Abstract
Immune-related adverse events (irAE) during the administration of immune-checkpoint inhibitors (ICIs) become more evident due to the increased use of these therapies. To remind the importance of early recognition of this phenomenon, we report a paradigmatic case characterized by severe systemic inflammatory myopathy and severe cardiac involvement that abruptly precipitated in an untoward ending after one single dose of Pembrolizumab.
Keywords
INTRODUCTION
Immune-checkpoint inhibitors (ICIs), including Pembrolizumab, have shown a remarkable benefit in the treatment of advanced and metastatic malignancies [1]. Pembrolizumab has been recently approved for the treatment of metastatic melanoma and advanced/refractory non-small-cell lung cancer [2, 3].
Immune-related adverse events (irAE) during the administration of ICIs are becoming more evident with the spread of these therapies. Most of irAE are of low-grade and can be managed successfully with only supportive care, while neuromuscular irAE can be extremely serious, requiring prompt diagnosis and aggressive treatment [4].
The specific characteristics of neuromuscular irAE have not been defined yet because of the low frequency (ranging from 1% to 2%) of cancer patients undergoing therapy with ICIs [5]. However, according to the literature, myositis and myasthenia-like syndrome seem the most representative neuromuscular irAE [6]. Morphological muscle features of patients with irAE myositis have been already described [7], while immunological features, in particular expression of Human Leukocyte Antigen type I (HLA I), anti-membrane attack complex (MAC) deposition and cell typing, have not been fully clarified.
The predictable future spread of ICIs could also lead to an increase of irAE and possibly related fatal events, especially in patients who develop necrotizing myopathy [8].
Our report falls within this scenario, as it is characterized by severe systemic inflammatory myopathy along with deep cardiac involvement, abruptly precipitated in an untoward ending after one single dose of Pembrolizumab.
CASE REPORT
The patient was a 60 years old man suffering from grade IV A pulmonary squamous cell carcinoma (p40+, Calretinin-, WT1-, Ck7-, CK20-) with pleural-pericardial involvement. The patient had a previous history of meningioma treated with radiotherapy, hypertension and ischemic cardiopathy. A previous cardiac echo-color Doppler test showed a good cardiac output; there were no signs of heart failure.
The patient received one single dose of pembrolizumab 200 mg and after 7 days he started complaining of neck pain and walking difficulties. He was admitted to emergency care and blood tests revealed Creatine kinase (CK) highly raised level (20.000 U/l); troponin rate was mildly elevated (40 ng/L–normal range 2–20 ng/l); the clinical examination showed head drop and limbs proximal weakness (deltoid 3/5, biceps brachii 4/5, iliopsoas 3/5 quadriceps 3/5 at MRC scale).
EMG showed diffuse denervation along with myopathic signs suggestive of muscle inflammation. Repetitive nerve stimulation did not show any decrement. Blood tests for anti-acetylcholine receptor (AChR), anti-muscle-specific tyrosine kinase (MuSK) anti-Jo1, anti-SRP and anti-HMGCR autoantibodies turned out negative. Hydration was immediately started along with a daily high dose of steroids. Serum troponin before steroid administration was stable around 35 ng/L.
One day later, the patient started complaining of diplopia (without neither nystagmus nor overt gaze paralysis) and mild swallowing disturbances. Brain CT scan excluded major cerebral events; CK level decreased to 9000 U/L with mild strength improvement in upper limbs (deltoid 4/5); no sign of heart failure was detected, and control electrocardiograms were normal. The patient underwent a muscle biopsy in the left biceps muscle that showed a severe, acute inflammatory myopathy: the sample presented several necrotic-degenerating muscle fibers with massive cell infiltration (Fig. 1). Human Leukocyte Antigen type I (HLA I) positivity was detected by immunohistochemistry on the sarcolemma and in the cytoplasm of numerous fibers and in all the inflammatory cellular infiltrations (A). Anti-membrane attack complex (MAC) antibodies were positive in numerous capillaries (B). Cell typing revealed the presence of CD4+ and CD8 + T-cells (C e D) (Fig. 2). CD68+ macrophages were also detected (not shown).
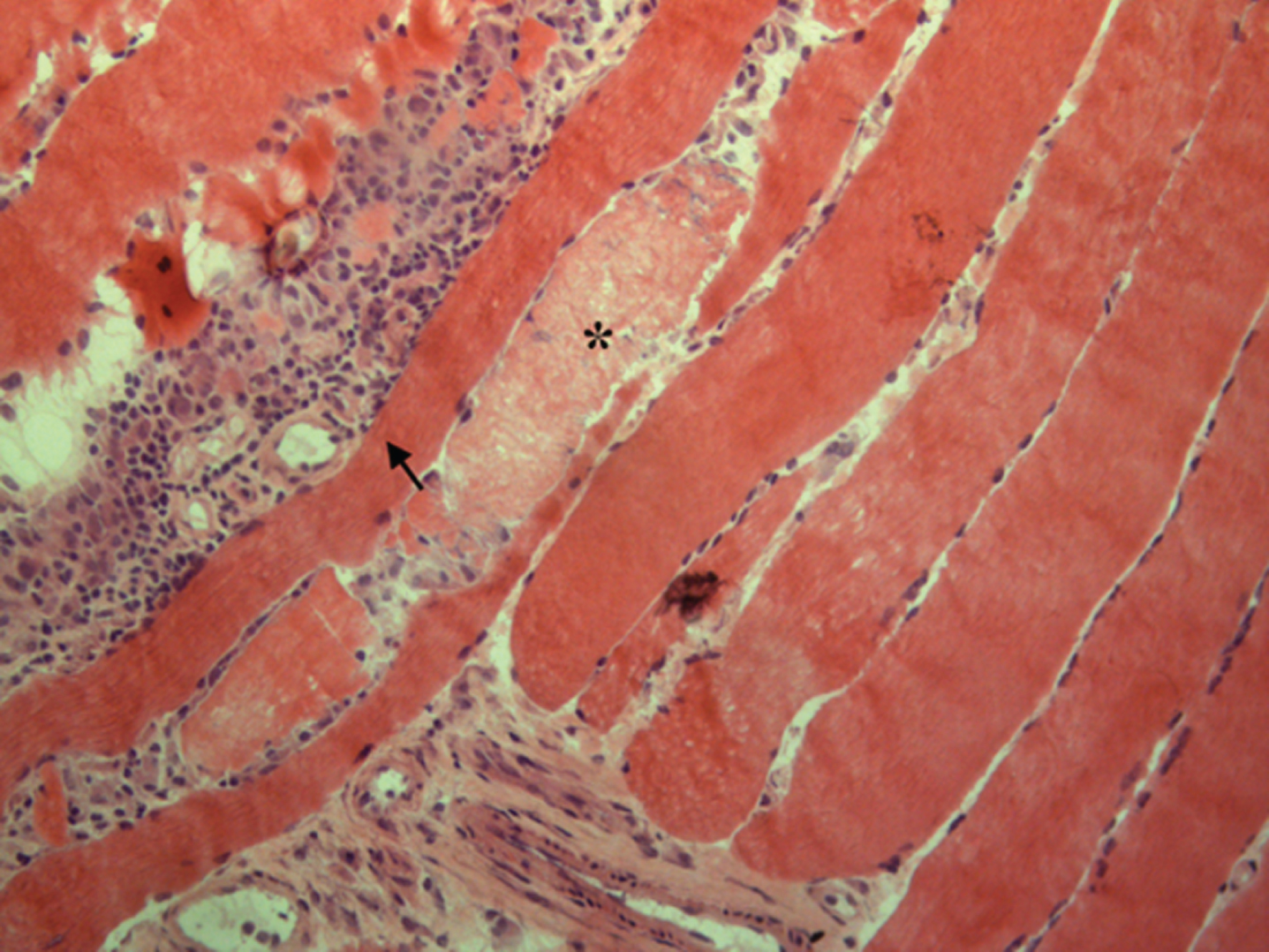
Histological findings with hematoxylin-eosin staining revealing some necrotic-degenerating muscle fibers (star) with a wide interstitial and perivascular inflammatory cellular infiltration (arrowhead).
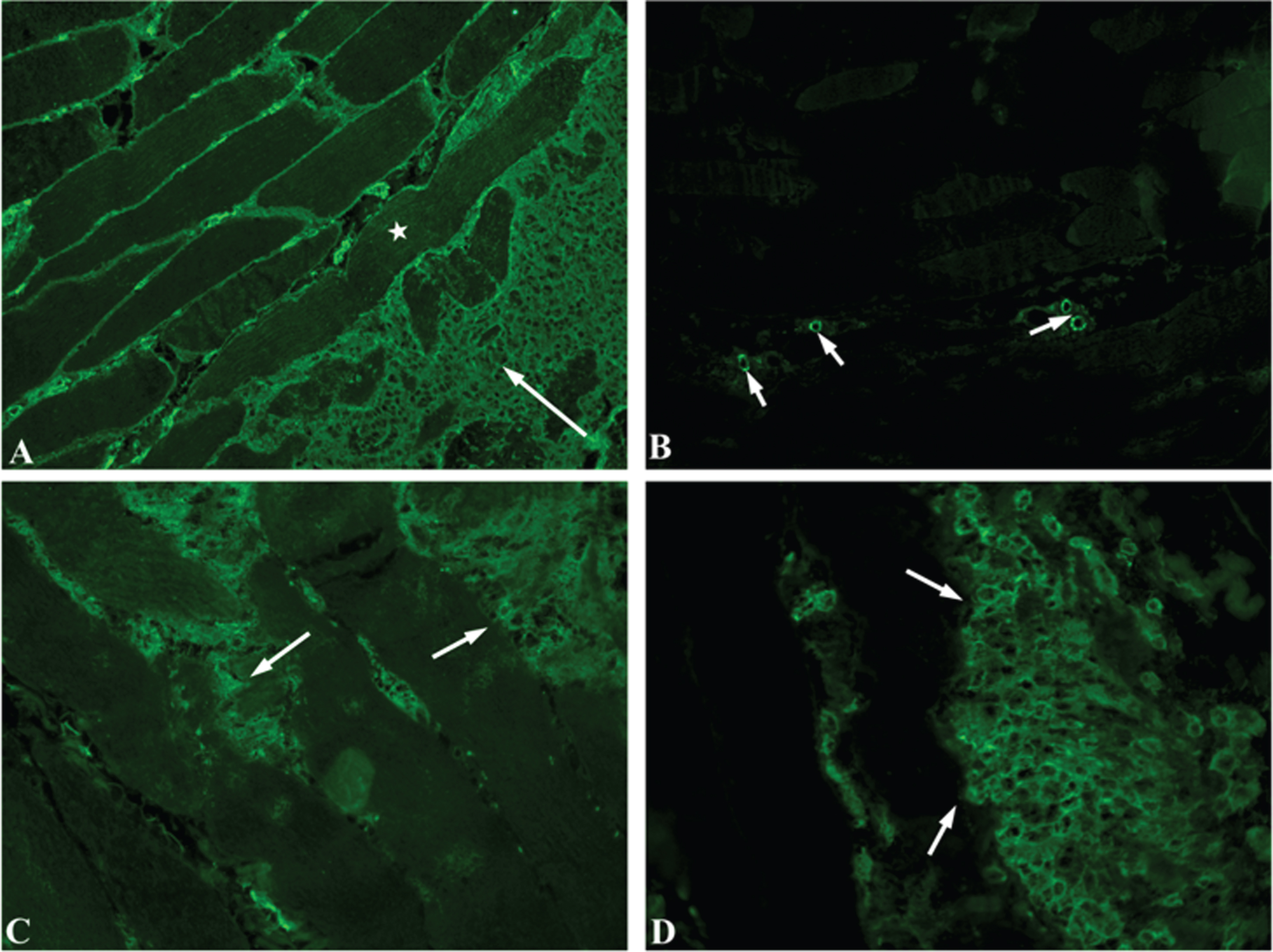
Indirect immunofluorescence. HLA-I positivity on the sarcolemma and in the cytoplasm of myofiber (star) and in the inflammatory cell infiltrate (arrow) (image A, magnification: 200×); MAC deposition on small blood vessels (arrows) (image B, magnification: 100×); CD4+ (image C, magnification 200×) and CD8+ (image D, magnification 400×) large endomysial T-cells infiltrates (arrows).
Two days after starting steroid therapy, the patient presented acute heart failure and was admitted in the intensive care unit; blood cardiac troponins were over 5000 times the normal levels. Despite of external pace maker implantation (due to sudden onset of atrio-ventricular block and severe reduction in heart-rate) and vaso-active support, the patient passed away one day later.
The last blood examination test showed reduction of CK (4688 U/L) and further increase in Troponin rate (70225 ng/L). A Doppler echocardiography performed at the time showed a severe reduction in cardiac output (estimated around 25%) and marked left ventricular apex hypo-akinesia and septal hypokinesia. No pericardial effusions were detected.
DISCUSSION AND CONCLUSION
This case emphasizes that patients taking pembrolizumab are at risk of developing necrotizing myositis and highlights the importance of early diagnosis and recognition of inflammatory myopathy during immunotherapy with ICIs. At the onset of overt muscular symptoms, our patient was immediately treated with high dose of corticosteroids; the initial muscle strength recovery was followed by an unexpected precipitating heart failure.
Although fatal cases of myocarditis induced by ICIs therapy have already been reported [9], they usually respond to high steroid doses. The case described here is noteworthy due to the rapid progression of cardiac impairment after steroid administration. It is possible that high dose of steroids contributed to exacerbate the previous ischemic cardiopathy, worsening the heart function already impaired by the underlying myocarditis.
This is, to our knowledge, the first case that, after one single dose of Pembrolizumab, rapidly evolved to a fatal outcome.
Seki et al. previously reported inflammatory myopathy in 4 patients after one single dose treatment of Pembrolizumab with recovery after immunosuppressive treatment [6]. Other authors described a patient that died for diffuse necrotic myositis two weeks after the second cycle of pembrolizumab [4].
So far, only few data are available concerning immunological features of Pembrolizumab-associated inflammatory myopathy [5, 6]. Muscle biopsy of our patient showed a severe inflammation characterised by T cells and macrophages, along with numerous necrotic-degenerating muscle fibers. While the exact pathogenesis underlying irAE myositis remains unclear, our immunological findings support the hypothesis that T-cell-mediated autoimmune response in muscles may be crucial [6].
Because of the potentially fatal nature of its adverse effects and the possibility of rapid progression, in the days following the administration of ICIs we recommend both close clinical and blood CK monitoring.
CONFLICT OF INTEREST
The authors have no conflict of interest to report
Footnotes
ACKNOWLEDGMENTS
None declared.
