observational study. The Ethics Committee of Xuanwu Hospital, Capital Medical University, has confirmed that there is no need for ethical approval.
Funding
This study received funding from the HUIZHI Talent Leadership Development Program of Xuanwu Hospital (Grant/Award Number: HZ2021PYLJ023) as well as Beijing Sail Plan for Talents Development (Grant/Award Number: ZYLX202114).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
