Abstract
Immune checkpoint inhibitors, such as pembrolizumab, have demonstrated substantial therapeutic efficacy in the treatment of cancer. However, immune-related adverse events, including myasthenia gravis (MG), present significant clinical challenges. This study presents the case of a 75-year-old male patient who developed generalized acetylcholine receptor antibody-positive MG following the first infusion of pembrolizumab for cholangiocarcinoma. The patient’s symptoms rapidly progressed to an impending myasthenic crisis (MGFA Class IVB) within 20 days of pembrolizumab administration. Eculizumab was used as a rescue therapy due to the unavailability of conventional treatments, resulting in rapid and significant symptom relief, with sustained improvement during maintenance therapy. The use of eculizumab as rescue therapy presents a viable treatment option for pembrolizumab-induced MG, owing to its rapid therapeutic effects.
Keywords
Introduction
Immune checkpoint inhibitors (ICIs) have demonstrated significant therapeutic efficacy in cancer treatment; however, they are known to induce immune-related adverse events (irAEs).1,2 Pembrolizumab, a monoclonal antibody ICI that binds to programmed cell death receptor 1 (PD-1), has been reported to induce myasthenia gravis (MG). 3 Traditional therapies for pembrolizumab-induced MG include glucocorticoids, intravenous immunoglobulin (IVIg), and plasma exchange. However, the clinical outcomes of these therapies remain unsatisfactory.
Here, we present a case of pembrolizumab-induced impending crisis in anti-acetylcholine receptor (AChR) antibody-positive MG. Initiating eculizumab treatment resulted in a rapid response and alleviation of clinical symptoms, with significant improvement in respiratory muscle function. This approach avoided the need for alternative treatments, potentially involving multiple rounds of medication adjustments. Therefore, using eculizumab as rescue therapy provides a viable option for treating pembrolizumab-induced MG due to its rapid therapeutic effects.
Case description
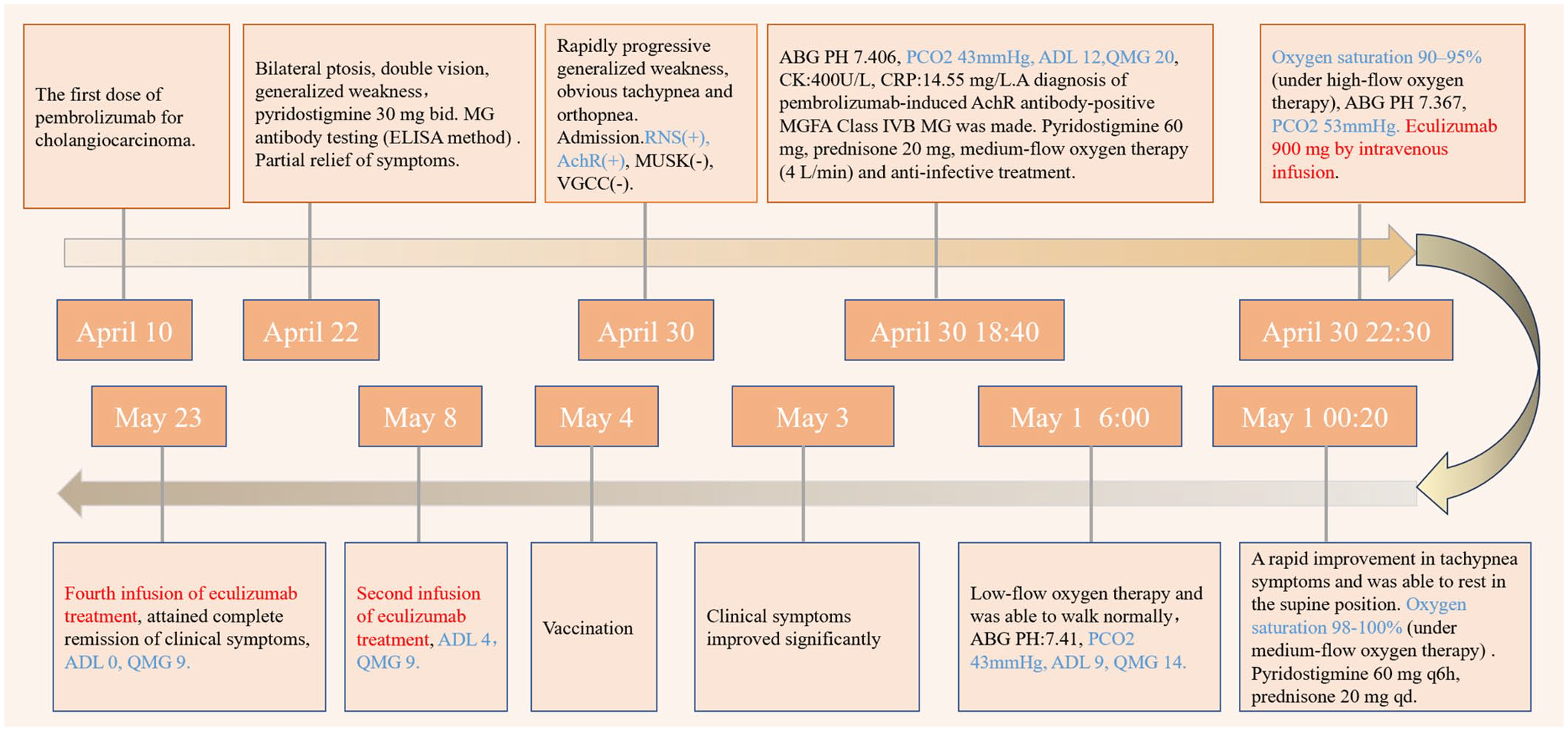
A 75-year-old male was admitted to our hospital on April 30, 2024, due to a 1-week history of ptosis and generalized weakness following the first infusion of pembrolizumab, with symptom worsening over the previous 2 days. One week prior to admission, the patient developed weakness in all limbs and increased fatigue after physical activity.
The patient required assistance while walking, was unable to lift both arms, and experienced neck weakness, bilateral ptosis, double vision, and tachypnea following physical activity. He also occasionally choked while drinking water, although he was able to swallow. The patient sought medical attention at a neurology outpatient clinic, where a diagnosis of MG was strongly considered. An MG antibody test was performed using an enzyme-linked immunosorbent assay (ELISA), and treatment with pyridostigmine (30 mg bid) was initiated, which alleviated the patient’s weakness. Two days prior to admission, his symptoms worsened: he became unable to walk, experienced pronounced tachypnea and dyspnea when lying down, could not sleep in the supine position, and developed orthopnea, which led to his hospitalization. On April 10, 2024, the patient received the first course of pembrolizumab for cholangiocarcinoma, along with capecitabine, an antineoplastic agent. He had a history of hepatitis B infection and cirrhosis for more than 10 years, treated with entecavir. In 2015, he underwent a hepatectomy for liver cancer and made a satisfactory recovery. During the physical examination, the patient was alert and spoke fluently but exhibited orthopnea and pronounced tachypnea. Bilateral ptosis was noted, along with an inability to adduct the right eye, diplopia, and weakness in neck extension. The Medical Research Council scale revealed 1/5 strength in neck flexors/extensors and 2/5 strength in the deltoids, biceps, triceps, quadriceps, and iliopsoas. The patient also tested positive for eyelid easy fatigability.
The repetitive nerve stimulation test yielded a positive result. Blood tests showed the following results: lactate, 2.80 mmol/L (elevated); procalcitonin, 0.095 ng/mL (elevated); interleukin-6, 30.23 pg/mL (elevated); quantitative C-reactive protein, 14.55 mg/L (elevated); creatine kinase (CK), 400 U/L (elevated); CK-MB, 2.73 μg/L (elevated); lactate dehydrogenase, 289 U/L (elevated); alanine aminotransferase, 55.7 U/L (elevated). Creatinine, routine blood tests, coagulation parameters, and pro-BNP were normal. Electrocardiography revealed sinus tachycardia, and echocardiography was normal. Chest-computed tomography showed reactive inflammation with atelectasis in the lower lobe of the right lung and a small amount of pleural effusion on the right side. Arterial blood gas (ABG) analysis performed at 18:40 showed a pH of 7.406, partial pressure of CO2 (pCO2) of 43 mmHg, and partial pressure of oxygen (pO2) of 87.2 mmHg. Scores on the Activities of Daily Living (ADL) and Quantitative Myasthenia Gravis (QMG) scales were 12 and 20 points, respectively. A positive serum AChR antibody test result of 35.35 nmol/L (<0.45 nmol/L, ELISA) was obtained. The patient tested negative for anti-MuSK, anti-Lrp4, anti-titin, and serum onconeural antibodies. Based on these findings, the diagnosis of pembrolizumab-induced AChR antibody-positive MGFA Class IVB MG was confirmed. Pyridostigmine (60 mg q6h), prednisone (20 mg), medium-flow oxygen therapy (4 L/min), and ceftriaxone for anti-infective treatment were immediately administered. The patient was only able to maintain orthopnea on exertion and exhibited gradual worsening of tachypnea. Oxygen saturation at the fingertip ranged from 90% to 95% under high-flow oxygen therapy. The patient had previously experienced severe gastrointestinal reactions while taking capecitabine. After increasing the oral dose of pyridostigmine, side effects such as abdominal pain and worsening nausea occurred. The patient refused further intramuscular or intravenous administration of pyridostigmine. A retest of ABG performed at 22:30 showed a pH of 7.367, pCO2 of 53 mmHg, and pO2 of 129 mmHg.
A diagnosis of an impending crisis state of severe MG was considered. Due to the late hour, it was difficult to promptly access blood products such as immunoglobulins or plasma. After thoroughly discussing the patient’s condition and obtaining informed consent from the patient and his family, we initiated treatment with eculizumab to help the patient safely navigate the impending crisis and avoid intubation. The patient was simultaneously administered eculizumab and prophylactic antibiotics, including benzathine penicillin (1.2 million U, once a week). The penicillin injection was administered to reduce the risk of severe meningococcal infection, as the patient had not completed meningococcal vaccination. Two hours after administration (00:20), improvement in his tachypnea was observed, and the patient was able to rest in the supine position. Oxygen therapy was reduced to medium flow. On the following day, an ABG retest revealed a pH of 7.41, pCO2 of 43 mmHg, and pO2 of 115 mmHg. The patient’s ADL and QMG scores improved to 9 and 14 points, respectively. The patient has opted to continue with regular eculizumab treatment, consisting of 900 mg for 4 consecutive weeks, followed by 1200 mg every 2 weeks.
Upon receiving treatment, the patient showed significant daily improvements in his symptoms. During the first 5 days of hospitalization (May 1–5), the patient required only low-flow oxygen therapy and was able to walk normally, assume the supine position, and resume ADL. His clinical symptoms improved markedly. He received a meningococcal vaccination and continued antibiotic prophylaxis with benzathine penicillin for an additional 14 days. Retesting of the AChR antibody using radioimmunoassay yielded a positive result of 4.31 nmol/L (<0.5 nmol/L). On May 8, a second infusion of eculizumab was administered.
Patient reassessment using the ADL and QMG scales revealed significant improvements, with scores of 4 and 9 points, respectively. Blood retesting showed normal levels of cardiac enzymes, C-reactive protein, and creatinine. The patient demonstrated substantial clinical improvement within 1 week, achieved complete remission of clinical symptoms by week 4 of eculizumab treatment, and continued to show sustained improvement during maintenance therapy with eculizumab. After discharge, the patient continued eculizumab treatment every 2 weeks, while the prednisone dose was gradually reduced over 1 month. Pyridostigmine was maintained at 60 mg every 8 h. Tumor treatment with capecitabine was continued, and ICIs were discontinued. The patient’s clinical course is shown in Figure 1, and the ADL and QMG scores following eculizumab treatment are presented in Figure 2.
Clinical course.

Assessments of ADL and QMG following eculizumab treatment.
We conducted a 6-month follow-up for the patient, during which intravenous infusions of 1200 mg of eculizumab were administered biweekly, and pyridostigmine was maintained at a dosage of 60 mg every 8 h. Tumor therapy with capecitabine was continued. Currently, the patient remains stable with minimal symptom expression, and there is no evidence of tumor recurrence. The patient demonstrated good treatment tolerance and did not experience any adverse events during the follow-up period.
Literature review
A literature search was performed using the MEDLINE/PubMed database. The search terms included “Myasthenia gravis” and “Immune checkpoint inhibitors” or “Programmed Cell Death 1” or “PD-1” or “pembrolizumab,” along with “Eculizumab” or “complement inhibitors.” The search was restricted to articles published in English between January 1986 and July 2024. Titles, abstracts, and full articles were reviewed, and the references of each article were examined to identify additional cases. A total of three articles, comprising four cases, were retrieved (see Table 1).
Cases of complement inhibitor therapy for PD-1-related MG.

AChR, acetylcholine receptor; ICI, immune checkpoint inhibitor; irAE, immune-related adverse events; IVIg, intravenous immunoglobulin; MG, myasthenia gravis; PD-1, programmed cell death receptor 1; QMG, Quantitative Myasthenia Gravis.
Discussion
Here, we report a case of pembrolizumab-induced impending crisis state in severe MG in which rapid improvement was achieved using eculizumab as an initial rescue therapy. This approach effectively prevented further deterioration of the patient’s condition caused by multiple rounds of potentially ineffective medication adjustments.
ICIs are immunomodulatory antibodies that enhance immune system function. ICIs targeting PD-1, programmed cell death ligand 1 (PD-L1), and cytotoxic T lymphocyte-associated antigen 4 have significantly improved clinical outcomes in various cancer types. 4 Pembrolizumab, a monoclonal antibody that binds to PD-1 and blocks its interactions with PD-L1 and PD-L2, alleviates PD-1 pathway-mediated inhibition of immune responses, including antitumor immunity.
While ICIs have contributed to improved survival in cancer patients, concerns have been raised regarding irAEs associated with their use,1,2 which can affect multiple organ systems. In particular, the incidence of neurological irAEs, such as MG, myopathy, and peripheral neuropathy, may range from 1% to 5%.3,5,6 Although relatively uncommon, ICIs may induce fulminant or even fatal toxic reactions. 7
MG is a rare but potentially life-threatening adverse reaction to ICIs frequently occurring in patients treated with anti-PD-1 agents, such as pembrolizumab and nivolumab.3,8,9 Approximately 0.12%–0.2% of patients receiving ICIs and approximately 14% of those with irAEs develop MG. A single-center study and literature review of ICI-related MG (ICI-MG) in the United States reported that among 65 patients diagnosed with ICI-MG, 82% had received anti-PD-1 therapy, and 97% developed ICI-MG (new onset or disease flare) after a median of 4 weeks following ICI initiation. 10 The positive rate of AChR antibodies in ICI-MG ranges from 50% to 70%. 11 The symptoms of ICI-MG are typically more severe than those of non-ICI-MG.10,12 ICI-MG is also associated with a higher incidence of overlapping myocarditis and myositis. The mortality rate for patients with myocardial or bulbar involvement can reach 60%, highlighting the association between neuromuscular irAEs and increased hospitalization and mortality rates. 13 Data on PD-1-related MG indicate that most patients require support for upper airway muscles and that many require multiple rounds of various immunotherapies, including plasma exchange, glucocorticoid pulse therapy, and IVIg pulse therapy. 12 Although rituximab has been used in the treatment of MG, 14 the outcomes have been unsatisfactory. 10
Eculizumab, a humanized monoclonal antibody that specifically inhibits the enzymatic cleavage of complement protein C5 and blocks the formation of the terminal complement complex, has been approved for the treatment of patients with AChR antibody-positive, refractory generalized MG. 15 Eculizumab binds strongly and specifically to C5, inhibiting its cleavage into C5a and C5b. This helps preserve the defense mechanisms of the proximal complement pathway, which are critical in treating complement-mediated diseases.15,16 A small prospective Chinese case series of impending myasthenic crises demonstrated a significant decrease in serum CH50 and soluble C5b-9 levels, while serum anti-AChR antibody levels, C1q and C5a levels, and peripheral lymphocyte proportions did not show significant changes, indicating the efficacy and safety of eculizumab. 16 Clinical reports have further confirmed the efficacy of eculizumab in improving clinical outcomes and enabling successful weaning from invasive ventilation in most patients with myasthenic crises.16–20 Concurrent systemic infection and delayed administration of eculizumab have been reported to limit complete functional recovery. However, two cases of refractory impending myasthenic crisis at the same center achieved minimal manifestations shortly after timely drug administration. 21 Due to its fast-acting therapeutic effects and crisis-rescue capability, eculizumab was used as the first-line treatment in our case to prevent disease exacerbation due to delayed administration. The rapid improvement of clinical symptoms and pCO2 levels in our patient following the initial dose further supported this treatment approach.
To date, four cases of complement inhibitor therapy for PD-1-related MG have been reported (Table 1). Pembrolizumab, which has low affinity for C1q and Fc receptors, exerts its therapeutic effects without activating antibody-dependent cellular cytotoxicity or complement-dependent cytotoxicity. It has therefore been hypothesized that eculizumab provides a mechanism-based treatment for MG in patients receiving pembrolizumab, without interfering with the latter’s action. 22 Our study is the first to report the direct use of eculizumab as a first-choice rescue therapy. The marked improvement in respiratory function within several hours of treatment is noteworthy, given that the average duration for significant improvement is typically 4 weeks. 21 After discussing with the patient and his family, we decided not to initiate conventional treatments, such as glucocorticoid pulse therapy, IVIg therapy, or plasma exchange. The patient underwent treatment for 6 months without complications, and the long-term therapeutic efficacy remained stable.
The patient exhibited a mild elevation in CK and CK-MB levels upon admission in our study, while both the electrocardiogram and cardiac ultrasound results were unremarkable. This was likely attributable to immune-related myositis and myocarditis induced by PD-1 inhibitors. Following the discontinuation of PD-1 inhibitor therapy and the initiation of eculizumab, the relevant biomarkers rapidly normalized, effectively mitigating the risk of progression to severe myositis or myocarditis.
The rapid response to eculizumab in our patient was of considerable significance. First, the damage to the postsynaptic membrane may be related to the duration of MG. One study reported that in an untreated patient with generalized MG of only 2 months, both the mean nerve terminal area and the mean postsynaptic membrane profile concentration were significantly reduced. 23 In our case, this was the patient’s first onset of ICI-induced MG (ICI-MG), and the disease course was only 8 days, suggesting that the function of the postsynaptic membrane was relatively preserved. Previous studies have reported a slightly lower efficacy of eculizumab treatment,15,16 which may be attributed to the late timing of add-on treatment following the failure of traditional rescue therapy. Further damage to the postsynaptic membrane may occur when conventional treatments are ineffective. Second, due to the rapid pharmacokinetic effect of eculizumab, serum C5 levels can be completely suppressed within 1 day after the initial dose administration.24,25
The balance between antitumor treatment and anti-MG therapy to achieve optimal therapeutic outcomes remains a challenge. Zadeh et al. 26 reported a case in which a patient died of cancer 1 year after experiencing improvement in PD-1-related MG. It remains unclear whether the use of complement inhibitors and steroids contributed to cancer progression. Additionally, biomarkers to predict the onset of ICI-MG remain scarce. In patients with preexisting autoimmune diseases, the risks and benefits must be carefully weighed. 27
Consensus on the optimal timing for reinitiating anti-PD-1 therapy remains absent and warrants further investigation. While previous case reports have not suggested that complement inhibitors may impact the antitumor efficacy of anti-PD-1 therapy, ICI therapy has been discontinued in most cases. In a single-center study and literature review, Safa et al. reported that of a total of 63 patients, discontinuation or withholding of ICI therapy was recommended for 61 (97%), while the remaining 2 continued using ICIs after their symptoms were resolved with steroid treatment. 10
Conclusion
We report a case of pembrolizumab-induced AChR-positive MG that was rapidly and effectively treated with eculizumab as rescue therapy. Our findings demonstrate that eculizumab, which produced a rapid and effective response, is a promising therapeutic option for pembrolizumab-related MG. However, further studies are warranted to validate these findings and provide deeper insights.
